Antipsychotica bij dementie
Uitgangsvraag
Wat is de rol van antipsychotica in de behandeling van neuropsychiatrische symptomen bij patiënten met dementie?
De module 'Antipsychotica bij dementie' bestaat uit de twee deelvragen:
- Wat is de effectiviteit van antipsychotica in de behandeling van neuropsychiatrische symptomen bij patiënten met dementie?
- Wat is het meest effectieve antipsychoticum als behandeling van neuropsychiatrische symptomen bij patiënten met dementie?
Aanbeveling
Algemeen
- Bespreek en overweeg het starten van een medicamenteuze behandeling enkel bij ernstige neuropsychiatrische symptomen (psychose, agitatie) en wanneer niet-farmacologische interventies afdoende zijn geprobeerd maar onvoldoende werkzaam zijn.
- Evalueer andere (somatische) oorzaken van psychotische klachten en sluit eventueel uit.
- Het cluster maakt geen onderscheid per type dementie, maar wees uiterst terughoudend met het gebruik van antipsychotica bij patiënten met Lewy body dementie en/of Parkinson dementie.
- Gezien het beperkte bewijs, maakt het cluster geen onderscheid in behandeling tussen agitatie en psychotische klachten.
Starten
- Start een antipsychoticum bij dementie uitsluitend na het bespreken van doelsymptomen en behandelduur met patiënt en (mantel)zorgverlener. Gebruik voor het kwantificeren van de doelsymptomen een gevalideerd meetinstrument, zoals de BPRS of NPI.
- Streef naar een zo kort mogelijke behandelperiode.
- Gezien het beperkte bewijs onthoudt het cluster zich van uitspraken betreffende de effectiviteit van middelen ten opzichte van elkaar. Overweeg op basis van het gunstigere bijwerkingenprofiel bij ernstige neuropsychiatrische symptomen en onvoldoende effect van niet-medicamenteuze interventies te starten met een lage dosis risperidon (0,5mg per dag tot maximaal 2 keer per dag 1mg).
- Monitor de klachten interdisciplinair en overweeg bij gedeeltelijke respons en goed verdragen van het medicijn, de medicatiedosering te verhogen.
- Evalueer na maximaal zes weken de symptomen met behulp van hetzelfde meetinstrument als voorafgaand aan het starten is gebruikt.
Stoppen
- Stop met de medicamenteuze behandeling indien dit geen of onvoldoende effect geeft op de doelsymptomen na zes weken.
- Stop de behandeling of verlaag de dosering van antipsychotica bij onacceptabele bijwerkingen in overleg met patiënt en (mantel)zorgverlener.
Overwegingen
Vergelijking 1 ‘Antipsychotica versus placebo’
Kwaliteit van bewijs
De overall kwaliteit van bewijs is zeer laag. Dit betekent dat we zeer onzeker zijn over het gevonden geschatte effect van de cruciale uitkomstmaten psychose, slaperigheid en extrapiramidale symptomen. Er is afgewaardeerd vanwege:
- Risk of Bias: methodologische beperkingen (hoog risico op bias op meerdere domeinen, waaronder selectie bias, attrition bias, reporting bias).
- Inconsistentie: conflicterende resultaten.
- Imprecisie: onnauwkeurigheid - brede betrouwbaarheidsintervallen (één grens van klinische relevantie wordt overschreden) en het lage aantal patiënten.
De geïncludeerde systematische review van Mühlbauer (2024) heeft enkele (methodologische) beperkingen die bijdragen aan inconsistenties tussen hun en onze bevindingen. Zo ontbreekt informatie over de beoordeling van de kwaliteit van het bewijs voor de belangrijke uitkomstmaten ‘carer burden’ en ‘cognitive function’. Daarnaast werden veel van de geïncludeerde studies beoordeeld als ‘hoog risico op bias in ten minste één domein,’ zonder dat hier altijd voldoende voor werd afgewaardeerd in de GRADE beoordeling. Dit kan hebben geleid tot een overschatting van de kracht van de bevindingen. Verder ontbreekt in de gepoolde resultaten een duidelijke specificatie van het onderzochte middel, wat het lastig maakt om de oorsprong van de waargenomen effecten te bepalen. Het is belangrijk bovenstaande kanttekeningen in het achterhoofd te houden bij het interpreteren van de resultaten.
Balans tussen gewenste en ongewenste effecten
Typische antipsychotica
Uit de literatuuranalyse blijkt dat in de vergelijkingen tussen typische antipsychotica ten opzichte van placebo (Mühlbauer, 2024) een klein klinisch relevant verschil werd gezien op de uitkomstmaten psychose en agitatie bij de typische antipsychotica, waarbij de bewijskracht is geclassificeerd als zeer laag. De uitkomstmaten somnolentie en extrapiramidale bijwerkingen waren klinisch relevant verschillend, in het nadeel van de typische antipsychotica waarbij de bewijskracht voor de uitkomstmaat somnolentie is geclassificeerd als laag, en de uitkomstmaat extrapiramidale verschijnselen als matig. In het algemeen is het hierbij wel van belang te benoemen dat de dosering van haloperidol hier nog een rol in kan spelen, gezien er in de (oudere) geincludeerde studies met hogere doseringen haloperidol (met meer kans op bijwerkingen) werd behandeld.
In Mühlbauer (2024) is gekeken naar 5 separate studies waarin haloperidol als typisch antipsychoticum werd onderzocht. Zoals hierboven reeds beschreven heeft deze systematische review zijn methodologische beperkingen.
Daarnaast betreft het dus een beperkt aantal studies waarin haloperidol is meegenomen, waarin ook sprak is van vrij veel heterogeniteit. In een beperkte groep is een brede dosisrange (van 0,5mg-12mg) en ook een sterk wisselende follow-up duur (3 tot 16 weken). Dit maakt dat de bewijskracht zeer laag is en ook niet zonder meer te extrapoleren is. Daarnaast zijn enkele geincludeerde studies in Mühlbauer (2024) van oudere datum. Er zijn bij het cluster geen recentere studies bekend die het effect van haloperidol onderzochten in deze populatie.
Bij de interpretatie van de resultaten van de typische antipsychotica is het belangrijk om op te merken dat naast de vijf haloperidol studies, slechts één andere studie is geïncludeerd wat het typisch antipyschoticum tiotixeen onderzocht (een middel dat niet in Nederland verkrijgbaar is). De getoonde resultaten in de forest plots zijn dus grotendeels te relateren aan haloperidol en kunnen niet zonder meer worden geëxtrapoleerd naar andere typische antipsychotica.
Uit de literatuuranalyse van de typische antipsychotica is te zien dat het effect op psychose door haloperidol beperkt is, met als belangrijkste nadelen somnolentie en extrapiramidale klachten. Dit is wat verwacht kan worden op basis van het werkingsmechanisme. Hierbij is de dosis range van de studies wel van belang, de hogere doseringen (zeker tot 12mg) worden in de Nederlandse klinische praktijk niet tot nauwelijks meer gebruikt. In de studies zou dit geleid kunnen hebben tot een overschatting van de bijwerkingen.
Atypische antipsychotica
In de literatuuranalyse voor atypische antipsychotica in vergelijking met placebo, is voor de antipsychotica gezamenlijk geen klinisch relevant verschil te zien op de uitkomstmaten psychose en agitatie. Wel was een klinisch relevant verschil in het nadeel van antipsychotica voor de bijwerkingen somnolentie en extrapiramidale symptomen. Voor deze beide uitkomsten werd de bewijskracht als laag geclassificeerd.
Voor de vergelijking van atypische antipsychotica met placebo, is wederom de systematische review van Mühlbauer (2024) gebruikt, met opnieuw de methodologische beperkingen zoals eerder beschreven.
In totaal zijn twaalf studies meegenomen voor de primaire uitkomstmaat psychose. Dertien studies zijn meegenomen in de bijwerking somnolentie en vijftien studies hebben extra piramidale symptomen meegenomen als uitkomstmaat.
Ook bij deze studies is het belangrijk op te merken dat er verschillende atypische antipsychotica met verschillende receptoraffiniteit, verschillende doseringen en variabele follow-up duur zijn gebruikt. Als geheel bekeken komt hier dus geen evident positief effect naar voren.
Vergelijking 2 ‘Antipsychotica onderling vergeleken’
Kwaliteit van bewijs
De overal bewijskracht is zeer laag. Dit betekent dat we zeer onzeker zijn over het gevonden geschatte effect van de cruciale uitkomstmaten psychose, somnolentie en extrapiramidale symptomen. Er is afgewaardeerd vanwege:
- Risk of Bias: methodologische beperkingen.
- Imprecisie: onnauwkeurigheid: brede betrouwbaarheidsintervallen en het lage aantal patiënten.
Balans tussen gewenste en ongewenste effecten
Net als bij de eerder beschreven systematische review bij de typische antipsychotica (Mühlbauer,2024), is de kwaliteit van het bewijs voor de onderlinge vergelijking tussen antipsychotica zeer laag (Lü, 2024).
Uiteindelijk zijn de verschillen tussen antipyschotica in deze module bestudeerd aan de hand van drie studies waarbij een head-to-head vergelijking heeft plaatsgevonden: Rainer (2007), Schneider (2006) en Tario (2006). De NMA (Lü, 2024) omschrijft slechts drie closed loops (i.e. head-to-head comparisons). Dit betreft vergelijkingen tussen:
- Quetiapine versus risperidon (Rainer, 2007).
- Olanzapine versus quetiapine versus risperidon (Schneider, 2006).
- Quetiapine versus haloperidol (Tariot, 2006).
Alle studies hebben hierbij een verschillend doseringsregime en een andere follow-up duur. Tevens zijn in deze studies met name voor quetiapine en haloperidol veel hogere doseringen gebruikt dan in Nederland gebruikelijk bij neuropsychiatrische symptomen bij dementie.
Omdat er binnen de studies verschillende meetschalen werden gebruikt en niet altijd een gemiddelde SD is gerapporteerd, konden resultaten niet gepoold worden. Hierdoor waren de resultaten lastig te vergelijken en heeft dit met name tot zwakke beschrijvingen geleid, waarbij er mogelijk of misschien sprake is van een klein verschil. Nergens is een klinisch relevant verschil gevonden. Derhalve is op basis hiervan geen sterke uitspraak te doen betreft voorkeur voor het ene of het andere antipsychoticum.
Om op de cruciale uitkomstmaten psychose, somnolentie en extrapiramidale klachten, nog wel een indruk te kunnen krijgen van de verschillen per middel zijn de effecten per atypisch antipsychoticum versus placebo nog uitgesplitst in forest plots (zie figuur S1-3 in de bijlage). Hieruit komt naar voren dat de verschillen klein zijn. In het algeheel lijkt qua effect op psychose risperidon iets effectiever te zijn dan de andere atypische antipsychotica, met wel een risico op bijwerkingen in de vorm van somnolentie en extrapiramidale symptomen, die vergelijkbaar zijn met de andere onderzochte en in Nederland beschikbare atypische antipsychotica. In de vergelijking met de analyse op typische antipsychotica (met name haloperidol) lijkt het effect van deze op de uitkomstmaat psychose iets effectiever dan risperidon, echter lijkt het risico op somnolentie en extrapiramidale symptomen groter te zijn.
In het geheel is geen duidelijk verschil terug te zien betreffende effectiviteit op verschillende ingangsklachten (bijvoorbeeld psychotische klachten versus agitatie). Daarom is dit onderscheid in de aanbeveling losgelaten en is er dus voor gekozen om voor beide ingangsklachten één middel te adviseren. Dit met de kanttekening dat hierbij vooral naar het bijwerkingenprofiel is gekeken (EPS en somnolentie) en daarnaast praktische ervaring met het middel. In zijn geheel lijkt risperidon dan ietwat gunstiger naar voren te komen. Omdat het effect beperkt is met wel een reëel risico op bijwerkingen is het de overweging om altijd met een lage dosering te starten (i.e. 0,5mg per dag, tot een maximum van twee keer per dag 1mg) (ephor.).
Het is belangrijk om te benadrukken dat bij het raadplegen van andere literatuur (Verenso, 2018; NICE, 2018) er vanuit dezelfde beschikbare bronnen/ bekeken literatuur verschillende aanbevelingen geformuleerd zijn. In het algemeen is er dus geen consensus over wat het middel van eerste keus moet zijn. Dit impliceert des te meer dat de onderlinge verschillen zeer klein zijn.
Type dementie
In de beschreven studies zijn patiënten met Lewy body dementie en Parkinson dementie uitgesloten. Er kan daarom geen uitspraak worden gedaan over de effecten van behandeling met antipsychotica bij deze populatie. Daarnaast is het gebruik van antidopaminerge medicatie (met name typische antipsychotica) bij deze typen van dementie relatief gecontra-indiceerd gezien het risico op verergering van extrapyramidale symptomen, hierbij is terughoudendheid in het gebruik geboden. Bij een noodzaak tot behandeling wordt er verwezen naar de richtlijn ‘Ziekte van parkinson’ (NVN, 2020).
Overig
Het onderwerp ‘medicamenteuze behandeling van neuropsychiatrische symptomen’ wordt uitgewerkt in verschillende modules, gefocust en onderverdeeld op de behandeling per farmacotherapeutische groep (zie Medicatie neuropsychiatrische symptomen). Een bepaald doelsymptoom kan dus in meerdere modules zijn uitgewerkt, waarbij per module een aanbeveling wordt gedaan voor een keuze binnen deze farmacotherapeutische groep. Er kan hiermee geen uitspraak worden gedaan over een vergelijking van de effecten tussen de verschillende farmacotherapeutische groepen.
Algemeen: Antipsychotica (vergelijking 1 en 2)
Waarden en voorkeuren van patiënten (en eventueel hun naasten/verzorgers)
De gewenste effecten op patiëntrelevante uitkomstmaten zijn klein. De ongewenste effecten op patiëntrelevante uitkomstmaten zijn aanwezig, maar niet volledig bekend.
Alles bij elkaar lijkt het totale effect op psychotische klachten bij alle middelen beperkt, een enkel middel laat een statistisch significant resultaat zien die methodologisch ook als een klein klinisch relevant effect kan worden bestempeld. Daartegenover staat het risico op bijwerkingen, waarbij mede vanwege het werkingsmechanisme extrapiramidale symptomen en somnolentie voor de hand liggen. Echter, vanwege de heterogeniteit in de studies, is het moeilijk om een exacte uitspraak te doen over hoe vaak bijwerkingen precies optreden. Wel is te zeggen dat er geen enorme verschillen tussen middelen te zien zijn.
Kostenaspecten
Gezien de lage kosten van de onderzochte antipsychotica, is zijn de kosten geen doorslaggevende factor en van grote invloed in het uitspreken van een voorkeur voor een bepaald middel. Bij het cluster zijn geen kosteneffectiviteit-studies bekend.
Gelijkheid ((health) equity/equitable)
Gezien de geringe kosten van de geneesmiddelen zijn deze niet van invloed op gelijkheid of toegankelijkheid voor patiënten in verschillende stadia van zorg, zoals thuiswonend, opgenomen in het ziekenhuis of woonachtig in een zorgcentrum.
Aanvaardbaarheid
Ethische aanvaardbaarheid
Er zijn geen ethische bezwaren om antipsychotica in te zetten conform de aanbevelingen. Hierbij is het wel van belang te benadrukken dat dit zo is gezien het kortdurende gebruik, waarbij de risico’s op negatieve uitkomsten die buiten de uitkomstmaten van deze zoekvraag (mortaliteit, qtc verlenging) beperkt is zoals ook beschreven in de Federatie richtlijn ‘Kortdurend antipsychoticagebruik’ (NVKG, 2019).
Duurzaamheid
Er zijn geen duurzaamheidsaspecten van invloed naar voren gekomen.
Haalbaarheid
In de praktijk is er veel ervaring met gebruik van antipsychotica bij de beschreven indicatie. Hierbij heeft risperidon ook reeds een specifieke indicatie bij het gebruik bij dementie. Daarnaast betreft het hier bekende geneesmiddelen waar binnen de relevante beroepsgroep al ervaring mee is opgedaan en die ook in de voorgaande versie van deze module zijn beschreven Daarom lijkt deze aanpassing wel haalbaar.
‘Antipsychotica versus placebo’ EN ‘Antipsychotica onderling vergeleken’
Rationale van de aanbeveling: weging van argumenten voor en tegen de interventies
De belangrijkste overweging betreft de afweging van resultaten op positieve effecten (psychose en agitatie) ten opzichte van bijwerkingen (EPS en somnolentie) zowel van antipsychotica versus placebo (vergelijking 1) als antipsychotica onderling (vergelijking 2). Gezien de (zeer) lage bewijskracht en de kleine verschillen in effectiviteit, laat het cluster het onderscheid in benadering van de verschillende doelsymptomen los.
Eindoordeel: Zwakke aanbeveling voor.
Onderbouwing
Cognitive issues associated with dementia are well established, but around 90% of the patients with dementia may also experience non-cognitive symptoms such as increased aggression, anxiety, apathy, agitation, depression, delusions, hallucinations and sleep disturbances (NICE, 2018).
Herewith, patients with (more advanced) dementia could experience neuropsychiatric symptoms (e.g. agitation, aggression, distress, psychosis). These symptoms could be caused by the progression of dementia, by a psychiatric cause or by somatic causes. These symptoms significantly impact patients, families, and carers. If these symptoms could be treated successfully, it would have a big impact on the quality of life for everyone concerned.
However, the effectiveness and specific type of antipsychotics is unclear. Therefore, this module aims to evaluate the effectiveness and identify the best antipsychotic treatments for managing neuropsychiatric symptoms in dementia patients.
Subquestion 1. Placebo comparisons
1.1 Typical antipsychotics
Population: Patients with dementia and neuropsychiatric symptoms
Intervention: typical antipsychotics; Comparator: Placebo
|
Outcome Timeframe |
Study results and measurements |
Absolute effect estimates |
Certainty of the evidence (Quality of evidence) |
Summary |
|
|
Placebo |
Typical antipsychotics |
||||
|
Psychosis (critical) |
Measured by: NPI-NH Scale: - Lower better Based on data from 240 participants in 2 studies |
Difference: SMD 0.29 lower (CI 95% 0.55 lower - 0.03 lower) |
Very low Due to very serious risk of bias and serious imprecision1 |
We are uncertain whether typical antipsychotics improve or worsen psychosis |
|
|
Somnolence (critical) |
Relative risk: 2.75 (CI 95% 0.85 - 8.92) Based on data from 466 participants in 3 studies |
7 per 100 |
19 per 100 |
Low Due to serious risk of bias and serious imprecision2 |
Typical antipsychotics may worsen somnolence |
|
Difference: 12 more per 100 (CI 95% 4 fewer - 26 more) |
|||||
|
Extrapyramidal symptoms (critical) |
Relative risk: 2.26 (CI 95% 1.58 - 3.23) Based on data from 467 participants in 3 studies |
15 per 100 |
33 per 100 |
Moderate Due to serious risk of bias3 |
Typical antipsychotics probably worsen extrapyramidal symptoms |
|
Difference: 18 more per 100 (CI 95% 8 more - 33 more) |
|||||
|
Agitation (important) |
Measured by: CMAI Scale: Lower better Based on data from 361 participants in 4 studies |
Difference: SMD 0.40 lower (CI 95% 0.77 lower - 0.03 lower) |
Very low Due to very serious risk of bias, and serious imprecision4 |
We are uncertain whether typical antipsychotics improve or worsen agitation |
|
|
Carer burden (important) |
Measured by: Scale: Lower better Based on data from 70 participants in 1 study |
Difference: MD 0.70 higher (CI 95% 3.65 lower - 5.05 higher) |
Very low Due to serious risk of bias and very serious imprecision5 |
We are uncertain whether typical antipsychotics improve or worsen carer burden |
|
|
Cognitive function (important) |
Measured by: Scale: - High better Based on data from 205 participants in 2 studies |
Difference: MD 0.23 lower (CI 95% 1.27 lower - 0.77 higher) |
Very low Due to serious risk of bias and very serious imprecision6 |
We are uncertain whether typical antipsychotics improve or worsen cognitive function |
|
|
Health-related quality of life (important) |
- |
- |
No GRADE (no evidence was found) |
No evidence was found regarding the effect of typical antipsychotics on health-related quality of life |
|
CMAI: Cohen-Mansfield Agitation Inventory; NPI - NH: Neuropsychiatric Inventory Home Version; SMD: Standardized mean difference; MD: Mean difference.
1. Risk of Bias: very serious. All studies were rated at high risk of bias in at least one of the following domains: selection bias (comparability of study groups), attrition bias (incomplete outcome data), and other bias (use of a run-in period); Imprecision: serious. Wide confidence intervals.
2. Risk of Bias: serious. Due to high dropout rates, baseline differences, and missing information on multiple domains; Imprecision: serious. Wide confidence intervals.
3. Risk of Bias: serious. Due to high dropout rates, baseline differences, and missing information on multiple domains.
4. Risk of Bias: very serious. All studies were rated at high risk of bias in at least one of the following domains: selection bias (comparability of study groups), attrition bias (incomplete outcome data), and other bias (use of a run-in period); Imprecision: serious. Confidence interval indicates both an important effect and an effect with no clinical relevance.
5. Risk of Bias: serious. Due to missing information on multiple domains and because the study was rated at high risk of bias at the following domain: other sources of bias (use of a run-in period, commercial funding). Imprecision: very serious. Wide confidence intervals (crossing one border of clinical relevance) and the low number of patients.
6. Risk of Bias: serious. Due to missing information on multiple domains and because the studies were rated at high risk of bias at the following domain: other sources of bias (use of a run-in period, commercial funding); Imprecision: serious. Wide confidence intervals and the low number of patients.
1.2 Atypical antipsychotics
Population: Patients with dementia and neuropsychiatric symptoms
Intervention: Atypical Antipsychotics; Comparator: Placebo
|
Outcome Timeframe |
Study results and measurements |
Absolute effect estimates |
Certainty of the evidence (Quality of evidence) |
Summary |
|
|
Placebo |
Atypical antipsychotics |
||||
|
Psychosis (critical) |
Measured by: NPI-NH Scale: 1 - 12 Lower better Based on data from 3364 participants in 12 studies Follow up 3 to 12 weeks |
Difference: SMD 0.11 lower (CI 95% 0.18 lower - 0.03 lower) |
Low Due to serious risk of bias and serious imprecision1 |
Atypical antipsychotics may have little or no effect on psychosis |
|
|
Somnolence (critical) |
Relative risk: 2.39 (CI 95% 1.67 - 4.4) Based on data from 3878 participants in 13 studies Follow up 3 to 12 weeks |
71 per 1000 |
150 per 1000 |
Low Due to serious risk of bias and serious imprecision2 |
Atypical antipsychotics may worsen somnolence |
|
Difference: 79 more per 1000 |
|||||
|
Extrapyramidal symptoms (critical) |
Relative risk: 1.39 (CI 95% 1.14 - 1.68) Based on data from 4180 participants in 15 studies Follow up 3 to 12 weeks |
82 per 1000 |
140 per 1000 |
Low Due to serious risk of bias and serious imprecision3 |
Atypical antipsychotics may worsen extrapyramidal symptoms |
|
Difference: 58 more per 1000 |
|||||
|
Agitation (important) |
Measured by: CMAI Scale: 29 - 203 High better Based on data from 1971 participants in 9 studies Follow up 3 to 12 weeks |
Difference: SMD 0.21 lower (CI 95% 0.30 lower - 0.12 lower) |
Low Due to serious risk of bias and serious imprecision4 |
Atypical antipsychotics may have little or no difference on agitation |
|
|
Health-related quality of life (important) |
Measured by: Scale: High better Based on data from 151 participants in 1 study |
Difference: MD 0.95 lower (CI 95% 6.04 lower - 4.14 higher) |
Very low Due to serious risk of bias, serious inconsistency and serious imprecision5 |
We are uncertain whether atypical antipsychotics improve or worsen health-related quality of life |
|
|
Cognitive function (important) |
Measured by: Scale: High better Based on data from 2698 participants in 11 studies |
Difference: SMD 0.11 higher (CI 95% 0.01 higher - 0.22 higher) |
Low Due to serious risk of bias and serious imprecision6 |
Atypical antipsychotics may have little or no difference on cognitive function |
|
|
Carer burden (important) |
- |
- |
No GRADE (no evidence was found) |
No evidence was found regarding the effect of atypical antipsychotics on carer burden |
|
CMAI: Cohen-Mansfield Agitation Inventory; NPI - NH: Neuropsychiatric Inventory Home Version; SMD: Standardized mean difference; MD: Mean difference.
1. Risk of Bias: serious. All studies were rated at high risk of bias in at least one of the following domains: selection bias (comparability of study groups), attrition bias (incomplete outcome data), reporting bias (selective outcome reporting), and other bias; Imprecision: serious. Wide confidence intervals.
2. Risk of bias: serious. All studies were rated at high risk of bias in at least one of the following domains: selection bias (comparability of study groups), attrition bias (incomplete outcome data), reporting bias (selective outcome reporting), and other bias; Imprecision: serious. Wide confidence intervals.
3. Risk of Bias: serious. All studies were rated at high risk of bias in at least one of the following domains: selection bias (comparability of study groups), attrition bias (incomplete outcome data), reporting bias (selective outcome reporting), and other bias; Imprecision: serious. Wide confidence intervals (crossing border of clinical relevance).
4. Risk of Bias: serious. All studies were rated at high risk of bias in at least one of the following domains: selection bias (comparability of study groups), attrition bias (incomplete outcome data), reporting bias (selective outcome reporting), and other bias; Imprecision: serious. Wide confidence intervals (crossing border of clinical relevance).
5. Risk of Bias: serious. Due to missing information about randomization procedure and blinding, and high risk of attrition bias (incomplete outcome data). Inconsistency: serious. Conflicting results. Imprecision: serious. Wide confidence intervals and the low number of patients.
6. Risk of Bias: serious. All studies were rated at high risk of bias in at least one of the following domains: selection bias (comparability of study groups), attrition bias (incomplete outcome data), reporting bias (selective outcome reporting), and other bias; Imprecision: serious. Wide confidence intervals (crossing border of clinical relevance).
Subquestion 2. Head-to-head comparisons
2.1 Typical antipsychotics
Population: Patients with dementia and neuropsychiatric symptoms
Intervention: Typical Antipsychotics; Comparator: Head-to-head
Not reported.
2.2 Atypical antipsychotics
Population: Patients with dementia and neuropsychiatric symptoms
Intervention: Atypical Antipsychotics; Comparator: Head-to-head
Quetiapine vs risperidone
|
Outcome Timeframe |
Study results and measurements |
Absolute effect estimates |
Certainty of the evidence (Quality of evidence) |
Summary |
|
|
risperidone |
Quetiapine |
||||
|
Psychosis (critical) |
Based on data from 128 participants in 2 studies Follow up 8 to 12 weeks |
Results do not show a difference in treatment. Since the studies used different scales, and not reported mean (SD), data could not be pooled. |
Low Due to serious risk of bias and serious imprecision1 |
Quetiapine may have little or no difference on psychosis compared to risperidone. |
|
|
Somnolence (critical) |
Relative risk: 0.05 (CI 95% -0.03 - 0.14) Based on data from 72 participants in 1 studies Follow up 8 weeks |
0 per 1000 |
53 per 1000 |
Low Due to serious risk of bias and serious imprecision2 |
Quetiapine may have little or no difference on somnolence compared to risperidone |
|
Difference: 53 fewer per 1000 (CI 95% 0 fewer - 0 fewer) |
|||||
|
Extrapyramidal symptoms (critical) |
Based on data from 72 participants in 1 studies Follow up 8 weeks |
The SAS score increased by 0.6 in the Quetiapine group, and 0.35 in the risperidone group. Mean (SD) was not reported. |
Low Due to serious risk of bias and serious imprecision3 |
Quetiapine may have little or no difference on extrapyramidal symptoms compared to risperidone |
|
|
Agitation (important) |
Based on data from 72 participants in 1 studies Follow up 8 weeks |
The CMAI score reduced by 3.54 in the Quetiapine group, and 5.51 in the risperidone group. Mean (SD) was not reported. |
Low Due to serious risk of bias and serious imprecision4 |
Quetiapine may have little or no difference on agitation compared to risperidone |
|
|
Carer burden (important) |
Based on data from 72 participants in 1 studies Follow up 8 weeks |
The NPI-NH score reduced by 2.5 in the Quetiapine group, and 3.6 in the risperidone group. Mean (SD) was not reported. |
Low Due to serious risk of bias and serious imprecision5 |
Quetiapine may have little or no difference on Carer Burden compared to risperidone |
|
|
Cognitive function (important) |
Based on data from 72 participants in 1 studies Follow up 8 weeks |
The MMSE score reduced by 0.03 in the Quetiapine group, and increased by 0.65 in the risperidone group. Mean (SD) was not reported. |
Low Due to serious risk of bias and serious imprecision6 |
Quetiapine may have little or no difference on Cognitive function compared to risperidone |
|
|
Health-related quality of life (important) |
- |
- |
No GRADE (no evidence was found) |
No evidence was found regarding the effect of Quetiapine on health-related quality of life |
|
SAS: Simpson Angus scale; CMAI: Cohen-Mansfield Agitation Inventory; NPI - NH: Neuropsychiatric Inventory Home Version; MMSE: Mini-Mental State Examination; MD: mean difference; SD: standardized difference
1. Risk of Bias: serious. Imprecision: serious. Wide confidence intervals.
2. Risk of Bias: serious. Imprecision: serious. Low number of patients.
3. Risk of Bias: serious. Imprecision: serious. Low number of patients.
4.Risk of Bias: serious. Imprecision: serious. Low number of patients.
5.Risk of Bias: serious. Imprecision: serious. Low number of patients.
6.Risk of Bias: serious. Imprecision: serious. Low number of patients.
Olanzapine vs Quetiapine
|
Outcome Timeframe |
Study results and measurements |
Absolute effect estimates |
Certainty of the evidence (Quality of evidence) |
Summary |
|
|
Olanzapine |
Quetiapine |
||||
|
Psychosis (critical) |
Based on data from 71 participants in 1 studies Follow up 12 weeks |
Psychosis was reported with BPRS and NPI-NH score. The MD for BPRS was 2.20 (95%CI: - 6.46 to 10.86) in favour of the Quetiapine group. The MD for NPI was 0.0 (95% CI: -4.64 to 4.64). |
Low Due to serious risk of bias and serious imprecision1 |
Quetiapine may have little or no difference on psychosis compared to Olanzapine |
|
|
Extrapyramidal symptoms (critical) Somnolence (critical) Agitation (important) Carer burden (important) Cognitive function (important) Health-related quality of life (important) |
- |
- |
No GRADE (no evidence was found) |
No evidence was found regarding the effect of Quetiapine on extrapyramidal symptoms, somnolence, agitation, carer burden, cognitive function, and health-related quality of life |
|
BPRS: Brief Psychiatric Rating Scale; NPI- NH: Neuropsychiatric Inventory Home Version; MD: mean difference.
1.Risk of Bias: serious. Imprecision: serious. Wide confidence intervals.
Olanzapine vs risperidone
|
Outcome Timeframe |
Study results and measurements |
Absolute effect estimates |
Certainty of the evidence (Quality of evidence) |
Summary |
|
|
risperidone |
Olanzapine |
||||
|
Psychosis (critical) |
Based on data from 71 participants in 1 studies Follow up 12 weeks |
Psychosis was reported with BPRS and NPI-NH score. The MD for BPRS was 2.40 (95%CI: - 5.58 to 10.18) in favour of the risperidone group. The MD for NPI was 1.10 (95% CI: -4.10 to 6.30) in favour of the risperidone group. |
Low Due to serious risk of bias and serious imprecision1 |
Olanzapine may have little or no difference on psychosis compared to risperidone |
|
|
Extrapyramidal symptoms (critical) Somnolence (critical) Agitation (important) Carer burden (important) Cognitive function (important) Health-related quality of life (important) |
- |
- |
No GRADE (no evidence was found) |
No evidence was found regarding the effect of Olanzapine on extrapyramidal symptoms, somnolence, agitation, carer burden, cognitive function, and health-related quality of life |
|
BPRS: Brief Psychiatric Rating Scale; NPI: Neuropsychiatric Inventory Home Version; MD: mean difference; 1.Risk of Bias: serious. Imprecision: serious. Wide confidence intervals.
Quetiapine vs Haloperidol
|
Outcome Timeframe |
Study results and measurements |
Absolute effect estimates |
Certainty of the evidence (Quality of evidence) |
Summary |
|
|
Haloperidol |
Quetiapine |
||||
|
Psychosis (critical) |
Based on data from 171 participants in 1 studies Follow up 10 weeks |
Psychosis was measured with BPRS and CGI-S score. The MD for BPRS was 1.93 (95%CI: - 1.36 to 5.22) in favour of the Haloperidol group. The MD for CGI-S was 0.08 (95% CI: -0.23 to 0.39 ) in favour of the Quetiapine group. |
Very low Due to serious risk of bias and very serious imprecision1 |
We are uncertain whether Quetiapine improves or worsens psychosis compared to Haloperidol |
|
|
Somnolence (critical) |
Relative risk: -0.11 (CI 95% -0.24 - 0.02) Based on data from 185 participants in 1 studies Follow up 10 weeks |
362 per 1000 |
253 per 1000 |
Low Due to serious risk of bias and serious imprecision2 |
Quetiapine may have little or no difference on somnolence compared to Haloperidol |
|
Difference: 109 fewer per 1000 |
|||||
|
Cognitive function (important) |
Measured by: MMSE Scale: - High better Based on data from 132 participants in 1 studies Follow up 10 weeks |
Difference: MD 0.52 higher (CI 95% 0.75 lower - 1.79 higher) |
Very low Due to serious risk of bias and very serious imprecision1 |
We are uncertain whether Quetiapine improves or worsens cognitive function compared to Haloperidol |
|
|
Agitation (important) |
Based on data from 161 participants in 1 studies Follow up 10 weeks |
Agitation was measured with BPRS-agitation and NPI-NH agitation score. The MD for BPRS was 0.23 (95%CI: - 0.82 to 1.82) in favour of the Haloperidol group. The MD for NPI-NH was 0.20 (95% CI: -1.36 to 1.76) in favour of the Quetiapine group. |
Very low Due to serious risk of bias and very serious imprecision1 |
We are uncertain whether Quetiapine improves or worsens agitation compared to Haloperidol |
|
|
Extrapyramidal symptoms (critical) Carer burden (important) Health-related quality of life (important) |
- |
- |
No GRADE (no evidence was found) |
No evidence was found regarding the effect of Quetiapine on extrapyramidal symptoms, carer burden, and health-related quality of life |
|
BPRS: Brief Psychiatric Rating Scale; CGI-S: Clinical Global Impressions–Severity of Illness; NPI - NH: Neuropsychiatric Inventory Home Version; MD: mean difference; SD: standardized difference
1. Risk of Bias: serious. Imprecision: very serious. Low number of patients and wide confidence intervals;
2. Risk of Bias: serious. Imprecision: serious. Low number of patients.
PICO 1. Description of studies
One systematic review was included in the analysis of the literature. Important study characteristics and results are summarized in Table 1. The assessment of the risk of bias is summarized in the risk of bias tables (under the tab ‘Evidence tabellen’).
Mühlbauer (2021) performed a Cochrane systematic review that assessed the efficacy and safety of antipsychotics for the treatment of agitation and psychosis in people with Alzheimer's disease and vascular dementia. On 7 January 2021, a search was performed in various international databases (ALOIS, the Cochrane Dementia and Cognitive Improvement Group's register, MEDLINE (Ovid Sp), Embase (Ovid SP), PsycINFO (Ovid SP), CINAHL (EBSCOhost), Web of Science Core Collection (ISI Web of Science), LILACS (BIREME), ClinicalTrials.gov and the World Health Organization's meta-register, and the International Clinical Trials Registry Portal). Studies that were included were randomized, placebo-controlled trials comparing the effects of antipsychotics and placebo for the treatment of agitation or psychosis in people with dementia due to Alzheimer's disease or vascular dementia. Authors excluded studies comparing different antipsychotics head-to-head and antipsychotic withdrawal trials.
The search yielded 8233 separate hits of which 24 trials met the eligibility criteria including 6090 patients. Six trials tested a typical antipsychotic (haloperidol, thiothixene), four for agitation and two for psychosis. Twenty trials tested an atypical antipsychotic (e.g. risperidone, olanzapine, aripiprazole, quetiapine), eight for agitation and twelve for psychosis. Pooled data were presented and included in the literature analyses. Reported outcomes measures were psychosis, adverse events (somnolence and extrapyramidal symptoms), agitation, carer burden or carer quality of life, and cognitive function.
PICO 2. Description of studies
One systematic review and network meta-analysis was included in the analysis of the literature. Important study characteristics and results are summarized in Table 2. The assessment of the risk of bias is summarized in the risk of bias tables (under the tab ‘Evidence tabellen’).
Lü (2024) conducted a systematic review and network meta-analysis on the efficacy, acceptability, and tolerability of second-generation antipsychotics, also known as atypical antipsychotics, on behavioural and psychological symptoms of dementia. A search was performed in four databases (PubMed, Embase, Web of Science, Cochrane Trial Register) from inception to December 2023. Patients were included with Alzheimer’s dementia (AD), vascular dementia, or mixed dementia. Patients with Parkinson’s dementia, Lewy body dementia, other mental comorbidities unrelated to dementia (e.g. depression, delirium, schizophrenia), or uncontrolled physical illness (e.g. any physical disease in the acute phase, cardiovascular- or cerebrovascular accidents) or poorly controlled chronic disease (eg, poorly controlled hypertension and diabetes, residual symptoms of cerebral vascular events convalescence) were excluded. There were no restrictions based on patient’s age or the severity of dementia.
The search yielded 874 separate hits of which 19 trials met the eligibility criteria for the NMA. Briefly, these studies were published between 1999 and 2023, and their sample sizes varied from 40 to 652 participants with a total of 6374 individuals with intervention lengths ranging from 6 weeks to 36 weeks.
The NMA only included three closed loops Quetiapine vs Olanzapine vs placebo, risperidone vs Quetiapine vs placebo and risperidone vs olanzapine vs placebo). Thus, three out of 19 studies included head-to-head comparison:
- Rainer, 2007: Quetiapine versus risperidone
- Schneider, 2006: Olanzapine versus Quetiapine versus risperidone
- Tariot, 2006: Quetiapine versus Haloperidol
The other studies included in Lü (2024) answered subquestion 1.1: placebo comparison, for which Muhlbauer (2021) was already included. In addition, Lü (2024) had an inadequate search strategy for inclusion as an NMA. Therefore, the three studies that performed head-to-head comparison were included rather than the full NMA.
Table 1. Characteristics of included studies
*For further details, see risk of bias table in the Appendix
** Lü (2024) reported low risk of bias for the study Schneider (2006). However, due to baseline differences in the NPI scores, the RoB for the outcome psychosis was concerned with moderate bias
*** Lü (2024) reported low risk of bias for the study Tariot (2006). However, due to missing information on the blinding and randomization, the RoB for this study was concerned with moderate bias
Results
1. Subquestion - placebo
1.1 Typical antipsychotics
Psychosis
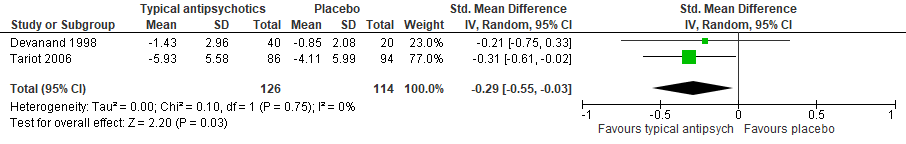
Two studies included in Mühlbauer (2021) reported on psychosis, presented in units of NPI-NH (higher is worse) (Devanand, 1998; Tariot, 2006). The analysis resulted in a SMD of -0.29 (95% CI: -0.55 to -0.03), favouring the typical antipsychotic group. This difference was considered a small clinically relevant effect. Results are shown in a forest plot (Figure 1).
Figure 1. Psychosis: Forest plot of the SMD on the effect of typical antipsychotics versus placebo
1 Devanand (1998): Haloperidol (low dose of 0.50-0.75 mg/day, or standard dose of 2-3 mg/day)
2 Tariot (2006): Haloperidol (0.5-12 mg/day)
Somnolence
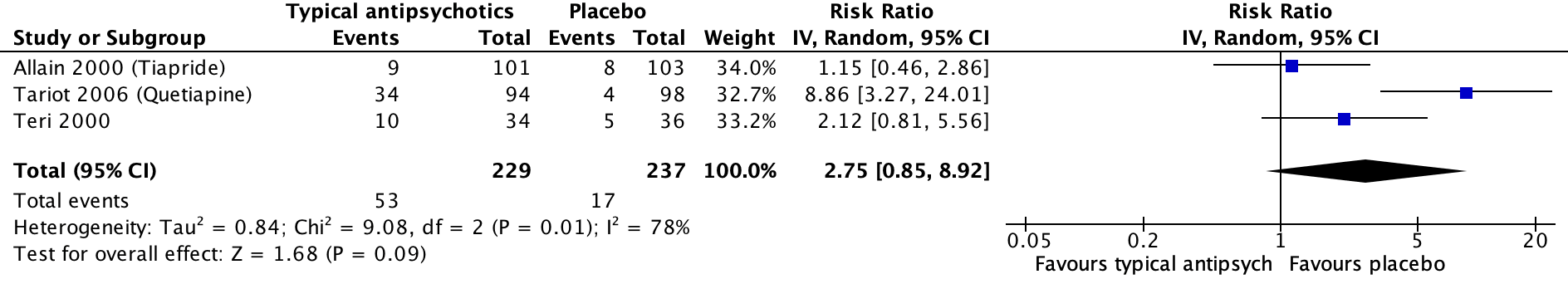
Three studies included in Mühlbauer (2021) reported on somnolence, which was assessed with different instruments (Allain, 2000; Tariot, 2006; Teri, 2000). In the antipsychotics group, 53 out of 229 patients experienced somnolence compared to 17 out of 237 patients in the placebo group. This resulted in a risk ratio of 2.75 (95% CI: 0.85 to 8.92) favouring the placebo group. This difference was clinically relevant. Results are shown in a forest plot (Figure 2).
Figure 2. Somnolence: Forest plot of the risk ratio on the effect of typical antipsychotics versus placebo
1 Allain (2000): Haloperidol (2 mg/day; dose could be progressively increased to a maximum of 6 mg/day)
2 Tariot (2006): Haloperidol (0.5-12 mg/day)
3 Teri (2000): Haloperidol (0.5-2 mg/day)
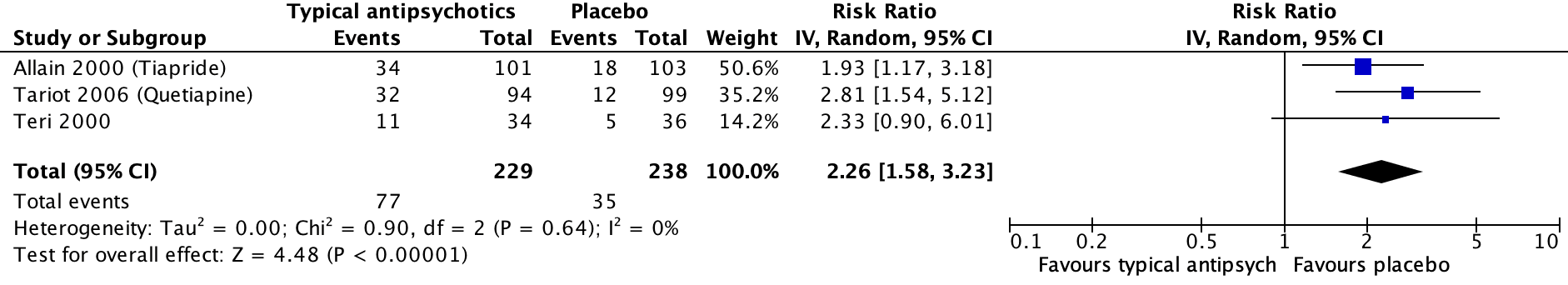
Extrapyramidal symptoms
Three studies included in the SR Mühlbauer (2021) reported on the outcome extrapyramidal symptoms, which was assessed with different instruments (Allain, 2000; Tariot, 2006; Teri, 2000). In the antipsychotics group, 77 out of 229 patients experienced extrapyramidal symptoms compared to 35 out of 238 patients in the placebo group. This resulted in a risk ratio of 2.26 (95% CI: 1.58 to 3.23) favouring the placebo group. This difference was clinically relevant. Results are shown in a forest plot (see Figure 3).
Figure 3. Extrapyramidal symptoms: Forest plot of the risk ratio on the effect of typical antipsychotics versus placebo
1 Allain (2000): Haloperidol (2 mg/day; dose could be progressively increased to a maximum of 6 mg/day)
2 Tariot (2006): Haloperidol (0.5-12 mg/day)
3 Teri (2000): Haloperidol (0.5-2 mg/day)
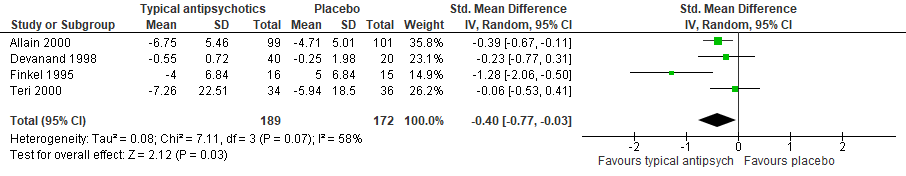
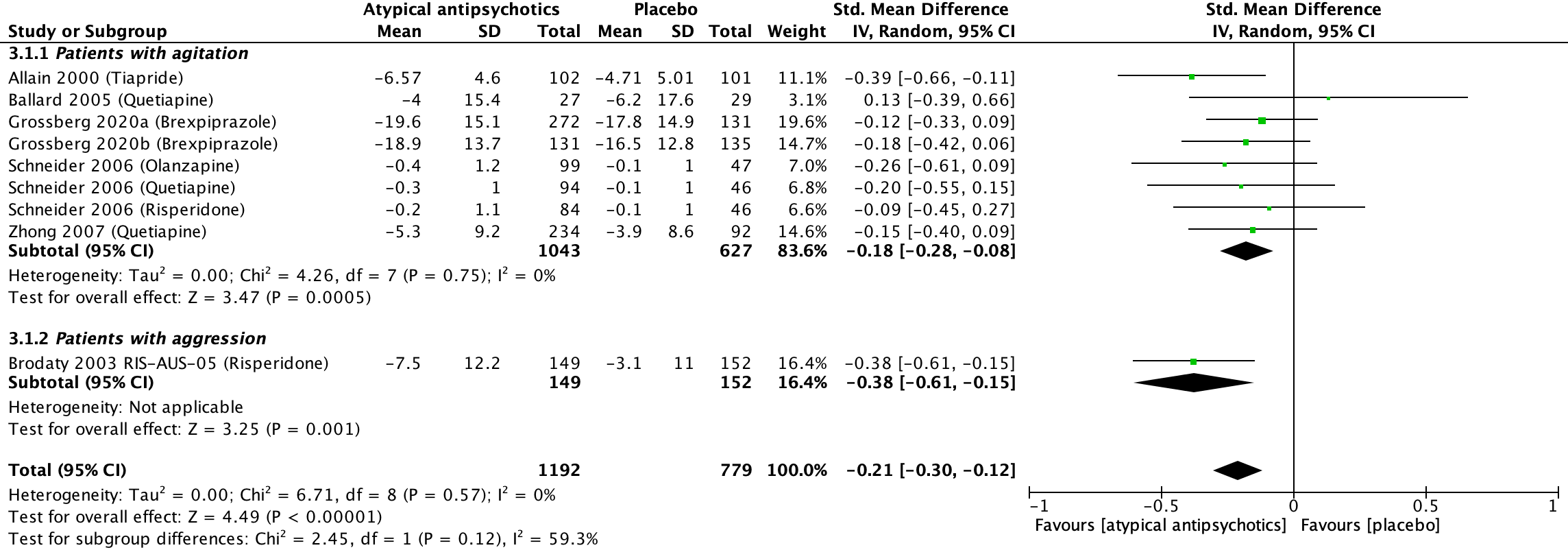
Agitation
Four studies included in Mühlbauer (2021) reported on the outcome agitation, presented in units on CMAI (higher is worse) (Allain, 2000; Devanand, 1998; Finkel, 1995; Teri, 2000). The analysis resulted in a SMD of -0.40 (95% CI: -0.77 to -0.03), favouring the typical antipsychotic group. This difference was considered a small clinically relevant effect. Results are shown in a forest plot (Figure 4).
Figure 4. Agitation: Forest plot of the SMD on the effect of typical antipsychotics versus placebo
1 Allain (2000): Haloperidol (2 mg/day; dose could be progressively increased to a maximum of 6 mg/day)
2 Devanand (1998): Haloperidol (low dose of 0.50-0.75 mg/day, or standard dose of 2-3 mg/day)
3 Finkel (1995): Thiothixene (mean dose 4.6 mg/day with a range of 0.25 to 18 mg/day)
4 Teri (2000): Haloperidol (0.5-2 mg/day)
Carer burden or carer quality of life
One study from the SR of Mühlbauer (2021) reported on caregiver burden (Teri, 2000). The mean difference of the caregiver burden with patients treated with typical antipsychotics (Haloperidol; 0.5-2 mg/day) (n=34, -1.88 ± 8.89) versus caregiver burden with patients treated with placebo (n=36, -2.58 ± 9.67), was 0.70 (95% CI: -3.65 to 5.05), favouring the placebo group. This difference was not clinically relevant.
Health-related quality of life
Mühlbauer (2021) did not report this outcome.
Cognitive function (important)
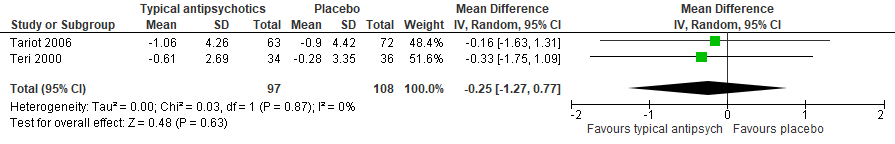
Two studies included in Mühlbauer (2021) reported on the outcome cognitive functioning, presented in units of MMSE (Tariot, 2006; Teri, 2000). The mean difference between patients treated with typical antipsychotics (n=97) and patients treated with placebo (n=108) was -0.25 (95% CI: -1.27 to 0.77). This difference was not clinically relevant. Results are shown in a forest plot (Figure 5).
Figure 5. Cognitive function: Forest plot of the mean difference on the effect of typical antipsychotics versus placebo
1 Tariot (2006): Haloperidol (0.5-12 mg/day)
2 Teri (2000): Haloperidol (0.5-2 mg/day)
1.2 Atypical antipsychotics
Psychosis
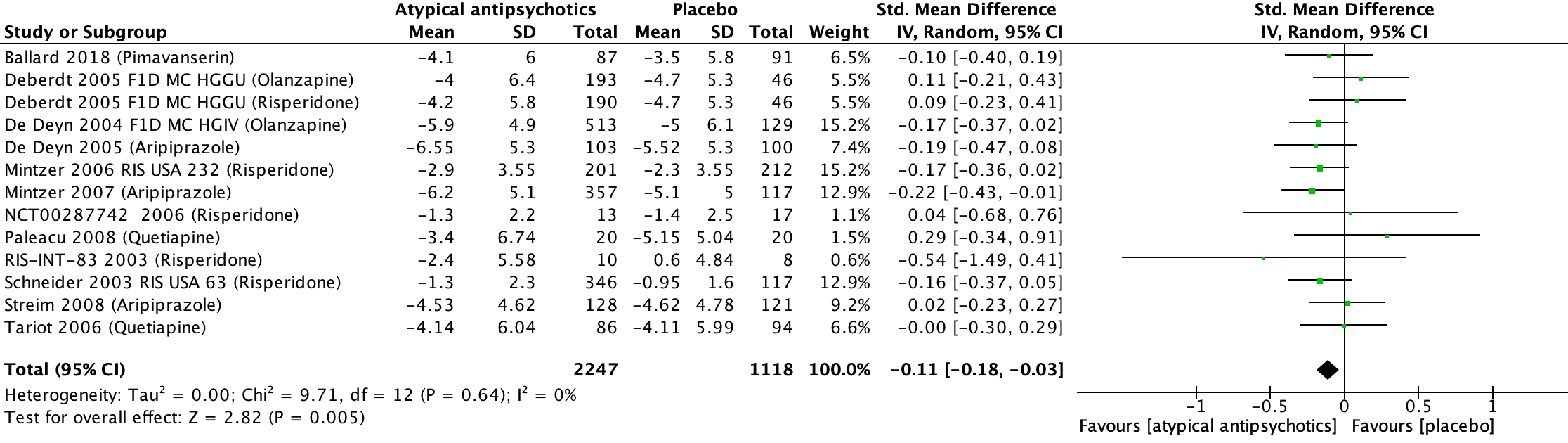
Twelve studies included in Mühlbauer (2021) reported on psychosis, presented in units of NPI-NH (higher is worse) (Ballard, 2018; Deberdt, 2005; De Deyn, 2004; De Deyn, 2005; Mintzer, 2006; Mintzer, 2007; NCT00187742, 2006; Paleacu, 2008; RIS-INT-83, 2003; Schneider, 2003; Streim, 2008; Tariot, 2006). The analysis resulted in a SMD of -0.11 (95% CI: -0.18 to -0.03), favouring the atypical antipsychotic group. This difference was not clinically relevant. Results are shown in a forest plot (Figure 6).
Figure 6. Psychosis: Forest plot of the SMD on the effect of atypical antipsychotics versus placebo
1 Ballard (2018): Pimavanserin (2x17 mg tablets daily)
2 Deberdt (2005) F1D MC HGGU: risperidone (0.5-2 mg/day)
3 Deberdt (2005) F1D MC HGGU: Olanzapine (2.5-10 mg/day)
4 De Deyn (2004) F1D MC HGIV: Olanzapine (1.0, 2.5, 5.0, or 7.5 mg/day)
5 De Deyn (2005): Aripiprazole (2-15 mg/day)
6 Mintzer (2006) RIS USA 232: risperidone (1.0-1.5 mg/day)
7 Mintzer (2007): Aripiprazole (2, 5, or 10 mg/day)
8 NCT00287742 (2006): risperidone (0.5-2 mg/day)
9 Paleacu (2008): Quetiapine (50-300 mg/day)
10 RIS-INT-83 (2003): risperidone (1-1.5 mg/day)
11 Schneider (2003) RIS USA 63: risperidone (1, 2, or 4 mg/day)
12 Streim (2008): Aripiprazole (2-15 mg/day)
13 Tariot (2006): Quetiapine (25-600 mg/day)
Somnolence
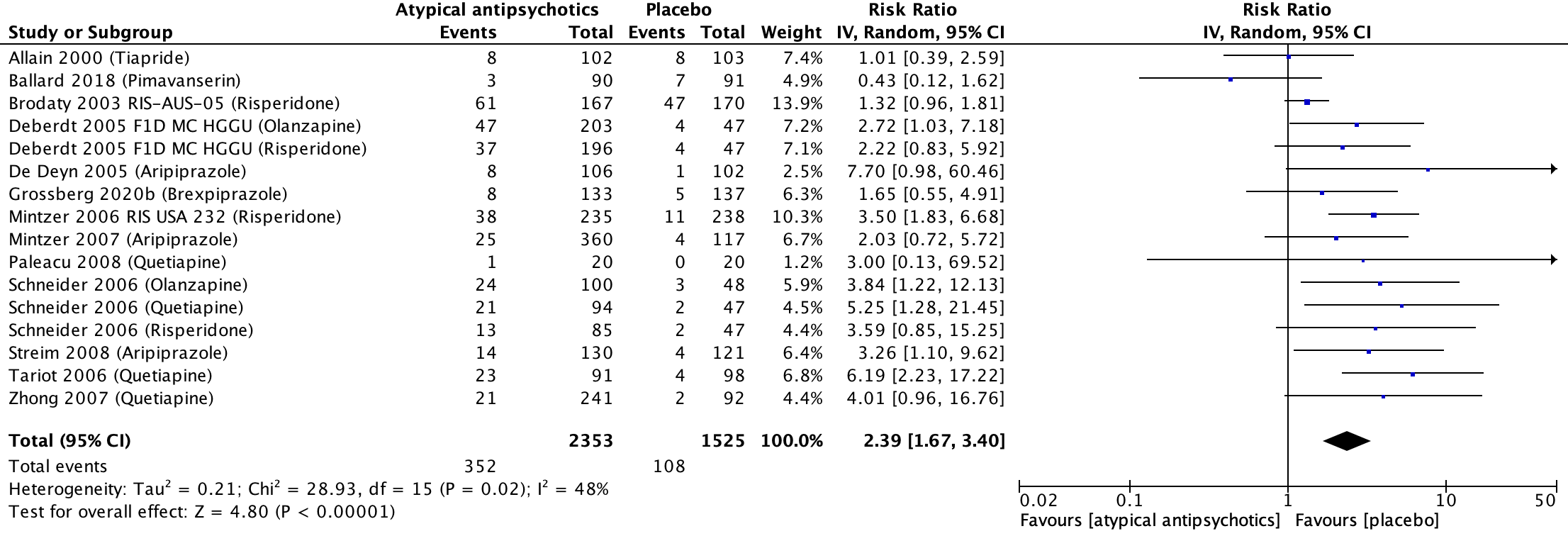
Thirteen studies included in Mühlbauer (2021) reported on somnolence, which was assessed with different instrument (Allain, 2000; Ballard, 2018; Brodaty, 2003; Deberdt, 2005; De Deyn, 2005; Grossberg, 2020b; Mintzer, 2006; Mintzer, 2007; Paleacu, 2008; Schneider, 2006; Streim, 2008; Tariot, 2006; Zhong, 2007). In the antipsychotics group, 352 out of 2353 patients experienced somnolence compared to 108 out of 1525 patients in the placebo group. This resulted in a risk ratio of 2.39 (95% CI: 1.67 to 3.40) favouring the placebo group. This difference was clinically relevant. Results are shown in a forest plot (see Figure 7).
Figure 7. Somnolence: Forest plot of the risk ratio on the effect of atypical antipsychotics versus placebo
1 Allain (2000): Tiapride (100 mg/day; dose could be progressively increased to a maximum of 300 mg/day)
2 Ballard (2018): Pimavanserin (2x17 mg tablets daily)
3 Brodaty (2003) RIS-AUS-05: risperidone (0.5-2 mg/day)
4 Deberdt (2005) F1D MC HGGU: risperidone (0.5-2 mg/day)
5 Deberdt (2005) F1D MC HGGU: Olanzapine (2.5-10 mg/day)
6 De Deyn (2005): Aripiprazole (2-15 mg/day)
7 Grossberg (2020b): Brexpiprazole (0.5-2 mg/day)
8 Mintzer (2006) RIS USA 232: risperidone (1.0-1.5 mg/day)
9 Mintzer (2007): Aripiprazole (2, 5, or 10 mg/day)
10 Paleacu (2008): Quetiapine (50-300 mg/day)
11 Schneider (2006): risperidone (tablets of 0.5 and 1.0 mg. Mean last dose 1.0 mg/day)
12 Schneider (2006): Quetiapine (tables of 25 and 50 mg. Mean last dose 56.6 mg/day)
13 Schneider (2006): Olanzapine (tablets of 2.5 and 5.0 mg. Mean last dose 5.5 mg/day)
14 Streim (2008): Aripiprazole (2-15 mg/day)
15 Tariot (2006): Quetiapine (25-600 mg/day)
16 Zhong (2007): Quetiapine (100-200 mg/day)
Extrapyramidal symptoms
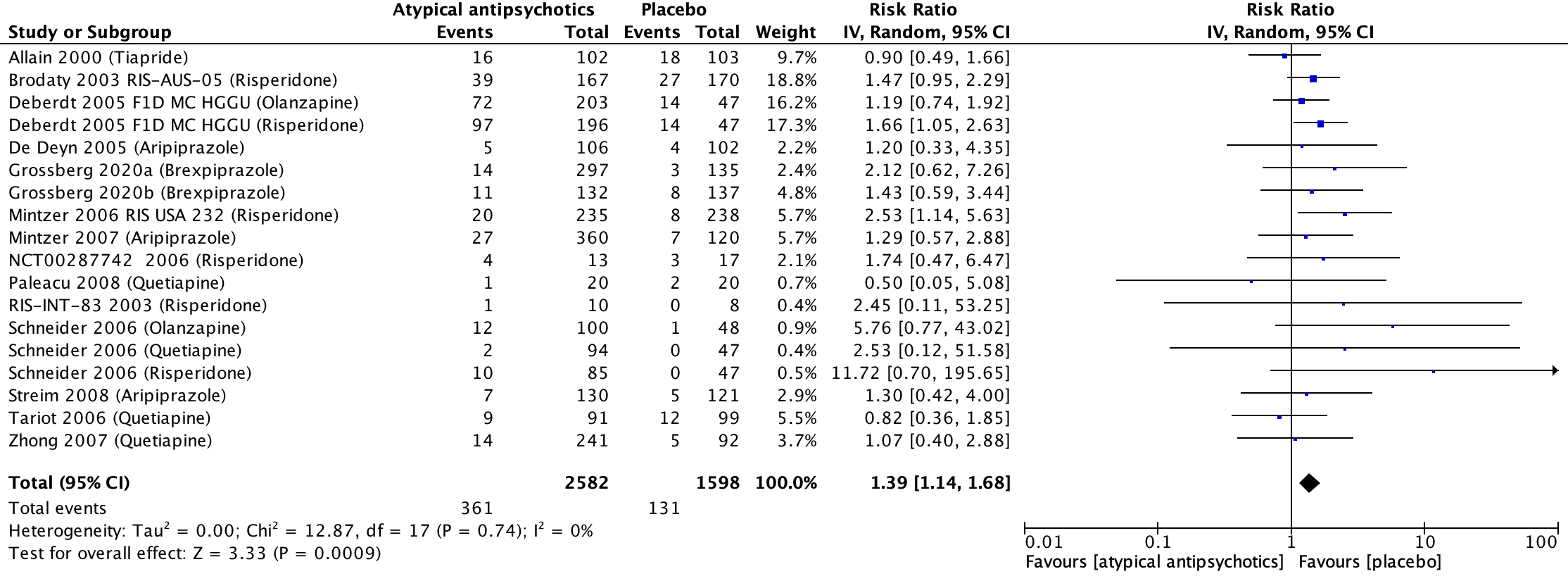
Fifteen studies included in the SR Mühlbauer (2021) reported on the outcome extrapyramidal symptoms, which was assessed with different instrument (Allain, 2000; Brodaty, 2003; Deberdt, 2005; De Deyn, 2005; Grossberg, 2020a; Grossberg, 2020b; Mintzer, 2006; Mintzer, 2007; NCT00187742, 2006; Paleacu, 2008; RIS-INT-83, 2003; Schneider, 2006; Streim, 2008; Tariot, 2006; Zhong, 2007). In the antipsychotics-group, 361 out of 2582 patients experienced extrapyramidal symptoms compared to 131 out of 1598 patients in the placebo group. This resulted in a risk ratio of 1.39 (95% CI: 1.14 to 1.68) favouring the placebo group. This difference was clinically relevant. Results are shown in a forest plot (see Figure 8).
Figure 8. Extrapyramidal symptoms: Forest plot of the risk ratio on the effect of atypical antipsychotics versus placebo
1 Allain (2000): Tiapride (100 mg/day; dose could be progressively increased to a maximum of 300 mg/day)
2 Brodaty (2003) RIS-AUS-05: risperidone (0.5-2 mg/day)
3 Deberdt (2005) F1D MC HGGU: Olanzapine (2.5-10 mg/day)
4 Deberdt (2005) F1D MC HGGU: risperidone (0.5-2 mg/day)
5 De Deyn (2005): Aripiprazole (2-15 mg/day)
6 Grossberg (2020a): Brexpiprazole (0.5-2 mg/day)
7 Grossberg (2020b): Brexpiprazole (0.5-2 mg/day)
8 Mintzer (2006) RIS USA 232: risperidone (1.0-1.5 mg/day)
9 Mintzer (2007): Aripiprazole (2, 5, or 10 mg/day)
10 NCT00287742 (2006): risperidone (0.5-2 mg/day)
11 Paleacu (2008): Quetiapine (50-300 mg/day)
12 RIS-INT-83 (2003): risperidone (1-1.5 mg/day)
13 Schneider (2006): risperidone (tablets of 0.5 and 1.0 mg. Mean last dose 1.0 mg/day)
14 Schneider (2006): Quetiapine (tables of 25 and 50 mg. Mean last dose 56.6 mg/day)
15 Schneider (2006): Olanzapine (tablets of 2.5 and 5.0 mg. Mean last dose 5.5 mg/day)
16 Streim (2008): Aripiprazole (2-15 mg/day)
17 Tariot (2006): Quetiapine (25-600 mg/day)
18 Zhong (2007): Quetiapine (100-200 mg/day)
Agitation
Seven studies included in Mühlbauer (2021) reported on the outcome agitation, presented in units on CMAI (higher is worse) (Allain, 2000; Ballard, 2005; Grossberg 2020a; Grossberg, 2020b; Schneider, 2006; Zhong, 2007). The analysis resulted in a SMD of -0.21 (95% CI: -0.30 to -0.12), favouring the atypical antipsychotic group. This difference was considered a small clinically relevant effect. Results are shown in a forest plot (Figure 9).
Figure 9. Agitation: Forest plot of the SMD on the effect of atypical antipsychotics versus placebo
1 Allain (2000): Tiapride (100 mg/day; dose could be progressively increased to a maximum of 300 mg/day)
2 Ballard (2005): Quetiapine (50-100 mg/day)
3 Grossberg (2020a): Brexpiprazole (0.5-2 mg/day)
4 Grossberg (2020b): Brexpiprazole (0.5-2 mg/day)
5 Schneider (2006): Olanzapine (tablets of 2.5 and 5.0 mg. Mean last dose 5.5 mg/day)
6 Schneider (2006): Quetiapine (tables of 25 and 50 mg. Mean last dose 56.6 mg/day)
7 Schneider (2006): risperidone (tablets of 0.5 and 1.0 mg. Mean last dose 1.0 mg/day)
8 Zhong (2007): Quetiapine (100-200 mg/day)
9 Brodaty (2003) RIS-AUS-05: risperidone (0.5-2 mg/day)
Carer burden or carer quality of life
Mühlbauer (2021) did not report this outcome.
Health-related quality of life
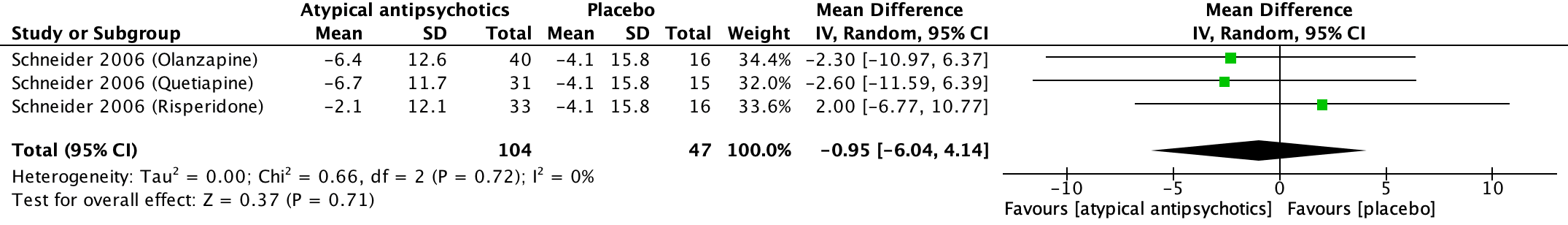
One study from the SR of Mühlbauer (2021) reported on caregiver burden (Schneider, 2006). This study studied three atypical antipsychotics: Olanzapine, Quetiapine and risperidone. Overall, the mean difference in health-related quality of life in patients treated with typical antipsychotics versus placebo was -0.95 (95%CI: -6.04 to 4.14). This difference was considered a small clinically relevant effect. Results are shown in a forest plot (Figure 10). The mean values in the figure have been converted to negative values to facilitate correct interpretation within the context of the figure.
Figure 10. Health-related quality of life: Forest plot of the mean difference on the effect of atypical antipsychotics versus placebo
1 Schneider (2006): risperidone (tablets of 0.5 and 1.0 mg. Mean last dose 1.0 mg/day)
2 Schneider (2006): Olanzapine (tablets of 2.5 and 5.0 mg. Mean last dose 5.5 mg/day)
3 Schneider (2006): Quetiapine (tables of 25 and 50 mg. Mean last dose 56.6 mg/day)
Cognitive function
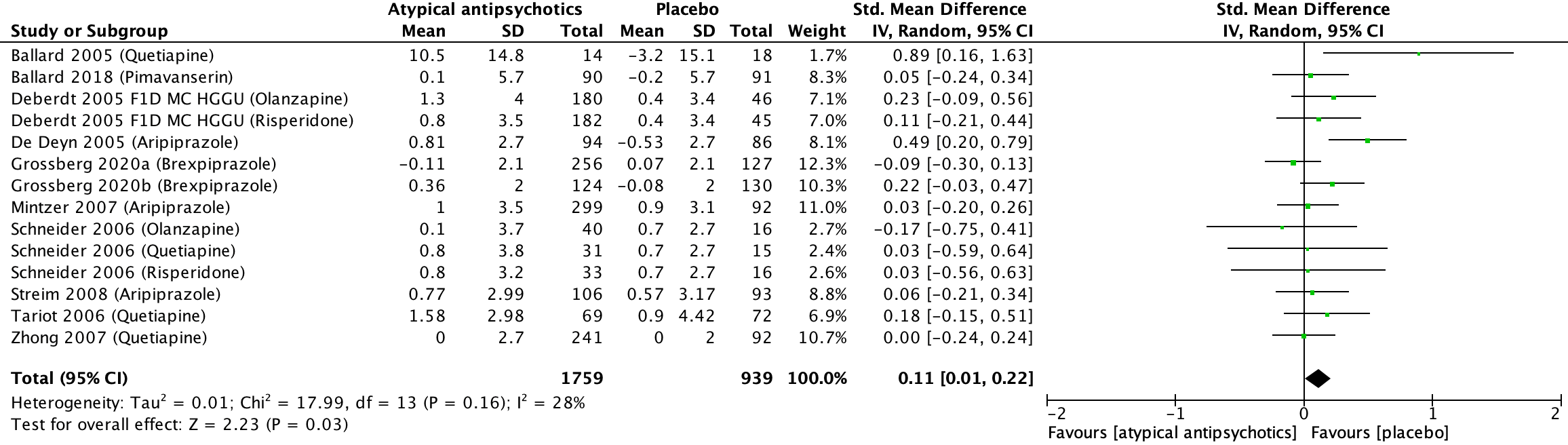
Eleven studies included in Mühlbauer (2021) reported on the outcome cognitive functioning, presented in units of MMSE (Ballard, 2005; Ballard, 2018; Deberdt, 2005; De Deyn, 2005; Grossberg 2020a; Grossberg, 2020b; Mintzer, 2007; Schneider, 2006; Streim, 2008; Tariot, 2006; Zhong, 2007). The SMD between patients treated with atypical antipsychotics (n=1759) and patients treated with placebo (n=939) was 0.11 (95% CI: 0.01 to 0.22). This difference was not clinically relevant. Results are shown in a forest plot (Figure 11). The mean values in the figure have been converted to facilitate correct interpretation within the context of the figure.
Figure 11. Cognitive function: Forest plot of the SMD on the effect of atypical antipsychotics versus placebo
1 Ballard (2005): Quetiapine (50-100 mg/day)
2 Ballard (2018): Pimavanserin (2x17 mg tablets daily)
3 Deberdt (2005) F1D MC HGGU: Olanzapine (2.5-10 mg/day)
4 Deberdt (2005) F1D MC HGGU: risperidone (0.5-2 mg/day)
5 De Deyn (2005): Aripiprazole (2-15 mg/day)
6 Grossberg (2020a): Brexpiprazole (0.5-2 mg/day)
7 Grossberg (2020b): Brexpiprazole (0.5-2 mg/day)
8 Mintzer (2007): Aripiprazole (2, 5, or 10 mg/day)
9 Schneider (2006): Quetiapine (tables of 25 and 50 mg. Mean last dose 56.6 mg/day)
10 Schneider (2006): Olanzapine (tablets of 2.5 and 5.0 mg. Mean last dose 5.5 mg/day)
11 Schneider (2006): risperidone (tablets of 0.5 and 1.0 mg. Mean last dose 1.0 mg/day)
12 Streim (2008): Aripiprazole (2-15 mg/day)
13 Tariot (2006): Quetiapine (25-600 mg/day)
14 Zhong (2007): Quetiapine (100-200 mg/day)
2. Subquestion - Head-to-head comparison
2.1 Typical antipsychotics
Lü (2024) does not report any head-to-head comparisons on typical antipsychotics. The comparison of the typical antipsychotic Haloperidol to the atypical antipsychotic Quetiapine is reported in 1.2.2.
2.2 Atypical antipsychotics
Psychosis
Quetiapine vs risperidone
Two studies reported on psychosis (Rainer, 2007; Schneider, 2006). The two studies used different scales, and Rainer (2007) did not report the mean (SD), thus data could not be pooled.
Rainer (2007) reported psychosis in units of NPI-NH, which decreased from 25.6 to 17.5 with Quetiapine (n=34; 50-400 mg/day) and from 25.7 to 16.6 with risperidone (n=31; 0.5-4 mg/day). Mean (SD) was not reported, thus the mean difference could not be calculated and clinical relevance could not be assessed.
Schneider (2006) reported psychosis with the BPRS and NPI. NPI score was reduced with 16.6 ± 18.3 in the Quetiapine group (n=31; mean last dose 56.5 mg/day) and 16.4 ± 15.0 in the risperidone group (n=32; mean last dose 1.0 mg/day). This resulted in a mean difference of 0.20 (95%CI: -8.84 to 9.24), in favour of the Quetiapine group, which was not clinically relevant. BPRS score was reduced with 7.4 ± 9.8 in the Quetiapine group (n=30) and 8.5 ± 12.2 in the risperidone group (n=32). This resulted in a mean difference of 1.10 (95%CI: -4.39 to 6.59), in favour of the risperidone group, which was not clinically relevant.
Olanzapine vs Quetiapine
One study reported on psychosis (Schneider, 2006) measured with the BPRS and NPI. The NPI score was reduced with 14.0 ± 18.7 point in the Olanzapine group (n=40; mean last dose 5.5 mg/day) and 16.6 ± 18.3 in the Quetiapine group (n=31; mean last dose 56.5 mg/day). This resulted in a mean difference of 2.20 (95%CI: -6.46 to 10.86), in favour of the Quetiapine group, which was not clinically relevant. BPRS score was reduced with 7.4 ± 9.8 in the Olanzapine group (n=40) and 7.4 ± 9.8 in the Quetiapine group (n=30). This resulted in a mean difference of 0.00 (95%CI: -4.64 to 4.64), which was not clinically relevant.
Olanzapine vs risperidone
One study reported on psychosis (Schneider, 2006) measured with the BPRS and NPI. NPI score was reduced with 14.0 ± 18.7 point in the Olanzapine group (n=40; mean last dose 5.5 mg/day) and 16.4 ± 15.0 in the risperidone group (n=32; mean last dose 1.0 mg/day). This resulted in a mean difference of 2.40 (95%CI: -5.58 to 10.18), in favour of the risperidone group, which was not clinically relevant. BPRS score was reduced with 7.4 ± 9.8 in the Olanzapine group (n=40) and 8.5 ± 12.2 in the risperidone group (n=32). This resulted in a mean difference of 1.10 (95%CI: -4.10 to 6.30), in favour of the risperidone group, which was not clinically relevant.
Quetiapine vs Haloperidol
One study reported on psychosis (Tariot, 2006), measured with the BPRS and CGI-S after 10 weeks. The BPRS score was reduced by 9.06 ± 11.07 in the Quetiapine group (n=85; 25-600 mg/day) and 7.13 ± 10.85 in the Haloperidol group (n=86; 0.5-12 mg/day). This resulted in a mean difference of 1.93 (95%CI: -1.36 to 5.22), in favour of the Haloperidol group, which was not clinically relevant. The CGI-S score was reduced by 0.60 ± 1.05 in the Quetiapine group (n=85) and 0.52 ± 1.00 in the Haloperidol group (n=86). This resulted in a mean difference of 0.08 (95%CI: -0.23 to 0.39), in favour of the Quetiapine group, which was not clinically relevant.
Somnolence
Quetiapine vs risperidone
One study reported on somnolence (Rainer, 2007). Two of the 38 patients with Quetiapine (50-400 mg/day) (5.3%) experienced somnolence compared to zero of the 34 patients with risperidone (0.5-4 mg/day) (0%). This resulted in a risk ratio of -0.05 (95%CI: -0.14 to 0.03), in favour of the risperidone group, which was not clinically relevant.
Quetiapine vs Haloperidol
One study reported on somnolence (Tariot, 2006). 23 of the 91 patients with Quetiapine (25-600 mg/day) (25.3%) experienced somnolence compared to 34 of the 94 patients with Haloperidol (0.5-12 mg/day) (36.2%). This resulted in a risk ratio of -0.11 (95%CI: -0.24 to 0.02), in favour of the Quetiapine group, which was not clinically relevant.
Extrapyramidal symptoms
Quetiapine vs risperidone
One study reported on extrapyramidal symptoms (Rainer, 2007). The incidence of extrapyramidal symptoms, as measured by the mean change from baseline to endpoint in SAS scores, increased with a score of (5.00 – 5.06) 0.06 in the Quetiapine group (n=38; 50-400 mg/day) and a score of (5.68 – 6.03) 0.35 in the risperidone group (n=34; 0.5-4 mg/day). Mean (SD) was not reported, thus the mean difference could not be calculated and clinical relevance could not be assessed.
Agitation
Quetiapine vs risperidone
One study reported on agitation (Rainer, 2007). Both Quetiapine and risperidone showed some efficacy against agitation, as shown by a reduction in CMAI scores from baseline to Week 8, with no significant differences between treatments (Quetiapine, 59.21 to 55.67, n=33 (50-400 mg/day); risperidone, 54.48 to 48.97, n=31 (0.5-4 mg/day)). Mean (SD) was not reported, thus the mean difference could not be calculated and clinical relevance could not be assessed.
Quetiapine vs Haloperidol
One study reported on agitation (Tariot, 2006), measured with the BPRS-agitation and NPI-NH agitation. After 10 weeks the BPRS agitation scale was reduced by 2.14 ± 3.58 in the Quetiapine group (n=85; 25-600 mg/day) and 2.37 ± 3.43 in the Haloperidol group (n=86; 0.5-12 mg/day). This resulted in a mean difference of 0.23 (95%CI: -0.82 to 1.28), in favour of the Haloperidol group, which was not clinically relevant. After 10 weeks the NPI-NH agitation scale was reduced by 2.63 ± 4.99 in the Quetiapine group (n=86) and 2.43 ± 4.45 in the Haloperidol group (n=86). This resulted in a mean difference of 0.20 (95%CI: -1.36 to 1.76), in favour of the Quetiapine group, which was not clinically relevant.
Carer burden or carer quality of life
Quetiapine vs risperidone
One study reported on burden to the carer, presented in units of NPI-NH (Rainer, 2007). The NPI score was reduced from 30.2 to 27.7 with Quetiapine (n=34; 50-400 mg/day) and from 30.3 to 26.7 with risperidone (n=31; 0.5-4 mg/day). Mean (SD) was not reported, thus de mean difference could not be calculated and clinical relevance could not be assessed.
Health-related quality of life
None of the studies reported on health-related quality of life
Cognitive function
Quetiapine vs risperidone
One study reported on cognitive function (Rainer, 2007) measured with MMSE. The MMSE score remained stable from baseline to Week 8 with Quetiapine (n=30; 50-400 mg/day) (18.50 to 18.47) and risperidone (n=26; 0.5-4 mg/day) (18.00 to 18.65). Mean (SD) was not reported, thus the mean difference could not be calculated and clinical relevance could not be assessed.
Quetiapine vs Haloperidol
One study reported on cognitive function (Tariot 2006) measured with MMSE. The MMSE score was reduced by 1.58 ± 2.98 in the Quetiapine group (n=69; 25-600 mg/day) and 1.06 ± 4.26 in the Haloperidol group (n=63; 0.5-12 mg/day). This resulted in a mean difference of 0.52 (95%CI: -0.75 to 1.79), in favour of the Quetiapine group, which was not clinically relevant.
A systematic review of the literature was performed to answer the following question(s):
PICO 1 What is the effectiveness of antipsychotics in comparison to placebo in patients with dementia and neuropsychiatric symptoms?
PICO 2 What is the effectiveness of various antipsychotics in patients with dementia and neuropsychiatric symptoms?
| Patients | Patients with dementia (e.g. Alzheimer, vascular, Parkinson, LBD) and neuropsychiatric symptoms (e.g. agitation, aggression, distress, psychosis) |
| Intervention | Antipsychotics (e.g. clozapine, risperidone, haloperidol, pipamperon) |
| Control 1 | Placebo |
| Control 2 | Comparison between antipsychotics |
| Outcomes | Psychosis, adverse events, agitation, carer burden or care quality of life, health-related quality of life, cognitive function |
| Other selection citeria | Study design: systematic reviews |
Relevant outcome measures
The guideline panel considered psychosis and adverse events as critical outcome measures for decision making; and agitation, carer burden or carer quality of life, health-related quality of life, and cognitive function as important outcome measures for decision making.
A priori, the guideline panel did not define the outcome measures listed above but used the definitions used in the studies.
For all outcome measures, the guideline panel defined a 25% difference for dichotomous outcomes (0.8 ≥ RR ≥ 1.25) and 0.5 SD for continuous outcomes as a minimal clinically (patient) important difference.
If studies used different rating instruments (e.g. psychosis could be measured with NPI, BEHAVE-AD) to measure the same outcome measure, then the standardized mean difference (SMD) is calculated. According to Cohen, SMD values of 0.2-0.5 represent a small clinically relevant effect, values of 0.5-0.8 a medium clinically relevant effect, and values >0.8 represent a large clinically relevant effect (Cohen, 1988).
Search and select (Methods)
The databases Medline (via OVID) and Embase (via Embase.com) were searched with relevant search terms from 2010 until 30 August 2024. The detailed search strategy is listed under the tab ‘Literature search strategy’. The systematic literature search resulted in 4086 hits. Studies were selected based on the following criteria:
- Systematic reviews in which searches were performed in at least two databases, with a detailed search strategy, risk of bias assessment and results of individual studies available, randomized controlled trials or (observational) comparative studies.
- Full-text English or Dutch language publication and
- Studies according to the PICO.
Initially, 31 studies were selected based on title and abstract screening. After reading the full text, 29 studies were excluded (see the exclusion table under the tab ‘Evidence tabellen’). For example, reasons for exclusion were a less recent search (Ma, 2014), search performed in a Chinese database in which the Chinese full text articles were not accessible (Zhang, 2024), insufficient methodological quality (Gerlach, 2020). Eventually, two systematic reviews were selected for inclusion:
- Subquestion 1 Placebo comparison: Mühlbauer (2021).
- Subquestion 2 Head-to-head comparison: Lü (2024).
- Cohen, J. (1988). Statistical Power Analysis for the Behavioral Sciences (2nd ed.). Hillsdale, NJ: Lawrence Erlbaum Associates, Publishers.
- Farmacotherapeutisch Kompas (z.d.). Risperidon. Zorginsitituut Nederland. Geraadpleegd op 11-3-2025 van https://www.farmacotherapeutischkompas.nl/bladeren/preparaatteksten/r/risperidon#doseringen.
- Lü W, Liu F, Zhang Y, He X, Hu Y, Xu H, Yang X, Li J, Kuang W. Efficacy, acceptability and tolerability of second-generation antipsychotics for behavioural and psychological symptoms of dementia: a systematic review and network meta-analysis. BMJ Ment Health. 2024 Jul 30;27(1):e301019. doi: 10.1136/bmjment-2024-301019. PMID: 39079887; PMCID: PMC11293415.
- Mühlbauer V, Möhler R, Dichter MN, Zuidema SU, Köpke S, Luijendijk HJ. Antipsychotics for agitation and psychosis in people with Alzheimer's disease and vascular dementia. Cochrane Database Syst Rev. 2021 Dec 17;12(12):CD013304. doi: 10.1002/14651858.CD013304.pub2. PMID: 34918337; PMCID: PMC8678509.
Risk of bias
1. Subquestion – placebo
Risk of bias assessment performed by Mühlbauer (2021) is shown below.
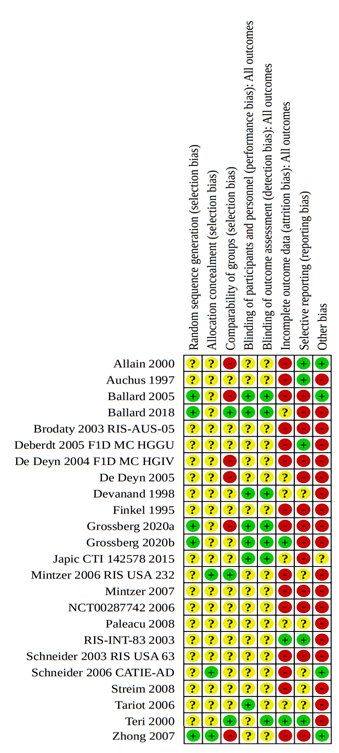
2. Subquestion - Head-to-head comparison
Risk of bias assessment performed by Lü (2020) is shown below.
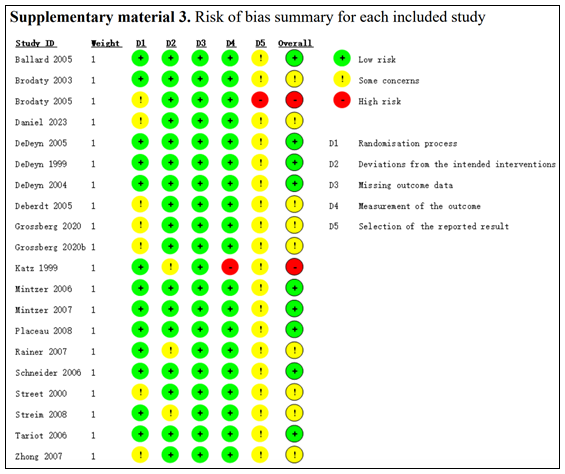
Table S1. Recommendations regarding the use of antipsychotics in people with dementia based on other guidelines
|
Study |
Based on |
Authors’ conclusions and/or recommendations |
|
Mühlbauer, 2021 |
Systematic review of 24 RCTs including 6090 people: - six studies tested typical antipsychotics, mostly haloperidol; - 20 studies tested atypical antipsychotics, such as risperidone, olanzapine, and aripiprazole; and - two studies tested both typical and atypical antipsychotics. |
There is some evidence that typical antipsychotics might decrease agitation and psychosis slightly in patients with dementia. Atypical antipsychotics reduce agitation in dementia slightly, but their effect on psychosis in dementia is negligible. The apparent effectiveness of the drugs seen in daily practice may be explained by a favourable natural course of the symptoms, as observed in the placebo groups. Both drug classes increase the risk of somnolence and other adverse events. If antipsychotics are considered for sedation in patients with severe and dangerous symptoms, this should be discussed openly with the patient and legal representative. |
|
Mok, 2024 |
Population based matched cohort study in adults (≥50 years) with a diagnosis of dementia (n=173 910, 63.0% women). Each new antipsychotic user (n=35 339, 62.5% women) was matched with up to 15 non-users using incidence density sampling. |
Antipsychotic use in people with dementia was associated with increased risks of stroke, venous thromboembolism, myocardial infarction, heart failure, fracture, pneumonia, and acute kidney injury, compared with non-use, but not ventricular arrhythmia. Relative hazards were highest for pneumonia, acute kidney injury, stroke, and venous thromboembolism, and absolute risk and risk difference between antipsychotic users and their matched comparators was largest for pneumonia Risks of these wide-ranging adverse outcomes need to be considered before prescribing antipsychotic drugs to people with dementia. |
|
NICE, 2018 |
*Ma (2014) > SR (de Deyn, 2005; Mintzer, 2007; Streim, 2008; Street, 2000; de Deyn, 2004; Deberdt, 2005; Ballard, 2005; Schneider, 2006; Tariot, 2006; Zhong, 2007; Kurlan, 2007; Paleacu, 2008; de Deyn, 1999; Katz, 1999; Brodaty, 2003; Mintzer, 2006)
*Verhey (2006) > RCT (olanzapine vs haloperidol) *Ballard (2008): antipsychotic withdrawal vs continuation *Ballard (2009): antipsychotic withdrawal vs continuation *Ballard (2015): antipsychotics vs memantine *Holmes (2007): risperidone vs rivastigmine *Moulton (2014): SR (pharmacological treatment vs placebo) in people with Huntington’s disease *Pan (2014): SR (antipsychotic withdrawal vs continuation) |
Agitation, aggression, distress and psychosis
Before starting non-pharmacological or pharmacological treatment for distress in people living with dementia, conduct a structured assessment to:
As initial and ongoing management, offer psychosocial and environmental interventions to reduce distress in people living with dementia. Only offer antipsychotics, for people living with dementia who are either:
Be aware that for people with dementia with Lewy bodies or Parkinson’s disease dementia, antipsychotics can worsen the motor features of the condition, and in some cases cause severe antipsychotic sensitivity reactions. For more guidance, see the advice on managing delusions and hallucinations in the NICE guideline on Parkinson’s disease. Be aware that interventions may need to be modified for people living with dementia
Before starting antipsychotics, discuss the benefits and harms with the person and their family members or carers (as appropriate). Consider using a decision aid to support this discussion.
When using antipsychotics:
Stop treatment with antipsychotics:
Ensure that people living with dementia can continue to access psychosocial and environmental interventions for distress while they are taking antipsychotics and after they have stopped taking them.
For people living with dementia who experience agitation or aggression, offer personalised activities to promote engagement, pleasure and interest.
Do not offer valproate to manage agitation or aggression in people living with dementia, unless it is indicated for another condition. |
|
Verenso, 2018 |
Negen (reviews van) RCT’s voldeden aan de selectiecriteria: eenmaal een conventioneel antipsychoticum (Tariot, 2006); negenmaal een atypisch antipsychoticum in acht RCT's (Streim, 2008; Mintzer, 2007; Mintzer, 2006; Tariot, 2006; De Deyn, 2005; Deberdt, 2005; De Deyn, 2004; Satterlee, 1995) en eenmaal een atypisch antipsychoticum bij mensen met dementie bij de ziekte van Parkinson (Rabey, 2007). |
Module: Behandeling van psychotisch gedrag bij mensen met dementie
|
|
Farmacotherapeutisch Kompas (zie link)*
*Geraadpleegd op 14 februari 2024. |
NHG-Standaard Dementie, 2020. Via richtlijnen.nhg.org. Verenso. Probleemgedrag bij mensen met dementie, 2018. Via verenso.nl. NHG-Behandelrichtlijn Geneesmiddelen en zuurstof in spoedeisende situaties, 2023. Via richtlijnen.nhg.org. Ephor. Antipsychotica (bij agitatie/agressie en psychose bij dementie). Geraadpleegd april 2020, via ephorapp.nl. |
In een crisissituatie bij dementie is het soms nodig kortdurend antipsychotica toe te dienen. Bij heftige agitatie, angst of agressie bij dementie heeft kortdurende medicamenteuze behandeling een plaats indien de patiënt ondanks de-escalerende technieken onderzoek of behandeling weigert en hiermee zichzelf of zijn omgeving schade berokkent. In crisissituaties zonder psychotisch gedrag komt een kortwerkende benzodiazepine (lorazepam) in aanmerking. In crisissituaties met psychotisch gedrag heeft haloperidol de voorkeur (niet bij ‘Lewy body’-dementie en de ziekte van Parkinson).
Crisissituatie bij dementie Het volgende stappenplan is van toepassing bij crisissituaties met heftige agitatie, angst of agressie. Stap 1. Bespreek niet-medicamenteus beleid. Ga naar stap 2 bij onvoldoende effect van niet-medicamenteus behandelen.
Stap 2. Start medicamenteuze behandeling: Acute agressie of ernstige agitatie mét psychotisch gedrag
Haloperidol is gecontra-indiceerd bij ‘Lewy body’-dementie, Parkinson-dementie en frontotemporale dementie vanwege de kans op extrapiramidale verschijnselen en een verlengd QT-interval. Zie voor behandeling het stappenplan ‘Lewy body’-dementie en Parkinson-dementie. Behandel alleen met psychofarmaca in acute situaties, als niet-medicamenteuze interventies (nog) niet voldoende effectief zijn. Schakel zo nodig de crisisdienst in of overleg met een specialist ouderengeneeskunde of ouderenpsychiater. Doseer zo laag en kortdurend als mogelijk. Hanteer een maximale behandelduur van drie maanden. Het gebruik van clorazepinezuur bij acute of ernistige agitatie zónder psychotisch gedrag wordt afgerden, Stap 3. Start probleemanalyse van het probleemgedrag.Het gebruik ban clorazepinezuur bij acute of ernstige agitatie zónder psychotisch gedrag wordt afgeraden. |
Table of excluded studies
|
Reference |
Reason for exclusion |
|
Seibert M, Mühlbauer V, Holbrook J, Voigt-Radloff S, Brefka S, Dallmeier D, Denkinger M, Schönfeldt-Lecuona C, Klöppel S, von Arnim CAF. Efficacy and safety of pharmacotherapy for Alzheimer's disease and for behavioural and psychological symptoms of dementia in older patients with moderate and severe functional impairments: a systematic review of controlled trials. Alzheimers Res Ther. 2021 Jul 16;13(1):131. doi: 10.1186/s13195-021-00867-8. PMID: 34271969; PMCID: PMC8285815. |
Less recent compared to Lü (2024) and Mühlbauer (2021); Wrong intervention (very broad PICO; only one study on antipsychotics: Tariot, 2006). |
|
Chu CS, Yang FC, Tseng PT, Stubbs B, Dag A, Carvalho AF, Thompson T, Tu YK, Yeh TC, Li DJ, Tsai CK, Chen TY, Ikeda M, Liang CS, Su KP. Treatment Efficacy and Acceptability of Pharmacotherapies for Dementia with Lewy Bodies: A Systematic Review and Network Meta-Analysis. Arch Gerontol Geriatr. 2021 Sep-Oct;96:104474. doi: 10.1016/j.archger.2021.104474. Epub 2021 Jul 2. PMID: 34256210. |
Less recent compared to Lü (2024) and Mühlbauer (2021); small population, broad intervention. |
|
Vredeveld EJ, Hulshof TA, Zuidema SU, Luijendijk HJ. Subjective Versus Objective Outcomes of Antipsychotics for the Treatment of Neuropsychiatric Symptoms Associated with Dementia. CNS Drugs. 2019 Sep;33(9):933-942. doi: 10.1007/s40263-019-00654-y. PMID: 31473979; PMCID: PMC6776492. |
Wrong comparison for the PICO of this module. |
|
Marinheiro G, Dantas JM, Mutarelli A, Menegaz de Almeida A, Monteiro GA, Zerlotto DS, Telles JPM. Efficacy and safety of brexpiprazole for the treatment of agitation in Alzheimer's disease: a meta-analysis of randomized controlled trials. Neurol Sci. 2024 Oct;45(10):4679-4686. doi: 10.1007/s10072-024-07576-8. Epub 2024 May 20. PMID: 38763935. |
Wrong I for the PICO of this module. |
|
Cheung G, Stapelberg J. Quetiapine for the treatment of behavioural and psychological symptoms of dementia (BPSD): a meta-analysis of randomised placebo-controlled trials. N Z Med J. 2011 Jun 10;124(1336):39-50. PMID: 21946743. |
Less recent compared to Mühlbauer (2021); small I. |
|
Shotbolt P, Samuel M, David A. Quetiapine in the treatment of psychosis in Parkinson's disease. Ther Adv Neurol Disord. 2010 Nov;3(6):339-50. doi: 10.1177/1756285610389656. PMID: 21179595; PMCID: PMC3002640. |
Wrong P for the PICO of this module.; insufficient methodological quality (no search strategy available, data not pooled, no risk of bias assessment). |
|
Yunusa I, Rashid N, Demos GN, Mahadik BS, Abler VC, Rajagopalan K. Comparative Outcomes of Commonly Used Off-Label Atypical Antipsychotics in the Treatment of Dementia-Related Psychosis: A Network Meta-analysis. Adv Ther. 2022 May;39(5):1993-2008. doi: 10.1007/s12325-022-02075-8. Epub 2022 Mar 5. PMID: 35247186; PMCID: PMC9056477. |
Less recent compared to Lü (2024); insufficient methodological quality (no search strategy available, data not pooled, no risk of bias assessment). |
|
Huang YY, Teng T, Giovane CD, Wang RZ, Suckling J, Shen XN, Chen SD, Huang SY, Kuo K, Cai WJ, Chen KL, Feng L, Zhang C, Liu CY, Li CB, Zhao QH, Dong Q, Zhou XY, Yu JT. Pharmacological treatment of neuropsychiatric symptoms of dementia: a network meta-analysis. Age Ageing. 2023 Jun 1;52(6):afad091. doi: 10.1093/ageing/afad091. PMID: 37381843. |
Less recent compared to Lü (2024) |
|
Huang YY, Teng T, Shen XN, Chen SD, Wang RZ, Zhang RQ, Dou KX, Zhong XL, Wang J, Chen KL, Zhao QH, Tan L, Dong Q, Zhou XY, Yu JT. Pharmacological treatments for psychotic symptoms in dementia: A systematic review with pairwise and network meta-analysis. Ageing Res Rev. 2022 Mar;75:101568. doi: 10.1016/j.arr.2022.101568. Epub 2022 Jan 17. PMID: 35051646. |
Less recent compared to Lü (2024) and Mühlbauer (2021) |
|
Tan L, Tan L, Wang HF, Wang J, Tan CC, Tan MS, Meng XF, Wang C, Yu JT. Efficacy and safety of atypical antipsychotic drug treatment for dementia: a systematic review and meta-analysis. Alzheimers Res Ther. 2015 Apr 20;7(1):20. doi: 10.1186/s13195-015-0102-9. Retraction in: Alzheimers Res Ther. 2016 Jul 07;8(1):28. doi: 10.1186/s13195-016-0197-7. PMID: 25897331; PMCID: PMC4404265. |
Article is retracted. |
|
Mischel N, Rakesh G, Gupta S, Muzyk A, Masand P. Pharmacological agents to treat severe neuropsychiatric symptoms in major neurocognitive disorders. Ann Clin Psychiatry. 2020 May;32(2):128-141. PMID: 32343284. |
Full text is not available. |
|
Gerlach LB, Kales HC. Pharmacological Management of Neuropsychiatric Symptoms of Dementia. Curr Treat Options Psychiatry. 2020 Dec;7(4):489-507. doi: 10.1007/s40501-020-00233-9. Epub 2020 Sep 2. PMID: 33344107; PMCID: PMC7742723. |
Insufficient methodological quality (no search strategy available, data not systematically presented, data not pooled, no risk of bias assessment). |
|
Seitz DP, Gill SS, Herrmann N, Brisbin S, Rapoport MJ, Rines J, Wilson K, Le Clair K, Conn DK. Pharmacological treatments for neuropsychiatric symptoms of dementia in long-term care: a systematic review. Int Psychogeriatr. 2013 Feb;25(2):185-203. doi: 10.1017/S1041610212001627. Epub 2012 Oct 19. PMID: 23083438; PMCID: PMC3544545. |
Insufficient methodological quality (no search strategy available, data not systematically presented, data not pooled). |
|
Yunusa I, Alsumali A, Garba AE, Regestein QR, Eguale T. Assessment of Reported Comparative Effectiveness and Safety of Atypical Antipsychotics in the Treatment of Behavioral and Psychological Symptoms of Dementia: A Network Meta-analysis. JAMA Netw Open. 2019 Mar 1;2(3):e190828. doi: 10.1001/jamanetworkopen.2019.0828. PMID: 30901041; PMCID: PMC6583313. |
Less recent compared to Lü (2024). |
|
Ma H, Huang Y, Cong Z, Wang Y, Jiang W, Gao S, Zhu G. The efficacy and safety of atypical antipsychotics for the treatment of dementia: a meta-analysis of randomized placebo-controlled trials. J Alzheimers Dis. 2014;42(3):915-37. doi: 10.3233/JAD-140579. PMID: 25024323. |
Less recent compared to Mühlbauer (2021). |
|
Jin B, Liu H. Comparative efficacy and safety of therapy for the behavioral and psychological symptoms of dementia: a systemic review and Bayesian network meta-analysis. J Neurol. 2019 Oct;266(10):2363-2375. doi: 10.1007/s00415-019-09200-8. Epub 2019 Jan 21. PMID: 30666436. |
Wrong intervention for the PICO of this module (too broad; limited distinguishion between antipsychotics). |
|
Tampi RR, Tampi DJ, Balachandran S, Srinivasan S. Antipsychotic use in dementia: a systematic review of benefits and risks from meta-analyses. Ther Adv Chronic Dis. 2016 Sep;7(5):229-45. doi: 10.1177/2040622316658463. Epub 2016 Jul 15. PMID: 27583123; PMCID: PMC4994396. |
Insufficient methodological quality (no crude data, no systematic results, no quality assessment, data not pooled). |
|
Wang J, Yu JT, Wang HF, Meng XF, Wang C, Tan CC, Tan L. Pharmacological treatment of neuropsychiatric symptoms in Alzheimer's disease: a systematic review and meta-analysis. J Neurol Neurosurg Psychiatry. 2015 Jan;86(1):101-9. doi: 10.1136/jnnp-2014-308112. Epub 2014 May 29. PMID: 24876182. |
Population unclear, wrong intervention (broad) for the PICO of this module. |
|
Kongpakwattana K, Sawangjit R, Tawankanjanachot I, Bell JS, Hilmer SN, Chaiyakunapruk N. Pharmacological treatments for alleviating agitation in dementia: a systematic review and network meta-analysis. Br J Clin Pharmacol. 2018 Jul;84(7):1445-1456. doi: 10.1111/bcp.13604. Epub 2018 May 14. PMID: 29637593; PMCID: PMC6005613. |
Wrong intervention for the PICO of this module (too broad; no distinguishing for antipsychotics). |
|
Zhang Z, Zhang X, Xu L. Comparative efficacy and safety of olanzapine and risperidone in the treatment of psychiatric and behavioral symptoms of Alzheimer's disease: Systematic review and meta-analysis. Medicine (Baltimore). 2024 Jul 5;103(27):e35663. doi: 10.1097/MD.0000000000035663. PMID: 38968479; PMCID: PMC11224812. |
Includes 23 studies of which 18 are from a Chinese database and not accessible. |
|
Maglione M, Maher AR, Hu J, et al. Off-Label Use of Atypical Antipsychotics: An Update [Internet]. Rockville (MD): Agency for Healthcare Research and Quality (US); 2011 Sep. (Comparative Effectiveness Reviews, No. 43.) Available from: https://www.ncbi.nlm.nih.gov/books/NBK66081/ |
Wrong design (report). |
|
Tampi RR, Tampi DJ. Efficacy and tolerability of benzodiazepines for the treatment of behavioral and psychological symptoms of dementia: a systematic review of randomized controlled trials. Am J Alzheimers Dis Other Demen. 2014 Nov;29(7):565-74. doi: 10.1177/1533317514524813. PMID: 25551131; PMCID: PMC10852883. |
Wrong intervention for the PICO of this module (very broad), data not pooled; each study had a different I and C. |
|
Watt, J.A., Goodarzi, Z., Veroniki, A.A. et al. Safety of pharmacologic interventions for neuropsychiatric symptoms in dementia: a systematic review and network meta-analysis. BMC Geriatr 20, 212 (2020). https://doi.org/10.1186/s12877-020-01607-7 |
Wrong outcomes for the PICO of this module. |
|
Ureste, P., Cheng, C. (2022). The Efficacy and Safety of Atypical Antipsychotics for the Treatment of Dementia: A Meta-analysis of Randomized Placebo-Controlled Trials. In: Tampi, R.R., Tampi, D.J., Young, J.J., Balasubramaniam, M., Joshi, P. (eds) Essential Reviews in Geriatric Psychiatry. Springer, Cham. https://doi.org/10.1007/978-3-030-94960-0_4 |
Chapter based on Ma (2014). |
|
Rao A, Suliman A, Story G, Vuik S, Aylin P, Darzi A. Meta-analysis of population-based studies comparing risk of cerebrovascular accident associated with first- and second-generation antipsychotic prescribing in dementia. Int J Methods Psychiatr Res. 2016 Dec;25(4):289-298. doi: 10.1002/mpr.1509. Epub 2016 Apr 27. PMID: 27121795; PMCID: PMC6860234. |
Includes observational studies only. |
|
Zhai Y, Yin S, Zhang D. Association between Antipsychotic Drugs and Mortality in Older Persons with Alzheimer's Disease: A Systematic Review and Meta-Analysis. J Alzheimers Dis. 2016 Mar 31;52(2):631-9. doi: 10.3233/JAD-151207. PMID: 27031490. |
Wrong outcomes for the PICO of this module, wrong comparison for the PICO of this module. |
|
Yunusa I, Rashid N, Abler V, Rajagopalan K. Comparative Efficacy, Safety, Tolerability, and Effectiveness of Antipsychotics in The Treatment of Dementia-Related Psychosis (DRP): A Systematic Literature Review. J Prev Alzheimers Dis. 2021;8(4):520-533. doi: 10.14283/jpad.2021.48. PMID: 34585228. |
No pooled data. |
|
Xue XJ, He RL, Li WX, Xin JW, Ye QY, Chen XC, Pan XD. [Safety of atypical antipsychotics in the treatment of behavioral and psychological symptoms in dementia: a meta-analysis]. Zhonghua Yi Xue Za Zhi. 2018 Jul 3;98(25):2030-2036. Chinese. doi: 10.3760/cma.j.issn.0376-2491.2018.25.013. PMID: 29996606. |
Full text is not available. |
|
Nawzad S, Cahn W, Abdullah-Koolmees H. The efficacy of antipsychotics in the treatment of physical aggressive behavior in patients with dementia in nursing homes. Ther Adv Psychopharmacol. 2022 May 16;12:20451253221097452. doi: 10.1177/20451253221097452. PMID: 35600754; PMCID: PMC9118396. |
Includes only 1 RCT and 1 other study. |
Beoordelingsdatum en geldigheid
Publicatiedatum : 03-11-2025
Beoordeeld op geldigheid : 23-10-2025
Samenstelling werkgroep
Voor het ontwikkelen van de richtlijnmodule is in 2021 een multidisciplinair cluster ingesteld. Het cluster Cognitieve stoornissen en dementie bestaat uit meerdere richtlijnen (zie hier de actuele clusterindeling). De stuurgroep bewaakt het proces van modulair onderhoud binnen het cluster. De expertisegroepsleden brengen hun expertise in, indien nodig. De volgende personen uit het cluster zijn betrokken geweest bij de herziening van deze module:
Clusterstuurgroepleden
- Dhr. prof. dr. M.G.M. (Marcel) Olde Rikkert (voorzitter), klinisch geriater; NVKG
- Dhr. prof. dr. A.R. (Tony) Absalom, anesthesioloog; NVA
- Dhr. dr. J.H.J.M. (Jeroen) de Bresser, radioloog; NVvR
- Mevr. dr. I.K. (Indrag) Lampe, psychiater; NVvP
- Mevr. R. (Renske) Leistra, patiëntvertegenwoordiger; Alzheimer Nederland
- Mevr. prof. dr. B.C. (Barbara) van Munster, internist; NIV
- Dhr. prof. dr. E. (Edo) Richard, neuroloog; NVN
- Mevr. prof. dr. Ir. C. (Charlotte) Teunissen, klinisch chemicus; NVKC
- Dhr. dr. R.A.W. (Ronald) Verhagen, orthopedisch chirurg; NOV
Betrokken clusterexpertisegroepleden
- Dhr. dr. A.P.A. (Auke) Appelman, radioloog; NVvR
- Mevr. dr. R.L. (Rozemarijn) van Bruchem-van Visser, internist ouderengeneeskunde; NIV
- Dhr. drs. B.P.H. (Bas) ter Brugge, specialist ouderengeneeskunde; Verenso
- Dhr. dr. J.A.H.R. (Jurgen) Claassen, klinisch geriater; NVKG
- Dhr. dr. P.L.J. (Paul) Dautzenberg, klinisch geriater; NVKG
- Mevr. dr. M.E.A. (Marlise) van Eersel, internist; NIV
- Mevr. drs. C.M. (Christa) de Geus, neurogeneticus; NVKG
- Mevr. dr. E.M. (Elsmarieke van de Giessen), radioloog; NVNG
- Mevr. drs. M. (Marjolein) Groeneveld, verpleegkundig Consulent Geriatrie, klinisch epidemioloog; V&VN
- Mevr. drs. H. (Hiltsje) Heemskerk, klinisch geriater; NVKG
- Dhr. dr. R.B. (Rients) Huitema, klinisch neuropsycholoog; NIP
- Dhr. drs. A. (Ali) Lahdidioui, internist; NIV
- Dhr. dr. J. (Jules) Lavalaye, nucleair geneeskundige; NVNG
- Mevr. drs. L. (Lieke) Mitrov, ziekenhuisapotheker; NVZA
- Mevr. dr. M. (Marieke) Perry, huisarts/onderzoeker; NHG
- Dhr. dr. G. (Gerwin) Roks, neuroloog; NVN
- Mevr. dr. T.R. (Rikje) Ruiter, internist; NIV
- Mevr. A.J.B.P. (Astrid) Schoonbrood, ergotherapeut; EN
- Mevr. dr. N. (Niki) Schoonenboom, neuroloog; NVN
- Dhr. dr. H. (Harro) Seelaar, neuroloog; NVN
- Dhr. dr. K.S. (Koen) Simons, intensivist-internist; NVIC
- Mevr. drs. M.M.E. (Marlies) Sleegers-Kerkenaar, klinisch geriater; NVKG
- Mevr. drs. VCJ (Vera) van Stek-Smits, neuropsycholoog-gezondheidszorgpsycholoog; NIP
- Mevr. dr. E.M. (Eveline) Veltman, psychiater; NVvP
- Dhr. drs. D. (Dave) Verkaik, AIOS geriatrie; NVKG
- Mevr. prof. dr. M. (Meike) Vernooij, radioloog; NVvR
- Dhr. dr. E.G.B. (Jort) Vijverberg, neuroloog; NVN
- Mevr. dr. M.A. (Marjolein) Wijngaarden, internist; NIV
Met ondersteuning van
- Mevr. dr. C.T.J. (Charlotte) Michels, senior adviseur, Kennisinstituut van de Federatie Medisch Specialisten
- Mevr. dr. L.C. (Lotte) Houtepen, adviseur, Kennisinstituut van de Federatie Medisch Specialisten
- Mevr. drs. L.C. (Laura) van Wijngaarden, junior adviseur, Kennisinstituut van de Federatie Medisch Specialisten
Belangenverklaringen
Een overzicht van de belangen van de clusterleden en het oordeel over het omgaan met eventuele belangen vindt u in onderstaande tabel. De ondertekende belangenverklaringen zijn op te vragen bij het secretariaat van het Kennisinstituut van de Federatie Medisch Specialisten via secretariaat@kennisinstituut.nl.
Clusterstuurgroepleden
|
Clusterlid |
Functie |
Nevenfuncties |
Gemelde belangen |
Ondernomen actie |
|
Olde Rikkert* |
Hoogleraar Geriatrie, Radboudumc, Nijmegen |
Hoofdredacteur Nederlands Tijdschrift voor Geneeskunde |
Geen; uitsluitend ZonMw gefinancierd onderzoek dat overheidsbelang centraal stelt. Sinds 2017 geen farma-onderzoek meer. |
Geen restrictie |
|
Absalom |
Hoogleraar Anesthesiologie, UMCG, Groningen |
Consultancy werkzaamheden (betaald, alle betalingen aan UMCG) 6. Consultancy werk voor Becton Dickinson (Eysins, Switzerland) en Terumo (Tokyo, Japan) – technische advies over spuitpompen. Niet gerelateerd aan dementie/ MCI/ delier. |
Extern gefinancierd onderzoeken, maar financier heeft geen belangen bij de richtlijn. * Rigel Pharmaceuticals (San Francisco, USA) (PAST) * The Medicines Company (Parsippany, NJ, USA)(PAST) |
Geen restricties, omdat adviseurswerk niet gerelateerd is aan de afbakening van het cluster |
|
De Bresser |
- Neuroradioloog 1.0fte, LUMC, Leiden |
Geen |
Mijn onderzoek wordt mede gesponsord door Alzheimer Nederland. Deze financier heeft geen belang bij bepaalde uitkomsten van de richtlijn. |
Geen restrictie |
|
Lampe |
Psychiater, OLVG Ziekenhuis, Amsterdam |
Geen |
Geen |
Geen restrictie |
|
Leistra |
Belangenbehartiger, Alzheimer Nederland |
Casemanager Dementie bij de King Arthur Groep |
Geen |
Geen restrictie |
|
Richard |
Hoogleraar neurologie: * afdeling neurologie Radboudumc (0.8fte) * afdeling Public and Occupational Health Amsterdam UMC (0.2 fte) |
- Neuroloog-onderzoeker AmsterdamUMC, locatie AMC, gastvrijheidsaanstelling. - Hoofdredacteur Leerboek Neurologie (Bohn Staffleu van Loghem) |
Geen, uitsluitend onderzoek financiering van non-profit instellingen (e.g. ZonMw, Europese Commissie). |
Geen restrictie |
|
Teunissen |
Hoofd Neurochemisch laboratorium, Afdeling Klinische Chemie, AmsterdamUMC, lokatie VUmc, Amsterdam |
*Adviseur voor educatief blad: Mednet Neurologie (betaald). * Alle betalingen zijn aan het AmsterdamUMC. |
*Wetenschappelijke samenwerking met ADxNeurosciences, Olink, Quanterix, Roche in kader van o.a. Marie Curie subsidie. Het doel van het Marie Curie project is om een nieuwe generatie van onderzoekers in het biomarker veld op te leiden, tot experts in alle aspecten van biomarker onderzoek. Aan het einde van de ontwikkeling van biomarkers zullen deze via bedrijven op de markt moeten komen, en omdat niet alle wetenschappers uiteindelijk in de academische wereld blijven, is het belangrijk dat jonge onderzoekers ervaring opdoen met de manier van onderzoek doen in het bedrijfsleven. In marie curie projecten gebeurt dat door een deel van het onderzoek bij een bedrijf uit te voeren. *Het Neurochemisch laboratorium doet contractresearch voor Acumen, ADx Neurosciences, AC-Immune, Alamar, Aribio, Axon Neurosciences, Beckman-Coulter, BioConnect, Bioorchestra, Brainstorm Therapeutics, Celgene, Cognition Therapeutics, EIP Pharma, Eisai, Eli Lilly, Fujirebio, Instant Nano Biosensors, Novo Nordisk, Olink, PeopleBio, Quanterix, Roche, Toyama, Vivoryon. Dit zijn meestal biomarker-analyses voor hun trials, bij inclusies en als uitkomstmaten. *Grants: Research of CET is supported by the European Commission (Marie Curie International Training Network, grant agreement No 860197 (MIRIADE) and TAME, Innovative Medicines Initiatives 3TR (Horizon 2020, grant no 831434) EPND ( IMI 2 Joint Undertaking (JU), grant No. 101034344) and JPND (bPRIDE, CCAD), European Partnership on Metrology, co-financed from the European Union’s Horizon Europe Research and Innovation Programme and by the Participating States ((22HLT07 NEuroBioStand), CANTATE project funded by the Alzheimer Drug Discovery Foundation, Alzheimer Association, Michael J Fox Foundation, Health Holland, the Dutch Research Council (ZonMW), Alzheimer Drug Discovery Foundation, The Selfridges Group Foundation, Alzheimer Netherlands. CT is recipient of ABOARD, which is a public-private partnership receiving funding from ZonMW (#73305095007) and Health~Holland, Topsector Life Sciences & Health (PPP-allowance; #LSHM20106). CT is recipient of TAP-dementia, a ZonMw funded project (#10510032120003) in the context of the Dutch National Dementia Strategy. |
Geen restrictie |
|
Van Munster |
* Hoogleraar Interne Geneeskunde, Ouderengeneeskunde/Geriatrie, UMCG, Groningen. *Plaatsvervangend opleider Geriatrie, UMCG, Groningen. |
- 2020 – heden Voorzitter Alzheimer Centrum Groningen (alle functies zijn onbetaald) |
*2022 ZONMw: Young Onset Dementia- INCLUDED: Advance care planning. 2022 ZEGG/ZONMw: "The impact of a comprehensive geriatric assessment including advance care planning in acutely hospitalized frail patients with cognitive disorders: the GOAL study" |
Geen restrictie |
|
Verhagen |
Orthopedisch chirurg/opleider in Tergooi MC |
Geen |
Geen |
Geen restrictie |
Clusterexpertisegroepleden
Richtlijn Dementie: Module ‘Antipsychotica bij dementie’
|
Clusterlid |
Functie |
Nevenfuncties |
Gemelde belangen |
Ondernomen actie |
|
Mitrov |
Ziekenhuisapotheker, Flevoziekenhuis, Almere |
Special Interest Group Farmacotherapie bij ouderen (NVKG/NVZA), onbetaald |
Geen |
Geen restrictie |
|
Verkaik |
Aios geriatrie, betaalde functie. In opleiding vanuit Gelre ziekenhuizen Apeldoorn en Zutphen. |
Deelname pvc commissie als aios lid voor kwaliteits visitaties (onbetaald) |
Geen |
Geen restrictie |
Richtlijn Dementie: Module ‘Antidepressiva bij dementie’
|
Clusterlid |
Functie |
Nevenfuncties |
Gemelde belangen |
Ondernomen actie |
|
Heemskerk |
Klinisch geriater, Arkin Ouderen (GGZ, Amsterdam) |
NVKG, commissie ouderenpsychiatrie |
Geen |
Geen restrictie |
|
Veltman |
Psychiater, UMCU |
Geen |
Geen |
Geen restrictie |
Richtlijn MCI: Module 'Biomarkers’
|
Clusterlid |
Functie |
Nevenfuncties |
Gemelde belangen |
Ondernomen actie |
|
Van de Giessen |
Radioloog (aandachtsgebeid nucleaire geneeskunde), Amsterdam UMC |
- NVNG, commissie onderwijs en themagroep neuro |
Beoordelingen PET scans voor Ixico (betaald aan instituut) Contract research voor Roche (betaald aan instituut) Onderzoeksfinanciering van non-profit instelingen (o.a. ZonMw, Alzheimer Nederland, Hersenstichting, Health~Holland, KWF) |
Geen restrictie |
|
Richard |
Hoogleraar neurologie: * afdeling neurologie Radboudumc (0.8fte) * afdeling Public and Occupational Health Amsterdam UMC (0.2 fte) |
- Neuroloog-onderzoeker AmsterdamUMC, locatie AMC, gastvrijheidsaanstelling. - Hoofdredacteur Leerboek Neurologie (Bohn Staffleu van Loghem) |
Geen, uitsluitend onderzoek financiering van non-profit instellingen (e.g. ZonMw, Europese Commissie). |
Geen restrictie |
Richtlijn MCI: Module ‘Leefstijladviezen’
|
Clusterlid
|
Functie |
Nevenfuncties |
Gemelde belangen |
Ondernomen actie |
|
Groeneveld |
Verpleegkundig consulent geriatrie (16 uur) Verpleegkundig onderzoeker Waardengedreven zorg (16 uur) |
Geen
|
Extern gefinancieerd onderzoek: * Catharina onderzoeksfonds: Prestatie en acceptatie van een predictiemodel voor vallen in het ziekenhuis (rol als projectleider) *KIPZ subsidie t/m 31 december: Prestatie en acceptatie van een predictiemodel voor vallen in het ziekenhuis (ja) |
Geen restrictie |
|
Ruiter |
*Internist ouderen geneeskunde en klinische farmacologie Maasstad Ziekenhuis 0,8fte *Post-doctoraal onderzoeker & Epidemioloog B Erasmus MC dept. Epidemiologie 0,1 fte * Lid van het College ter beoordeling van geneesmiddelen |
*Lid raad van toezicht Stichting Landelijk Wonen Klein Houtdijk (Onbetaald). *Secretaris Forum Visitatorum NIV (Onbetaald). *Redactielid Tijdschrift Geriatrie en Gerontologie (Onbetaald). * Voorzitter commissie beroepsbelangen kerngroep ouderengeneeskunde NIV. |
Geen |
Geen restrictie |
|
Claassen |
Universitair hoofddocent en klinisch geriater (1.0 fte), Radboudumc, Nijmegen |
Geen |
*Fase 3 onderzoek Novo Nordisk (EVOKE, NCT04777396. Wereldwijde geneesmiddelenstudie (semaglutide) over Alzheimer. Rol als lokale PI. Middel komt niet aan bod in huidige vijf modules. *MOCIA-project, gefinancierd door ZonMW (www.mocia.nl). *ABOARD onderzoek (gefinancierd door ZonMW en Health Holland, projectnummer: 73305095007) |
Geen restrictie |
Richtlijn MCI: Module ‘Follow-up’
|
Clusterlid |
Functie |
Nevenfuncties |
Gemelde belangen |
Ondernomen actie |
|
Claassen |
Universitair hoofddocent en klinisch geriater (1.0 fte), Radboudumc, Nijmegen |
Geen |
*Fase 3 onderzoek Novo Nordisk (EVOKE, NCT04777396. Wereldwijde geneesmiddelenstudie (semaglutide) over Alzheimer. Rol als lokale PI. Middel komt niet aan bod in huidige vijf modules. *MOCIA-project, gefinancierd door ZonMW (www.mocia.nl). *ABOARD onderzoek (gefinancierd door ZonMW en Health Holland, projectnummer: 73305095007) |
Geen restrictie |
|
Perry |
* Huisarts, Huisartsenpraktijk Velp, 0.5 fte * Senior-onderzoeker afdelingen eerstelijnsgeneeskunde en Geriatrie van het Radboudumc en bij het Radboud Alzheimer Centrum, 0.5 fte |
Auteur hoofdstuk Vergeetachtigheid in Álledaagse klachten 2020 (onkostenvergoeding) Commissielid werkgroep multidisciplinaire richtlijn Dementie 2018 - 2020 (onkostenvergoeding) Commissielid werkgroep heziening NHG standaard Dementie 2017 - 2020 (onkostenvergoeding) Commissielid werkgroep Addendum MCI bij multidisciplinaire richtlijn Dementie 20 1 6-20 1 B (onkostenvergoeding) Auteur online nascholing dementie Accredidact huisartsen 2016 en doktersassistenten 2017 (betaald) Auteur twee boekhoofdstukken dementie (palliatieve zorg en diagnostische verrichtingen) 2017 en 2018 in opdracht van het NHG (onkostenvergoeding) Auteur hoofdtsuk Vergeetachtigheid (Alledaagse klachten in de huisartsgeneeskunde) 2020 Onkostenvergoeding Expert bij www.dementie.nl tot heden (vrijwillig) Columnist Alz (donateursblaadje Alzheimer Nederland) tot 2016 (vrijwillig) |
Projectleider DementieNet (financiering door Giekes-Strijbis fonds, Alzheimer Nederland en ZonMw) Andere ZonMw/Memorabel projecten: - Decidem (anticiperende besluitvorming met mensen met dementie door huisartsen), medeprojectleider - Crisisreductie in de dementiezorg, medeaanvrager - SHiMMy en SHARED (relatie sociale gezondheid en ontstaan en progressie van dementie), medeaanvrager - S-Decided (gezamelijke besluitvorming bij diagnostiek bij geheugenklachten), medeaanvrager
Diverse projecten omtrent transmurale/interprofessionele aanpak van advance care planning van Zorginstituut Nederland, Stoffels-Hornstra fonds, ZonMw EPOS: Extramurale praktijkontwikkeling specialist ouderengeneeskunde UNICITY: dementie op jonge leeftijd, oa signalering door huisartsen en onderscheid met depressie, burn-out
*DementieNet aanpak, generieke benadering voor lokale eerstelijns netwerkzorg *Expert bij www.dementie.nl, website van Alzheimer Nederland (vrijwillig) *Columnist Alz (donateursblaadje Alzheimer Nederland) tot 2016 (vrijwillig) *Diverse malen gastspreker bij verschillende Alzheimer Cafés (vrijwillig) |
Geen restrictie |
|
Roks |
Neuroloog ETZ Tilburg |
METC Brabant, lid en vice voorzitter (onkostenvergoeding) |
Geen |
Geen restrictie |
Algemene herziening: Nieuwe concept indeling (richtlijn Dementie en richtlijn MCI)
|
Clusterlid |
Functie |
Nevenfuncties |
Gemelde belangen |
Ondernomen actie |
|
Lampe |
Psychiater, OLVG ziekenhuis |
Geen |
Geen |
Geen restrictie |
|
Olde Rikkert* |
Hoogleraar Geriatrie, Radboudumc, Nijmegen |
Hoofdredacteur Nederlands Tijdschrift voor Geneeskunde |
Geen; uitsluitend ZonMw gefinancierd onderzoek dat overheidsbelang centraal stelt. Sinds 2017 geen farma-onderzoek meer. |
Geen restrictie |
|
Perry |
* Huisarts, Huisartsenpraktijk Velp, 0.5 fte * Senior-onderzoeker afdelingen eerstelijnsgeneeskunde en Geriatrie van het Radboudumc en bij het Radboud Alzheimer Centrum, 0.5 fte |
Auteur hoofdstuk Vergeetachtigheid in Álledaagse klachten 2020 (onkostenvergoeding) Commissielid werkgroep multidisciplinaire richtlijn Dementie 2018 - 2020 (onkostenvergoeding) Commissielid werkgroep heziening NHG standaard Dementie 2017 - 2020 (onkostenvergoeding) Commissielid werkgroep Addendum MCI bij multidisciplinaire richtlijn Dementie 20 1 6-20 1 B (onkostenvergoeding) Auteur online nascholing dementie Accredidact huisartsen 2016 en doktersassistenten 2017 (betaald) Auteur twee boekhoofdstukken dementie (palliatieve zorg en diagnostische verrichtingen) 2017 en 2018 in opdracht van het NHG (onkostenvergoeding) Auteur hoofdtsuk Vergeetachtigheid (Alledaagse klachten in de huisartsgeneeskunde) 2020 Onkostenvergoeding Expert bij www.dementie.nl tot heden (vrijwillig) Columnist Alz (donateursblaadje Alzheimer Nederland) tot 2016 (vrijwillig) |
Projectleider DementieNet (financiering door Giekes-Strijbis fonds, Alzheimer Nederland en ZonMw) Andere ZonMw/Memorabel projecten: - Decidem (anticiperende besluitvorming met mensen met dementie door huisartsen), medeprojectleider - Crisisreductie in de dementiezorg, medeaanvrager - SHiMMy en SHARED (relatie sociale gezondheid en ontstaan en progressie van dementie), medeaanvrager - S-Decided (gezamelijke besluitvorming bij diagnostiek bij geheugenklachten), medeaanvrager
Diverse projecten omtrent transmurale/interprofessionele aanpak van advance care planning van Zorginstituut Nederland, Stoffels-Hornstra fonds, ZonMw EPOS: Extramurale praktijkontwikkeling specialist ouderengeneeskunde UNICITY: dementie op jonge leeftijd, oa signalering door huisartsen en onderscheid met depressie, burn-out
*DementieNet aanpak, generieke benadering voor lokale eerstelijns netwerkzorg *Expert bij www.dementie.nl, website van Alzheimer Nederland (vrijwillig) *Columnist Alz (donateursblaadje Alzheimer Nederland) tot 2016 (vrijwillig) *Diverse malen gastspreker bij verschillende Alzheimer Cafés (vrijwillig) |
Geen restrictie |
|
Richard |
Hoogleraar neurologie: * afdeling neurologie Radboudumc (0.8fte) * afdeling Public and Occupational Health Amsterdam UMC (0.2 fte) |
- Neuroloog-onderzoeker AmsterdamUMC, locatie AMC, gastvrijheidsaanstelling. - Hoofdredacteur Leerboek Neurologie (Bohn Staffleu van Loghem) |
Geen, uitsluitend onderzoek financiering van non-profit instellingen (e.g. ZonMw, Europese Commissie). |
Geen restrictie |
Inbreng patiëntenperspectief
Kwalitatieve raming van mogelijke financiële gevolgen in het kader van de Wkkgz
Bij de richtlijnmodule voerden de clusterleden conform de Wet kwaliteit, klachten en geschillen zorg (Wkkgz) een kwalitatieve raming uit om te beoordelen of de aanbevelingen mogelijk leiden tot substantiële financiële gevolgen. Bij het uitvoeren van deze beoordeling is de richtlijnmodule op verschillende domeinen getoetst (zie het stroomschema bij Werkwijze).
Uit de kwalitatieve raming blijkt dat er waarschijnlijk geen substantiële financiële gevolgen zijn, zie onderstaande tabel.
| Module |
Uitkomst raming |
Toelichting |
|
Antipsychotica bij dementie |
Geen substantiële financiële gevolgen |
Hoewel uit de toetsing volgt dat de aanbeveling(en) breed toepasbaar zijn (>40.000 patiënten), volgt uit de toetsing dat het overgrote deel van de zorgverleners al aan de norm voldoet, het geen nieuwe manier van zorgverlening betreft, het geen toename in het aantal voltijdsequivalenten of wijziging in het opleidingsniveau van zorgverleners betreft. Er worden daarom geen financiële gevolgen verwacht. |
Werkwijze
Voor meer details over de gebruikte richtlijnmethodologie verwijzen wij u naar de Werkwijze. Relevante informatie voor de ontwikkeling/herziening van deze richtlijnmodule is hieronder weergegeven.
Zoekverantwoording
Algemene informatie
|
Cluster/richtlijn: Cognitieve stoornissen en Dementie |
|
|
Uitgangsvraag/modules: UV1 Wat is de rol van antipsychotica in de behandeling van neuro-psychiatrische symptomen bij patiënten met dementie? |
|
|
Database(s): Embase.com, Ovid/Medline, Ovid/PsycInfo |
Datum: 30 augustus 2024 |
|
Periode: vanaf 2010 |
Talen: geen restrictie |
|
Literatuurspecialist: Alies Oost |
|
|
BMI-zoekblokken: voor verschillende opdrachten wordt (deels) gebruik gemaakt van de zoekblokken van BMI-Online https://blocks.bmi-online.nl/ Deduplication: voor het ontdubbelen is gebruik gemaakt van http://dedupendnote.nl:9777/ |
|
|
Toelichting: Voor deze vraag is gezocht op de elementen:
àHet sleutelartikel wordt niet gevonden met de search omdat termen voor neuro-psychiatrische symptomen ontbreken. In overleg met de adviseur is besloten dat deze niet uit de search hoeft te komen. →Er is besloten om in eerste instantie alleen de SRs te screenen. |
|
|
Te gebruiken voor richtlijntekst: In de databases Embase.com, Ovid/Medline en Ovid/PsycInfo is op 30 augustus 2024 systematisch gezocht naar systematische reviews, RCTs en observationele studies vanaf 2010 over antipsychotica bij neuro-psychiatrische symptomen bij patiënten met dementie. De literatuurzoekactie leverde 1055 unieke treffers op. |
|
Zoekopbrengst
|
|
EMBASE |
OVID/MEDLINE |
OVID/PSYCINFO |
Ontdubbeld |
|
SR |
899 |
302 |
261 |
1055* |
|
RCT |
1132 |
422 |
175 |
1269 |
|
Observationele studies |
1709 |
523 |
277 |
1762 |
|
Totaal |
3740 |
1247 |
713 |
4086 |
*in Rayyan
Zoekstrategie
Embase.com
|
No. |
Query |
Results |
|
#1 |
'dementia'/exp OR 'parkinson disease'/exp OR dement*:ti,ab,kw OR parkinson*:ti,ab,kw OR alzheimer*:ti,ab,kw OR ((lewy* NEAR/2 bod*):ti,ab,kw) OR dlbd:ti,ab,kw OR lbd:ti,ab,kw OR ftd:ti,ab,kw OR ftld:ti,ab,kw OR ((pick* NEAR/3 (complex OR disease* OR syndrome*)):ti,ab,kw) |
764377 |
|
#2 |
'behavioral and psychological symptoms of dementia'/exp OR 'behavior'/de OR 'behavior change'/exp OR 'behavior disorder'/de OR 'abnormal behavior'/exp OR 'disruptive behavior'/exp OR 'behavior control'/exp OR 'emotion'/exp OR 'agitation'/exp OR 'aggression'/exp OR 'psychosis'/exp OR agitat*:ti,ab,kw OR irritab*:ti,ab,kw OR aggress*:ti,ab,kw OR anger:ti,ab,kw OR angr*:ti,ab,kw OR tantrum*:ti,ab,kw OR rage:ti,ab,kw OR violen*:ti,ab,kw OR hostil*:ti,ab,kw OR distress*:ti,ab,kw OR anxiety:ti,ab,kw OR fear:ti,ab,kw OR restless*:ti,ab,kw OR psychosis:ti,ab,kw OR psychoses:ti,ab,kw OR psychotic*:ti,ab,kw OR paranoia:ti,ab,kw OR neuropsychiatr*:ti,ab,kw OR 'neuro psychiatr*':ti,ab,kw OR neurobehav*:ti,ab,kw OR 'neuro behav*':ti,ab,kw OR (((behaviour* OR behavior*) NEAR/3 (symptom* OR manifestation* OR chang* OR aberra* OR abnormal OR disrupt* OR condition* OR problem* OR disturb*)):ti,ab,kw) |
2512654 |
|
#3 |
'neuroleptic agent'/exp OR neuroleptic*:ti,ab,kw OR antipsychotic*:ti,ab,kw OR 'anti psychotic*':ti,ab,kw OR ((major NEAR/3 (tranquilliz* OR tranquiliz*)):ti,ab,kw) OR 'clozapine'/exp OR 'clozapin*':ti,ab,kw OR 'risperidone'/exp OR 'risperidon*':ti,ab,kw OR 'Haloperidol'/exp OR 'Haloperidol':ti,ab,kw OR haloperidol:ti,ab,kw OR 'pipamperone'/exp OR 'dipiperon*':ti,ab,kw OR 'pipamperon*':ti,ab,kw |
343981 |
|
#4 |
#1 AND #2 AND #3 NOT ('conference abstract'/it OR 'editorial'/it OR 'letter'/it OR 'note'/it) NOT (('animal'/exp OR 'animal experiment'/exp OR 'animal model'/exp OR 'nonhuman'/exp) NOT 'human'/exp) AND [2010-2024]/py |
6834 |
|
#5 |
'meta analysis'/exp OR 'meta analysis (topic)'/exp OR metaanaly*:ti,ab OR 'meta analy*':ti,ab OR metanaly*:ti,ab OR 'systematic review'/de OR 'cochrane database of systematic reviews'/jt OR prisma:ti,ab OR prospero:ti,ab OR (((systemati* OR scoping OR umbrella OR 'structured literature') NEAR/3 (review* OR overview*)):ti,ab) OR ((systemic* NEAR/1 review*):ti,ab) OR (((systemati* OR literature OR database* OR 'data base*') NEAR/10 search*):ti,ab) OR (((structured OR comprehensive* OR systemic*) NEAR/3 search*):ti,ab) OR (((literature NEAR/3 review*):ti,ab) AND (search*:ti,ab OR database*:ti,ab OR 'data base*':ti,ab)) OR (('data extraction':ti,ab OR 'data source*':ti,ab) AND 'study selection':ti,ab) OR ('search strategy':ti,ab AND 'selection criteria':ti,ab) OR ('data source*':ti,ab AND 'data synthesis':ti,ab) OR medline:ab OR pubmed:ab OR embase:ab OR cochrane:ab OR (((critical OR rapid) NEAR/2 (review* OR overview* OR synthes*)):ti) OR ((((critical* OR rapid*) NEAR/3 (review* OR overview* OR synthes*)):ab) AND (search*:ab OR database*:ab OR 'data base*':ab)) OR metasynthes*:ti,ab OR 'meta synthes*':ti,ab |
1057327 |
|
#6 |
'clinical trial'/exp OR 'randomization'/exp OR 'single blind procedure'/exp OR 'double blind procedure'/exp OR 'crossover procedure'/exp OR 'placebo'/exp OR 'prospective study'/exp OR rct:ab,ti OR random*:ab,ti OR 'single blind':ab,ti OR 'randomised controlled trial':ab,ti OR 'randomized controlled trial'/exp OR placebo*:ab,ti |
4097198 |
|
#7 |
'major clinical study'/de OR 'clinical study'/de OR 'case control study'/de OR 'family study'/de OR 'longitudinal study'/de OR 'retrospective study'/de OR 'prospective study'/de OR 'comparative study'/de OR 'cohort analysis'/de OR ((cohort NEAR/1 (study OR studies)):ab,ti) OR (('case control' NEAR/1 (study OR studies)):ab,ti) OR (('follow up' NEAR/1 (study OR studies)):ab,ti) OR (observational NEAR/1 (study OR studies)) OR ((epidemiologic NEAR/1 (study OR studies)):ab,ti) OR (('cross sectional' NEAR/1 (study OR studies)):ab,ti) |
8383159 |
|
#8 |
'case control study'/de OR 'comparative study'/exp OR 'control group'/de OR 'controlled study'/de OR 'controlled clinical trial'/de OR 'crossover procedure'/de OR 'double blind procedure'/de OR 'phase 2 clinical trial'/de OR 'phase 3 clinical trial'/de OR 'phase 4 clinical trial'/de OR 'pretest posttest design'/de OR 'pretest posttest control group design'/de OR 'quasi experimental study'/de OR 'single blind procedure'/de OR 'triple blind procedure'/de OR (((control OR controlled) NEAR/6 trial):ti,ab,kw) OR (((control OR controlled) NEAR/6 (study OR studies)):ti,ab,kw) OR (((control OR controlled) NEAR/1 active):ti,ab,kw) OR 'open label*':ti,ab,kw OR (((double OR two OR three OR multi OR trial) NEAR/1 (arm OR arms)):ti,ab,kw) OR ((allocat* NEAR/10 (arm OR arms)):ti,ab,kw) OR placebo*:ti,ab,kw OR 'sham-control*':ti,ab,kw OR (((single OR double OR triple OR assessor) NEAR/1 (blind* OR masked)):ti,ab,kw) OR nonrandom*:ti,ab,kw OR 'non-random*':ti,ab,kw OR 'quasi-experiment*':ti,ab,kw OR crossover:ti,ab,kw OR 'cross over':ti,ab,kw OR 'parallel group*':ti,ab,kw OR 'factorial trial':ti,ab,kw OR ((phase NEAR/5 (study OR trial)):ti,ab,kw) OR ((case* NEAR/6 (matched OR control*)):ti,ab,kw) OR ((match* NEAR/6 (pair OR pairs OR cohort* OR control* OR group* OR healthy OR age OR sex OR gender OR patient* OR subject* OR participant*)):ti,ab,kw) OR ((propensity NEAR/6 (scor* OR match*)):ti,ab,kw) OR versus:ti OR vs:ti OR compar*:ti OR ((compar* NEAR/1 study):ti,ab,kw) OR (('major clinical study'/de OR 'clinical study'/de OR 'cohort analysis'/de OR 'observational study'/de OR 'cross-sectional study'/de OR 'multicenter study'/de OR 'correlational study'/de OR 'follow up'/de OR cohort*:ti,ab,kw OR 'follow up':ti,ab,kw OR followup:ti,ab,kw OR longitudinal*:ti,ab,kw OR prospective*:ti,ab,kw OR retrospective*:ti,ab,kw OR observational*:ti,ab,kw OR 'cross sectional*':ti,ab,kw OR cross?ectional*:ti,ab,kw OR multicent*:ti,ab,kw OR 'multi-cent*':ti,ab,kw OR consecutive*:ti,ab,kw) AND (group:ti,ab,kw OR groups:ti,ab,kw OR subgroup*:ti,ab,kw OR versus:ti,ab,kw OR vs:ti,ab,kw OR compar*:ti,ab,kw OR 'odds ratio*':ab OR 'relative odds':ab OR 'risk ratio*':ab OR 'relative risk*':ab OR 'rate ratio':ab OR aor:ab OR arr:ab OR rrr:ab OR ((('or' OR 'rr') NEAR/6 ci):ab))) |
15351559 |
|
#9 |
#4 AND #5 - SR |
899 |
|
#10 |
#4 AND #6 NOT #9 - RCT |
1132 |
|
#11 |
#4 AND (#7 OR #8) NOT (#9 OR #10) - observationeel |
1709 |
|
#12 |
#9 OR #10 OR #11 |
3740 |
Ovid/Medline
|
# |
Searches |
Results |
|
1 |
exp Dementia/ or Parkinsonian Disorders/ or dement*.ti,ab,kf. or parkinson*.ti,ab,kf. or alzheimer*.ti,ab,kf. or (lewy* adj2 bod*).ti,ab,kf. or dlbd.ti,ab,kf. or lbd.ti,ab,kf. or ftd.ti,ab,kf. or ftld.ti,ab,kf. or (pick* adj3 (complex or disease* or syndrome*)).ti,ab,kf. |
477445 |
|
2 |
Behavior/ or Behavioral Symptoms/ or Social Behavior/ or exp Problem Behavior/ or exp Behavior Control/ or exp Affect/ or exp Anger/ or exp Violence/ or exp Hostility/ or exp Aberrant Motor Behavior in Dementia/ or Psychotic Disorders/ or agitat*.ti,ab,kf. or irritab*.ti,ab,kf. or aggress*.ti,ab,kf. or anger.ti,ab,kf. or angr*.ti,ab,kf. or tantrum*.ti,ab,kf. or rage.ti,ab,kf. or violen*.ti,ab,kf. or hostil*.ti,ab,kf. or distress*.ti,ab,kf. or anxiety.ti,ab,kf. or fear.ti,ab,kf. or restless*.ti,ab,kf. or psychosis.ti,ab,kf. or psychoses.ti,ab,kf. or psychotic*.ti,ab,kf. or paranoia.ti,ab,kf. or (neuropsychiatr* or 'neuro psychiatr*' or neurobehav* or 'neuro behav*').ti,ab,kf. or ((behaviour* or behavior*) adj3 (symptom* or manifestation* or chang* or aberra* or abnormal or disrupt* or condition* or problem* or disturb*)).ti,ab,kf. |
1385630 |
|
3 |
exp Antipsychotic Agents/ or neuroleptic*.ti,ab,kf. or antipsychotic*.ti,ab,kf. or 'anti psychotic*'.ti,ab,kf. or (major adj3 (tranquilliz* or tranquiliz*)).ti,ab,kf. or exp clozapine/ or 'clozapin*'.ti,ab,kf. or exp risperidone/ or 'risperidon*'.ti,ab,kf. or exp Haloperidol/ or 'Haloperidol'.ti,ab,kf. or haloperidol.ti,ab,kf. or 'dipiperon*'.ti,ab,kf. or 'pipamperon*'.ti,ab,kf. |
161211 |
|
4 |
(1 and 2 and 3) not (comment/ or editorial/ or letter/) not ((exp animals/ or exp models, animal/) not humans/) |
4096 |
|
5 |
limit 4 to yr="2010 -Current" |
2111 |
|
6 |
meta-analysis/ or meta-analysis as topic/ or (metaanaly* or meta-analy* or metanaly*).ti,ab,kf. or systematic review/ or cochrane.jw. or (prisma or prospero).ti,ab,kf. or ((systemati* or scoping or umbrella or "structured literature") adj3 (review* or overview*)).ti,ab,kf. or (systemic* adj1 review*).ti,ab,kf. or ((systemati* or literature or database* or data-base*) adj10 search*).ti,ab,kf. or ((structured or comprehensive* or systemic*) adj3 search*).ti,ab,kf. or ((literature adj3 review*) and (search* or database* or data-base*)).ti,ab,kf. or (("data extraction" or "data source*") and "study selection").ti,ab,kf. or ("search strategy" and "selection criteria").ti,ab,kf. or ("data source*" and "data synthesis").ti,ab,kf. or (medline or pubmed or embase or cochrane).ab. or ((critical or rapid) adj2 (review* or overview* or synthes*)).ti. or (((critical* or rapid*) adj3 (review* or overview* or synthes*)) and (search* or database* or data-base*)).ab. or (metasynthes* or meta-synthes*).ti,ab,kf. |
770602 |
|
7 |
exp clinical trial/ or randomized controlled trial/ or exp clinical trials as topic/ or randomized controlled trials as topic/ or Random Allocation/ or Double-Blind Method/ or Single-Blind Method/ or (clinical trial, phase i or clinical trial, phase ii or clinical trial, phase iii or clinical trial, phase iv or controlled clinical trial or randomized controlled trial or multicenter study or clinical trial).pt. or random*.ti,ab. or (clinic* adj trial*).tw. or ((singl* or doubl* or treb* or tripl*) adj (blind$3 or mask$3)).tw. or Placebos/ or placebo*.tw. |
2770345 |
|
8 |
Epidemiologic studies/ or case control studies/ or exp cohort studies/ or Controlled Before-After Studies/ or Case control.tw. or cohort.tw. or Cohort analy$.tw. or (Follow up adj (study or studies)).tw. or (observational adj (study or studies)).tw. or Longitudinal.tw. or Retrospective*.tw. or prospective*.tw. or consecutive*.tw. or Cross sectional.tw. or Cross-sectional studies/ or historically controlled study/ or interrupted time series analysis/ [Onder exp cohort studies vallen ook longitudinale, prospectieve en retrospectieve studies] |
4813383 |
|
9 |
Case-control Studies/ or clinical trial, phase ii/ or clinical trial, phase iii/ or clinical trial, phase iv/ or comparative study/ or control groups/ or controlled before-after studies/ or controlled clinical trial/ or double-blind method/ or historically controlled study/ or matched-pair analysis/ or single-blind method/ or (((control or controlled) adj6 (study or studies or trial)) or (compar* adj (study or studies)) or ((control or controlled) adj1 active) or "open label*" or ((double or two or three or multi or trial) adj (arm or arms)) or (allocat* adj10 (arm or arms)) or placebo* or "sham-control*" or ((single or double or triple or assessor) adj1 (blind* or masked)) or nonrandom* or "non-random*" or "quasi-experiment*" or "parallel group*" or "factorial trial" or "pretest posttest" or (phase adj5 (study or trial)) or (case* adj6 (matched or control*)) or (match* adj6 (pair or pairs or cohort* or control* or group* or healthy or age or sex or gender or patient* or subject* or participant*)) or (propensity adj6 (scor* or match*))).ti,ab,kf. or (confounding adj6 adjust*).ti,ab. or (versus or vs or compar*).ti. or ((exp cohort studies/ or epidemiologic studies/ or multicenter study/ or observational study/ or seroepidemiologic studies/ or (cohort* or 'follow up' or followup or longitudinal* or prospective* or retrospective* or observational* or multicent* or 'multi-cent*' or consecutive*).ti,ab,kf.) and ((group or groups or subgroup* or versus or vs or compar*).ti,ab,kf. or ('odds ratio*' or 'relative odds' or 'risk ratio*' or 'relative risk*' or aor or arr or rrr).ab. or (("OR" or "RR") adj6 CI).ab.)) |
5771545 |
|
10 |
5 and 6 - SR |
302 |
|
11 |
(5 and 7) not 10 - RCT |
422 |
|
12 |
(5 and (8 or 9)) not (10 or 11) - observationeel |
523 |
|
13 |
10 or 11 or 12 |
1247 |
Ovid/PsycInfo
|
# |
Searches |
Results |
|
1 |
exp Dementia/ or exp Alzheimer Disease/ or exp Parkinsons Disease/ or dement*.ti,ab,id. or parkinson*.ti,ab,id. or alzheimer*.ti,ab,id. or (lewy* adj2 bod*).ti,ab,id. or dlbd.ti,ab,id. or lbd.ti,ab,id. or ftd.ti,ab,id. or ftld.ti,ab,id. or (pick* adj3 (complex or disease* or syndrome*)).ti,ab,id. |
160980 |
|
2 |
exp Antisocial Behavior/ or exp Social Behavior/ or exp Behavior Problems/ or exp Behavior Change/ or exp Emotions/ or exp Affect Regulation/ or exp Psychosis/ or agitat*.ti,ab,id. or irritab*.ti,ab,id. or aggress*.ti,ab,id. or anger.ti,ab,id. or angr*.ti,ab,id. or tantrum*.ti,ab,id. or rage.ti,ab,id. or violen*.ti,ab,id. or hostil*.ti,ab,id. or distress*.ti,ab,id. or anxiety.ti,ab,id. or fear.ti,ab,id. or restless*.ti,ab,id. or psychosis.ti,ab,id. or psychoses.ti,ab,id. or psychotic*.ti,ab,id. or paranoia.ti,ab,id. or (neuropsychiatr* or 'neuro psychiatr*' or neurobehav* or 'neuro behav*').ti,ab,id. or ((behaviour* or behavior*) adj3 (symptom* or manifestation* or chang* or aberra* or abnormal or disrupt* or condition* or problem* or disturb*)).ti,ab,id. |
2150552 |
|
3 |
exp Neuroleptic Drugs/ or neuroleptic*.ti,ab,id. or antipsychotic*.ti,ab,id. or 'anti psychotic*'.ti,ab,id. or (major adj3 (tranquilliz* or tranquiliz*)).ti,ab,id. or exp clozapine/ or 'clozapin*'.ti,ab,id. or exp risperidone/ or 'risperidon*'.ti,ab,id. or exp Haloperidol/ or 'Haloperidol'.ti,ab,id. or haloperidol.ti,ab,id. or 'dipiperon*'.ti,ab,id. or 'pipamperon*'.ti,ab,id. |
58486 |
|
4 |
1 and 2 and 3 |
3050 |
|
5 |
limit 4 to yr="2010 -Current" |
1360 |
|
6 |
((literature review or systematic review or meta analysis).md. or "literature review"/ or meta analysis/ or (((meta adj2 analy*) or metaanaly* or (synthes* adj2 (literature* or research* or studies or data)) or (pooled and analys*) or ((data adj1 pool*) and studies) or medline or medlars or embase or cinahl or scisearch or psychlit or psyclit or cinhal or cancerlit or cochrane or bids or pubmed or ovid or ((hand or manual or database* or computer*) adj1 search*) or (electronic adj1 (database* or data base or data bases))).ti,ab,id. or (review* or overview).ti. or (bibliograph* or relevant journals or ((review* or overview*) adj9 (systematic* or methodologic* or quantitativ* or research* or literature* or studies or trial* or effective*))).ab.)) not (((retrospective* or record* or case* or patient*) adj1 review*) or ((patient* or review*) adj1 chart*)).ti,ab,id. |
501267 |
|
7 |
exp clinical trial/ or randomized controlled trial/ or randomized controlled trials as topic/ or Random Allocation/ or Double-Blind Method/ or Single-Blind Method/ or (clinical trial, phase i or clinical trial, phase ii or clinical trial, phase iii or clinical trial, phase iv or controlled clinical trial or randomized controlled trial or multicenter study or clinical trial).pt. or random*.ti,ab. or (clinic* adj trial*).tw. or ((singl* or doubl* or treb* or tripl*) adj (blind$3 or mask$3)).tw. or Placebos/ or placebo*.tw. |
306831 |
|
8 |
Epidemiologic studies/ or case control studies/ or exp Cohort Analysis/ or Controlled Before-After Studies/ or Case control.tw. or cohort*.tw. or Cohort analy$.tw. or (Follow up adj (study or studies)).tw. or (observational adj (study or studies)).tw. or Longitudinal.tw. or Retrospective*.tw. or prospective*.tw. or consecutive*.tw. or Cross sectional.tw. or Cross-sectional studies/ or historically controlled study/ or interrupted time series analysis/ |
493363 |
|
9 |
5 and 6 - SR |
261 |
|
10 |
(5 and 7) not 9 - RCT |
175 |
|
11 |
(5 and 8) not (9 or 10) - observationeel |
277 |
|
12 |
9 or 10 or 11 |
713 |