Paracetamol
Uitgangsvraag
In hoeverre is een dosisaanpassing nodig bij het gebruik van paracetamol bij patiënten met chronisch risicovol alcoholgebruik en pijn?
Aanbeveling
Schrijf bij patiënten met chronisch risicovol alcoholgebruik paracetamol voor volgens de pijnladder. Geef, indien nodig, paracetamol in de gebruikelijke dosering.
Overwegingen
Voor- en nadelen van de interventie en de kwaliteit van het bewijs
Voor de literatuursamenvatting zijn vier RCT’s gevonden die het effect van een therapeutische dosering paracetamol (4 gram/dag) hebben onderzocht t.o.v. placebo op het risico op hepatotoxiciteit en pijn in patiënten met chronisch risicovol alcoholgebruik. Van deze studies includeerden drie RCT’s patiënten die vlak voor inclusie zijn gestopt met drinken (Dart, 2010; Kuffner, 2001; Kuffner, 2007). De laatste RCT includeerde patiënten met een matig alcoholgebruik (1-3 glazen per dag), waarbij de patiënten tijdens de studie hun alcoholgebruik continueerden (Heard, 2007). De geïncludeerde studies rapporteerden alleen over het risico op hepatotoxiciteit, middels serum ALT, AST en bilirubine waarden, alsmede de INR. Hierbij zijn met name de gemiddelde verschillen in concentraties van de biomarkers en de INR na follow-up gerapporteerd. Doordat dit geen informatie geeft over het percentage patiënten met een klinische relevante verhoging van de markers of de INR, kunnen de bewijskracht van de resultaten niet beoordeeld worden aan de hand van de GRADE-methodiek. Bij de studies die dit wel rapporteerden (Kuffner, 2001; Kuffner, 2007), is de GRADE-methodiek wel toegepast en is ervoor specifiek de groep patiënten die recent voor studieaanvang zijn gestopt met drinken, bewijs gevonden met een zeer lage bewijskracht voor de uitkomstmaat hepatotoxiciteit. Dit vanwege het risico op bias, indirectheid (door met name gemiddelde waardes te rapporteren) en zeer brede betrouwbaarheidsintervallen m.b.t. het aantal events van patiënten met een klinische relevante verhoging in biomarkers. Voor de uitkomstmaat pijnreductie is geen bewijs gevonden. Omdat de bewijskracht voor het percentage patiënten met een klinisch relevante verhoging van de markers (Kuffner 2001, Kuffner, 2007) zeer laag is, zijn de resultaten over gemiddelde stijging van ALAT, ASAT, bilirubine en INR uit de studies wel meegenomen in de overwegingen.
Hepatotoxiciteit van paracetamol uit zich in (sterk) verhoogde serum transaminases als gevolg van hepatocellulaire schade, wat in het ergste geval kan leiden tot leverfunctiestoornissen en leverfalen. Omdat ALAT meer in de lever voorkomt dan ASAT, is dit leverenzym een gevoeligere maat voor leverschade. In de beschreven studies is het ALAT ook het beste onderzocht als uitkomstmaat. Afwijkingen in de leverfunctieparameters (bilirubine, INR) treden pas op nadat er veel leverschade is (dus na een flinke stijging in de transaminases). Direct na stoppen van alcohol hebben patiënten bij gebruik van paracetamol theoretisch het hoogste risico op hepatotoxiciteit. CYP2E1 is dan mogelijk nog geïnduceerd terwijl alcohol niet meer aanwezig is om te competeren met paracetamol voor CYP2E1 binding. De inductie van CYP2E1 is al significant afgenomen op dag 3 na stoppen, en neemt nog enigszins verder af tot dag 8 na stoppen (Oneta, 2002). Geen van de geïncludeerde studies die keek naar recent abstinente patiënten onderzocht de patiënten langer dan een week. Echter, na dag 3 is de inductie al flink verminderd en alle studies onderzochten de leverenzymen de eerste 4 dagen en één studie nog tot en met dag 7. Naast de duur van de inductie heeft mogelijk de hoeveelheid en duur van het alcoholgebruik voorafgaand aan de studie effect op de mate van inductie. In vrijwel alle studies dronk het grootste deel van de patiënten al meer dan een maand. De hoeveelheid alcohol inname per dag stond niet gerapporteerd, echter alle studies bij recent abstinente patiënten vonden plaats in de setting van een verslavingskliniek dus wordt aangenomen dat de alcoholinname substantieel was.
Naast alcoholgebruik zijn er ook andere factoren mogelijk van invloed op het risico van hepatotoxiciteit bij gebruik van paracetamol. Bij patiënten die chronisch ondervoed zijn, kan een verminderde glutathionvoorraad er mogelijk voor zorgen dat de hepatotoxische metaboliet minder goed onschadelijk kan worden gemaakt (Caparrotta, 2017). In de geïncludeerde studies wordt BMI genoemd onder patiëntkarakteristieken en bleken met name patiënten met een normaal BMI geïncludeerd te zijn. De studie van Kuffner (2007) vond geen relatie tussen de BMI of gradaties van ondervoeding en verhogingen in ALAT. Het was echter onduidelijk hoeveel patiënten ondervoed waren. Kuffner (2007) vond ook geen relatie tussen plasma glutathion concentratie en ALAT, ASAT of INR.
Een andere mogelijk risicofactor voor hepatotoxiciteit is een al bestaande leverinsufficiëntie in de vorm van levercirrose. Bij patiënten met cirrose neemt de CYP2E1-activiteit echter niet toe, maar juist af vanwege het verminderd functioneren van de lever. In deze richtlijn is bewust gekozen deze patiëntengroep te excluderen vanwege de al bestaande adviezen op dit gebied (geneesmiddelenbijlevercirrose.nl).
Subgroepen
Recente alcohol onthouding
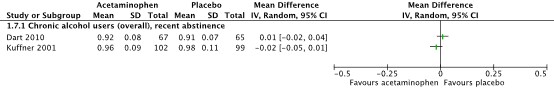
Er is in alle drie RCTs geen verschil gevonden tussen de controle- en interventiegroepen met betrekking tot de gemiddelde ALAT, bilirubine en INR-waardes op dag 2, 4, 6 en 7 bij gebruik van paracetamol (of placebo) in een dosering van 4 gram per dag gedurende enkele dagen. Gemiddelde ASAT-waardes waren op dag 2 en 4 ook vergelijkbaar. In één studie (Dart, 2010) werd echter op dag 6 en 7 een hogere ASAT-waarde t.o.v. controle gezien. Deze waarde was echter niet hoger dan de baseline ASAT-waarde in de interventiegroep en was klinisch niet relevant verhoogd.
Kuffner (2001) en Kuffner (2007) rapporteren het percentage van patiënten met stijgingen van leverenzymen (ALAT, ASAT, en bilirubine) t.o.v. ULN in beide groepen. Beide studies laten duidelijk zien dat stijgingen in de enzymen tot >3x ULN zich heel weinig voordeden. Daarnaast moet opgemerkt worden dat in deze laatste studie (Kuffner 2007), de verhoogde ALAT in de interventiegroep onder andere voorkwam op dag 0 (= baseline, voor gestart was met het gebruik van paracetamol). Deze stijging is dus niet toe te schrijven aan het gebruik van paracetamol.
Samenvattend laten de data zien dat het kortdurend gebruik van 4 gram paracetamol per dag bij recent abstinente chronische alcoholgebruikers geen klinisch relevant effect lijkt te hebben op leverenzymen en leverfunctieparameters. Er is dan ook geen sterke onderbouwing gevonden voor het aanpassen van de maximale dosering paracetamol bij deze patiëntengroep. In één studie werd er geen relatie gevonden tussen BMI en de mate van ondervoeding en een verhoging van ALAT-waarden (Kuffner, 2007). Het was echter niet duidelijk hoeveel patiënten in deze studie ondervoed waren, waardoor er over deze groep geen advies gegeven kan worden (zie kennisvraag).
Matig alcoholgebruik
In één studie (Heard, 2007) werden patiënten onderzocht die hun alcoholgebruik continueerden (1-3 consumpties per dag), waarbij theoretisch het risico op hepatotoxiciteit dus iets lager ligt dan in voorgaande groep omdat alcohol nog steeds competeert met paracetamol voor CYP2E1-binding. De studie van Heard liet zien dat de gemiddelde waarde van ALAT, ASAT en bilirubine niet verschilde tussen beide groepen op dag 4 van de behandeling met paracetamol. Op dag 11 van het onderzoek (1 dag na staken paracetamol) was het ALAT en ASAT gemiddeld hoger in de paracetamolgroep, echter dit was nog steeds marginaal en lager dan de ULN, dus wordt als niet klinisch relevant beoordeeld. Hoewel er mogelijk alsnog patiënten met een sterke verhoogde waarde zouden kunnen zijn, lijkt deze studie erop te wijzen dat gebruik van paracetamol in therapeutische dosering naast het drinken van 1-3 alcoholconsumpties niet voor hepatotoxiciteit zorgt. Omdat dit maar één studie is, is het bewijs zeer beperkt.
Waarden en voorkeuren van patiënten (en evt. hun verzorgers)
Patiënten zijn gebaat bij adequate pijnbehandeling met zo min mogelijk bijwerkingen. Paracetamol is hiervoor in de pijnladder bij alle patiënten de eerste keuze pijnstiller. Zoals in deze richtlijn onderbouwd, zijn er bij het gebruik van de therapeutische dosering paracetamol geen directe aanwijzingen voor een hoger risico op hepatotoxiciteit bij patiënten met chronisch risicovol alcoholgebruik. Het geven van een lagere dosering lijkt dan ook niet direct noodzakelijk.
Kosten (middelenbeslag)
De kosten van paracetamol zijn dermate laag dat dit geen rol speelt in de besluitvorming. Daarnaast is het geen extra middel dat bij andere patiëntgroepen niet gegeven wordt.
Aanvaardbaarheid, haalbaarheid en implementatie
Patiënten met chronisch risicovol alcoholgebruik een dosering van vier gram paracetamol per dag voorschrijven lijkt goed aanvaardbaar en haalbaar aangezien dit de gebruikelijke dosering is en er dus standaard zorg kan worden verleend.
In deze richtlijn is niet onderzocht of dit doseeradvies ook kan worden gehandhaafd bij de specifieke subgroep van patiënten met cirrose. Voor de laatste subgroep verwijst de werkgroep naar Geneesmiddelen bij levercirrose.
Gelet op bovenstaande overwegingen adviseert de werkgroep om paracetamol voor te schrijven volgens de pijnladder van de WHO. Hierbij dienen de besproken (mogelijke) risico’s in acht te worden genomen.
Rationale van de aanbeveling: weging van argumenten voor en tegen de interventies
De GRADE conclusie voor het effect van paracetamol op een stijging >3x ULN van transaminases of >ULN van leverfunctieparameters in de gevonden literatuur is zeer laag. De overige resultaten in de literatuur lieten echter geen aanwijzingen zien voor een klinisch relevante stijging van ALAT, ASAT, bilirubine of INR zien. Op basis daarvan ziet de werkgroep geen reden om een toxisch effect van een gebruikelijke dosering paracetamol te verwachten bij patiënten met chronisch alcoholgebruik.
Onderbouwing
Achtergrond
There is uncertainty about the use and possible dose adjustments of pain medication in patients with chronic alcohol use. Alcohol induces the enzyme CYP2E1 which potentially increases the amount of paracetamol metabolized into its hepatotoxic metabolite (NAPQI) and subsequently the risk of liver damage. Alcohol itself also binds to CYP2E1 and theoretically, during active alcohol use, alcohol and paracetamol compete for binding sites and the risk is of hepatotoxicity is not much higher. However, in the 5-8 days after discontinuation of alcohol, paracetamol is the only substrate of CYP2E1 and risk of hepatotoxicity may be increased. Because of this interaction based on old literature, it has always been recommended to use a maximum of 2 grams of paracetamol per day in patients with chronic alcohol consumption. This is often not performed in practice and it is therefore questioned whether there is literature that supports the need for a dose adjustment.
Conclusies / Summary of Findings
Hepatoxicity (recent alcohol abstinence)
|
Very low GRADE
No GRADE |
The evidence is very uncertain about the effect of a therapeutic dose of paracetamol on serum ALT, AST or total bilirubin levels compared with another dose or placebo in patients with chronic alcohol use (recent alcohol abstinence) and pain (without cirrhosis).
No GRADE conclusion could be drawn about the effect of a therapeutic dose of paracetamol on INR compared with another dose or placebo in patients with chronic alcohol use (recent alcohol abstinence) and pain (without cirrhosis).
Source: Kuffner (2001), Kuffner (2007) |
Hepatoxicity (moderate, continuous alcohol use)
|
No GRADE |
No GRADE conclusion could be drawn about the effect of a therapeutic dose of paracetamol on serum ALT, AST, total bilirubin or INR compared with another dose or placebo in patients with chronic alcohol use (moderate, continuous alcohol use) and pain (without cirrose).
Source: - |
Pain reduction
|
No GRADE |
No evidence was found regarding the effect of a therapeutic dose of paracetamol on pain reduction compared with another dose or placebo in patients with chronic alcohol use and pain (without cirrhosis).
Source: - |
Samenvatting literatuur
Description of studies
Dart (2010) performed a double-blind, randomized controlled trial to determine the effect of paracetamol on serum liver tests in newly abstinent patients with alcohol abuse. A total of 142 adult alcohol-abusing patients who entered the alcohol detoxification center were included in the study, of which 74 were randomized into the intervention group and received 4 grams of paracetamol per day for five days. The 68 patients in the control group received a placebo. The study groups were similar regarding mean age (I: 45.7 years ± 9.4, C: 46.8 years ± 8.3) and proportion of males (I: 91%, C: 94%) and BMI (I: 25.0 ± 4.1, C: 24.1 ± 4.0) . The duration of the current drinking episode was similar among the study groups, with 67% of the patients having a duration of ≥1 month in both study groups. Follow-up time was seven days after start of treatment. Study outcomes of interest include serum alanine aminotransferase (ALT), aspartate aminotransferase (AST) and total bilirubin levels and the international normalized ratio (INR). The study was limited due to a relatively high rate of withdrawal and the use of a per-protocol analysis.
Heard (2007) performed a double-blind, randomized controlled trial to determine the effect of ten days of the maximal therapeutic dose of paracetamol on ALT activity in patients who consume one to three alcohol beverages per day. A total of 156 healthy adults with an average daily consumption of one to three alcohol beverages were included in the study, of which 104 were randomized into the intervention group and received 4 grams of paracetamol per day for ten days. The participants in the control group received a placebo. All participants were instructed to continue with their usual alcohol consumption pattern. The study groups were similar regarding mean age (I: 32.6 years ± 9.8, C: 34.0 years ± 13.4) and proportion of males (I: 52%, C: 58%). The average number of daily drinks during the study was 2.08 ± 0.73 days in the paracetamol group, compared to 2.24 ± 0.92 in the placebo group. Mean BMI for both study groups was not reported (only categories), although it was reported that only one patient in the paracetamol group was underweight. Follow-up time was eleven days after start of treatment. Study outcomes of interest include serum ALT, AST and total bilirubin levels and the INR. The overall risk of bias was low.
Kuffner (2001) performed a double-blind, randomized controlled trial to determine if hepatic injury is associated with the maximal therapeutic dosing of paracetamol in patients with chronic alcohol abuse immediately after cessation of alcohol intake. A total of 230 adult patients who entered the alcohol detoxification facility were included in the study, of which 118 patients were randomized into the intervention group and received 4 grams of paracetamol per day for two days (after abstinence of alcohol use). The 112 patients in the control group received a placebo. The study groups were similar regarding mean age (I: 43.9 years ± 8.6, C: 45.0 years ± 8.2), proportion of males (I: 89%, C: 90%) and BMI (I: 23.6 ± 4.6, C: 23.4 ± 3.5). The duration of the current drinking episode was similar among the study groups, with over 40% of the patients having a duration of ≥1 month (I: 42%, C: 44%). Follow-up time was four days after start of treatment. Study outcomes of interest included serum ALT and AST levels and the international normalized ratio (INR). The study was limited due to a relatively high rate of withdrawal and the use of a per-protocol analysis.
Kuffner (2007) performed a double-blind, randomized controlled trial to determine the effect of a 3-day course of paracetamol on the serum ALT, AST and bilirubin levels, INR, as well as the effect on glutathione plasma levels. A total of 443 adult alcoholic patients who entered the alcohol detoxification center were included in the study, of which 308 were randomized into the intervention group and received 4 grams of paracetamol per day for three days (after abstinence of alcohol use). The 135 patients in the control group received a placebo. The study groups were similar regarding mean age (I: 43 years ± 9, C: 44 years ± 8), proportion of males (I: 92%, C: 95%) and BMI (I: 25.0 ± 5, C: 25 ± 4). The duration of the current drinking episode was similar among the study groups, with ≥50% of the patients having a duration of ≥1 month (I: 50%, C: 56%). Follow-up time was four days after start of treatment. Study outcomes of interest include serum ALT levels. The study was limited by incomplete data on serum AST and total bilirubin levels and INR (see Evidence tables), a relatively high rate of withdrawal and the use of a per-protocol analysis.
Results
Hepatotoxicity
• Alanine aminotransferase (ALT)
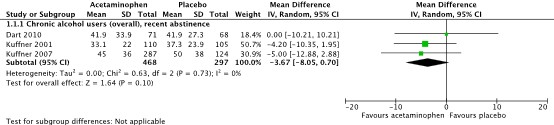
All included studies reported on serum ALT levels in international units (IU) per liter (Dart, 2010; Heard, 2007; Kuffner, 2001; Kuffner, 2007). All but the study of Heard (2007) reported results of serum ALT levels on day 2 after start of treatment. Pooled results, all on chronic alcohol users with recent abstinence, show a mean difference (MD, 95%CI) of -3.67 IU/L (-8.05 to 0.70), see Figure 1.
Figure 1. Serum alanine aminotransferase (ALT) levels (IU/L) on day 2 after start of treatment.
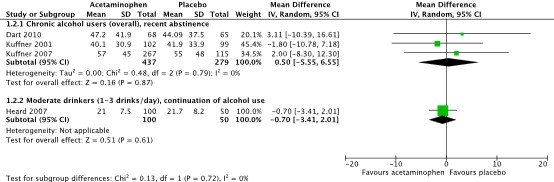
All included studies also reported results of serum ALT levels on day 4 after start of treatment (Dart, 2010; Heard, 2007; Kuffner, 2001; Kuffner, 2007). Pooled results for the group of chronic alcohol users with recent abstinence show a MD (95%CI) of 0.50 IU/L (-5.55 to 6.55), see Figure 2. For the group moderate drinkers with continuation of alcohol use (Heard, 2007), results show a MD (95%CI) of -0.70 IU/L (-3.41 to 2.01).
Figure 2. Serum alanine aminotransferase (ALT) levels (IU/L) on day 4 after start of treatment.
Two of the included studies also reported on results of serum ALT levels at other timepoints (Dart, 2010; Heard, 2007). Dart (2010) reported results of respectively one and two days after the treatment was stopped (day 6 and day 7). Heard (2007) reported results of one day after the treatment was stopped (day 11). Table 1 provides an overview of these results.
Table 2. Serum alanine aminotransferase (ALT) levels (IU/L) at day 6, 7 and 11 after start of treatment.
|
Study |
Day 6 Mean serum ALT level (IU/L) |
Day 7 ‘’ |
Day 11 ‘’ |
|
Chronic alcohol users (overall), recent abstinence |
|||
|
Dart, 2010 |
I: 55.8 (SD 44.8) C: 49.3 (42.1) |
I: 61.8 (50.4) C: 49.2 (42.4) |
|
|
Moderate drinkers (1-3 drinks/day), continuation of alcohol use |
|||
|
Heard, 2007 |
|
|
I: 30.0 (19.6) C: 21.6 (7.9) |
|
MD (95%CI) |
6.50 (-8.44 to 21.44) |
12.60 (-3.39 to 28.59) |
8.40 (3.98 to 12.82) |
Kuffner (2001) also reported on the percentage of patients with a serum ALT or AST of >120 IU/L at any timepoint during the study. This occurred in 4/102 (3.9%) patients in the paracetamol group, compared to 5/99 (5.1%) patients in the placebo group. The corresponding risk ratio (RR) is 0.78 (95%CI 0.21 to 2.81) in favor of the paracetamol group, which is clinically relevant. Next, Kuffner (2001) also reported on the percentage of patients with a serum ALT or AST of >1000 IU/L or an INR >1.5 at any timepoint during the study. This occurred in 0/102 (0%) patients in the paracetamol group, compared to 1/99 (1%) patients in the placebo group. The corresponding risk ratio (RR) is 0.32 (95%CI 0.01 to 7.85), which is clinically relevant.
Kuffner (2007) reported on the percentage of patients with a serum ALT of >3x ULN at any timepoint during the study. This occurred in 24/308 (7.8%) patients in the paracetamol group, compared to 8/135 (5.9%) patients in the placebo group. The corresponding risk ratio (RR) is 1.31 (95%CI 0.61 to 2.85) in favor of the placebo group, which is clinically relevant. However, in the paracetamol group, the percentage of patients with a serum ALT of >3x ULN was mainly increased already at baseline and day 5. Therefore, the increase in ALT levels could not be considered as effect of paracetamol.
• Aspartate aminotransferase (AST)
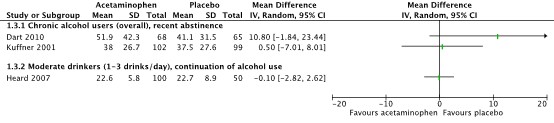
Three of the included studies reported on serum AST levels in international units (IU) per liter (Dart, 2010; Heard, 2007; Kuffner, 2001). Kuffner (2007) did not report full data on serum AST levels. The results were ‘highly concordant’ with data for serum ALT levels. Two of the included studies reported results of serum AST levels on day 2 after start of treatment, see Figure 3.
Figure 3. Serum aspartate aminotransferase (AST) levels (IU/L) on day 2 after start of treatment.
All studies reported results of serum AST levels on day 4 after start of treatment, see Figure 4.
Figure 4. Serum aspartate aminotransferase (AST) levels (IU/L) on day 4 after start of treatment.
Two of the included studies also reported on results of serum AST levels at timepoints after treatment period (Dart, 2010; Heard, 2007). Dart (2010) reported results of respectively one and two days after the treatment was stopped (day 6 and day 7). Heard (2007) reported results of one day after the treatment was stopped (day 11). Table 2 provides an overview of these results.
Table 3. Serum aspartate aminotransferase (AST) levels (IU/L) at day 6, 7 and 11 after start of treatment.
|
Study |
Day 6 Mean serum AST level (IU/L) |
Day 7 ‘’ |
Day 11 ‘’ |
|
Chronic alcohol users (overall), recent abstinence |
|||
|
Dart, 2010 |
I: 60.0 (SD 42.5) C: 38.9 (28.3) |
I: 51.5 (37.3) C: 36.5 (25.9) |
|
|
Moderate drinkers (1-3 drinks/day), continuation of alcohol use |
|||
|
Heard, 2007 |
|
|
I: 28.5 (24.7) C: 21.8 (5.3) |
|
MD (95%CI) |
21.10 (8.72 to 33.48) |
15.00 (3.95 to 26.05) |
6.70 (1.64 to 11.76) |
• Total bilirubin
Two of the included studies reported on serum total bilirubin levels in mmol/L (Dart, 2010; Heard, 2007). Dart reported on results of serum total bilirubin levels on day 2 after start of treatment. In the paracetamol group, the mean serum total bilirubin level (SD) was 16.55 (10.88) compared to 13.94 (6.74) in the placebo group. The corresponding MD (95%CI) is 2.61 (-0.37 to 5.59).
Both studies reported results of serum total bilirubin levels on day 4 after start of treatment, see Figure 5.
Figure 5. Serum total bilirubin levels (μmol/L) on day 4 after start of treatment.
Both Dart (2010) and Heard (2007) also reported on results of serum total bilirubin levels at timepoints after treatment period. Dart (2010) reported results of respectively one and two days after the treatment was stopped (day 6 and day 7). Heard (2007) reported results of one day after the treatment was stopped (day 11). Table 3 provides an overview of these results.
Table 3. Serum total bilirubin) levels (mmol/L) at day 6, 7 and 11 after start of treatment.
|
Study |
Day 6 Mean serum bilirubin level (mmol/L) |
Day 7 ‘’ |
Day 11 ‘’ |
|
Chronic alcohol users (overall), recent abstinence |
|||
|
Dart, 2010 |
I: 8.29 (3.93) C: 8.59 (4.51) |
I: 9.05 (4.29) C: 8.62 (3.67) |
|
|
Moderate drinkers (1-3 drinks/day), continuation of alcohol use |
|||
|
Heard, 2007 |
|
|
I: 10.60 (7.52) C: 10.60 (7.52) |
|
MD (95%CI) |
-0.30 (-1.76 to 1.16) |
0.43 (-0.94 to 1.80) |
0.00 (-2.55 to 2.55) |
Kuffner (2007) also reported on the percentage of patients with bilirubin level of >ULN at any timepoint during the study. This occurred in 20/308 (6.4%) patients in the paracetamol group, compared to 6/135 (4.4%) patients in the control group. However, as in the same study no clinically relevant increase in ALT or AST levels were seen, these results will not be further considered.
• International normalized ratio (INR)
Two of the included studies reported on the INR (Dart, 2010; Kuffner, 2001). In the study by Kuffner (2007), full data on INR was not reported. ‘No significant change’ in results between the paracetamol and placebo group were seen in this study.
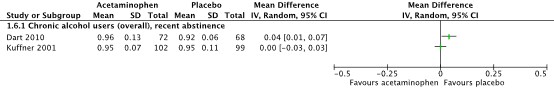
Both studies reported results of INR on day 2 after start of treatment, see Figure 6.
Figure 6. International normalized ratio (INR) on day 2 after start of treatment.
Both studies reported results of INR on day 4 after start of treatment, see Figure 7.
Figure 7. International normalized ratio (INR) on day 4 after start of the intervention.
Dart (2010) also reported results on INR of respectively one and two days after the treatment was stopped (day 6 and day 7). Table 4 provides an overview of these results.
Table 4. International normalized ratio (INR) at day 6 and 7 after start of treatment.
|
Study |
Day 6 INR |
Day 7 ‘’ |
|
Dart, 2010 |
I: 0.93 (0.09) C: 0.93 (0.07) |
I: 0.92 (0.06) C: 0.93 (0.08) |
|
MD (95%CI) |
0.00 (-0.03 to 0.03 |
-0.01 (-0.03 to 0.01) |
Pain reduction
None of the included studies reported on the outcome measure pain reduction.
Level of evidence of the literature
The level of evidence came from RCTs and therefore started as high. For the conclusions, the level of evidence was graded separately for the group of chronic alcohol users with recent abstinence compared to the group of moderate users with continuation of alcohol use.
Hepatotoxicity (recent alcohol abstinence)
As only mean differences in the serum levels of ALT/AST, total levels of bilirubin and the INR were reported and no percentage of patients with a clinically relevant event, no GRADE conclusion could be drawn for these results. The GRADE conclusion for the outcome measures serum ALT, AST and total bilirubin was based on the results by Kuffner (2001) who did report on those outcome measures. The results of Kuffner (2007) on percentage of patients with >3x ULN for ALT are also not further considered for the GRADE conclusion, as the increase in ALT levels were already seen at baseline.
The level of evidence regarding the outcome measures ALT and AST was downgraded by 3 levels to very low because of study limitations (relatively high loss-to-follow-up in study groups, -1 level for risk of bias), applicability (results are not reported for percentage of patients with serum ALT of >2x ULN, -1 level for indirectness) and number of included patients (confidence interval of the effect estimate crosses both upper and lower boundary for clinical relevance, -2 levels for imprecision).
The level of evidence regarding the outcome measure bilirubin was downgraded by 3 levels to very low because of study limitations (relatively high loss-to-follow-up in study groups, -1 level for risk of bias) and number of included patients (confidence interval of the effect estimate crosses both upper and lower boundary for clinical relevance, -2 levels for imprecision).
Hepatoxicity (moderate continuous alcohol use)
As only mean differences in the serum levels of ALAT/ASAT, total levels of bilirubin and the INR were reported and no percentage of patients with a clinically relevant event, no GRADE conclusion could be drawn.
Pain reduction
No evidence was found on the effect of a therapeutic dose of paracetamol on the outcome measure pain reduction, compared with another dose or placebo in patients with chronic alcohol abuse and pain, without cirrhosis. Therefore, the level of evidence could not be assessed.
Zoeken en selecteren
A systematic review of the literature was performed to answer the following question:
What is the effect of a dose adjustment of paracetamol regarding the risk of hepatoxicity and pain reduction in adult patients with chronic alcohol use and pain (without cirrhosis)?
P (Patients): adult patients with chronic alcohol use and pain, without cirrhosis
I (Intervention): paracetamol (acetaminophen) in therapeutic dose
C (Comparison): other dose of paracetamol (acetaminophen) or placebo
O (Outcomes): risk of hepatotoxicity, pain reduction
Relevant outcome measures
The guideline development group considered risk of hepatotoxicity as a critical outcome measure for decision making; and pain reduction as an important outcome measure for decision making.
Regarding the outcome measure risk of hepatoxicity, the working group used the following definitions: serum alanine aminotransferase (ALT), serum aspartate aminotransferase (AST), total bilirubin and international normalized ratio (INR). Regarding the outcome measure pain reduction, the working groups used the definitions as stated in the studies.
Furthermore, a follow-up period of ≤4 days was considered as short-term follow-up and >4 days was considered as long-term follow-up.
The working group defined the following minimal clinically important differences for continuous outcomes:
- ALT: 3x ULN
- AST: 3x ULN
- Bilirubin: above ULN
- INR: above ULN
The above boundaries for clinical relevance were chosen, as liver damage may occur above the chosen levels. Regarding clinical relevance, the working group states that total bilirubin and INR should only be considered in case results for ALT and AST levels are clinically relevant.
For dichotomous outcomes, the working group defined a 10% difference (RR < 0.91 or > 1.10) as a minimal clinically (patient) important difference for all outcomes.
Search and select (Methods)
The databases Medline (via OVID) and Embase (via Embase.com) were searched with relevant search terms from 2000 until 12 November 2023. The detailed search strategy is depicted under the tab Methods. The systematic literature search resulted in 551 hits. Studies were selected based on the following criteria systematic reviews and RCTs according to the PICO. A total of eight studies were initially selected based on title and abstract screening. After reading the full text, four studies were excluded (see the table with reasons for exclusion under the tab Methods), and four studies were included.
Results
Four studies were included in the analysis of the literature. Important study characteristics and results are summarized in the evidence tables. The assessment of the risk of bias is summarized in the risk of bias tables.
Referenties
- Caparrotta TM, Antoine DJ, Dear JW. Are some people at increased risk of paracetamol-induced liver injury? A critical review of the literature. Eur J Clin Pharmacol. 2018 Feb;74(2):147-160. doi: 10.1007/s00228-017-2356-6. Epub 2017 Oct 24. PMID: 29067481; PMCID: PMC5765191.
- Dart RC, Green JL, Kuffner EK, Heard K, Sproule B, Brands B. The effects of paracetamol () on hepatic tests in patients who chronically abuse alcohol - a randomized study. Aliment Pharmacol Ther. 2010 Aug;32(3):478-86. doi: 10.1111/j.1365-2036.2010.04364.x. Epub 2010 May 20. PMID: 20491750.
- Heard K, Green JL, Bailey JE, Bogdan GM, Dart RC. A randomized trial to determine the change in alanine aminotransferase during 10 days of paracetamol (acetaminophen) administration in subjects who consume moderate amounts of alcohol. Aliment Pharmacol Ther. 2007 Jul 15;26(2):283-90. doi: 10.1111/j.1365-2036.2007.03368.x. PMID: 17593074.
- Kuffner EK, Dart RC, Bogdan GM, Hill RE, Casper E, Darton L. Effect of maximal daily doses of acetaminophen on the liver of alcoholic patients: a randomized, double-blind, placebo-controlled trial. Arch Intern Med. 2001 Oct 8;161(18):2247-52. doi: 10.1001/archinte.161.18.2247. PMID: 11575982.
- Kuffner EK, Green JL, Bogdan GM, Knox PC, Palmer RB, Heard K, Slattery JT, Dart RC. The effect of acetaminophen (four grams a day for three consecutive days) on hepatic tests in alcoholic patients--a multicenter randomized study. BMC Med. 2007 May 30;5:13. doi: 10.1186/1741-7015-5-13. PMID: 17537264; PMCID: PMC1894983.
- Oneta CM, Lieber CS, Li J, Rüttimann S, Schmid B, Lattmann J, Rosman AS, Seitz HK. Dynamics of cytochrome P4502E1 activity in man: induction by ethanol and disappearance during withdrawal phase. J Hepatol. 2002 Jan;36(1):47-52. doi: 10.1016/s0168-8278(01)00223-9. PMID: 11804663.
Evidence tabellen
Evidence table for intervention studies (randomized controlled trials and non-randomized observational studies [cohort studies, case-control studies, case series])
|
Study reference |
Study characteristics |
Patient characteristics |
Intervention (I) |
Comparison / control (C)
|
Follow-up |
Outcome measures and effect size |
Comments |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Dart, 2010 |
Type of study: RCT, double-blind
Setting and country: Alcohol detoxification centre, United States
Funding and conflicts of interest: Non-commercial funding and no potential conflicts of interest. |
Inclusion criteria: Adult alcohol-abusing patients. History of positive alcohol screen by breath analyser at admission and duration of current drinking episode for ≥ seven days.
Exclusion criteria: Paracetamol overuse as indication by baseline serum level > 132.4 mmol/L, history of ingesting > 4g/day in any of the four preceding days prior to study enrolment, baseline serum ALT or AST level >200 IU ⁄ L, baseline INR > 1.5, positive urine pregnancy test
N total at baseline: Intervention: 74 Control: 68
Important prognostic factors2: Mean age ± SD: I: 45.7 ± 9.4 C: 46.8 ± 8.3
Sex: I: 67 (91%) males C: 64 (94%) males
Body mass index (mean ± SD) I: 25.0 ± 4.1 C: 24.1 ± 4.0
Drinking behaviour Duration of current drinking episode (% of study population) I: 1-2 weeks: 12% 2-4 weeks: 20% 1-6 months: 30% >6 months: 37% C: 1-2 weeks: 16% 2-4 weeks: 18% 1-6 months: 27% >6 months: 40%
Groups comparable at baseline. |
Acetaminophen (paracetamol) 4 g/day for five days
|
Placebo |
Length of follow-up: Seven days
Loss-to-follow-up: Intervention: 8/74 (11%) at day 7 Reasons: withdrawal (4), unrelated adverse events (3), protocol violation (1) Control: 4/68 (6%) ‘’ Reasons: withdrawal (4)
Incomplete outcome data: See loss-to-follow-up
|
Alanine aminotransferase (ALT) Mean (SD) in IU/L
Aspartate aminotransferase (AST) Mean (SD) in IU/L
Total bilirubin Mean (SD) in mmol/L
International normalized ratio (INR) Mean (SD)
|
Authors report that the data do not support the conclusion that paracetamol can cause severe liver injury in alcohol-abusing patients.
Per-protocol analysis. Difference in loss-to-follow-up is not due to treatment-related events, according to authors. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Heard, 2007 |
Type of study: RCT, double-blind
Setting and country: Health care centre, United States
Funding and conflicts of interest: Non-commercial funding and no potential conflicts of interest. |
Inclusion criteria: Healthy adults with a history of moderate alcohol consumption (in preceding two months), including at least one alcoholic beverage in the 48 hours prior to enrolment (to demonstrate continuous use).
Exclusion criteria: Adults who responded to be alcoholic, average consumption of >3 drinks/day, paracetamol overuse as indication by baseline serum level > 132.4 mmol/L, history of ingesting > 4g/day in any of the four preceding days prior to study enrolment, baseline serum ALT or AST level >50 IU ⁄ L, clinical intoxication, psychiatric impairment or inability to give informed consent, hypersensitivity to paracetamol, pregnancy test, enrolment in another trial in preceding 3 months.
N total at baseline: Intervention: 104 Control: 52
Important prognostic factors2: Mean age ± SD: I: 32.6 (9.8) C: 34 (13.4)
Sex: I: 58% males C: 52% males
Body mass index (mean ± SD) Mean BMI not reported. One patient in the intervention group underweight.
Drinking behaviour Daily drinks during study period (mean ± SD) I: 2.08 ± 0.73 C: 2.24 ± 0.92
Groups comparable at baseline. |
Acetaminophen (paracetamol) 4 g/day for ten days
|
Placebo |
Length of follow-up: Eleven days
Loss-to-follow-up: Intervention: 4/104 (4%) at day 11 Reasons: missed study medications (1), adverse event (3) Control: 2/52 (4%) ‘’ Reasons: withdrawal request (1), adverse event (1)
|
Alanine aminotransferase (ALT) Mean (SD) in IU/L
Aspartate aminotransferase (AST) Mean (SD) in IU/L
Total bilirubin Mean (SD) in mmol/L
|
Authors conclude paracetamol may lead to mild elevation of serum ALT levels, but there is no evidence of liver injury. Per-protocol analysis.
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Kuffner, 2001 |
Type of study: RCT, double-blind
Setting and country: Alcohol detoxification centre, United States
Funding and conflicts of interest: Non-commercial funding. Potential conflicts of interest not reported. |
Inclusion criteria: Adult patients entering alcohol detoxification facility.
Exclusion criteria: Baseline serum acetaminophen level of > 20 mg/L, history of allergy to acetaminophen, history of ingesting > 4g/day in any of the four preceding days prior to study enrolment, baseline serum ALT or AST level >120 U/L, baseline INR > 1.5, positive urine pregnancy test, enrolment in other trial within previous 3 months, clinical alcohol intoxication at time of first dose of study medication
N total at baseline: Intervention: 118 Control: 112
Important prognostic factors2: Mean age ± SD: I: 43.9 ± 8.6 C: 45.0 ± 8.2
Sex: I: 91 (89%) males C: 89 (90%) males
Body mass index (mean ± SD) I: 23.6 ± 4.6 C: 23.4 ± 3.5
Drinking behaviour Duration of current drinking episode (% of study population) I: <1 week: 24% 1-4 weeks: 34% 1-6 months: 17% >6 months: 25% C: <1 week: 24% 1-4 weeks: 27% 1-6 months: 20% >6 months: 24%
Groups comparable at baseline. |
Acetaminophen (paracetamol) 4 g/day for two days
|
Placebo |
Length of follow-up: Four days
Loss-to-follow-up: Intervention: 16/118 (14%) at day 4 Reasons: withdrawal (16) Control: 13/112 (12%) ‘’ Reasons: withdrawal (13)
Incomplete outcome data: See loss-to-follow-up
|
Alanine aminotransferase (ALT) Mean (SD) in IU/L
Aspartate aminotransferase (AST) Mean (SD) in IU/L
Percentage of patients with serum ALT/AST of >120 IU/L at any timepoint Intervention: 4/102 (3.9%) Control: 5/99 (5.1%)
Percentage of patients with serum ALT/AST of >1000 IU/L or INR >1.5 at any timepoint Intervention: 0/102 (0%) Control: 1/99 (1%)
International normalized ratio (INR) Mean (SD)
|
Authors conclude that no evidence was found regarding liver injury when using the maximal therapeutic dose of paracetamol.
Per-protocol analysis. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Kuffner, 2007 |
Type of study: RCT, double-blind
Setting and country: Alcohol detoxification centre, United States
Funding and conflicts of interest: Non-commercial funding. Potential conflicts of interest not reported. |
Inclusion criteria: Adult patients entering alcohol detoxification facility.
Exclusion criteria: Baseline serum acetaminophen level of > 20 mg/L, history of allergy to acetaminophen, history of ingesting > 4g/day in any of the four preceding days prior to study enrolment, baseline serum ALT or AST level >120 U/L, baseline INR > 1.5, positive urine pregnancy test, enrolment in other trial within previous 3 months, clinical alcohol intoxication at time of first dose of study medication
N total at baseline: Intervention: 308 Control: 135
Important prognostic factors2: Mean age ± SD: I: 43 ± 9 C: 44 ± 8
Sex: I: 284 (92%) males C: 128 (95%) males
Body mass index (mean ± SD) I: 25 ± 5 C: 25 ± 4
Drinking behaviour Duration of current drinking episode (% of study population) I: <1 week: 18% 1-4 weeks: 32% >1 month: 50% C: <1 week: 16% 1-4 weeks: 27% >1 month: 56%
Groups comparable at baseline. |
Acetaminophen (paracetamol) 4 g/day for three days
|
Placebo |
Length of follow-up: Four days
Loss-to-follow-up: Intervention: 50/308 (16%) at day 5 Reasons: withdrawal (48), adverse event (2) Control: 21/135 (16%) ‘’ Reasons: withdrawal (18), adverse event (3)
Incomplete outcome data: See loss-to-follow-up
|
Alanine aminotransferase (ALT) Mean (SD) in IU/L
Percentage of patients with serum ALT of >3x ULN at any timepoint Intervention: 24/308 (7.8%) Control: 8/135 (5.9%)
Aspartate aminotransferase (AST) Full data not reported. ‘Highly concordant’ with ALT levels.
Total bilirubin Full data not reported. ‘No significant change’.
Percentage of patients with serum bilirubin of >ULN at any timepoint Intervention: 20/308 (6.4%) Control: 6/135 (4.4%)
International normalized ratio (INR) Full data not reported. ‘No significant change’.
Although no outcome measure for current literature research, plasma glutathione levels (mechanism for liver hepatotoxicity) increased from 2.17 ± 0.97 to 2.27 ± 0.85 μM in the acetaminophen group and from 1.90 ± 0.68 to 2.02 ± 0.74 μM in the control group.
|
Authors report that use of maximal therapeutic dose of paracetamol is safe and does not cause liver injury in alcoholics.
Only data for ALT is reported. Data for AST is ‘highly concordant’ with data of ALT and is therefore not reported. For mean INR, it is only reported that no significant change was seen. Per-protocol analysis. |
Risk of bias table for intervention studies (randomized controlled trials; based on Cochrane risk of bias tool and suggestions by the CLARITY Group at McMaster University)
|
Study reference
(first author, publication year) |
Was the allocation sequence adequately generated?
Definitely yes Probably yes Probably no Definitely no |
Was the allocation adequately concealed?
Definitely yes Probably yes Probably no Definitely no |
Blinding: Was knowledge of the allocated interventions adequately prevented?
Were patients blinded?
Were healthcare providers blinded?
Were data collectors blinded?
Were outcome assessors blinded?
Were data analysts blinded?
Definitely yes Probably yes Probably no Definitely no |
Was loss to follow-up (missing outcome data) infrequent?
Definitely yes Probably yes Probably no Definitely no |
Are reports of the study free of selective outcome reporting?
Definitely yes Probably yes Probably no Definitely no |
Was the study apparently free of other problems that could put it at a risk of bias?
Definitely yes Probably yes Probably no Definitely no |
Overall risk of bias If applicable/necessary, per outcome measure
LOW Some concerns HIGH
|
|
Dart, 2010 |
Definitely yes;
Reason: Computer generated block randomization |
Definitely yes;
Reason: Study packets and medication were prepared and number by personnel blinded to allocation. |
Definitely yes;
Reason: Double-blind study design. Patients and personnel blinded. |
Probably yes
Reason: Loss-to-follow-up was more frequent in intervention group compared to control group (11% vs 6%), although clearly stated that the single serious adverse event in intervention group (pancytopenia, end stage liver disease and psoriasis) was due to previous disease. Use of per-protocol analysis. |
Definitely yes
Reason: All relevant outcomes were reported; |
Definitely yes;
Reason: No other problems noted |
Some concerns (all outcomes)
Reason: Percentage of requested withdrawals relatively high in intervention group |
|
Heard, 2007 |
Definitely yes;
Reason: Computer generated block randomization |
Definitely yes;
Reason: Investigators enrolling and evaluating the subjects were unaware of treatment group. |
Definitely yes;
Reason: Double-blind study design. Patients and personnel blinded |
Definitely yes;
Reason: Loss-to-follow-up infrequent (4%) in both study groups. Use of per protocol analysis. |
Definitely yes
Reason: All relevant outcomes were reported; |
Definitely yes;
Reason: No other problems noted |
LOW |
|
Kuffner, 2001 |
Definitely yes;
Reason: Computer generated randomization |
Definitely yes;
Reason: Personnel was blinded for randomization sequence, only known by investigator not involved in patient care |
Definitely yes;
Reason: Double-blind study design. Patients and personnel blinded. |
Probably no
Reason: Loss-to-follow-up was 14% and 12% in resp. intervention and control group. in both groups, all due to withdrawal requested by patient. Use of per-protocol analysis. |
Definitely yes;
Reason: No other problems noted |
Definitely yes;
Reason: No other problems noted |
Some concerns (all outcomes)
Reason: Percentage of requested withdrawal relatively high in both groups, use of per-protocol analysis. |
|
Kuffner, 2007 |
Definitely yes;
Reason: Computer generated randomization |
Definitely yes;
Reason: Personnel was blinded for randomization sequence, only known by investigator not involved in patient care |
Definitely yes;
Reason: Double-blind study design. Patients and personnel blinded. |
Probably no
Reason: Loss-to-follow-up was 16% in both groups, almost exclusively due to withdrawal requested by patient. Use of per-protocol analysis. |
Definitely no
Reason: Follow-up data of AST levels and INR not reported, due to ‘concordance with ALT levels’ (AST) or ‘no significant change’ (INR). |
Definitely yes;
Reason: No other problems noted |
Some concerns (all outcomes)
Reason: Follow-up data for several outcomes (AST, INR) not reported. Percentage of requested withdrawal relatively high in both groups |
Table of excluded studies
|
Reference |
Reason for exclusion |
|
Rumack B, Heard K, Green J, Albert D, Bucher-Bartelson B, Bodmer M, Sivilotti ML, Dart RC. Effect of therapeutic doses of acetaminophen (up to 4 g/day) on serum alanine aminotransferase levels in subjects consuming ethanol: systematic review and meta-analysis of randomized controlled trials. Pharmacotherapy. 2012 Sep;32(9):784-91. doi: 10.1002/j.1875-9114.2012.01122.x. Epub 2012 Jul 31. PMID: 22851428; PMCID: PMC3561770. |
Low quality review, without quality assessment and full search strategy. |
|
Bartels S, Sivilotti M, Crosby D, Richard J. Are recommended doses of acetaminophen hepatotoxic for recently abstinent alcoholics? A randomized trial. Clin Toxicol (Phila). 2008 Mar;46(3):243-9. doi: 10.1080/15563650701447020. PMID: 18344107. |
Outcome data only reported in graphs, exact data not retractable. |
|
Saseen JJ. Does acetaminophen affect liver function in alcoholic patients? J Fam Pract. 2003 Mar;52(3):187-8. PMID: 12620167. |
Commentary article on Kuffner (2001).
|
|
Louvet A, Ntandja Wandji LC, Lemaître E, Khaldi M, Lafforgue C, Artru F, Quesnel B, Lassailly G, Dharancy S, Mathurin P. Acute Liver Injury With Therapeutic Doses of Acetaminophen: A Prospective Study. Hepatology. 2021 May;73(5):1945-1955. doi: 10.1002/hep.31678. Epub 2021 Mar 25. PMID: 33306215. |
Incorrect study design (cohort study), comparison between therapeutic dose and overdose. |
Verantwoording
Beoordelingsdatum en geldigheid
Publicatiedatum : 30-09-2025
Beoordeeld op geldigheid : 10-09-2025
Algemene gegevens
De ontwikkeling/herziening van deze richtlijnmodule werd ondersteund door het Kennisinstituut van de Federatie Medisch Specialisten (www.demedischspecialist.nl/kennisinstituut) en werd gefinancierd uit [de Kwaliteitsgelden Medisch Specialisten (SKMS). De financier heeft geen enkele invloed gehad op de inhoud van de richtlijnmodule.
Samenstelling werkgroep
Voor het ontwikkelen van de richtlijnmodule is in 2022 een multidisciplinaire werkgroep ingesteld, bestaande uit vertegenwoordigers van alle relevante specialismen (zie hiervoor de Samenstelling van de werkgroep) die betrokken zijn bij de zorg voor patiënten met somatische complicaties als gevolg van risicovol alcoholgebruik.
Werkgroep
- Dr. H. van Soest, MDL-arts, Haaglanden MC, NVMDL (voorzitter)
- Dr. M.A. Lantinga, MDL-arts, Amsterdam UMC, NVMDL (vice-voorzitter)
- Dr. R.B. Takkenberg, MDL-arts, Amsterdam UMC, NVMDL
- Dr. H.J.M. de Jonge, MDL-arts, Jeroen Bosch Ziekenhuis, NVMDL
- Prof. dr. A.F.A. Schellekens, psychiater, Radboudumc, NVVP
- Drs. T. van Grinsven, verslavingsarts, Novadic Kentron, VVGN
- Dr. M. van Loon, spoedeisende hulparts, Haaglanden MC, NVSHA
- Drs. H. Lam, levertransplantatiechirurg, LUMC, NVvH (vanaf 2024)
- Drs. J.A. Willemse, directeur, Nederlandse Leverpatienten Vereniging
- Drs. P.J. van Dongen, ervaringsdeskundige en vrijwilliger, Hersenletsel.nl (tot 2023)
- H.M. Luttikhuis Msc, ziekenhuisapotheker, Slingeland Ziekenhuis, NVZA (vanaf 2023)
- Dr. R.A. Weersink, ziekenhuisapotheker, Deventer Ziekenhuis (tot 2024), NVZA (vanaf 2023)
- Dr. J. Blokzijl, MDL-arts, UMCG, NTV/LOL (vanaf 2025, specifiek voor module levertransplantatie)
Klankbordgroep
- J.E. de Haan, intensivist, Erasmus MC, NVIC
- Drs. A.T. Timmer, klinisch geriater, Tjongerschans Ziekenhuis, NVKG
- Dr. J.M.W. van den Ouweland, klinische chemie en endocrinologie, Canisius Wilhelmina Ziekenhuis, NVKC
- Dhr. A. van den Berg, ervaringsdeskundige, Stichting het Zwarte Gat (vanaf 2024)
Met ondersteuning van
- H. Olthuis-van Essen MSc, adviseur, Kennisinstituut van de Federatie Medisch Specialisten
- J.M.H. van der Hart MSc, adviseur, Kennisinstituut van de Federatie Medisch Specialisten
Belangenverklaringen
Een overzicht van de belangen van werkgroepleden en het oordeel over het omgaan met eventuele belangen vindt u in onderstaande tabel. De ondertekende belangenverklaringen zijn op te vragen bij het secretariaat van het Kennisinstituut van de Federatie Medisch Specialisten via secretariaat@kennisinstituut.nl.
Tabel 1 Gemelde (neven)functies en belangen werkgroep
|
Werkgroeplid |
Functie |
Nevenfuncties |
Gemelde belangen |
Ondernomen actie |
|
Dr. H. van Soest (voorzitter) |
MDL-arts, Haaglanden MC |
Voorzitter commissie hepatologie NVMDL; Voorzitter richtsnoer hepatitis C; Lid richtsnoer commissie hepatitis B; Lid medisch advies raad NLV; Lid visitatiecommissie NVMDL; Lid adviesraad MLDS/NAFLD-NL (initiation of a Dutch NAFLD consortium), lid werkgroep portale hypertensie |
Geen |
Geen restricties |
|
Dr. M.A. Lantinga (vice-voorzitter) |
MDL-arts, Amsterdam UMC, locatie VUmc |
Post Doc, Radboudumc (onbetaald), copromotor. Co-voorzitter portale hypertensie werkgroep, Nederlandse Vereniging van Hepatologie (NVH), onbetaald. Advisory Board - ERN RARE-LIVER // Training and Education Committee, onbetaald. |
Geen |
Geen restricties |
|
Dr. R.B. Takkenberg |
MDL-arts, Amsterdam UMC |
Deelname werkgroep tweede lijn, lid alcoholalliantie, gesprekken in het kader van nationaal preventieakkoord, deelname adviesraad over hepatitis Delta, Gilead |
ZonMw, Prevention of hepatic Encephalopathy by Administration of in patients with l, projectleider; Fonds-LVC, Neurofilament light chain and glial fibrillary acidic protein for hepatic encephalopathy, projectleider; KWF, Validation of a Short and Effective MRI Surveillance protocol for Hepatocellular Carcinoma Scr; MLDS, Initiation of a Dutch NAFLD consortium, geen projectleider |
Geen restricties (adviesraad betreft onderwerp dat niet relevant is voor deze richtlijnmodules) |
|
Dr. H.J.M. de Jonge |
MDL-arts, Jeroen Bosch Ziekenhuis |
Lid AIH werkgroep, lid werkgroep portale hypertensie, deelname werkgroep tweede lijn |
Geen |
Geen restricties |
|
Prof. dr. A.F.A. Schellekens |
Psychiater, Radboudumc |
Nationaal rapporteur verslavingen, adviescommissie alcohol van Gezondheidsraad |
ZonMw, onderzoek over verslaving, projectleider; NWO, onderzoek over verslaving, geen projectleider |
Geen restricties |
|
Drs. T. van Grinsven |
Verslavingsgeneeskundige, Novadic-Kentron |
Geen |
Dienstverband bij verslavingszorg Novadic-Kentron |
Geen restricties |
|
Dr. M. van Loon |
SEH-arts in Haaglanden MC; onderzoeker bij LUMC, afdeling Public Health en Eerstelijnsgeneeskunde |
Promotieonderzoek naar effectiviteit screening/haalbaarheid op SEH (afgerond); lid werkgroep 2e Lijn, samenwerkingsverband Vroegsignalering Alcoholstoornissen, onbetaald; Lid sectie Geriatrische Spoedzorg, NVSHA, scholing geven aan collega's m.b.t. acute geriatrie, betaald; de NVSHA vertegenwoordigen op landelijke bijeenkomsten, waarbij gesproken en beleid gemaakt wordt over geriatrische zorg in de acute keten, onbetaald; Instructeur voor aantal medische cursussen. |
Geen |
Geen restricties |
|
Drs. J.A. Willemse |
Directeur Nederlandse Leverpatienten Vereniging (betaald, tot 2024); bestuurslid/penningmeester Liver Patients International (onbetaald); bestuurslid ERN RARE LIVER (onbetaald) |
Deelname diverse commissies op gebied van leverziekten (onbetaald) |
Geen |
Geen restricties |
|
Drs. P.J. van Dongen (tot 2023) |
Patiëntenvertegenwoordiger bij Hersenletsel.nl (onbetaald) |
Geen |
Geen |
Geen restricties |
|
H.M. Luttikhuis Msc (vanaf 2023) |
Ziekenhuisapotheker in Slingelandziekenhuis |
Mede-eigenaar bij cooperatie Herenboerderij De Groote Modderkolk |
Geen |
Geen restricties |
|
Dr. R.A. Weersink (vanaf 2023) |
Ziekenhuisfarmacie, Deventer Ziekenhuis (tot 2024) |
Geen |
Geen |
Geen restricties |
|
Drs. H. Lam (vanaf 2024) |
Levertransplantatiechirurg, LUMC |
Geen |
Geen |
Geen restricties |
|
Dr. J. Blokzijl (vanaf 2025) |
MDL-arts, UMCG |
Geen |
Geen |
Geen restricties |
|
Klankbordgroeplid |
Functie |
Nevenfuncties |
Gemelde belangen |
Ondernomen actie |
|
J.E. de Haan |
Internist-intensivist |
Geen |
Geen |
Geen restricties |
|
Drs. A.T. Timmer |
Klinisch geriater in het Tjongerschans Ziekenhuis |
Geen |
Geen |
Geen restricties |
|
Dr. J.M.W. van den Ouweland |
Klinisch chemicus Dicoon, locatie Canisius-Wilhelmina Ziekenhuis, Nijmegen |
Geen |
Geen |
Geen restricties |
|
Lid algemeen bestuur Stichting het Zwarte Gat |
Lid cliëntenraad verslavingszorg; Lid werkgroepen Verslavingskunde Nederland; Lid expertcommissie MIND/LpGgz; Cliëntdeskundige bij twee zorgstandaarden, twee werkwijzers/richtlijnen voor alcohol/verslaving en werk en/of leefstijl, etc. |
Bij werken aan herstel van verslaving is er baat voor familie vanwege werk aan herstel. Verder zijn de uitkomsten altijd van nut bij naasten (b.v. cliënten die ik vertegenwoordig) |
Geen restricties |
Inbreng patiëntenperspectief
De werkgroep besteedde aandacht aan het patiëntenperspectief door het uitnodigen van patiëntenverenigingen voor de invitational conference, focusgroep en afgevaardigde patiëntenverenigingen in de werkgroep. Het verslag van de invitational conference is besproken in de werkgroep. De verkregen input is meegenomen bij het opstellen van de uitgangsvragen, de keuze voor de uitkomstmaten en bij het opstellen van de overwegingen. De conceptrichtlijn is tevens voor commentaar voorgelegd aan Hersenletsel.nl, de Nederlandse Leverpatienten Vereniging en Stichting het Zwarte Gat en de eventueel aangeleverde commentaren zijn bekeken en verwerkt.
Kwalitatieve raming van mogelijke financiële gevolgen in het kader van de Wkkgz
Bij de richtlijnmodule is conform de Wet kwaliteit, klachten en geschillen zorg (Wkkgz) een kwalitatieve raming uitgevoerd om te beoordelen of de aanbevelingen mogelijk leiden tot substantiële financiële gevolgen. Bij het uitvoeren van deze beoordeling is de richtlijnmodule op verschillende domeinen getoetst (zie het stroomschema op de Richtlijnendatabase).
Module |
Uitkomst raming |
Toelichting |
|
Module Pijnstilling - paracetamol |
geen financiële gevolgen |
Hoewel uit de toetsing volgt dat de aanbeveling breed toepasbaar is (5.000-40.000 patiënten), volgt ook uit de toetsing geen nieuwe manier van zorgverlening of andere organisatie van zorgverlening betreft. Er worden daarom geen substantiële financiële gevolgen verwacht. |
Werkwijze
Voor meer details over de gebruikte richtlijnmethodologie verwijzen wij u naar de Werkwijze. Relevante informatie voor de ontwikkeling/herziening van deze richtlijnmodule is hieronder weergegeven.
Zoekverantwoording
Literature search strategy
Algemene informatie
|
Richtlijn: Richtlijnmodules somatische complicaties van alcoholstoornissen |
|
|
In hoeverre is een dosisaanpassing nodig bij het gebruik van paracetamol bij patiënten met chronisch alcoholmisbruik en pijn? |
|
|
Database(s): Ovid/Medline, Embase |
Datum: 12-11-2023 |
|
Periode: 2000- |
Talen: nvt |
|
Literatuurspecialist: Eugenie Delvaux |
|
|
BMI zoekblokken: voor verschillende opdrachten wordt (deels) gebruik gemaakt van de zoekblokken van BMI-Online https://blocks.bmi-online.nl/ Bij gebruikmaking van een volledig zoekblok zal naar de betreffende link op de website worden verwezen. |
|
|
Toelichting: Voor deze vraag is op twee manieren gezocht met de volgende concepten: Paracetamol EN alcoholgebruik EN volwassenen Het eerste artikel van Lauterburg BH[1988] komt niet uit de search aangezien er vanaf 2000 gezocht dient te worden. Het 2e artikel komt uit de search. |
|
|
Te gebruiken voor richtlijnen tekst: In de databases Embase en Ovid/Medline is op 12-11-2023 met relevante zoektermen gezocht vanaf 2000 naar (systematische) reviews, RCT’s en observationele studies over het gebruik van paracetamol bij patiënten met chronische alcoholmisbruik en pijn?. De literatuurzoekactie leverde 551 unieke treffers op. |
|
Zoekopbrengst
|
|
EMBASE |
OVID/MEDLINE |
Ontdubbeld |
|
SRs |
73 |
11 |
78 |
|
RCTs |
80 |
37 |
76 |
|
Observationele studies |
451 |
70 |
397 |
|
Totaal |
|
|
551 |
Zoekstrategie
Embase
|
No. |
Query |
Results |
|
#14 |
#6 AND #11 |
451 |
|
#13 |
#6 AND #8 |
80 |
|
#12 |
#6 AND #7 |
73 |
|
#11 |
#9 OR #10 |
16428567 |
|
#10 |
'case control study'/de OR 'comparative study'/exp OR 'control group'/de OR 'controlled study'/de OR 'controlled clinical trial'/de OR 'crossover procedure'/de OR 'double blind procedure'/de OR 'phase 2 clinical trial'/de OR 'phase 3 clinical trial'/de OR 'phase 4 clinical trial'/de OR 'pretest posttest design'/de OR 'pretest posttest control group design'/de OR 'quasi experimental study'/de OR 'single blind procedure'/de OR 'triple blind procedure'/de OR (((control OR controlled) NEAR/6 trial):ti,ab,kw) OR (((control OR controlled) NEAR/6 (study OR studies)):ti,ab,kw) OR (((control OR controlled) NEAR/1 active):ti,ab,kw) OR 'open label*':ti,ab,kw OR (((double OR two OR three OR multi OR trial) NEAR/1 (arm OR arms)):ti,ab,kw) OR ((allocat* NEAR/10 (arm OR arms)):ti,ab,kw) OR placebo*:ti,ab,kw OR 'sham-control*':ti,ab,kw OR (((single OR double OR triple OR assessor) NEAR/1 (blind* OR masked)):ti,ab,kw) OR nonrandom*:ti,ab,kw OR 'non-random*':ti,ab,kw OR 'quasi-experiment*':ti,ab,kw OR crossover:ti,ab,kw OR 'cross over':ti,ab,kw OR 'parallel group*':ti,ab,kw OR 'factorial trial':ti,ab,kw OR ((phase NEAR/5 (study OR trial)):ti,ab,kw) OR ((case* NEAR/6 (matched OR control*)):ti,ab,kw) OR ((match* NEAR/6 (pair OR pairs OR cohort* OR control* OR group* OR healthy OR age OR sex OR gender OR patient* OR subject* OR participant*)):ti,ab,kw) OR ((propensity NEAR/6 (scor* OR match*)):ti,ab,kw) OR versus:ti OR vs:ti OR compar*:ti OR ((compar* NEAR/1 study):ti,ab,kw) OR (('major clinical study'/de OR 'clinical study'/de OR 'cohort analysis'/de OR 'observational study'/de OR 'cross-sectional study'/de OR 'multicenter study'/de OR 'correlational study'/de OR 'follow up'/de OR cohort*:ti,ab,kw OR 'follow up':ti,ab,kw OR followup:ti,ab,kw OR longitudinal*:ti,ab,kw OR prospective*:ti,ab,kw OR retrospective*:ti,ab,kw OR observational*:ti,ab,kw OR 'cross sectional*':ti,ab,kw OR cross?ectional*:ti,ab,kw OR multicent*:ti,ab,kw OR 'multi-cent*':ti,ab,kw OR consecutive*:ti,ab,kw) AND (group:ti,ab,kw OR groups:ti,ab,kw OR subgroup*:ti,ab,kw OR versus:ti,ab,kw OR vs:ti,ab,kw OR compar*:ti,ab,kw OR 'odds ratio*':ab OR 'relative odds':ab OR 'risk ratio*':ab OR 'relative risk*':ab OR 'rate ratio':ab OR aor:ab OR arr:ab OR rrr:ab OR ((('or' OR 'rr') NEAR/6 ci):ab))) |
14566050 |
|
#9 |
'major clinical study'/de OR 'clinical study'/de OR 'case control study'/de OR 'family study'/de OR 'longitudinal study'/de OR 'retrospective study'/de OR 'prospective study'/de OR 'comparative study'/de OR 'cohort analysis'/de OR ((cohort NEAR/1 (study OR studies)):ab,ti) OR (('case control' NEAR/1 (study OR studies)):ab,ti) OR (('follow up' NEAR/1 (study OR studies)):ab,ti) OR (observational NEAR/1 (study OR studies)) OR ((epidemiologic NEAR/1 (study OR studies)):ab,ti) OR (('cross sectional' NEAR/1 (study OR studies)):ab,ti) |
7924097 |
|
#8 |
'randomized controlled trial'/exp OR 'randomized controlled trial' OR random*:ti,ab OR (((pragmatic OR practical) NEAR/1 'clinical trial*'):ti,ab) OR ((('non inferiority' OR noninferiority OR superiority OR equivalence) NEAR/3 trial*):ti,ab) OR rct:ti,ab,kw |
2236499 |
|
#7 |
'meta analysis'/exp OR 'meta analysis (topic)'/exp OR metaanaly*:ti,ab OR 'meta analy*':ti,ab OR metanaly*:ti,ab OR 'systematic review'/de OR 'cochrane database of systematic reviews'/jt OR prisma:ti,ab OR prospero:ti,ab OR (((systemati* OR scoping OR umbrella OR 'structured literature') NEAR/3 (review* OR overview*)):ti,ab) OR ((systemic* NEAR/1 review*):ti,ab) OR (((systemati* OR literature OR database* OR 'data base*') NEAR/10 search*):ti,ab) OR (((structured OR comprehensive* OR systemic*) NEAR/3 search*):ti,ab) OR (((literature NEAR/3 review*):ti,ab) AND (search*:ti,ab OR database*:ti,ab OR 'data base*':ti,ab)) OR (('data extraction':ti,ab OR 'data source*':ti,ab) AND 'study selection':ti,ab) OR ('search strategy':ti,ab AND 'selection criteria':ti,ab) OR ('data source*':ti,ab AND 'data synthesis':ti,ab) OR medline:ab OR pubmed:ab OR embase:ab OR cochrane:ab OR (((critical OR rapid) NEAR/2 (review* OR overview* OR synthes*)):ti) OR ((((critical* OR rapid*) NEAR/3 (review* OR overview* OR synthes*)):ab) AND (search*:ab OR database*:ab OR 'data base*':ab)) OR metasynthes*:ti,ab OR 'meta synthes*':ti,ab |
976749 |
|
#6 |
#5 AND [1-1-2000]/sd |
1114 |
|
#5 |
#4 NOT (('adolescent'/exp OR 'child'/exp OR adolescent*:ti,ab,kw OR child*:ti,ab,kw OR schoolchild*:ti,ab,kw OR infant*:ti,ab,kw OR girl*:ti,ab,kw OR boy*:ti,ab,kw OR teen:ti,ab,kw OR teens:ti,ab,kw OR teenager*:ti,ab,kw OR youth*:ti,ab,kw OR pediatr*:ti,ab,kw OR paediatr*:ti,ab,kw OR puber*:ti,ab,kw) NOT ('adult'/exp OR 'aged'/exp OR 'middle aged'/exp OR adult*:ti,ab,kw OR man:ti,ab,kw OR men:ti,ab,kw OR woman:ti,ab,kw OR women:ti,ab,kw)) |
1427 |
|
#4 |
#3 NOT ('conference abstract'/it OR 'editorial'/it OR 'letter'/it OR 'note'/it) NOT (('animal'/exp OR 'animal experiment'/exp OR 'animal model'/exp OR 'nonhuman'/exp) NOT 'human'/exp) |
1486 |
|
#3 |
#1 AND #2 |
2534 |
|
#2 |
'paracetamol'/exp OR '4 acetamidophenol':ti,ab,kw OR '4 acetaminophenol':ti,ab,kw OR '4 acetylaminophenol':ti,ab,kw OR '4 hydroxyacetanilide':ti,ab,kw OR 'a-mol':ti,ab,kw OR 'abenol':ti,ab,kw OR 'acamol':ti,ab,kw OR 'acamoli forte suppositories for kids':ti,ab,kw OR 'acenol':ti,ab,kw OR 'acephen':ti,ab,kw OR 'acet suppositories':ti,ab,kw OR 'acetaflex':ti,ab,kw OR 'acetalgin':ti,ab,kw OR 'acetamino phenol':ti,ab,kw OR 'acetaminophen':ti,ab,kw OR 'acetaminophene':ti,ab,kw OR 'acetaminophenol':ti,ab,kw OR 'acetamol':ti,ab,kw OR 'acetomenophen':ti,ab,kw OR 'acetylaminophenol':ti,ab,kw OR 'adorem':ti,ab,kw OR 'afebrin':ti,ab,kw OR 'algiafin':ti,ab,kw OR 'algocit':ti,ab,kw OR 'algostase mono':ti,ab,kw OR 'algotropyl':ti,ab,kw OR 'alphagesic':ti,ab,kw OR 'altridexamol':ti,ab,kw OR 'alvedon':ti,ab,kw OR 'amadil':ti,ab,kw OR 'anacin 3':ti,ab,kw OR 'anaflon':ti,ab,kw OR 'analgiser':ti,ab,kw OR 'antidol':ti,ab,kw OR 'apamide':ti,ab,kw OR 'apap':ti,ab,kw OR 'apiredol':ti,ab,kw OR 'apiretal':ti,ab,kw OR 'apirex':ti,ab,kw OR 'apotel':ti,ab,kw OR 'arax':ti,ab,kw OR 'arthralgen':ti,ab,kw OR 'atamel':ti,ab,kw OR 'ben-u-ron':ti,ab,kw OR 'benuron':ti,ab,kw OR 'biogesic':ti,ab,kw OR 'biogesic suspension':ti,ab,kw OR 'bodrex':ti,ab,kw OR 'calapol':ti,ab,kw OR 'calodol':ti,ab,kw OR 'calonal':ti,ab,kw OR 'calpol':ti,ab,kw OR 'causalon':ti,ab,kw OR 'cemol':ti,ab,kw OR 'christamol':ti,ab,kw OR 'claradol':ti,ab,kw OR 'clocephen':ti,ab,kw OR 'cp 500':ti,ab,kw OR 'cp500':ti,ab,kw OR 'cupanol':ti,ab,kw OR 'dafalgan':ti,ab,kw OR 'dafalgancaps':ti,ab,kw OR 'dafalganhop':ti,ab,kw OR 'dafalgantabs':ti,ab,kw OR 'daga':ti,ab,kw OR 'daleron':ti,ab,kw OR 'daro':ti,ab,kw OR 'datril':ti,ab,kw OR 'defebryl':ti,ab,kw OR 'depon':ti,ab,kw OR 'depyretin':ti,ab,kw OR 'difenatil':ti,ab,kw OR 'dirox':ti,ab,kw OR 'dismifen':ti,ab,kw OR 'disprol':ti,ab,kw OR 'dolal':ti,ab,kw OR 'dolex':ti,ab,kw OR 'dolex 500':ti,ab,kw OR 'dolflash':ti,ab,kw OR 'doliprane':ti,ab,kw OR 'dolipranecaps':ti,ab,kw OR 'dolitabs':ti,ab,kw OR 'dolko':ti,ab,kw OR 'dolocatil':ti,ab,kw OR 'dolofen':ti,ab,kw OR 'dololavi':ti,ab,kw OR 'dolomol':ti,ab,kw OR 'doloramol':ti,ab,kw OR 'dolorin':ti,ab,kw OR 'dolorol':ti,ab,kw OR 'dolostop':ti,ab,kw OR 'dolotemp':ti,ab,kw OR 'dolprone':ti,ab,kw OR 'dolstic':ti,ab,kw OR 'doltem':ti,ab,kw OR 'drilan':ti,ab,kw OR 'dristan af':ti,ab,kw OR 'dulsifeb':ti,ab,kw OR 'duorol':ti,ab,kw OR 'dymadon':ti,ab,kw OR 'efetamol':ti,ab,kw OR 'efferaldol flas':ti,ab,kw OR 'efferalgan':ti,ab,kw OR 'efferalgan 500':ti,ab,kw OR 'efferalgan forte':ti,ab,kw OR 'efferalganmed':ti,ab,kw OR 'efferalganodis':ti,ab,kw OR 'efferamol':ti,ab,kw OR 'efferelgan':ti,ab,kw OR 'enelfa':ti,ab,kw OR 'eneril':ti,ab,kw OR 'eraldor':ti,ab,kw OR 'eu med':ti,ab,kw OR 'exopon':ti,ab,kw OR 'expandol':ti,ab,kw OR 'febrectal':ti,ab,kw OR 'febridol':ti,ab,kw OR 'febrilix':ti,ab,kw OR 'fendon':ti,ab,kw OR 'fervex':ti,ab,kw OR 'fibrinol':ti,ab,kw OR 'fizamol':ti,ab,kw OR 'fortolin':ti,ab,kw OR 'galpharm six plus pain relief':ti,ab,kw OR 'gelocatil':ti,ab,kw OR 'geluprane 500':ti,ab,kw OR 'gewadal-pamol':ti,ab,kw OR 'gunaceta':ti,ab,kw OR 'headache strength allerest':ti,ab,kw OR 'hedex':ti,ab,kw OR 'helporal':ti,ab,kw OR 'holdofeb':ti,ab,kw OR 'infants` feverall':ti,ab,kw OR 'influbene c':ti,ab,kw OR 'influbene n':ti,ab,kw OR 'injectapap':ti,ab,kw OR 'janupap':ti,ab,kw OR 'kamolas':ti,ab,kw OR 'kyofen':ti,ab,kw OR 'lekadol':ti,ab,kw OR 'lekadol direkt':ti,ab,kw OR 'lekadol forte':ti,ab,kw OR 'lemgrip':ti,ab,kw OR 'lemsip cold and flu':ti,ab,kw OR 'letamol':ti,ab,kw OR 'liquiprin':ti,ab,kw OR 'lotemp':ti,ab,kw OR 'lupocet':ti,ab,kw OR 'lupocet forte':ti,ab,kw OR 'lyteca':ti,ab,kw OR 'malidens':ti,ab,kw OR 'mandanol':ti,ab,kw OR 'medamol':ti,ab,kw OR 'medipyrin':ti,ab,kw OR 'meforagesic':ti,ab,kw OR 'megapar':ti,ab,kw OR 'megapar forte':ti,ab,kw OR 'metagesic':ti,ab,kw OR 'metalid':ti,ab,kw OR 'mexalen':ti,ab,kw OR 'milidon 500':ti,ab,kw OR 'minopan':ti,ab,kw OR 'mobistix instant':ti,ab,kw OR 'mypara':ti,ab,kw OR 'n acetyl 4 aminophenol':ti,ab,kw OR 'n acetyl para aminophenol':ti,ab,kw OR 'n-acetyl-p-aminophenol':ti,ab,kw OR 'nalgesik':ti,ab,kw OR 'napamol':ti,ab,kw OR 'napap':ti,ab,kw OR 'naprex':ti,ab,kw OR 'nebs':ti,ab,kw OR 'nektol 500':ti,ab,kw OR 'neocitran':ti,ab,kw OR 'neodalmin':ti,ab,kw OR 'neopap':ti,ab,kw OR 'nevral':ti,ab,kw OR 'nideran':ti,ab,kw OR 'nilapur':ti,ab,kw OR 'nirolex febbre e dolore':ti,ab,kw OR 'nisidinasol':ti,ab,kw OR 'nobedon':ti,ab,kw OR 'nuridol':ti,ab,kw OR 'nysacetol':ti,ab,kw OR 'ofirmev':ti,ab,kw OR 'pacemol':ti,ab,kw OR 'pacimol':ti,ab,kw OR 'padolieve':ti,ab,kw OR 'padolieve max':ti,ab,kw OR 'pamal':ti,ab,kw OR 'pamol':ti,ab,kw OR 'pamol f':ti,ab,kw OR 'pamol flash':ti,ab,kw OR 'panacit':ti,ab,kw OR 'panadol':ti,ab,kw OR 'panadol actifast':ti,ab,kw OR 'panadol advance':ti,ab,kw OR 'panadol extra strength':ti,ab,kw OR 'panadol fasttabs':ti,ab,kw OR 'panadol forte':ti,ab,kw OR 'panadol hot':ti,ab,kw OR 'panadol max strength cold and flu':ti,ab,kw OR 'panadol novum':ti,ab,kw OR 'panadol optizorb':ti,ab,kw OR 'panadol pore':ti,ab,kw OR 'panadol rapid':ti,ab,kw OR 'panadol soluble':ti,ab,kw OR 'panadol soluble max':ti,ab,kw OR 'panadol zapp':ti,ab,kw OR 'panamax':ti,ab,kw OR 'panaprex':ti,ab,kw OR 'panasorb':ti,ab,kw OR 'panasorbe':ti,ab,kw OR 'panodil':ti,ab,kw OR 'para acetamidophenol':ti,ab,kw OR 'para acetylaminophenol':ti,ab,kw OR 'para hydroxyacetanilide':ti,ab,kw OR 'para suppo':ti,ab,kw OR 'para-hot':ti,ab,kw OR 'para-supps':ti,ab,kw OR 'para-tabs':ti,ab,kw OR 'paraceon':ti,ab,kw OR 'paracet':ti,ab,kw OR 'paraceta 500':ti,ab,kw OR 'paracetalgin':ti,ab,kw OR 'paracetaminophenol':ti,ab,kw OR 'paracetamol':ti,ab,kw OR 'paracetamol ester':ti,ab,kw OR 'paracetamole':ti,ab,kw OR 'paracut':ti,ab,kw OR 'paradolor':ti,ab,kw OR 'parafizz':ti,ab,kw OR 'parafusiv':ti,ab,kw OR 'parageniol':ti,ab,kw OR 'paragin':ti,ab,kw OR 'paralen':ti,ab,kw OR 'paralen rapid':ti,ab,kw OR 'paralief':ti,ab,kw OR 'paralink':ti,ab,kw OR 'paralyoc':ti,ab,kw OR 'paramax':ti,ab,kw OR 'paramax forte':ti,ab,kw OR 'paramax junior':ti,ab,kw OR 'paramax rap':ti,ab,kw OR 'paramax rapid':ti,ab,kw OR 'paramegal':ti,ab,kw OR 'paramidol':ti,ab,kw OR 'paramolan':ti,ab,kw OR 'parapaed':ti,ab,kw OR 'parapaed junior':ti,ab,kw OR 'parapaed six plus':ti,ab,kw OR 'paraphar':ti,ab,kw OR 'parapo':ti,ab,kw OR 'parapyrex':ti,ab,kw OR 'paraserts':ti,ab,kw OR 'parasolve':ti,ab,kw OR 'paratabs':ti,ab,kw OR 'paraveganio':ti,ab,kw OR 'paravescent':ti,ab,kw OR 'paravict':ti,ab,kw OR 'parvid':ti,ab,kw OR 'pasolind':ti,ab,kw OR 'pasolind n':ti,ab,kw OR 'paximol':ti,ab,kw OR 'pe-tam':ti,ab,kw OR 'pedicetamol':ti,ab,kw OR 'pedipan':ti,ab,kw OR 'penral-night':ti,ab,kw OR 'perfalgan':ti,ab,kw OR 'phenaphen':ti,ab,kw OR 'phizamol':ti,ab,kw OR 'pinex':ti,ab,kw OR 'piros':ti,ab,kw OR 'polarfen':ti,ab,kw OR 'predimol':ti,ab,kw OR 'prompt':ti,ab,kw OR 'prosinus':ti,ab,kw OR 'protalgon':ti,ab,kw OR 'puernol':ti,ab,kw OR 'pyrigesic':ti,ab,kw OR 'raperon':ti,ab,kw OR 'rapidol':ti,ab,kw OR 'relaphen':ti,ab,kw OR 'reliv':ti,ab,kw OR 'remedol':ti,ab,kw OR 'revanin':ti,ab,kw OR 'rhinapen elixir':ti,ab,kw OR 'rhodapap':ti,ab,kw OR 'rowalief':ti,ab,kw OR 'roxamol gelcaps':ti,ab,kw OR 'rubophen':ti,ab,kw OR 'salzone':ti,ab,kw OR 'sanador':ti,ab,kw OR 'sedarene adultes':ti,ab,kw OR 'sedarene enfants':ti,ab,kw OR 'sedes a':ti,ab,kw OR 'serimol':ti,ab,kw OR 'setamol':ti,ab,kw OR 'sinaspril':ti,ab,kw OR 'sinebriv':ti,ab,kw OR 'sinedol':ti,ab,kw OR 'sinpro':ti,ab,kw OR 'snp 810':ti,ab,kw OR 'snp 830':ti,ab,kw OR 'snp 840':ti,ab,kw OR 'snp810':ti,ab,kw OR 'snp830':ti,ab,kw OR 'snp840':ti,ab,kw OR 'solpaone':ti,ab,kw OR 'sootheze six plus':ti,ab,kw OR 'supofen':ti,ab,kw OR 'tabalgin':ti,ab,kw OR 'tachipirin':ti,ab,kw OR 'tachipirina':ti,ab,kw OR 'taganopain':ti,ab,kw OR 'tapar':ti,ab,kw OR 'tempra':ti,ab,kw OR 'tempte':ti,ab,kw OR 'temzzard':ti,ab,kw OR 'termadec febbre e dolore':ti,ab,kw OR 'termalgin':ti,ab,kw OR 'termofren':ti,ab,kw OR 'teva-mexalen':ti,ab,kw OR 'therimin':ti,ab,kw OR 'tipol':ti,ab,kw OR 'tipol max':ti,ab,kw OR 'tralgon':ti,ab,kw OR 'tralgon elixir':ti,ab,kw OR 'tramil':ti,ab,kw OR 'treuphadol':ti,ab,kw OR 'turpan':ti,ab,kw OR 'tylenol':ti,ab,kw OR 'tylenol forte':ti,ab,kw OR 'tylenol nr 1':ti,ab,kw OR 'tylex':ti,ab,kw OR 'valadol':ti,ab,kw OR 'wegmal':ti,ab,kw OR 'winadol':ti,ab,kw OR 'winasorb':ti,ab,kw OR 'xebramol':ti,ab,kw OR 'xumadol':ti,ab,kw OR 'zipamol':ti,ab,kw OR 'zolben':ti,ab,kw OR 'zydinol':ti,ab,kw |
241631 |
|
#1 |
'alcoholism'/exp OR 'drinking behavior'/exp OR (((addiction OR dependen* OR polyneuropathy* OR 'use disorder*' OR 'induced disorder*' OR related OR individual OR neuropath* OR polyneuritis OR chronic) NEAR/4 alcohol*):ti,ab,kw) OR alcoholic:ti,ab,kw OR dipsomania:ti,ab,kw OR dipsomaniac:ti,ab,kw OR 'ethanol dependence':ti,ab,kw OR (((alcohol OR controlled OR behavior OR behavior OR habit OR pattern OR social) NEAR/4 drinking):ti,ab,kw) |
313094 |
Ovid/Medline
|
# |
Searches |
Results |
|
12 |
6 and 9 |
70 |
|
11 |
6 and 8 |
37 |
|
10 |
6 and 7 |
11 |
|
9 |
Epidemiologic studies/ or case control studies/ or exp cohort studies/ or Controlled Before-After Studies/ or Case control.tw. or cohort*.tw. or Cohort analy$.tw. or (Follow up adj (study or studies)).tw. or (observational adj (study or studies)).tw. or Longitudinal.tw. or Retrospective*.tw. or prospective*.tw. or consecutive*.tw. or Cross sectional.tw. or Cross-sectional studies/ or historically controlled study/ or interrupted time series analysis/ |
4624404 |
|
8 |
exp clinical trial/ or randomized controlled trial/ or exp clinical trials as topic/ or randomized controlled trials as topic/ or Random Allocation/ or Double-Blind Method/ or Single-Blind Method/ or (clinical trial, phase i or clinical trial, phase ii or clinical trial, phase iii or clinical trial, phase iv or controlled clinical trial or randomized controlled trial or multicenter study or clinical trial).pt. or random*.ti,ab. or (clinic* adj trial*).tw. or ((singl* or doubl* or treb* or tripl*) adj (blind$3 or mask$3)).tw. or Placebos/ or placebo*.tw. |
2656363 |
|
7 |
meta-analysis/ or meta-analysis as topic/ or (metaanaly* or meta-analy* or metanaly*).ti,ab,kf. or systematic review/ or cochrane.jw. or (prisma or prospero).ti,ab,kf. or ((systemati* or scoping or umbrella or "structured literature") adj3 (review* or overview*)).ti,ab,kf. or (systemic* adj1 review*).ti,ab,kf. or ((systemati* or literature or database* or data-base*) adj10 search*).ti,ab,kf. or ((structured or comprehensive* or systemic*) adj3 search*).ti,ab,kf. or ((literature adj3 review*) and (search* or database* or data-base*)).ti,ab,kf. or (("data extraction" or "data source*") and "study selection").ti,ab,kf. or ("search strategy" and "selection criteria").ti,ab,kf. or ("data source*" and "data synthesis").ti,ab,kf. or (medline or pubmed or embase or cochrane).ab. or ((critical or rapid) adj2 (review* or overview* or synthes*)).ti. or (((critical* or rapid*) adj3 (review* or overview* or synthes*)) and (search* or database* or data-base*)).ab. or (metasynthes* or meta-synthes*).ti,ab,kf. |
707078 |
|
6 |
limit 5 to yr="2000-Current" |
242 |
|
5 |
4 not ((exp animals/ or exp models, animal/) not humans/) not (letter/ or comment/ or editorial/) |
440 |
|
4 |
3 not (child* or schoolchild* or infan* or adolescen* or pediatri* or paediatr* or neonat* or boy or boys or boyhood or girl or girls or girlhood or youth or youths or baby or babies or toddler* or childhood or teen or teens or teenager* or newborn* or postneonat* or postnat* or puberty or preschool* or suckling* or picu or nicu or juvenile?).tw. |
564 |
|
3 |
1 and 2 |
601 |
|
2 |
exp Acetaminophen/ or acetaminophen.ti,ab,kf. or paracetamol.ti,ab,kf. or panadol.ti,ab,kf. |
34194 |
|
1 |
exp Alcohol-Related Disorders/ or exp Drinking Behavior/ or (((addiction or dependen* or polyneuropathy* or 'use disorder*' or 'induced disorder*' or related or individual or neuropath* or polyneuritis or chronic) adj4 (ethanol or alcohol*)) or (((alcohol or controlled or behavior or behavior or habit or pattern or social) adj4 drinking) or alcoholic or dipsomania or dipsomaniac)).ti,ab,kf. |
258950 |