Fontan-circulatie
Uitgangsvraag
Welk antistollingsbeleid dient te worden gevolgd rondom de behandeling van een Fontan-circulatie?
Aanbeveling
Aanbeveling-1
Behandel kinderen zo snel als mogelijk na het complementeren van de Fontan circulatie met therapeutisch ongefractioneerde heparine (of LMWH) en ga als orale intake weer gestart is over op een VKA of DOAC: behandel kinderen met een Fontan circulatie zonder kunstklep* gedurende drie tot zes maanden postoperatief met VKA, waarbij gestreefd wordt naar een INR 2.0 – 3.0, of een DOAC in therapeutische dosering, op basis van een afweging van voor- en nadelen bij een individuele patiënt. Zie voor de dosering het Kinderformularium (behandeling VTE dosis).
Aanbeveling-2
Behandel kinderen met een Fontan circulatie zonder kunstklep* na zes maanden met een DOAC (rivaroxaban) in profylactische dosering of acetylsalicylzuur voor onbepaalde tijd. Zie voor de dosering het Kinderformularium (DOAC: Fontan profylaxe).
Aanbeveling-3
Behandel kinderen met een Fontan circulatie zonder kunstklep* na zes maanden met risicofactoren (bilaterale VCS, kleine pulmonaaltakken/VCS, stent in pulmonaaltakken, niet afgesloten blind-eindigende pulmonalisstomp, stasis in hypoplastische ventrikel, open fenestratie) met VKA, waarbij gestreefd wordt naar een INR 2.0 – 3.0, of een DOAC in therapeutische dosering, op basis van een afweging van voor- en nadelen bij een individuele patiënt.
Aanbeveling-4
Behandel kinderen met een Fontan circulatie met een kunstklep levenslang met VKA, waarbij gestreefd wordt naar een INR 2.0 – 3.0 (bij aortaklep) of INR 2.5 - 3.5 (in atrioventriculaire kleppositie).
*voor Fontan patiënten met een kunstklep zie module Kunstkleppen
Overwegingen
Balans tussen gewenste en ongewenste effecten
Er is een literatuuronderzoek verricht naar de vergelijking tussen verschillende soorten tromboseprofylaxe bij kinderen met een indicatie voor een Fontan circulatie voor verschillende uitkomstmaten. In de literatuuranalyse zijn tien studies beschreven, waarvan twee studies met een follow-up van ≤1 jaar (Ankola, 2021; McCrindle, 2021) en acht studies met een follow-up van >1 jaar (Egbe, 2017; Kawamatsu, 2021; Al-Jazairi, 2019; Iyengar, 2016; Pessotti, 2014; Seipelt, 2002; McCrindle, 2013; Potter, 2013).
Trombotische complicaties
Acetylsalicylzuur was in studies met een follow-up van één jaar of korter geassocieerd met een klinisch relevant hoger risico op trombotische complicaties dan warfarine (Ankola, 2021) en rivaroxaban in profylactische dosering (McCrindle, 2021), maar een klinisch relevant lager risico dan enoxaparine (Ankola, 2021). Warfarine toonde een klinisch relevant lager risico dan enoxaparine (Ankola, 2021).
In zeven studies met een langere follow-up werd acetylsalicylzuur vergeleken met warfarine en bleek het risico op trombotische complicaties hoger bij acetylsalicylzuur (Al-Jazairi, 2019; Egbe, 2017; Iyengar, 2016; McCrindle, 2013; Pessotti, 2014; Potter, 2013; Seipelt, 2002). DOACs verminderden het risico op trombotische complicaties in vergelijking met plaatjesaggregatieremmers en vitamine K-antagonisten, maar verschilden niet significant van de combinatie van beide (Kawamatsu, 2021). Daarnaast waren plaatjesaggregatieremmers geassocieerd met een lager risico op trombotische complicaties dan vitamine K-antagonisten, terwijl ze een hoger risico hadden in vergelijking met de combinatie van een plaatjesaggregatieremmer en een anticoagulans. Vitamine K-antagonisten lieten op hun beurt een hoger risico zien dan deze combinatie.
Bloedingscomplicaties
Voor bloedingen toonde een korte follow-upstudie dat rivaroxaban een hoger risico op majeure bloedingen had dan acetylsalicylzuur, terwijl acetylsalicylzuur geassocieerd was met een hoger risico op klinisch relevante niet-majeure bloedingen (McCrindle, 2021).
Bij langere follow-up werd geen verschil in majeure bloedingen gevonden tussen acetylsalicylzuur en warfarine, maar acetylsalicylzuur gaf minder non-majeure en mineure bloedingen (Al-Jazairi, 2019; McCrindle, 2013). DOACs verlaagden het risico op majeure bloedingen vergeleken met andere anticoagulantia, maar verhoogden in sommige gevallen het risico op mineure bloedingen (Kawamatsu, 2021).
Gebruik van acetylsalicylzuur had een verhoogd risico op ischemische beroerte in vergelijking met rivaroxaban in een korte follow-upstudie (McCrindle, 2021), terwijl er bij langere follow-up geen bewijs werd gevonden. Voor kwaliteit van leven en bijwerkingen werd geen bewijs gevonden, behalve in één korte follow-upstudie, waarin geen klinisch relevant verschil werd vastgesteld tussen acetylsalicylzuur en rivaroxaban in profylactische dosering (McCrindle, 2021).
Sterfte
Voor sterfte werd in studies met een korte follow-up geen bewijs gevonden. Bij langere follow-up waren de resultaten tegenstrijdig: McCrindle (2013) rapporteerde een hoger risico bij acetylsalicylzuur, terwijl Pessotti (2014) een hoger risico bij warfarine vond.
Samenvattend is er bewijs dat het geven van thromboprofylaxe geïndiceerd is bij patiënten met een 1-kamer hart en een Fontan circulatie. Met name in het eerste jaar na Fontan completering en bij risico factoren is er een verhoogd risico op trombo-embolische complicaties die wordt verlaagd door thrombo-profylaxe.
Er is door thromboprofylaxe een verhoogd risico op blauwe plekken en bloedingen. Dit geldt voor alle medicaties en er is geen bewijs voor de superioriteit van 1 van de medicaties. Wel is er mogelijk bij menstruerende patiënten bij DOACs (met name rivaroxaban) een verhoogd risico op heftiger bloedverlies bij de menstruatie.
Ten aanzien van de vorige versie van de richtlijn is de introductie van DOACs ter preventie van trombo-embolische complicaties van belang. Sinds de introductie van DOACs zijn er meerdere studies verricht bij kinderen ter behandeling en preventie van trombo-embolische processen. In deze richtlijn zijn alleen de primaire studies van DOACs bij kinderen met een Fontan circulatie opgenomen en beoordeeld (Kawamatsu, 2021; McCrindle, 2021).
Er bestaan twee andere studies in kinderen met een hartaandoening, waarin een DOAC wordt vergeleken met andere strategieën ter preventie van trombo-embolische processen: Portman (2022) testte prospectief edoxaban versus ‘standard of care’ (met name vitamine K antagonisten) in kinderen met een hartaandoening (44% na Fontan operatie) en concludeerde dat edoxaban is een mogelijk alternatief als tromboseprofylaxe met lage frequentie van relevante bloedingen en trombo-embolische complicaties. Payne (2023) onderzocht retrospectief apixaban in de behandeling en het voorkomen van trombose in kinderen met een hartaandoening (45% met “single ventricle congenital heart disease”) en concludeerde dat het gebruik van apixaban goed mogelijk is met een lage frequentie van complicaties.
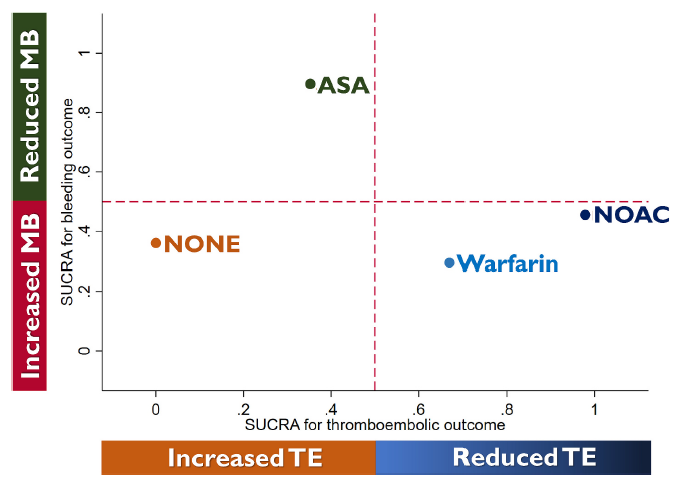
Daarnaast zijn er twee meta-analyses verschenen van de patiënten (zowel kinderen als volwassenen) met een Fontan circulatie (Van den Eynde, 2023; Sethasathien, 2024). In de meest recente meta-analyse wordt aangegeven dat DOACs het meest gunstige profiel hebben in de balans tussen voorkomen van trombose en het risico op (ernstige) bloedingen (zie figuur 1).
Figuur 1.
Alle tromboseprofylaxe werden gegradeerd op basis van hun werkzaamheid om trombo-embolische voorvallen en ernstige bloedingen te voorkomen, op basis van hun oppervlakte onder de cumulatieve rangschikkingscurve. Overgenomen uit Sethasathien, 2024
Kwaliteit van bewijs
De overall kwaliteit van bewijs is zeer laag. Dit betekent dat we zeer onzeker zijn over het gevonden geschatte effect van de cruciale uitkomstmaten.
Er is afgewaardeerd vanwege
- Risk of Bias: methodologische beperkingen in de studies (in RCTs onder andere geen blindering van de medicatie en in sommige gevallen onduidelijk beschreven randomisatieprocedure; in observationele studies onder andere onduidelijkheid over de exclusie van patiënten met trombotische complicaties bij de start van de studie, en het niet (consistent) corrigeren voor confounders in de statistische analyses).
- Imprecisie: onnauwkeurigheid, omdat het betrouwbaarheidsinterval beide grenzen van klinische relevantie overschrijdt.
Waarden en voorkeuren van patiënten en ouders/verzorgers
Bespreek in de spreekkamer of de gewenste effecten die op groepsniveau zijn onderzocht opwegen tegen de ongewenste effecten die op groepsniveau zijn onderzocht (voor zover gerapporteerd) en hoe die zich verhouden tot de waarden en voorkeuren van de individuele patiënt en ouders/verzorgers. De afweging is voor iedereen anders. Ook moeten er enkele leefregels worden meegegeven. Bij enkele antistolling moet geadviseerd worden om geen high impact sporten uit te voeren en geen tatoeage te laten zetten (in verband met lelijke genezing van de tatoeage). Daarnaast moet geadviseerd worden om het gebruik van antistollingsmedicatie altijd te melden voorafgaand aan een operatieve ingreep of tandextractie en bij vaccinaties. Bij dubbele antistolling moet geadviseerd worden om samen met de arts leefregels af te stemmen. Het is belangrijk om aandacht te besteden aan de emotionele en praktische aspecten van het langdurig gebruik van antistolling. Zie voor een overzicht van de belangrijkste kenmerken (werking, toedieningsvorm, monitoring, wat te doen bij bloeding of overdosering) van de verschillende antistollingsmedicatie in de bijlage Overzicht antistollingsmedicatie.
Tijdens de ontwikkeling van deze module is gebleken dat er binnen deze module noodzaak is voor het ontwikkelen van een keuzehulp: antistollingsmedicatie bij patiënten met een Fontan circulatie
Kostenaspecten
Ten aanzien van de kosten van de medicatie is behandeling met VKA verreweg het goedkoopst ~0.01 euro per dag, tan aanzien van acetylsalicylzuur (1 euro/per dag) en DOAC-rivaroxaban (~4 euro/dag). Waarbij bij VKA er meer kosten zijn ten aanzien controle en dosering op de kinderleeftijd.
Gelijkheid ((health) equity/equitable)
Het gebruik van VKA, acetylsalicylzuur en DOACs leiden naar verwachting niet tot een verschil van gezondheidsgelijkheid gezien de beperkte kosten en goede verkrijgbaarheid van het middel, hoewel DOACs op dit moment duidelijk duurder zijn t.o.v. de ander anticoagulantia maar ten aanzien van de VKA het voordeel hebben dat het niet gecontroleerd hoeft te worden.
Aanvaardbaarheid
Ethische aanvaardbaarheid
De interventie lijkt aanvaardbaar voor de betrokkenen. Er zijn geen ethische bezwaren voor het gebruik van tromboseprofylaxe.
Duurzaamheid
Geneesmiddelen, in dit geval antistollingsmedicatie, hebben waarschijnlijk minder impact op het milieu dan extra ziekenhuisopnames, die nodig zijn bij het optreden van trombotische complicaties.
Haalbaarheid
De interventie is haalbaar. De interventie is over het algemeen al standaardzorg in de praktijk.
Rationale van aanbeveling-1: weging van argumenten voor en tegen de interventies
Ondanks dat de kwaliteit van het bewijs laag is, is er duidelijke overeenstemming dat antistollingsmedicatie na het completeren van de Fontan circulatie noodzakelijk is. Hoewel bloedingscomplicaties ongewenst zijn, wegen de potentiële gevolgen van trombose zwaarder, waardoor tromboseprofylaxe sterk aanbevolen wordt.
In de literatuur wordt het risico op trombose het hoogst geacht in de periode van drie tot zes maanden na de operatie. We hebben op basis van expert opinion gekozen voor de aanbeveling om na zes maanden het type (acetylsalicylzuur) of dosering van de anticoagulantia (DOAC) aan te passen.
Rationale van aanbeveling-2: weging van argumenten voor en tegen de interventies
Ondanks dat de kwaliteit van het bewijs laag is, is er duidelijke overeenstemming dat antistollingsmedicatie na het completeren van de Fontan circulatie noodzakelijk is. Hoewel bloedingscomplicaties ongewenst zijn, wegen de potentiële gevolgen van trombose zwaarder, waardoor tromboseprofylaxe sterk aanbevolen wordt.
In de literatuur wordt het risico op trombose het hoogst geacht in de periode van drie tot zes maanden na de operatie. We hebben op basis van expert opinion gekozen voor de aanbeveling om na zes maanden het type (acetylsalicylzuur) of dosering van de anticoagulantia (DOAC) aan te passen.
Rationale van aanbeveling-3: weging van argumenten voor en tegen de interventies
Ondanks dat de kwaliteit van het bewijs laag is, is er duidelijke overeenstemming dat antistollingsmedicatie na het completeren van de Fontan circulatie noodzakelijk is. Hoewel bloedingscomplicaties ongewenst zijn, wegen de potentiële gevolgen van trombose zwaarder, waardoor tromboseprofylaxe sterk aanbevolen wordt.
Vanuit de beschikbare literatuur is evident dat een bilaterale VCS, kleine pulmonaaltakken/VCS, stent in pulmonaaltakken, niet afgesloten blind-eindigende pulmonalisstomp, stasis in hypoplastische ventrikel een verhoogd risico op trombose geven. Hoewel niet in alle literatuur een onderscheid wordt gemaakt ten aanzien van genoemde risicofactoren voor trombose bevelen we op basis van expert opinion aan dat bij de patiënten met genoemde risicofactoren het advies voor therapeutische dosering van orale anticoagulantia (VKA of DOAC) ook na zes maanden wordt voortgezet, waarbij de DOAC een gunstiger profiel lijkt te hebben in de balans tussen voorkomen van trombose en het risico op (ernstige) bloedingen.
Rationale van aanbeveling-4: weging van argumenten voor en tegen de interventies voor
Deze rationale en aanbeveling is conform de module Kunstkleppen van deze richtlijn. Antistollingsmanagement bij kinderen na een hartklepvervanging vereist een zorgvuldige, geïndividualiseerde benadering, waarbij de voordelen van trombo-embolische preventie worden afgewogen tegen de risico’s van bloedingen. Vitamine K-antagonisten blijven de hoeksteen van de therapie voor patiënten met mechanische hartkleppen.
Onderbouwing
Achtergrond
The Fontan-circulation is the palliative approach for patients born with a congenital cardiac defect in which only one ventricle is capable to sustain the systemic circulation. Systemic venous return is redirected to the pulmonary circulation in a 2-step approach by connecting the superior caval vein to the pulmonary artery-system (see module Glenn-shunt). The final step to create the Fontan circulation is to connect the inferior caval vein to the pulmonary circulation, either by a lateral tunnel, partly incorporating atrial tissue in the Fontan circulation, or by placing a rigid Gortex tube between the ICV and the pulmonary arteries. In both approaches a fenestration can be made between the Fontan-pathway and the atrium, creating a right-to-left shunt.
The risk of thrombosis is increased after Fontan completion with reported incidences of venous thrombosis ranging from 4 to 19% and the incidence of stroke ranging from 3 to 19% in children with thrombo-prophylaxis (Attard, 2018). Of note, the risk of thrombosis seems to be highest during the first year after Fontan completion (Attard, 2018).
Several pathophysiological mechanisms play a role in the increased risk of thrombosis in single ventricle patients with a Fontan circulation, including endothelial dysfunction, abnormal blood flow, hypercoagulability (Attard, 2018).
Because of the increased risk for thrombosis and thromboembolic events there is consensus that single ventricle patients with a Fontan circulation need some form of thromboprophylactic therapy, in which decreasing the risk of thromboembolic events outweighs the risk of increased bleeding. However, in single ventricle patients with a Fontan circulation several thromboprophylactic regimens were studied and used but it is currently unclear which thromboprophylactic strategy is most favorable for children (Van den Eynde, 2023).
Conclusies / Summary of Findings
1. Studies with follow-up ≤ 1 year
Table 3. Summary of Findings – Comparison of thromboprophylaxis A versus thromboprophylaxis B with outcomes thrombosis, hemorrhage, mortality, ischemic stroke, quality of life (QOL) and adverse events
Population: Children with an indication for the Fontan procedure
Intervention: Thromboprophylaxis A (specified in table)
Comparator: Thromboprophylaxis B (specified in table)
Click here to see both tables in a document
|
Outcome |
Study results and measurements |
Absolute effect estimates |
Certainty of the Evidence (Quality of evidence) |
Summary |
||
|
Aspirin versus warfarin |
||||||
|
|
|
Aspirin |
Warfarin |
|
|
|
|
Thrombotic complications (critical) |
Relative risk: 1.26 (95% CI 0.25 to 6.29)
Based on data from 155 participants in 1 study |
5 per 100 |
4 per 100 |
Very low |
The evidence is very uncertain about the effect of aspirin on thrombotic complications, when compared with warfarin in children with an indication for the Fontan procedure
(Ankola, 2021) |
|
|
Difference: 1 more per 100 (95% CI 6 fewer to 8 more) |
||||||
|
- |
- |
No GRADE |
No evidence was found regarding the effect of aspirin on hemorrhage, mortality, ischemic stroke, quality of life (QOL), and adverse events, when compared with warfarin in children with an indication for the Fontan procedure |
||
|
Aspirin versus enoxaparin |
||||||
|
|
|
Aspirin |
Enoxaparin |
|
|
|
|
Thrombotic complications (critical) |
Relative risk: 0.63 (95% CI 0.08 to 4.99)
Based on data from 116 participants in 1 study |
5 per 100 |
8 per 100 |
Very low |
The evidence is very uncertain about the effect of aspirin on thrombotic complications, when compared with enoxaparin in children with an indication for the Fontan procedure
(Ankola, 2021) |
|
|
Difference: 3 fewer per 100 (95% CI 18 fewer to 12 more) |
||||||
|
- |
- |
|
No evidence was found regarding the effect of aspirin on hemorrhage, mortality, ischemic stroke, quality of life (QOL), and adverse events, when compared with enoxaparin in children with an indication for the Fontan procedure |
||
|
Warfarin versus enoxaparin |
||||||
|
|
|
Warfarin |
Enoxaparin |
|
|
|
|
Thrombotic complications (critical) |
Relative risk: 0.50 (95% CI 0.05 to 5.10)
Based on data from 65 participants in 1 study |
4 per 100 |
8 per 100 |
Very low |
The evidence is very uncertain about the effect of warfarin on thrombotic complications, when compared with enoxaparin in children with an indication for the Fontan procedure
(Ankola, 2021) |
|
|
Difference: 4 fewer per 100 (95% CI 19 fewer to 12 more) |
||||||
|
- |
- |
No GRADE |
No evidence was found regarding the effect of warfarin on hemorrhage, mortality, ischemic stroke, quality of life (QOL), and adverse events, when compared with enoxaparin in children with an indication for the Fontan procedure |
||
|
Aspirin versus rivaroxaban |
||||||
|
|
|
Aspirin |
Rivaroxaban |
|
|
|
|
Thrombotic complications (critical) |
Relative risk: 3.76 (95% CI 0.35 to 40.04)
Based on data from 98 participants in 1 study |
6 per 100 |
2 per 100 |
Very low |
The evidence is very uncertain about the effect of aspirin on thrombotic complications, when compared with rivaroxaban in children with an indication for the Fontan procedure
(McCrindle, 2021) |
|
|
Difference: 4 more per 100 (95% CI 4 fewer to 13 more) |
||||||
|
Hemorrhage (important) |
Major bleeding |
Relative risk: 0.62 (95% CI 0.03 to 14.80)
Based on data from 98 participants in 1 study |
0 per 100 |
2 per 100 |
Very low |
The evidence is very uncertain about the effect of aspirin on major bleeding, when compared with rivaroxaban in children with an indication for the Fontan procedure
(McCrindle, 2021) |
|
Difference: 2 fewer per 100 (95% CI 7 fewer to 4 more) |
||||||
|
Clinically relevant nonmajor bleeding |
Relative risk: 1.41 (95% CI 0.34 to 5.95)
Based on data from 98 participants in 1 study |
9 per 100 |
6 per 100 |
|
The evidence is very uncertain about the effect of aspirin on clinically relevant nonmajor bleeding, when compared with rivaroxaban in children with an indication for the Fontan procedure
(McCrindle, 2021) |
|
|
Difference: 3 fewer per 100 (95% CI 9 fewer to 14 more) |
||||||
|
Trivial bleeding |
Relative risk: 1.08 (95% CI 0.61 to 1.91)
Based on data from 98 participants in 1 study |
35 per 100 |
33 per 100 |
Very low |
The evidence is very uncertain about the effect of aspirin on trivial bleeding, when compared with rivaroxaban in children with an indication for the Fontan procedure
(McCrindle, 2021) |
|
|
Difference: 2 more per 100 (95% CI 17 fewer to 22 more) |
||||||
|
Ischemic stroke (important) |
Relative risk: 5.57 (95% CI 0.23 to 133.19)
Based on data from 98 participants in 1 study |
3 per 100 |
0 per 100 |
Very low |
The evidence is very uncertain about the effect of aspirin on ischemic stroke, when compared with rivaroxaban in children with an indication for the Fontan procedure
(McCrindle, 2021) |
|
|
Difference: 3 more per 100 (95% CI 4 fewer to 10 more) |
||||||
|
Adverse events (important) |
Relative risk: 0.99 (95% CI 0.84 to 1.18)
Based on data from 98 participants in 1 study |
85 per 100 |
86 per 100 |
Moderate
|
There is likely little to no effect of aspirin on adverse events, when compared with rivaroxaban in children with an indication for the Fontan procedure
(McCrindle, 2021) |
|
|
Difference: 1 fewer per 100 (95% CI 15 fewer to 14 more) |
||||||
|
- |
- |
No GRADE |
No evidence was found regarding the effect of aspirin on mortality, and quality of life (QOL), when compared with rivaroxaban in children with an indication for the Fontan procedure |
||
1. Risk of bias: some concerns (-1 level). No exclusion criteria were defined, and it is unclear whether the outcomes of interest were not present at the start of the study. Imprecision: very serious (-2 levels). The confidence interval crosses both borders of clinical relevance.
2. Risk of bias: some concerns (-1 level). No exclusion criteria were defined, and it is unclear whether the outcomes of interest were not present at the start of the study. Imprecision: very serious (-2 levels). The confidence interval crosses both borders of clinical relevance.
3. Risk of bias: some concerns (-1 level). No exclusion criteria were defined, and it is unclear whether the outcomes of interest were not present at the start of the study. Imprecision: very serious (-2 levels). The confidence interval crosses both borders of clinical relevance.
4. Risk of bias: some concerns (-1 level). Method of randomization is unclear, no reporting on allocation concealment, and the study was not blinded. Imprecision: very serious (-2 levels). The confidence interval crosses both borders of clinical relevance.
5. Risk of bias: some concerns (-1 level). Method of randomization is unclear, no reporting on allocation concealment, and the study was not blinded. Imprecision: very serious (-2 levels). The confidence interval crosses both borders of clinical relevance.
6. Risk of bias: some concerns (-1 level). Method of randomization is unclear, no reporting on allocation concealment, and the study was not blinded. Imprecision: very serious (-2 levels). The confidence interval crosses both borders of clinical relevance.
7. Risk of bias: some concerns (-1 level). Method of randomization is unclear, no reporting on allocation concealment, and the study was not blinded. Imprecision: very serious (-2 levels). The confidence interval crosses both borders of clinical relevance.
8. Risk of bias: some concerns (-1 level). Method of randomization is unclear, no reporting on allocation concealment, and the study was not blinded. Imprecision: very serious (-2 levels). The confidence interval crosses both borders of clinical relevance.
9. Risk of bias: some concerns (-1 level). Method of randomization is unclear, no reporting on allocation concealment, and the study was not blinded.
2. Studies with follow-up > 1 year
Table 4. Summary of Findings – Comparison of thromboprophylaxis A versus thromboprophylaxis B with outcomes thrombosis, hemorrhage, mortality, ischemic stroke, quality of life (QOL) and adverse events
Population: Children with an indication for the Fontan procedure
Intervention: Thromboprophylaxis A (specified in table)
Comparator: Thromboprophylaxis B (specified in table)
|
Outcome |
Study results and measurements |
Absolute effect estimates |
Certainty of the Evidence (Quality of evidence) |
Summary |
|||
|
Aspirin versus warfarin |
|||||||
|
|
|
Aspirin |
Warfarin |
|
|
||
|
Thrombotic complications (critical) |
Relative risk: 1.42 (95% CI 0.35 to 40.04)
Based on data from 1533 participants in 7 studies |
19 per 100 |
9 per 100 |
|
The evidence is very uncertain about the effect of aspirin on thrombotic complications, when compared with warfarin in children with an indication for the Fontan procedure
(Al-Jazairi, 2019; Egbe, 2017; Iyengar, 2016; McCrindle, 2013; Pessotti, 2014; Potter, 2013; Seipelt, 2002) |
||
|
Difference: 5 more per 100 (95% CI 9 fewer to 19 more) |
|||||||
|
Hemorrhage (important) |
Major bleeding |
Relative risk: 0.89 (95% CI 0.74 to 1.08)
Based on data from 147 participants in 2 studies |
12 per 100 |
14 per 100 |
Very low |
The evidence is very uncertain about the effect of aspirin on major bleeding, when compared with warfarin in children with an indication for the Fontan procedure
(Al-Jazairi, 2019; McCrindle, 2013) |
|
|
Difference: 0 fewer per 100 (95% CI 11 fewer to 10 more) |
|||||||
|
Clinically relevant nonmajor bleeding |
Relative risk: 0.45 (95% CI 0.20 to 1.01)
Based on data from 32 participants in 1 study |
31 per 100 |
69 per 100 |
|
The evidence is very uncertain about the effect of aspirin on clinically relevant nonmajor bleeding, when compared with warfarin in children with an indication for the Fontan procedure
(Al-Jazairi, 2019) |
||
|
Difference: 38 fewer per 100 (95% CI 70 fewer to 5 more) |
|||||||
|
Minor bleeding |
Relative risk: 0.57 (95% CI 0.09 to 3.61)
Based on data from 243 participants in 2 studies |
27 per 100 |
49 per 100 |
Very low |
The evidence is very uncertain about the effect of aspirin on minor bleeding, when compared with warfarin in children with an indication for the Fontan procedure
(Al-Jazairi, 2019; McCrindle, 2013) |
||
|
Difference: 22 fewer per 100 (95% CI 53 fewer to 10 more) |
|||||||
|
Mortality (important) |
Relative risk: 2.84 (95% CI 0.12 to 68.36)
Based on data from 111 participants in 1 study |
2 per 100 |
0 per 100 |
Very low |
The evidence is very uncertain about the effect of aspirin on mortality, when compared with warfarin in children with an indication for the Fontan procedure
(McCrindle, 2013; Pessotti, 2014) |
||
|
Difference: 2 more per 100 (95% CI 3 fewer to 7 more) |
|||||||
|
Relative risk: 0.20 (95% CI 0.01 to 3.85)
Based on data from 30 participants in 1 study |
0 per 100 |
13 per 100 |
|||||
|
Difference: 13 fewer per 100 (95% CI 33 fewer to 6 more) |
|||||||
|
- |
- |
No GRADE |
No evidence was found regarding the effect of aspirin on ischemic stroke, quality of life (QOL), and adverse events, when compared with warfarin in children with an indication for the Fontan procedure |
|||
|
DOACs versus antiplatelets |
|||||||
|
|
|
DOACs |
Antiplatelets |
|
|
||
|
Thrombotic complications (critical) |
Relative risk: 0.17 (95% CI 0.01 to 3.18)
Based on data from 79 participants in 1 study |
0 per 100 |
7 per 100 |
Very low |
The evidence is very uncertain about the effect of DOACs on thrombotic complications, when compared with antiplatelets in children with an indication for the Fontan procedure
(Kawamatsu, 2021) |
||
|
Difference: 7 fewer per 100 (95% CI 16 fewer to 2 more) |
|||||||
|
Hemorrhage (important) |
Major bleeding |
Relative risk: 0.40 (95% CI 0.04 to 3.66)
Based on data from 79 participants in 1 study |
3 per 100 |
7 per 100 |
|
The evidence is very uncertain about the effect of DOACs on major bleeding, when compared with antiplatelets in children with an indication for the Fontan procedure
(Kawamatsu, 2021) |
|
|
Difference: 4 fewer per 100 (95% CI 14 fewer to 5 more) |
|||||||
|
Minor bleeding |
Relative risk: 1.19 (95% CI 0.08 to 18.43)
Based on data from 79 participants in 1 study |
3 per 100 |
2 per 100 |
Very low |
The evidence is very uncertain about the effect of DOACs on minor bleeding, when compared with antiplatelets in children with an indication for the Fontan procedure
(Kawamatsu, 2021) |
||
|
Difference: 0 per 100 (95% CI 7 fewer to 7 more) |
|||||||
|
- |
- |
No GRADE |
No evidence was found regarding the effect of DOACs on mortality, ischemic stroke, quality of life (QOL), and adverse events, when compared with antiplatelets in children with an indication for the Fontan procedure |
|||
|
DOACs versus VKAs |
|||||||
|
|
|
DOACs |
VKAs |
|
|
||
|
Thrombotic complications (critical) |
Relative risk: 0.10 (95% CI 0.01 to 1.80)
Based on data from 77 participants in 1 study |
0 per 100 |
12 per 100 |
|
The evidence is very uncertain about the effect of DOACs on thrombotic complications, when compared with VKAs in children with an indication for the Fontan procedure
(Kawamatsu, 2021) |
||
|
Difference: 12 fewer per 100 (95% CI 23 fewer to 1 fewer) |
|||||||
|
Hemorrhage (important) |
Major bleeding |
Relative risk: 0.19 (95% CI 0.02 to 1.50)
Based on data from 77 participants in 1 study |
3 per 100 |
15 per 100 |
Very low |
The evidence is very uncertain about the effect of DOACs on major bleeding, when compared with VKAs in children with an indication for the Fontan procedure
(Kawamatsu, 2021) |
|
|
Difference: 12 fewer per 100 (95% CI 24 fewer to 0 more) |
|||||||
|
Minor bleeding |
Relative risk: 0.38 (95% CI 0.04 to 3.49)
Based on data from 77 participants in 1 study Follow-up: 95 months (mean) |
3 per 100 |
7 per 100 |
|
The evidence is very uncertain about the effect of DOACs on major bleeding, when compared with VKAs in children with an indication for the Fontan procedure
(Kawamatsu, 2021) |
||
|
Difference: 5 fewer per 100 (95% CI 14 fewer to 5 more) |
|||||||
|
- |
- |
No GRADE |
No evidence was found regarding the effect of DOACs on mortality, ischemic stroke, quality of life (QOL), and adverse events, when compared with VKAs in children with an indication for the Fontan procedure |
|||
|
DOACs versus combination of an antiplatelet and anticoagulant |
|||||||
|
|
|
DOACs |
Combination of an antiplatelet and anticoagulant |
|
|
||
|
Thrombotic complications (critical) |
Relative risk: 1
Based on data from 50 participants in 1 study |
0 per 100 |
0 per 100 |
Very low |
The evidence is very uncertain about the effect of DOACs on thrombotic complications, when compared with a combination of an antiplatelet and anticoagulant in children with an indication for the Fontan procedure
(Kawamatsu, 2021) |
||
|
Difference: 0 fewer per 100
|
|||||||
|
Hemorrhage (important) |
Major bleeding |
Relative risk: 0.05 (95% CI 0.01 to 0.35)
Based on data from 50 participants in 1 study Follow-up: 95 months (mean) |
3 per 100 |
57 per 100 |
Very low |
The evidence is very uncertain about the effect of DOACs on major bleeding, when compared with a combination of an antiplatelet and an anticoagulant in children with an indication for the Fontan procedure
(Kawamatsu, 2021) |
|
|
Difference: 54 fewer per 100 (95% CI 81 fewer to 28 fewer) |
|||||||
|
Minor bleeding |
Relative risk: 1.22 (95% CI 0.05 to 28.21)
Based on data from 50 participants in 1 study Follow-up: 95 months (mean) |
3 per 100 |
0 per 100 |
Very low |
The evidence is very uncertain about the effect of DOACs on major bleeding, when compared with a combination of an antiplatelet and an anticoagulant in children with an indication for the Fontan procedure
(Kawamatsu, 2021) |
||
|
Difference: 3 more per 100 (95% CI 8 fewer to 14 more) |
|||||||
|
- |
- |
No GRADE |
No evidence was found regarding the effect of DOACs on mortality, ischemic stroke, quality of life (QOL), and adverse events, when compared with a combination of an antiplatelet and anticoagulant in children with an indication for the Fontan procedure |
|||
|
Antiplatelets versus VKAs |
|||||||
|
|
|
Antiplatelets |
VKAs |
|
|
||
|
Thrombotic complications (critical) |
Relative risk: 0.57 (95% CI 0.15 to 2.24)
Based on data from 84 participants in 1 study Follow-up: 95 months (mean) |
7 per 100 |
12 per 100 |
Very low |
The evidence is very uncertain about the effect of antiplatelets on thrombotic complications, when compared with VKAs in children with an indication for the Fontan procedure
(Kawamatsu, 2021) |
||
|
Difference: 5 fewer per 100 (95% CI 18 fewer to 7 more) |
|||||||
|
Hemorrhage (important) |
Major bleeding |
Relative risk: 0.48 (95% CI 0.13 to 1.78)
Based on data from 84 participants in 1 study Follow-up: 95 months (mean) |
7 per 100 |
15 per 100 |
Very low |
The evidence is very uncertain about the effect of antiplatelets on major bleeding, when compared with VKAs in children with an indication for the Fontan procedure
(Kawamatsu, 2021) |
|
|
Difference: 8 fewer per 100 (95% CI 21 fewer to 7 more) |
|||||||
|
Minor bleeding |
Relative risk: 0.32 (95% CI 0.03 to 2.93)
Based on data from 84 participants in 1 study Follow-up: 95 months (mean) |
2 per 100 |
7 per 100 |
Very low |
The evidence is very uncertain about the effect of antiplatelets on minor bleeding, when compared with VKAs in children with an indication for the Fontan procedure
(Kawamatsu, 2021) |
||
|
Difference: 5 fewer per 100 (95% CI 14 fewer to 4 more) |
|||||||
|
- |
- |
No GRADE |
No evidence was found regarding the effect of antiplatelets on mortality, ischemic stroke, quality of life (QOL), and adverse events, when compared with VKAs in children with an indication for the Fontan procedure |
|||
|
Antiplatelets versus combination of an antiplatelet and anticoagulant |
|||||||
|
|
|
Antiplatelets |
Combination of an antiplatelet and anticoagulant |
|
|
||
|
Thrombotic complications (critical) |
Relative risk: 2.39 (95% CI 0.03 to 2.93)
Based on data from 57 participants in 1 study Follow-up: 95 months (mean) |
7 per 100 |
0 per 100 |
Very low |
The evidence is very uncertain about the effect of antiplatelets on thrombotic complications, when compared with a combination of an antiplatelet and an anticoagulant in children with an indication for the Fontan procedure
(Kawamatsu, 2021) |
||
|
Difference: 7 more per 100 (95% CI 5 fewer to 19 more) |
|||||||
|
Hemorrhage (important) |
Major bleeding |
Relative risk: 0.12 (95% CI 0.04 to 0.40)
Based on data from 57 participants in 1 study Follow-up: 95 months (mean) |
7 per 100 |
57 per 100 |
Very low |
The evidence is very uncertain about the effect of antiplatelets on major bleeding, when compared with a combination of an antiplatelet and an anticoagulant in children with an indication for the Fontan procedure
(Kawamatsu, 2021) |
|
|
Difference: 50 fewer per 100 (95% CI 77 fewer to 23 fewer) |
|||||||
|
Minor bleeding |
Relative risk: 1.02 (95% CI 0.04 to 23.78)
Based on data from 57 participants in 1 study Follow-up: 95 months (mean) |
2 per 100 |
0 per 100 |
Very low |
The evidence is very uncertain about the effect of antiplatelets on major bleeding, when compared with a combination of an antiplatelet and an anticoagulant in children with an indication for the Fontan procedure
(Kawamatsu, 2021) |
||
|
Difference: 2 fewer per 100 (95% CI 8 fewer to 13 more) |
|||||||
|
- |
- |
No GRADE |
No evidence was found regarding the effect of antiplatelets on mortality, ischemic stroke, quality of life (QOL), and adverse events, when compared with a combination of an antiplatelet and anticoagulant in children with an indication for the Fontan procedure |
|||
|
VKAs versus combination of an antiplatelet and anticoagulant |
|||||||
|
|
|
VKAs |
Combination of an antiplatelet and anticoagulant |
|
|
||
|
Thrombotic complications (critical) |
Relative risk: 3.93 (95% CI 0.23 to 66.86)
Based on data from 55 participants in 1 study Follow-up: 95 months (mean) |
12 per 100 |
0 per 100 |
Very low |
The evidence is very uncertain about the effect of VKAs on thrombotic complications, when compared with a combination of an antiplatelet and an anticoagulant in children with an indication for the Fontan procedure
(Kawamatsu, 2021) |
||
|
Difference: 12 more per 100 (95% CI 1 fewer to 26 more) |
|||||||
|
Hemorrhage (important) |
Major bleeding |
Relative risk: 0.26 (95% CI 0.11 to 0.61)
Based on data from 55 participants in 1 study Follow-up: 95 months (mean) |
15 per 100 |
57 per 100 |
Very low |
The evidence is very uncertain about the effect of VKAs on major bleeding, when compared with a combination of an antiplatelet and an anticoagulant in children with an indication for the Fontan procedure
(Kawamatsu, 2021) |
|
|
Difference: 43 fewer per 100 (95% CI 71 fewer to 14 fewer) |
|||||||
|
Minor bleeding |
Relative risk: 2.50 (95% CI 0.14 to 45.62)
Based on data from 55 participants in 1 study Follow-up: 95 months (mean) |
7 per 100 |
0 per 100 |
Very low |
The evidence is very uncertain about the effect of VKAs on minor bleeding, when compared with a combination of an antiplatelet and an anticoagulant in children with an indication for the Fontan procedure
(Kawamatsu, 2021) |
||
|
Difference: 7 fewer per 100 (95% CI 5 fewer to 20 more) |
|||||||
|
- |
- |
No GRADE |
|
|||
1. Risk of bias: some concerns (-1 level). It is unclear whether the outcomes of interest were not present at the start of the study, confounder adjustment is not consistently conducted across all study, method of randomization is unclear, no blinding, and no reporting on allocation concealment. Imprecision: very serious (-2 levels). The confidence interval crosses both borders of clinical relevance.
2. Risk of bias: some concerns (-1 level). It is unclear whether the outcome of interest was not present at the start of the study, unclear reporting on multivariable statistical analyses, method of randomization unclear, and no blinding. Imprecision: serious (-1 level). The confidence interval crosses one border of clinical relevance.
3. Risk of bias: some concerns (-1 level). It is unclear whether the outcome of interest was not present at the start of the study and unclear reporting on multivariable statistical analyses. Imprecision: serious (-1 level). The confidence interval crosses one border of clinical relevance.
4. Risk of bias: some concerns (-1 level). It is unclear whether the outcome of interest was not present at the start of the study, unclear reporting on multivariable statistical analyses, method of randomization is unclear, and no blinding. Imprecision: very serious (-2 levels). The confidence interval crosses both borders of clinical relevance.
5. Risk of bias: some concerns (-1 level). Method of randomization is clear, no blinding, and no reporting on allocation concealment. Imprecision: very serious (-2 levels). The confidence interval crosses both borders of clinical relevance.
6. Risk of bias: some concerns (-1 level). It is unclear whether the outcome of interest was not present at the start of the study, no proper confounder adjustment was conducted, co-interventions differed between the intervention groups. Imprecision: very serious (-2 levels). The confidence interval crosses both borders of clinical relevance.
7. Risk of bias: some concerns (-1 level). It is unclear whether the outcome of interest was not present at the start of the study, no proper confounder adjustment was conducted, co-interventions differed between the intervention groups. Imprecision: very serious (-2 levels). The confidence interval crosses both borders of clinical relevance.
8. Risk of bias: some concerns (-1 level). It is unclear whether the outcome of interest was not present at the start of the study, no proper confounder adjustment was conducted, co-interventions differed between the intervention groups. Imprecision: very serious (-2 levels). The confidence interval crosses both borders of clinical relevance.
9. Risk of bias: some concerns (-1 level). It is unclear whether the outcome of interest was not present at the start of the study, no proper confounder adjustment was conducted, co-interventions differed between the intervention groups. Imprecision: very serious (-2 levels). The confidence interval crosses both borders of clinical relevance.
10. Risk of bias: some concerns (-1 level). It is unclear whether the outcome of interest was not present at the start of the study, no proper confounder adjustment was conducted, co-interventions differed between the intervention groups. Imprecision: very serious (-2 levels). The confidence interval crosses both borders of clinical relevance.
11. Risk of bias: some concerns (-1 level). It is unclear whether the outcome of interest was not present at the start of the study, no proper confounder adjustment was conducted, co-interventions differed between the intervention groups. Imprecision: very serious (-2 levels). The confidence interval crosses both borders of clinical relevance.
12. Risk of bias: some concerns (-1 level). It is unclear whether the outcome of interest was not present at the start of the study, no proper confounder adjustment was conducted, co-interventions differed between the intervention groups.
13. Risk of bias: some concerns (-1 level). It is unclear whether the outcome of interest was not present at the start of the study, no proper confounder adjustment was conducted, co-interventions differed between the intervention groups. Imprecision: very serious (-2 levels). The confidence interval crosses both borders of clinical relevance.
14. Risk of bias: some concerns (-1 level). It is unclear whether the outcome of interest was not present at the start of the study, no proper confounder adjustment was conducted, co-interventions differed between the intervention groups. Imprecision: very serious (-2 levels). The confidence interval crosses both borders of clinical relevance.
15. Risk of bias: some concerns (-1 level). It is unclear whether the outcome of interest was not present at the start of the study, no proper confounder adjustment was conducted, co-interventions differed between the intervention groups. Imprecision: very serious (-2 levels). The confidence interval crosses both borders of clinical relevance.
16. Risk of bias: some concerns (-1 level). It is unclear whether the outcome of interest was not present at the start of the study, no proper confounder adjustment was conducted, co-interventions differed between the intervention groups. Imprecision: very serious (-2 levels). The confidence interval crosses both borders of clinical relevance.
17. Risk of bias: some concerns (-1 level). It is unclear whether the outcome of interest was not present at the start of the study, no proper confounder adjustment was conducted, co-interventions differed between the intervention groups. Imprecision: very serious (-2 levels). The confidence interval crosses both borders of clinical relevance.
18. Risk of bias: some concerns (-1 level). It is unclear whether the outcome of interest was not present at the start of the study, no proper confounder adjustment was conducted, co-interventions differed between the intervention groups. Imprecision: very serious (-2 levels). The confidence interval crosses both borders of clinical relevance.
19. Risk of bias: some concerns (-1 level). It is unclear whether the outcome of interest was not present at the start of the study, no proper confounder adjustment was conducted, co-interventions differed between the intervention groups.
20. Risk of bias: some concerns (-1 level). It is unclear whether the outcome of interest was not present at the start of the study, no proper confounder adjustment was conducted, co-interventions differed between the intervention groups. Imprecision: very serious (-2 levels). The confidence interval crosses both borders of clinical relevance.
21. Risk of bias: some concerns (-1 level). It is unclear whether the outcome of interest was not present at the start of the study, no proper confounder adjustment was conducted, co-interventions differed between the intervention groups. Imprecision: very serious (-2 levels). The confidence interval crosses both borders of clinical relevance.
22. Risk of bias: some concerns (-1 level). It is unclear whether the outcome of interest was not present at the start of the study, no proper confounder adjustment was conducted, co-interventions differed between the intervention groups.
23. Risk of bias: some concerns (-1 level). It is unclear whether the outcome of interest was not present at the start of the study, no proper confounder adjustment was conducted, co-interventions differed between the intervention groups. Imprecision: very serious (-2 levels). The confidence interval crosses both borders of clinical relevance.
Samenvatting literatuur
Description of studies
A total of ten studies were included in the analysis of the literature. Important study characteristics and results are summarized in table 2. The assessment of the risk of bias is summarized in the risk of bias tables (under the tab ‘Evidence tabellen’).
1. Studies with follow-up ≤ 1 year
Ankola (2021) conducted a single-center retrospective cohort study in patients who underwent the Fontan procedure between January 1, 2006 and December 31, 2016. No further inclusion and exclusion criteria were defined.
McCrindle (2021) conducted an RCT in children with single-ventricle physiology who had the Fontan procedure within four months before enrollment. Further inclusion criteria were age between 2 and 8 years of age and being clinically stable and able to tolerate or nasogastric feedings. Exclusion criteria were having thrombosis, a history of gastrointestinal disease or surgery associated with impaired absorption, active bleeding or high risk of bleeding contraindicating antiplatelet or anticoagulant therapy, including history of intracranial hemorrhage, or contraindications to aspirin or rivaroxaban.
2. Studies with follow-up > 1 year
Egbe (2017) conducted a retrospective cohort study in adult patients (> 18 years) with a history of a Fontan operation at Mayi Clinic, Rochester, Minnesota, from January 1994 to June 2014 who were identified from the hospital’s patient database using free-text search software. Patients with fewer than 12 months’ follow-up after the beginning of the study period were excluded.
Kawamatsu (2021) conducted a retrospective cohort study at three centers specializing in the care of adult patients with congenital heart disease in Japan: St. Luke’s International Hospital, Chiba Prefectural Cardiovascular Center, and Tsukuba University Hospital. Patients over 15 years of age with Fontan circulation who visited any of the three institutions from April 2015 to March 2018 were included. No exclusion criteria were defined.
Al-Jazairi (2019) conducted a retrospective cohort study in patients who underwent Fontan procedures and were identified from the Congenital Heart Defects Registry of the King Faisal Specialist Hospital and Research Centre. Included patients underwent Fontan procedures that took place at King Faisal Specialist Hospital and Research Centre, Riyadh, between 1985 and 2010. Exclusion criteria were patients who died, were lost to follow-up, or had a Fontan redo within one year of the first Fontan procedure.
Iyengar (2016) conducted a cohort study in children who underwent the Fontan procedure, (specifically the extracardiac conduit procedure) and were registered in the Australia and New Zealand Fontan registry. Exclusion criteria were death prior to discharge, patients referred from outside Australia or New Zealand, having mechanical prosthetic valves, patients who were transplanted or died during the first year after the Fontan procedure, patients who were lost to follow-up for any period of more than five years, and patients with < 1 year of follow-up available.
Pessotti (2014) conducted a single-center RCT in children who underwent total cavopulmonary operation with extracardiac conduit in the period between 2008 and 2011. Inclusion criteria were (1) children with heart with univentricular morphology in staged programming, for total cavopulmonary shunt operation. Exclusion criteria were (1) some aspect of anatomical or angiographic features that contraindicated the cavopulmonary operation, including a) main ventricular ejection fraction less than 60%, or significant impairment of atrioventricular valve related to the predominant ventricle, (b) serious anatomical alteration of the pulmonary venous return (untreatable), (c) anatomy of severely unfavorable pulmonary tree, (2) Inability to perform outpatient treatment, (3) any medical condition that prevented the randomization, for example, any indication that the patient used necessarily one of the two drugs evaluated, or contraindication to the use of them, (4) refusal of legal guardian to sign the Post-information Consent.
Seipelt (2002) conducted a cohort study in patients who underwent modified Fontan procedure for various types of univentricular heart conditions. No further inclusion and exclusion criteria were defined.
McCrindle (2013) performed a secondary analysis of an RCT conducted at 6 centers in patients who underwent the Fontan procedure. Exclusion criteria were a recognized indication for long-term anticoagulation; patient characteristics increasing the risk of hemorrhagic complications; known contraindication for heparin, warfarin, or acetylsalicylic acid (ASA); and the inability to supervise therapy because of social or geographic circumstances.
Potter (2013) conducted a cohort study in children who underwent the Fontan procedure and were identified from the New England Fontan registry, which included all patients who lived in the New England area, were born before 1985, and had Fontan surgery between April 1973 and July 1991 at Boston Children's Hospital. Exclusion criteria were surgery limited to cavopulmonary shunts, including those patients with an interrupted inferior vena cava and azygos extension to asuperior vena cava who underwent a bidirectional cavopulmonary shunt, and patients with early post-operative death (< 30 days) or a thromboembolic event within the first 14 days of Fontan surgery.
Table 2. Characteristics of included studies
*For further details, see risk of bias table in the appendix
1Only overall age was reported
2Only overall sex was reported
3Only overall sample size was reported
4Ischemic stroke is described as a separate outcome in this summary of the literature
5Derived from Monagle, 2011 (original article)
Results
1.Thrombotic complications
1.1. Studies with follow-up ≤ 1 year
Aspirin versus warfarin
Ankola (2021) reported the outcome thrombotic complications for the comparison of aspirin and warfarin with a follow-up of 30 days. The risk of thrombotic complications was higher in patients receiving aspirin (5/103; 4.9%) compared to those receiving warfarin (2/52; 3.8%), with RR = 1.26 (95% CI 0.25 to 6.29). The difference between the two groups is considered clinically relevant.
Aspirin versus enoxaparin
Ankola (2021) compared aspirin and enoxaparin for the outcome thrombotic complications with a follow-up of 30 days. Patients treated with aspirin had a lower risk of thrombotic complications (5/103; 4.9%) compared to those treated with enoxaparin (1/13; 7.7%), with RR = 0.63 (95% CI 0.08 to 4.99). The difference between the two groups is considered clinically relevant.
Warfarin versus enoxaparin
Ankola (2021) compared aspirin and enoxaparin for the outcome thrombotic complications with a follow-up of 30 days. Treatment with warfarin was associated with a lower risk of thrombotic complications (2/52; 3.8%) compared to treatment with enoxaparin (1/13; 7.7%), with RR = 0.50 (95% CI 0.05 to 5.10). The difference between the two groups is considered clinically relevant.
Aspirin versus rivaroxaban
McCrindle (2021) compared aspirin and rivaroxaban for the outcome thrombotic complications, with a follow-up period of 12 months. Patients treated with aspirin had a higher risk of thrombotic complications (2/34; 5.9%) compared to patients treated with rivaroxaban (1/64; 1.6%), with RR = 3.76 (95% CI 0.35 to 40.04). The difference between the two groups is considered clinically relevant.
1.2. Studies with follow-up > 1 year
Aspirin versus warfarin
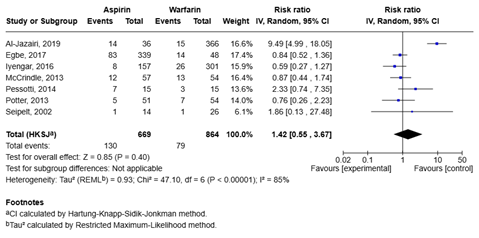
Seven studies (Al-Jazairi, 2019; Egbe, 2017; Iyengar, 2016; McCrindle, 2013; Pessotti, 2014; Potter, 2013; Seipelt, 2002) compared aspirin and warfarin for the outcome thrombotic complications. Treatment with aspirin was associated with a higher risk of thrombotic complications compared to treatment with warfarin, with RR = 1.42 (95% CI 0.55 to 3.67; figure 2). The difference between the two groups is considered clinically relevant.
Figure 2. Forest plot of aspirin versus warfarin for the outcome thrombotic complications
Al-Jazairi (2019) reported an incidence rate of 7.82 events per 1000 patient years for warfarin, versus 5.83 per 1000 patient years for aspirin, giving an incidence rate ratio of 1.34 (95% CI 0.68 to 2.60). The difference in thrombotic complications between the groups is clinically relevant.
Iyengar (2016) reported a hazard ratio (adjusted for factors including among others sex, age at Fontan and number of pre-Fontan procedures) of 2.3 (95% CI 0.7 to 7.4) for the comparison of warfarin and aspirin, meaning that warfarin is associated with a higher risk of thrombotic complications compared to aspirin. This is a clinically relevant difference.
Seipelt (2002) reported a higher incidence rate of thrombotic complications for patients receiving aspirin (1.6 events per 100 patient years) compared to those receiving warfarin (1.1 per 100 patient years), giving an incidence rate ratio of 1.45. The difference between the groups is clinically relevant.
DOACs versus antiplatelets
Kawamatsu (2021) compared DOACs and antiplatelets for the outcome thrombotic complications, with a mean follow-up of 95 ± 64 months. Treatment with DOACs was associated with a lower risk of thrombotic complications (0/36; 0%) compared to treatment with antiplatelets (3/43; 7.0%), with RR = 0.17 (95% CI 0.01 to 3.18). The difference between the two groups is considered clinically relevant.
The incidence rate of thrombotic complications was 0% per patient year for DOACs, compared to 0.68% per patient year for antiplatelets, again reflecting a clinically relevant difference between the groups. The analysis was adjusted for duration after the Fontan procedure.
DOACs versus VKAs
Kawamatsu (2021) compared DOACs and VKAs for the outcome thrombotic complications, with a mean follow-up of 95 ± 64 months. Treatment with DOACs was associated with a lower risk of thrombotic complications (0/36; 0%) compared to treatment with VKAs (5/41; 12.2%), with RR = 0.10 (95% CI 0.01 to 1.80). The difference between the two groups is considered clinically relevant.
The incidence rate of thrombosis was 0% per patient year for DOACs, compared to 1.7% per patient year for VKAs. This is a clinically relevant difference between the groups.
DOACs versus combination of an antiplatelet and anticoagulant
Kawamatsu (2021) compared DOACs and a combination of an antiplatelet and anticoagulant for the outcome thrombotic complications, with a mean follow-up of 95 ± 64 months. No thrombotic complications occurred in either of the two groups.
Antiplatelets versus VKAs
Kawamatsu (2021) compared antiplatelets and VKAs for the outcome thrombotic complications, with a mean follow-up of 95 ± 64 months. Treatment with antiplatelets was associated with a lower risk of thrombotic complications (3/43; 7.0%) compared to treatment with VKAs (5/41; 12.2%), with RR = 0.57 (95% CI 0.15 to 2.24). The difference between the two groups is considered clinically relevant.
The incidence rate of thrombotic complications was 0.7% per patient year for antiplatelets, compared to 1.7% for VKAs. This is a clinically relevant difference between the groups.
Antiplatelets versus combination of an antiplatelet and anticoagulant
Kawamatsu (2021) compared antiplatelets and VKAs for the outcome thrombotic complications, with a mean follow-up of 95 ± 64 months. Treatment with antiplatelets was associated with a higher risk of thrombotic complications (3/43; 7.0%) compared to treatment with a combination of an antiplatelet and anticoagulant (0/14; 0%), with RR = 2.39 (95% CI 0.13 to 43.57). The difference between the two groups is considered clinically relevant.
The incidence rate of thrombotic complications was 0.7% per patient year for antiplatelets compared to 0% per patient year for the combination of an antiplatelet and anticoagulant, which is a clinically relevant difference.
VKAs versus combination of an antiplatelet and anticoagulant
Kawamatsu (2021) compared VKAs and a combination of an antiplatelet and anticoagulant for the outcome thrombotic complications, with a mean follow-up of 95 ± 64 months. Treatment with VKAs was associated with a higher risk of thrombotic complications (5/41; 12.2%) compared to treatment with a combination of an antiplatelet and anticoagulant (0/14; 0%), with RR = 3.93 (95% CI 0.23 to 66.86). The difference between the two groups is considered clinically relevant.
The incidence rate of thrombotic complications was 1.7% per patient year for VKAs, compared to 0% per patient year for the combination of an antiplatelet and anticoagulant. This is a clinically relevant difference.
2. Hemorrhage
2.1. Studies with follow-up ≤ 1 year
2.1.1. Major bleeding
Aspirin versus rivaroxaban
McCrindle (2021) reported that no major bleeding events occurred in the aspirin group, while one major bleeding event occurred in the rivaroxaban group (1/64; 1.6%) during a 12-month follow-up period, with RR = 0.62 (95% CI 0.03 to 14.80). The difference between the two groups is clinically relevant.
2.1.2. Clinically relevant nonmajor bleeding
Aspirin versus rivaroxaban
McCrindle (2021) reported a higher proportion of clinically relevant nonmajor bleedings (3/34; 9%) in the aspirin group compared to the rivaroxaban group (4/64; 6%) during a 12-month follow-up period, with RR = 1.41 (95% CI 0.34 to 5.95). The difference between the groups is considered clinically relevant.
2.1.3. Trivial bleeding events
Aspirin versus rivaroxaban
McCrindle (2021) reported that the proportion of trivial bleedings was higher in the group of patients treated with aspirin (12/34; 35%) compared to the patients treated with rivaroxaban (21/64; 33%) during a 12-month follow-up period, with RR = 1.08 (95% CI 0.61 to 1.91). The difference between the groups is not considered clinically relevant.
2.2. Studies with follow-up > 1 year
2.2.1. Major bleeding
Aspirin versus warfarin
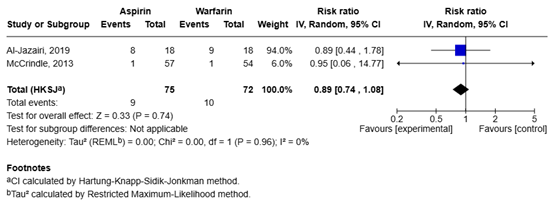
Al-Jazairi (2019) compared aspirin and warfarin for the outcome major bleeding. The risk of major bleeding was lower in patients treated with aspirin (8/18; 44.4%) compared to patients treated with warfarin (9/18; 50%), with RR = 0.89 (95% CI 0.44 to 1.78). The difference is not considered clinically relevant.
Additionally, Al-Jazairi (2019) reported an incidence rate of 3.70 events per 1000 patient years for warfarin, versus 2.91 per 1000 patient years for aspirin, giving an incidence rate ratio of 1.34 (95% CI 0.68 to 2.60). Warfarin was associated with a higher incidence rate of major bleeding events compared to aspirin, the difference between the groups is clinically relevant.
McCrindle (2013) compared aspirin and warfarin for the outcome major bleeding. In both groups one major bleeding event occurred, the risk was 2% (1/57) in the aspirin group and 2% (1/54) in the warfarin group, with RR = 0.95 (95% CI 0.06 to 14.77). The difference is not clinically relevant.
The pooled relative risk for the comparison of aspirin and warfarin is 0.89 (95% CI 0.74 to 1.08), meaning that aspirin is associated with a lower risk of major bleeding events compared to warfarin (Figure 3). The difference is not considered to be clinically relevant.
Figure 3. Forest plot of aspirin versus warfarin for the outcome major bleeding
DOACs versus antiplatelets
Kawamatsu (2021) compared DOACs and antiplatelets for the outcome major bleeding, with a mean follow-up of 95 ± 64 months. The proportion of major bleeding events was lower in the group of patients treated with DOACs (1/36; 2.8%) compared to the group treated with antiplatelets (3/43; 7.0%), with RR = 0.40 (95% CI 0.04 to 3.66). This represents a clinically relevant difference.
The incidence rate of major bleeding was 0.6% per patient year for DOACs, compared to 5.1% per patient year for antiplatelets. This is a clinically relevant difference. The analyses was adjusted for duration after the Fontan procedure.
DOACs versus VKAs
Kawamatsu (2021) compared DOACs and VKAs for the outcome major bleeding, with a mean follow-up of 95 ± 64 months. Treatment with DOACs was associated with a lower risk of major bleeding events (1/36; 2.8%) compared to treatment with VKAs (6/41; 14.6%), with RR = 0.19 (95% CI 0.02 to 1.50). The difference between the groups is clinically relevant.
The incidence rate of major bleeding was 0.6% per patient year for DOACs, compared to 2.0% per patient year for VKAs, which is a clinically relevant difference.
DOACs versus combination of an antiplatelet and an anticoagulant
Kawamatsu (2021) compared DOACs and a combination of an antiplatelet and an anticoagulant for the outcome major bleeding, with a mean follow-up of 95 ± 64 months. Treatment with DOACs was associated with a lower risk of major bleeding events (1/36; 2.8%) compared to treatment with a combination of an antiplatelet and an anticoagulant (8/14; 57.1%), with RR = 0.05 (95% CI 0.01 to 0.35). The difference between the groups is clinically relevant.
The incidence rate of major bleeding was 0.6% per patient year for DOACs, compared to 5.1% for the combination of an antiplatelet and an anticoagulant. The difference is clinically relevant.
Antiplatelets versus VKAs
Kawamatsu (2021) compared antiplatelets and VKAs for the outcome major bleeding, with a mean follow-up of 95 ± 64 months. Treatment with antiplatelets was associated with a lower risk of major bleeding events (3/43; 7.0%) compared to treatment with VKAs (6/41; 14.6%), with RR = 0.48 (95% CI 0.13 to 1.78). The difference between the groups is clinically relevant.
The incidence rate of major bleeding was 0.7% per patient year for antiplatelets, compared to 2.0% per patient year for VKAs, which is a clinically relevant difference.
Antiplatelets versus combination of an antiplatelet and an anticoagulant
Kawamatsu (2021) compared antiplatelets and a combination of an antiplatelet and an anticoagulant for the outcome major bleeding, with a mean follow-up of 95 ± 64 months. Treatment with antiplatelets was associated with a lower risk of major bleeding events (3/43; 7.0%) compared to treatment with a combination of an antiplatelet and an anticoagulant (8/14; 57.1%), with RR = 0.12 (95% CI 0.04 to 0.40). This represents a clinically relevant difference between the groups.
The incidence rate of major bleeding was 0.7% per patient year for antiplatelets, compared to 5.1% per patient year for the combination of an antiplatelet and an anticoagulant, again reflecting a clinically relevant difference.
VKAs versus combination of an antiplatelet and an anticoagulant
Kawamatsu (2021) compared VKAs and a combination of an antiplatelet and an anticoagulant for the outcome major bleeding, with a mean follow-up of 95 ± 64 months. Treatment with VKAs was associated with a lower risk of major bleeding events (6/41; 14.6%) compared to treatment with a combination of an antiplatelet and an anticoagulant (8/14; 57.1%), with RR = 0.26 (95% CI 0.11 to 0.61). This is a clinically relevant difference.
The incidence rate of major bleeding was 2.0% per patient year for VKAs, compared to 5.1% per patient year for the combination of an antiplatelet and an anticoagulant. The difference is clinically relevant.
2.2.2. Minor bleeding
Aspirin versus warfarin
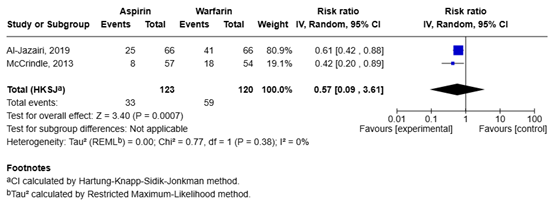
Al-Jazairi (2019) compared aspirin and warfarin for the outcome clinically relevant nonmajor bleeding. Treatment with aspirin was associated with a lower risk of minor bleeding events (25/66; 37.9%) compared to treatment with warfarin (41/66; 62.1%), with RR = 0.61 (95% CI 0.42 to 0.88). The difference between the groups is clinically relevant.
McCrindle (2013) compared aspirin and warfarin for the outcome minor bleeding with a follow-up period of 2.5 years. Treatment with aspirin was associated with a lower risk of minor bleeding events (8/57; 14%) compared to treatment with warfarin (18/54; 33%), with RR = 0.42 (95% CI 0.20 to 0.89). The difference between the groups is clinically relevant.
The pooled relative risk for the comparison of aspirin and warfarin is 0.57 (95% CI 0.09 to 3.61), meaning that aspirin is associated with a lower risk of minor bleeding events compared to warfarin (Figure 4). The difference is considered to be clinically relevant.
Figure 4. Forest plot of aspirin versus warfarin for the outcome minor bleeding
DOACs versus antiplatelets
Kawamatsu (2021) compared DOACs and antiplatelets for the outcome minor bleeding, with a mean follow-up of 95 ± 64 months. Treatment with DOACs was associated with a higher risk of minor bleeding events (1/36; 2.8%) compared to treatment with antiplatelets (1/43; 2.3%), with RR = 1.19 (95% CI 0.08 to 18.43). The difference between the groups is not considered clinically relevant.
DOACs versus VKAs
Kawamatsu (2021) compared DOACs and VKAs for the outcome minor bleeding, with a mean follow-up of 95 ± 64 months. Treatment with DOACs was associated with a lower risk of minor bleeding events (1/36; 2.8%) compared to treatment with VKAs (3/41; 7.3%), with RR = 0.38 (95% CI 0.04 to 3.49). The difference between the groups is clinically relevant.
DOACs versus combination of an antiplatelet and an anticoagulant
Kawamatsu (2021) compared DOACs and a combination of an antiplatelet and an anticoagulant for the outcome minor bleeding, with a mean follow-up of 95 ± 64 months. Treatment with DOACs was associated with a higher risk of minor bleeding events (1/36; 2.8%) compared to treatment with a combination of an antiplatelet and anticoagulant (0/14; 0%), with RR = 1.22 (95% CI 0.05 to 28.21). This is not a clinically relevant difference between the groups.
Antiplatelets versus VKAs
Kawamatsu (2021) compared antiplatelets and VKAs for the outcome minor bleeding, with a mean follow-up of 95 ± 64 months. Treatment with antiplatelets was associated with a lower risk of minor bleeding events (1/43; 2.3%) compared to treatment with VKAs (3/41; 7.3%), with RR = 0.32 (95% CI 0.03 to 2.93). The difference between the groups is clinically relevant.
Antiplatelets versus combination of an antiplatelet and anticoagulant
Kawamatsu (2021) compared antiplatelets and a combination of an antiplatelet and anticoagulant for the outcome minor bleeding, with a mean follow-up of 95 ± 64 months. Treatment with antiplatelets was associated with a higher risk of minor bleeding events (1/43; 2.3%) compared to treatment with a combination of a antiplatelets and anticoagulants (0/14; 0%), with RR = 1.02 (95% CI 0.04 to 23.78). The difference is not clinically relevant.
VKAs versus combination of an antiplatelet and anticoagulant
Kawamatsu (2021) compared VKAs and a combination of an antiplatelet and anticoagulant for the outcome minor bleeding, with a mean follow-up of 95 ± 64 months. Treatment with VKAs was associated with a higher risk of minor bleeding events (3/41; 7.3%) compared to treatment with a combination of an antiplatelet and anticoagulant (0/14; 0%), with RR = 2.50 (95% CI 0.14 to 45.62). This is considered a clinically relevant difference.
2.2.3. Clinically relevant nonmajor bleeding
Aspirin versus warfarin
Al-Jazairi (2019) compared aspirin and warfarin for the outcome clinically relevant nonmajor bleeding. Treatment with aspirin was associated with a higher risk of clinically relevant nonmajor bleeding events (5/16; 31.3%) compared to treatment with warfarin (11/16; 68.8%), with RR = 0.45 (95% CI 0.20 to 1.01). The difference between the groups is clinically relevant.
3. Mortality
3.1. Studies with follow-up ≤ 1 year
No evidence was found for the outcome mortality.
3.2. Studies with follow-up > 1 year
Aspirin versus warfarin
Pessotti (2014) compared aspirin and warfarin for the outcome mortality, with a follow-up period of 24 months and showed that no patients died in the aspirin group, compared to two in the warfarin group (2/15; 13.3%), with RR = 0.20 (95% CI 0.01 to 3.85). The difference between the groups is considered clinically relevant.
McCrindle (2013) compared aspirin and warfarin for the outcome mortality, with a follow-up period of 2.5 years and showed that one patient died in the aspirin group (1/57; 2%), compared to no patients in the warfarin group (0%), with RR = 2.84 (95% CI 0.12 to 68.36). The difference between the groups is clinically relevant.
4. Ischemic stroke
4.1. Studies with follow-up ≤ 1 year
Aspirin versus rivaroxaban
McCrindle (2021) reported that ischemic stroke occurred in one patient in the aspirin group (1/34; 3%), while no patients in the rivaroxaban group had an ischemic stroke, RR = 5.6 (95% CI 0.23 to 133.19). The difference between the groups is considered to be clinically relevant.
4.2. Studies with follow-up > 1 year
No evidence was found for the outcome ischemic stroke.
5. Quality of life (QOL)
No evidence was found for the outcome quality of life (QOL).
6. Adverse events
6.1. Studies with follow-up ≤ 1 year
Aspirin versus rivaroxaban
McCrindle (2021) reported that the proportion of patients experiencing at least one adverse effect during the study period was similar in both groups (29/34; 85% in patients receiving aspirin and 55/64; 86% in patients treated with rivaroxaban), with RR = 0.99 (95% CI 0.84 to 1.18).
6.2. Studies with follow-up > 1 year
No evidence was found for the outcome adverse events.
Zoeken en selecteren
A systematic review of the literature was performed to answer the following question(s):
What are the favorable and unfavorable effects of the use of thromboprophylaxis A compared with the use of thromboprophylaxis B for children with an indication for the Fontan procedure?
Table 1. PICO
| Patients | Children with an indication for the Fontan procedure |
| Intervention |
Thromboprophylaxis A
|
| Control | Thromboprophylaxis B |
| Outcomes | Ischemic stroke, thrombotic complications (including pulmonary embolism), hemorrhage, mortality, quality of life (QOL), adverse events |
| Other selection criteria |
Study design: systematic reviews, meta-analysis, randomized controlled trials, other comparative studies Period: 2014 |
Relevant outcome measures
The guideline panel considered thrombotic complications (including pulmonary embolism) as a critical outcome measure for decision making; and the other outcomes as important outcome measures for decision making.
A priori, the guideline panel did not define the outcome measures listed above but used the definitions used in the studies.
The guideline panel defined a difference of 25% (0.8 ≥ RR ≥ 1.25) as minimal clinically (patient) important difference.
Search and select (Methods)
A systematic literature search was performed by a medical information specialist using the following bibliographic databases: Embase.com and Ovid/Medline. Both databases were searched from 2014 (publication of former Dutch guideline) to 9 July 2024 for systematic reviews, RCTs and observational studies. Systematic searches were completed using a combination of controlled vocabulary/subject headings (e.g., Emtree-terms, MeSH) wherever they were available and natural language keywords. The overall search strategy was derived from two primary search concepts: (1) Fontan circulation; (2) thromboprophylaxis/anticoagulants. Duplicates were removed using EndNote software. After deduplication a total of 479 records were imported for title/abstract screening.
Studies were selected based on the following criteria:
- Study design: systematic reviews in which searches were performed in at least two databases, with a detailed search strategy, risk of bias assessment and results of individual studies available, randomized controlled trials, or (observational) comparative studies;
- Full-text English or Dutch language publication;
- Studies according to the PICO; and
- Studies including at least twenty participants (ten per group).
Initially, 24 studies were selected based on title and abstract screening. After reading the full text, 17 studies were excluded (see the exclusion table under the tab ‘Evidence tabellen’), and seven studies were included.
In addition to the systematic literature search, we also screened the title and abstract of four additional studies that were included in the conclusions of the module on Fontan in the TRAMPOLINE protocol (former Dutch guideline). One study (Khairy, 2008) did not fit the PICO (no comparison between thromboprophylactic strategies was made). After reading the full text, three studies were included in this summary of literature (Seipelt, 2002; McCrindle, 2013; Potter, 2013).
Referenties
- Al-Jazairi AS, Al Alshaykh HA, Di Salvo G, De Vol EB, Alhalees ZY. Assessment of Late Thromboembolic Complications Post-Fontan Procedure in Relation to Different Antithrombotic Regimens: 30-Years' Follow-up Experience. Ann Pharmacother. 2019 Aug;53(8):786-793. doi: 10.1177/1060028019829860. Epub 2019 Feb 21. PMID: 30788973.
- Ankola AA, Anderson BR, Choudhury TA, Rochelson E, Corda R, Bacha EA, Smerling AJ. Early Thromboprophylaxis Initiation is Associated With Reduced Fontan Thromboses in the Early Postoperative Period. Semin Thorac Cardiovasc Surg. 2021 Autumn;33(3):806-813. doi: 10.1053/j.semtcvs.2020.12.008. Epub 2021 Jan 12. PMID: 33444768.
- Attard C, Huang J, Monagle P, Ignjatovic V. Pathophysiology of thrombosis and anticoagulation post Fontan surgery. Thromb Res. 2018 Dec;172:204-213. doi: 10.1016/j.thromres.2018.04.011. Epub 2018 Apr 14. PMID: 29685556.
- Egbe AC, Connolly HM, Niaz T, Yogeswaran V, Taggart NW, Qureshi MY, Poterucha JT, Khan AR, Driscoll DJ. Prevalence and outcome of thrombotic and embolic complications in adults after Fontan operation. Am Heart J. 2017 Jan;183:10-17. doi: 10.1016/j.ahj.2016.09.014. Epub 2016 Oct 4. PMID: 27979032.
- Iyengar AJ, Winlaw DS, Galati JC, Wheaton GR, Gentles TL, Grigg LE, Justo RN, Radford DJ, Attard C, Weintraub RG, Bullock A, Sholler GS, Celermajer DS, d'Udekem Y; Australia and New Zealand Fontan Registry. No difference between aspirin and warfarin after extracardiac Fontan in a propensity score analysis of 475 patients. Eur J Cardiothorac Surg. 2016 Nov;50(5):980-987. doi: 10.1093/ejcts/ezw159. Epub 2016 May 26. PMID: 27229665.
- Kawamatsu N, Ishizu T, Machino-Ohtsuka T, Masuda K, Horigome H, Takechi F, Tateno S, Fukuda T, Kijima Y, Shiina Y, Komiyama N, Niwa K, Hiramatsu Y, Ieda M. Direct oral anticoagulant use and outcomes in adult patients with Fontan circulation: A multicenter retrospective cohort study. Int J Cardiol. 2021 Mar 15;327:74-79. doi: 10.1016/j.ijcard.2020.11.024. Epub 2020 Nov 19. PMID: 33220361.
- McCrindle BW, Manlhiot C, Cochrane A, Roberts R, Hughes M, Szechtman B, Weintraub R, Andrew M, Monagle P; Fontan Anticoagulation Study Group. Factors associated with thrombotic complications after the Fontan procedure: a secondary analysis of a multicenter, randomized trial of primary thromboprophylaxis for 2 years after the Fontan procedure. J Am Coll Cardiol. 2013 Jan 22;61(3):346-53. doi: 10.1016/j.jacc.2012.08.1023. Epub 2012 Dec 12. PMID: 23246393.
- McCrindle BW, Michelson AD, Van Bergen AH, Suzana Horowitz E, Pablo Sandoval J, Justino H, Harris KC, Jefferies JL, Miriam Pina L, Peluso C, Nessel K, Lu W, Li JS; UNIVERSE Study Investigators *. Thromboprophylaxis for Children Post-Fontan Procedure: Insights From the UNIVERSE Study. J Am Heart Assoc. 2021 Nov 16;10(22):e021765. doi: 10.1161/JAHA.120.021765. Epub 2021 Sep 24. Erratum in: J Am Heart Assoc. 2021 Dec 21;10(24):e020766. doi: 10.1161/JAHA.120.020766. PMID: 34558312; PMCID: PMC8751951.
- Payne RM, Burns KM, Glatz AC, Male C, Donti A, Brandão LR, Balling G, VanderPluym CJ, Bu'Lock F, Kochilas LK, Stiller B, Cnota JF 2nd, Rahkonen O, Khan A, Adorisio R, Stoica S, May L, Burns JC, Saraiva JFK, McHugh KE, Kim JS, Rubio A, Chía-Vazquez NG, Meador MR, Dyme JL, Reedy AM, Ajavon-Hartmann T, Jarugula P, Carlson-Taneja LE, Mills D, Wheaton O, Monagle P. Apixaban for Prevention of Thromboembolism in Pediatric Heart Disease. J Am Coll Cardiol. 2023 Dec 12;82(24):2296-2309. doi: 10.1016/j.jacc.2023.10.010. PMID: 38057072.
- Pessotti CF, Jatene MB, Jatene IB, Oliveira PM, Succi FM, Moreira Vde M, Lopes RW, Pedra SR. Comparative trial of the use of antiplatelet and oral anticoagulant in thrombosis prophylaxis in patients undergoing total cavopulmonary operation with extracardiac conduit: echocardiographic, tomographic, scintigraphic, clinical and laboratory analysis. Rev Bras Cir Cardiovasc. 2014 Oct-Dec;29(4):595-605. doi: 10.5935/1678-9741.20140111. PMID: 25714215; PMCID: PMC4408824.
- Potter BJ, Leong-Sit P, Fernandes SM, Feifer A, Mayer JE Jr, Triedman JK, Walsh EP, Landzberg MJ, Khairy P. Effect of aspirin and warfarin therapy on thromboembolic events in patients with univentricular hearts and Fontan palliation. Int J Cardiol. 2013 Oct 9;168(4):3940-3. doi: 10.1016/j.ijcard.2013.06.058. Epub 2013 Jul 17. PMID: 23870650.
- Portman MA, Jacobs JP, Newburger JW, Berger F, Grosso MA, Duggal A, Tao B, Goldenberg NA; ENNOBLE-ATE Trial Investigators. Edoxaban for Thromboembolism Prevention in Pediatric Patients With Cardiac Disease. J Am Coll Cardiol. 2022 Dec 13;80(24):2301-2310. doi: 10.1016/j.jacc.2022.09.031. Epub 2022 Oct 31. PMID: 36328157.
- Seipelt RG, Franke A, Vazquez-Jimenez JF, Hanrath P, von Bernuth G, Messmer BJ, Mühler EG. Thromboembolic complications after Fontan procedures: comparison of different therapeutic approaches. Ann Thorac Surg. 2002 Aug;74(2):556-62. doi: 10.1016/s0003-4975(02)03677-9. PMID: 12173844.
- Sethasathien S, Phinyo P, Sittiwangkul R, Silvilairat S. Comparative effectiveness among thromboprophylaxis strategies after the Fontan operation: A systematic review and network meta-analysis. Thromb Res. 2024 Sep;241:109093. doi: 10.1016/j.thromres.2024.109093. Epub 2024 Jul 8. PMID: 39003950.
- Van den Eynde J, Possner M, Alahdab F, Veldtman G, Goldstein BH, Rathod RH, Hoskoppal AK, Saraf A, Feingold B, Alsaied T. Thromboprophylaxis in Patients With Fontan Circulation. J Am Coll Cardiol. 2023 Jan 31;81(4):374-389. doi: 10.1016/j.jacc.2022.10.037. PMID: 36697138; PMCID: PMC11040452.
Evidence tabellen
Risk of bias tables
Risk of bias table for interventions studies (cohort studies based on risk of bias tool by the CLARITY Group at McMaster University)
|
|
Selection of participants
Was selection of exposed and non-exposed cohorts drawn from the same population?
|
Exposure
Can we be confident in the assessment of exposure?
|
Outcome of interest
Can we be confident that the outcome of interest was not present at start of study?
|
Confounding-assessment
Can we be confident in the assessment of confounding factors?
|
Confounding-analysis
Did the study match exposed and unexposed for all variables that are associated with the outcome of interest or did the statistical analysis adjust for these confounding variables?
|
Assessment of outcome
Can we be confident in the assessment of outcome?
|
Follow up
Was the follow up of cohorts adequate? In particular, was outcome data complete or imputed?
|
Co-interventions
Were co-interventions similar between groups?
|
Overall Risk of bias
|
|
Definitely yes, probably yes, probably no, definitely no |
Definitely yes, probably yes, probably no, definitely no |
Definitely yes, probably yes, probably no, definitely no |
Definitely yes, probably yes, probably no, definitely no |
Definitely yes, probably yes, probably no, definitely no |
Definitely yes, probably yes, probably no, definitely no |
Definitely yes, probably yes, probably no, definitely no |
Definitely yes, probably yes, probably no, definitely no |
Low, Some concerns, High |
|
|
Ankola, 2021 |
Definitely yes;
Reason: |
Definitely yes;
Reason: Medication use was registered for all patients |
Probably no;
Reason: no exclusion criteria were defined
|
Definitely yes;
Reason: |
Probably yes;
Reason: |
Definitely yes;
Reason: |
Definitely yes;
Reason: Outcome data was available for all patients |
No information |
Some concerns;
Reason: |
|
Al-Jazairi, 2019 |
Definitely yes;
Reason: |
Definitely yes;
Reason: Medication use was registered for all patients |
Probably no;
Reason: Not reported and not part of exclusion criteria |
Definitely yes;
Reason: Relevant confounders were assessed and reported |
Probably no;
Reason: No clear reporting on confounder adjustment through statistical analyses |
Definitely yes;
Reason: |
Definitely yes;
Reason: Outcome data was available for all patients
|
No information |
Some concerns;
Reason: |
|
Kawamatsu, 2021 |
Definitely yes;
Reason: |
Definitely yes;
Reason: Medication use was registered for all patients |
Probably no;
Reason: Not reported and not part of exclusion criteria |
Definitely yes;
Reason: Relevant confounders were assessed and reported |
Probably no;
Reason: Limited confounder adjustment was conducted in the statistical analyses |
Definitely yes;
Reason: |
Definitely yes;
Reason: Outcome data was available for all patients
|
Definitely no;
Reason: |
Some concerns;
Reason: |
|
Egbe, 2017 |
Definitely yes;
Reason: |
Definitely yes;
Reason: Medication use was registered for all patients |
Probably no;
Reason: Not reported and not part of exclusion criteria |
Definitely yes;
Reason: Relevant confounders were assessed and reported |
Probably no;
Reason: Multivariable statistical analyses were conducted, but not specifically for the comparison of aspirin and warfarin (both aspirin and warfarin were included as separate variables) |
Definitely yes;
Reason: |
Definitely yes;
Reason: Outcome data was available for all patients
|
No information |
Some concerns;
Reason: |
|
Iyengar, 2016 |
Definitely yes;
Reason: |
Definitely yes;
Reason: Medication use was registered for all patients |
Probably no;
Reason: Not reported and not part of exclusion criteria |
Definitely yes;
Reason: Relevant confounders were assessed and reported |
Definitely yes;
Reason: |
Definitely yes;
Reason: |
Definitely yes;
Reason: Outcome data was available for all patients
|
Probably yes;
Reason: Co-interventions were similar |
Some concerns;
Reason: |
|
Potter, 2013 |
Definitely yes;
Reason: |
Definitely yes;
Reason: Medication use was registered for all patients |
Definitely yes;
Reason: patients with very early postoperative thrombotic events were excluded |
Definitely yes;
Reason: Relevant confounders were assessed and reported |
Probably no;
Reason: |
Definitely yes;
Reason: |
Definitely yes;
Reason: Outcome data was available for all patients
|
No information |
Some concerns;
Reason: |
|
Seipelt, 2002 |
Probably yes;
Reason: |
Definitely yes;
Reason: Medication use was registered for all patients |
Probably no;
Reason: Not reported and not part of exclusion criteria |
Probably yes;
Reason: |
Probably no;
Reason: It is stated that multivariable analyses were conducted, but the results of these analyses are not clearly reported |
Definitely yes;
Reason: |
Definitely yes;
Reason: Outcome data was available for all patients
|
No information |
Some concerns;
Reason: |
Risk of bias table for intervention studies (randomized controlled trials; based on Cochrane risk of bias tool and suggestions by the CLARITY Group at McMaster University)
|
Study reference
(first author, publication year) |
Was the allocation sequence adequately generated?
Definitely yes Probably yes Probably no Definitely no |
Was the allocation adequately concealed?
Definitely yes Probably yes Probably no Definitely no |
Blinding: Was knowledge of the allocated interventions adequately prevented?
Were patients blinded?
Were healthcare providers blinded?
Were data collectors blinded?
Were outcome assessors blinded?
Were data analysts blinded?
Definitely yes Probably yes Probably no Definitely no |
Was loss to follow-up (missing outcome data) infrequent?
Definitely yes Probably yes Probably no Definitely no |
Are reports of the study free of selective outcome reporting?
Definitely yes Probably yes Probably no Definitely no |
Was the study apparently free of other problems that could put it at a risk of bias?
Definitely yes Probably yes Probably no Definitely no |
Overall risk of bias If applicable/necessary, per outcome measure
LOW Some concerns HIGH
|
|
McCrindle, 2021 |
Probably no;
Reason: Method of randomization is not reported |
Probably no;
Reason: Unclear |
Definitely no;
Reason: All medication use was open label |
Definitely yes;
Reason: No loss to follow-up |
Definitely yes;
Reason: |
Definitely yes;
Reason: No other problems were noted |
Some concerns;
Reason: Method of randomization is unclear, no reporting on allocation concealment, and no blinding |
|
McCrindle, 2013 |
Probably no;
Reason: Method of randomization is not clearly reported |
Probably yes;
Reason: Randomization was performed centrally |
Definitely no;
Reason: All medication use was open label |
Definitely yes;
Reason: No loss to follow-up |
Definitely yes;
Reason: |
Definitely yes;
Reason: No other problems were noted |
Some concerns;
Reason: Method of randomization is unclear, and no blinding |
|
Pessotti, 2014 |
Definitely yes;
Reason: |
Probably no;
Reason: Not reported |
Definitely no;
Reason: Not reported |
Definitely yes;
Reason: No loss to follow-up |
Definitely yes;
Reason: |
Definitely yes;
Reason: No other problems were noted |
Some concerns;
Reason: No reporting on allocation concealment and blinding |
Table of excluded studies
|
Reference |
Reason for exclusion |
|
Kazerouninia A, Georgekutty J, Kendsersky P, Byrne RD, Seto B, Chu PY, Wang Y, Rodriguez FH 3rd, Smith C, Saraf A, Lloyd MS, Frischhertz BP, Parekh DR, Ermis PR, Franklin WJ, Lam WW. A Multisite Retrospective Review of Direct Oral Anticoagulants Compared to Warfarin in Adult Fontan Patients. Cardiovasc Drugs Ther. 2023 Jun;37(3):519-527. doi: 10.1007/s10557-021-07298-5. Epub 2022 Jan 13. PMID: 35022950; PMCID: PMC11040449.
|
Adult age at start treatment |
|
Pujol C, Müssigmann M, Schiele S, Nagdyman N, Niesert AC, Kaemmerer H, Ewert P, Tutarel O. Direct oral anticoagulants in adults with congenital heart disease - a single centre study. Int J Cardiol. 2020 Feb 1;300:127-131. doi: 10.1016/j.ijcard.2019.09.077. Epub 2019 Oct 11. PMID: 31668654. |
Results not described separately for patients who underwent Fontan procedure |
|
Singh D, Jerrom T, Issitt R, Saxena R. Early thromboembolic complications after total cavopulmonary connection/Fontan operation in children: A five year single centre review. Progress in Pediatric Cardiology [Internet]. 2021 Jan 22;63:101344. Available from: https://doi.org/10.1016/j.ppedcard.2021.101344 |
Combination of treatments received by children without event is unclear |
|
Alsaied T, Alsidawi S, Allen CC, Faircloth J, Palumbo JS, Veldtman GR. Strategies for thromboprophylaxis in Fontan circulation: a meta-analysis. Heart. 2015 Nov;101(21):1731-7. doi: 10.1136/heartjnl-2015-307930. Epub 2015 Aug 28. PMID: 26319122. |
No quality assessment was conducted, Embase was not searched, available comparisons are limited (only aspirin vs. warfarin) |
|
Attard C, Monagle PT, d'Udekem Y, Mackay MT, Briody J, Cordina R, Hassan EB, Simm P, Rice K, Ignjatovic V; ANZ Fontan Registry Research group. Long-term outcomes of warfarin versus aspirin after Fontan surgery. J Thorac Cardiovasc Surg. 2021 Oct;162(4):1218-1228.e3. doi: 10.1016/j.jtcvs.2020.12.102. Epub 2021 Jan 5. PMID: 33563422. |
Cross-sectional study, no follow-up |
|
Deshaies C, Hamilton RM, Shohoudi A, Trottier H, Poirier N, Aboulhosn J, Broberg CS, Cohen S, Cook S, Dore A, Fernandes SM, Fournier A, Kay J, Mondésert B, Mongeon FP, Opotowsky AR, Proietti A, Ting J, Zaidi A, Khairy P; Alliance for Adult Research in Congenital Cardiology (AARCC). Thromboembolic Risk After Atriopulmonary, Lateral Tunnel, and Extracardiac Conduit Fontan Surgery. J Am Coll Cardiol. 2019 Aug 27;74(8):1071-1081. doi: 10.1016/j.jacc.2019.06.051. PMID: 31439217. |
Wrong design |
|
Firdouse M, Agarwal A, Chan AK, Mondal T. Thrombosis and thromboembolic complications in fontan patients: a literature review. Clin Appl Thromb Hemost. 2014 Jul;20(5):484-92. doi: 10.1177/1076029613520464. Epub 2014 Jan 24. PMID: 24463598. |
No comparison between outcomes for thromboprophylaxis A versus thromboprophylaxis B |
|
Heidendael JF, Engele LJ, Bouma BJ, Dipchand AI, Thorne SA, McCrindle BW, Mulder BJM. Coagulation and Anticoagulation in Fontan Patients. Can J Cardiol. 2022 Jul;38(7):1024-1035. doi: 10.1016/j.cjca.2022.01.028. Epub 2022 Feb 3. Erratum in: Can J Cardiol. 2023 Nov;39(11):1731. doi: 10.1016/j.cjca.2023.09.001. PMID: 35122939. |
Descriptive review of studies. The relevant studies that are described in this review were also found with the current literature search. |
|
Hosoi M, Ohbayashi M, Miyahara Y, Fujii T, Kise H, Kogo M. Risk Factors for Thromboembolic and Bleeding Events in Patients After the Fontan Operation (Insights from the National Database of Health Insurance Claims of Japan). Am J Cardiol. 2023 Nov 15;207:399-406. doi: 10.1016/j.amjcard.2023.08.153. Epub 2023 Sep 30. PMID: 37782971. |
Some patients were adult at time of Fontan procedure, no separate description of results |
|
Kleitsioti P, Theodoridis X, Papanastasiou CΑ, Karvounis H, Giannopoulos A, Giannakoulas G. Antiplatelet Versus Anticoagulation Treatment in Patients With Fontan Operation: A Systematic Review and Meta-Analysis. Cardiol Rev. 2025 Jan-Feb 01;33(1):17-21. doi: 10.1097/CRD.0000000000000550. Epub 2023 May 19. PMID: 37233451. |
Only the comparison antiplatelet therapy versus anticoagulants is available, Embase was not searched as part of the systematic literature search, reasons for exclusion of excluded studies not reported (per study) |
|
Maloku NA, Bejiqi NR, Mustafa NA, Tasic NV, Krasniqi NS. Anticoagulation therapy and thromboembolic complications in pediatric patients undergoing the Fontan procedure. Journal of Pharmaceutical Negative Results [Internet]. 2022 Nov 7;335–340. Available from: https://doi.org/10.47750/pnr.2022.13.s09.039
|
Wrong outcomes |
|
Miwa K, Iwai S, Nagashima T. Anticoagulation Therapy After the Fontan Procedure. Pediatr Cardiol. 2022 Aug;43(6):1271-1276. doi: 10.1007/s00246-022-02848-6. Epub 2022 Feb 25. PMID: 35217887. |
Descriptive study, no clear comparison for the outcome measures for group A versus group B (anticoagulation A versus B) |
|
Payne RM, Burns KM, Glatz AC, Male C, Donti A, Brandão LR, Balling G, VanderPluym CJ, Bu'Lock F, Kochilas LK, Stiller B, Cnota JF 2nd, Rahkonen O, Khan A, Adorisio R, Stoica S, May L, Burns JC, Saraiva JFK, McHugh KE, Kim JS, Rubio A, Chía-Vazquez NG, Meador MR, Dyme JL, Reedy AM, Ajavon-Hartmann T, Jarugula P, Carlson-Taneja LE, Mills D, Wheaton O, Monagle P. Apixaban for Prevention of Thromboembolism in Pediatric Heart Disease. J Am Coll Cardiol. 2023 Dec 12;82(24):2296-2309. doi: 10.1016/j.jacc.2023.10.010. PMID: 38057072. |
Wrong P, only part of the population underwent Fontan procedures and outcomes are not described separately for this subgroup of patients |
|
Portman MA, Jacobs JP, Newburger JW, Berger F, Grosso MA, Duggal A, Tao B, Goldenberg NA; ENNOBLE-ATE Trial Investigators. Edoxaban for Thromboembolism Prevention in Pediatric Patients With Cardiac Disease. J Am Coll Cardiol. 2022 Dec 13;80(24):2301-2310. doi: 10.1016/j.jacc.2022.09.031. Epub 2022 Oct 31. PMID: 36328157. |
Wrong P, only part of the population underwent Fontan procedures and outcomes are not described separately for this subgroup of patients |
|
Van den Eynde J, Possner M, Alahdab F, Veldtman G, Goldstein BH, Rathod RH, Hoskoppal AK, Saraf A, Feingold B, Alsaied T. Thromboprophylaxis in Patients With Fontan Circulation. J Am Coll Cardiol. 2023 Jan 31;81(4):374-389. doi: 10.1016/j.jacc.2022.10.037. PMID: 36697138; PMCID: PMC11040452. |
Inadequate search strategy |
|
Ohuchi H, Yasuda K, Miyazaki A, Ono S, Hayama Y, Negishi J, Noritake K, Mizuno M, Yamada O. Prevalence and predictors of haemostatic complications in 412 Fontan patients: their relation to anticoagulation and haemodynamics. Eur J Cardiothorac Surg. 2015 Mar;47(3):511-9. doi: 10.1093/ejcts/ezu145. Epub 2014 Apr 3. PMID: 24699205. |
Wrong design, prediction model |
|
Small AJ, Aboulhosn JA, Lluri G. Thromboprophylaxis in Adults With Atrio-Pulmonary Fontan. World J Pediatr Congenit Heart Surg. 2018 Sep;9(5):504-508. doi: 10.1177/2150135118772837. PMID: 30157726. |
Some patients were adult at time of Fontan procedure, no separate description of results |
Verantwoording
Beoordelingsdatum en geldigheid
Publicatiedatum : 21-04-2026
Beoordeeld op geldigheid : 21-04-2026
Algemene gegevens
De ontwikkeling/herziening van deze richtlijnmodule werd ondersteund door het Kennisinstituut van de Federatie Medisch Specialisten (www.demedischspecialist.nl/kennisinstituut) en werd gefinancierd uit de Stichting Kwaliteitsgelden Medisch Specialisten (SKMS). De financier heeft geen enkele invloed gehad op de inhoud van de richtlijnmodule.
Samenstelling werkgroep
Voor het ontwikkelen van de richtlijnmodule is in 2023 een multidisciplinaire werkgroep ingesteld, bestaande uit vertegenwoordigers van alle relevante specialismen (zie hiervoor de Samenstelling van de werkgroep) die betrokken zijn bij de zorg voor kinderen met een hartaandoening.
Werkgroep
- Dr. G. (Gert) van den Berg (voorzitter), kindercardioloog, werkzaam in het Erasmus MC Sophia Kinderziekenhuis te Rotterdam, NVK
- Dr. A. (Annelies) van der Hulst (vice-voorzitter), kindercardioloog, werkzaam in het Amsterdam UMC te Amsterdam, NVK
- Dr. C.H. (Heleen) van Ommen (vice-voorzitter), kinderarts-hematoloog, werkzaam in het Erasmus MC Sophia Kinderziekenhuis te Rotterdam, NVK
- Drs. H. (Hessel) Nijenhuis, kindercardioloog, werkzaam in het UMCG te Groningen, persoonlijke titel
- Drs. M. (Mirella) Molenschot, kindercardioloog, werkzaam in het UMC Utrecht/Wilhelmina Kinderziekenhuis te Utrecht, persoonlijke titel
- Drs. M.H. (Monique) Suijker, kinderarts-hematoloog, werkzaam in het Wilhelmina Kinderziekenhuis te Utrecht, persoonlijke titel
- Dr. P.P. Roeleveld, kinderarts-intensivist, werkzaam in het LUMC/Willem-Alexander Kinderziekenhuis te Leiden, NVK
- Drs. E. (Evangelia) Papathanasiou, kindercardioloog, werkzaam in het Radboudumc te Nijmegen, persoonlijke titel
- Dr. A.A.W. (Arno) Roest, kindercardioloog, werkzaam in het LUMC/Willem-Alexander Kinderziekenhuis te Leiden, NVK
- Dr. H. (Hanna) Talacua, cardio-thoracaal chirurg, werkzaam in het Wilhelmina Kinderziekenhuis te Utrecht, NVT
- Dr. L.M. (Léon) Putman, cardio-thoracaal chirurg, werkzaam in het Amsterdam UMC te Amsterdam, NVT
- Dr. J.B.A. (Judith) van de Meerakker, secretaris, werkzaam bij Patiëntenvereniging Aangeboren Hartafwijkingen te Haarlem
Klankbordgroep
- Drs. L. (Linda) van Wagenberg, kinderanesthesioloog-kinderintensivist, werkzaam in het UMC Utrecht te Utrecht, NVA
- Dr. C.E.J. (Pim) Sloots, kinderchirurg, werkzaam in het Erasmus MC te Rotterdam, NVvH
- E. (Esen) Doganer, beleidsmedewerker, werkzaam bij Stichting Kind & Zorg
- Drs. J. (Jolanda) van der Velden-Rodenburg, verpleegkundig specialist kindercardiologie, werkzaam in het UMC Utrecht te Utrecht, V&VN
Met ondersteuning van
- Dr. R. (Renee) Bolijn, adviseur, Kennisinstituut van de Federatie Medisch Specialisten
- Dr. T. (Tim) Christen, adviseur, Kennisinstituut van de Federatie Medisch Specialisten
- Drs. M. (Mischa) Lenaers, junior adviseur, Kennisinstituut van de Federatie Medisch Specialisten
Belangenverklaringen
De Code ter voorkoming van oneigenlijke beïnvloeding door belangenverstrengeling is gevolgd. Alle werkgroepleden hebben schriftelijk verklaard of zij in de laatste drie jaar directe financiële belangen (betrekking bij een commercieel bedrijf, persoonlijke financiële belangen, onderzoeksfinanciering) of indirecte belangen (persoonlijke relaties, reputatiemanagement) hebben gehad. Gedurende de ontwikkeling of herziening van een module worden wijzigingen in belangen aan de voorzitter doorgegeven. De belangenverklaring wordt opnieuw bevestigd tijdens de commentaarfase.
Een overzicht van de belangen van werkgroepleden en klankbordgroepleden en het oordeel over het omgaan met eventuele belangen vindt u in onderstaande tabel. De ondertekende belangenverklaringen zijn op te vragen bij het secretariaat van het Kennisinstituut van de Federatie Medisch Specialisten.
|
Naam |
Hoofdfunctie |
Nevenwerkzaamheden |
Gemelde belangen |
Ondernomen actie |
|
Werkgroep |
||||
|
Van den Berg* |
Kinderinterventiecardioloog ErasmusMC – Sophia kinderziekenhuis |
Advisory Board member conect4children Stichting. Advies over studie-opzet antistollingsmedicijn. Geen persoonlijke betaling, vergoeding voor geïnvesteerde tijd (enkele uren) zal tzt aan afdeling kindercardiologie uitgekeerd worden. |
Geen |
Geen restricties |
|
Van der Hulst |
kinderarts Amsterdam UMC |
Geen |
STAMINA: Stimulating Activitye in Minors with hAert disease |
Geen restricties |
|
Van de Meerakker |
Geen |
Secretaris Patiëntenvereniging Aangeboren Hartafwijkingen, onbetaald; Lid van de commissie voor subsidiebeoordelingen van Veelbelovende zorg, deelgebied patiëntparticipatie, ZonMw & ZiN, betaald; Lid van de commissie voor subsidiebeoordelingen van ZE&GG, deelgebied patiëntparticipatie, ZonMw, betaald; Lid expertgroep lymfoedeem, NLNet, onbetaald; Lid Raad van Advies Compressie Hulpmiddelenzorg, SEMH, betaald |
Geen |
Geen restricties |
|
Molenschot |
kindercardioloog UMC Utrecht/Wilhelmina Kinderziekenhuis |
Geen |
Geen |
Geen restricties |
|
Nijenhuis |
Kindercardioloog in het UMCG. |
Geen |
Geen relaties die baat hebben bij een bepaalde uitkomst. |
Geen restricties |
|
Van Ommen |
Kinderarts-hematoloog Hoofd afd. Kinderhematologie & Oncologie Erasmus MC Sophia |
Geen |
Boehringer Ingelheim, Consultant phase 4 study Dabigatran: observational study in children < 2 years of age with VTE treated with dabigatran. End date June 2022; Paid to the department of Pediatric Hematology (Erasmus MC): 2020: 560 euro/ 2022: 1902 euro.
Ik heb advies aan de farmaceut gegeven hoe deze fase 4 studie op te zetten, in welke landen, met welke artsen contact op te nemen voor particiaptie, etc etc. De vergoeding is aan de afdeling Kinderhematologie betaald. Deze vergoeding wordt gebruikt om master studenten/PhD studenten naar congressen te laten gaan om hun onderzoek te presenteren in poster of orale presentatie.
Fase 3 trial Effectiviteit en veiligheid van Edoxaban voor behandeling van trombose bij kinderen-afgesloten studie in 2022=Steering cie en local PI. Miscroscopic evaluation of clots in ECMO systems (PI). IPTN-ThromPED registry: international observational registry/study of children with thrombosis (chair IPTN). IPTN-ThromPED registry: international observational registry/study of children with thrombosis, in particular children with PE (chair IPTN).
Ik ben chair van de IPTN, International Pediatric Thrombosis Network. De IPTN heeft een internationale prospectieve registratie van kinderen met trombose sinds 2017. In 2023 heeft de IPTN eenmalig een unrestricted grant gekregen voor de Data Coordinating Center van de IPTN van Boerhinger Ingelheim en BMS. |
Geen restricties |
|
Papathanasiou |
kindercardioloog Radboudumc |
Geen |
Geen |
Geen restricties |
|
Putman |
Cardio-thoracaal chirurg Amsterdam UMC, 1.0FTE betaald |
Cardio-thoracaal chirurg LUMC, onbetaald |
Geen |
Geen restricties |
|
Roeleveld |
Kinderarts-intensivist Sectiehoofd, IC Kinderen Leids Universitair Medisch Centrum |
Lid van de stuurgroep van de "European Extracorporeal Life Support Organisation", onbetaald. |
Geen |
Geen restricties |
|
Roest |
LUMC, plv afdelingshoofd en kindercardioloog, betaald 1.0 fte |
Lid METC Leiden-Den Haag-Delft, onbetaald |
Leids Universiteits Fonds: Holoflow-project: visualisatie van bloedstromen bij aangeboren hartafwijkingen middels een hololens, projectleider. |
Geen restricties |
|
Suijker |
Kinderarts-hematoloog bij het Wilhelmina Kinderziekenhuis, Utrecht |
Antitrombotisch beleid: preventie van trombose bij kinderen met kanker; |
Ja, Parexel: Observationele studie naar gebruik van Dabigatran bij kinderen 0-2 jaar, betrokken als PI Dabigatran is al langer goed gekeurd en geregistreerd voor kinderen. Ik zou dabigatran niet anders beoordelen dan rivaroxaban. Ook in de Einstein studie was de groep 0-2 jaar klein. Op 29/12/2023 deelname beëindigd. Redenen hiervoor: - Dabigatran drank is niet leverbaar. Daarop werd studie protocol aangepast en leeftijdsgrens opgerekt naar 4 maanden. Dan kunnen de granules met fruit- of groentehap gegeven worden. - Begin december 2023 bleek dat de granules in Nederland (maar ook in België) niet vergoed worden. Ouders zouden dus zelf moeten bijbetalen. Wij hadden tot op heden ook nog geen patiënt geïncludeerd. |
Geen restricties |
|
Talacua |
Cardio-thoracaal chirurg WKZ Utrecht |
Geen |
Geen |
Geen restricties |
|
Klankbordgroep |
||||
|
Doganer |
Beleidsmedewerker Stichting Kind & Zorg (voorheen Stichting Kind en Ziekenhuis) |
Geen |
Geen |
Geen restricties |
|
Sloots |
Kinderchirurg, Erasmus MC / Sophia Kinderziekenhuis Rotterdam |
Geen |
Geen |
Geen restricties |
|
Van der Velden-Rodenburg |
verpleegkundig specialist Erasmus MC |
Geen |
Geen |
Geen restricties |
|
Van Wagenberg |
Kinderanesthesioloog- kinderintensivist UMC Utrecht/Wilhelmina kinderziekenhuis |
Geen |
Geen |
Geen restricties |
* Voorzitter van de werkgroep
Inbreng patiëntenperspectief
Er werd aandacht besteed aan het patiëntperspectief door het betrekken van een afgevaardigde van de Patiëntenvereniging Aangeboren Hartafwijkingen in de werkgroep en van een afgevaardigde van Stichting Kind & Zorg in de klankbordgroep. Tevens werd deze richtlijn aan deze patiëntenorganisaties ter commentaar voorgelegd.
Wkkgz & Kwalitatieve raming van mogelijke substantiële financiële gevolgen
Kwalitatieve raming van mogelijke financiële gevolgen in het kader van de Wkkgz
Bij de richtlijn is conform de Wet kwaliteit, klachten en geschillen zorg (Wkkgz) een kwalitatieve raming uitgevoerd of de aanbevelingen mogelijk leiden tot substantiële financiële gevolgen. Bij het uitvoeren van deze beoordeling zijn leidraadmodules op verschillende domeinen getoetst (zie het stroomschema op de Richtlijnendatabase).
|
Module |
Uitkomst raming |
Toelichting |
|
Fontan-circulatie |
Geen financiële gevolgen |
Uit de toetsing volgt dat de aanbeveling(en) niet breed toepasbaar zijn (<5.000 patiënten) en zal daarom naar verwachting geen substantiële financiële gevolgen hebben voor de collectieve uitgaven. |
Zoekverantwoording
Zoekstrategie
Embase.com
|
No. |
Query |
Results |
|
#1 |
'fontan procedure'/exp OR 'total cavopulmonary connection'/exp OR 'extracardiac total cavopulmonary connection'/exp OR 'extracardiac conduit'/exp OR 'lateral tunnel'/exp OR fontan:ti,ab,kw OR ((total NEAR/3 (cavopulmonary OR 'cavo pulmonary') NEAR/3 (anastomos* OR connection* OR shunt*)):ti,ab,kw) OR tcpc:ti,ab,kw OR 'extracardiac conduit':ti,ab,kw OR ecc:ti,ab,kw OR 'lateral tunnel':ti,ab,kw OR 'single ventricle':ti,ab,kw |
25824 |
|
#2 |
'thrombosis prevention'/exp OR 'anticoagulant agent'/exp OR 'anticoagulation'/exp OR 'anticoagulant therapy'/exp OR thromboprophyla*:ti,ab,kw OR ((thrombo* NEAR/3 (prophylaxis OR prophylactic OR prevention)):ti,ab,kw) OR 'anti coagulant*':ti,ab,kw OR 'anticoagulant*':ti,ab,kw OR 'anticoagulat*':ti,ab,kw OR 'anti coagulat*':ti,ab,kw OR 'antithrombotic*':ti,ab,kw OR 'anti thrombotic*':ti,ab,kw OR 'antithrombocytic*':ti,ab,kw OR 'anti thrombocytic*':ti,ab,kw OR 'antiplatelet agent*':ti,ab,kw OR 'antiplatelet drug*':ti,ab,kw OR 'platelet aggregation inhibitor*':ti,ab,kw OR 'platelet inhibitor*':ti,ab,kw OR 'platelet antagonist*':ti,ab,kw OR 'thrombocyte aggregation inhibiting agent*':ti,ab,kw OR 'thrombocyte aggregation inhibitor*':ti,ab,kw OR 'direct oral anticoagulant agent'/exp OR 'direct oral anticoagulant'/exp OR doac*:ti,ab,kw OR 'apixaban'/exp OR 'apixaban':ti,ab,kw OR 'rivaroxaban'/exp OR 'rivaroxaban':ti,ab,kw OR xarelto:ti,ab,kw OR 'edoxaban'/exp OR 'edoxaban':ti,ab,kw OR 'endoxaban':ti,ab,kw OR 'antivitamin k'/exp OR 'antivitamin k':ti,ab,kw OR 'anti vitamin k':ti,ab,kw OR 'vitamin k antagonist*':ti,ab,kw OR vka:ti,ab,kw OR 'choay':ti,ab,kw OR 'heparin'/exp OR 'low molecular weight heparin'/exp OR 'depolymerized heparin*':ti,ab,kw OR 'low molecular heparin*':ti,ab,kw OR 'low molecular weight heparin*':ti,ab,kw OR 'traxyparin*':ti,ab,kw OR 'unfractionated heparin*':ti,ab,kw OR 'heparin* lmw':ti,ab,kw OR 'lmw heparin*':ti,ab,kw OR lmwh:ti,ab,kw OR 'carbasalate calcium'/exp OR 'alcacyl':ti,ab,kw OR 'calcium carbasalate':ti,ab,kw OR 'carbasalate calcium':ti,ab,kw OR 'acetylsalicylic acid calcium'/exp OR 'acetylsalicylic acid calcium':ti,ab,kw OR 'ascal':ti,ab,kw OR 'calcium acetyl salicylate':ti,ab,kw OR 'calcium acetylsalicylate':ti,ab,kw OR 'disprin':ti,ab,kw OR 'acetylsalicylic acid'/exp OR 'acetyl* salicylic acid':ti,ab,kw OR 'acetylsalicylic acid':ti,ab,kw OR 'aspirin*':ti,ab,kw OR 'lysine acetylsalicylate'/exp OR 'acetylsalicylic acid lysine':ti,ab,kw OR 'aspegic':ti,ab,kw OR 'kardegic':ti,ab,kw OR 'lysine acetyl salicylate':ti,ab,kw OR 'lysine acetylsalicylate':ti,ab,kw OR 'lysine acetylsalicylic acid':ti,ab,kw OR 'clopidogrel'/exp OR 'clopidogrel':ti,ab,kw OR 'pcr 4099':ti,ab,kw OR 'pcr4099':ti,ab,kw OR 'plavix':ti,ab,kw OR 'nadroparin'/exp OR 'fraxiparin*':ti,ab,kw OR 'nadroparin*':ti,ab,kw OR 'tinzaparin'/exp OR 'logiparin':ti,ab,kw OR 'tinzaparin':ti,ab,kw OR 'enoxaparin'/exp OR 'clexane':ti,ab,kw OR 'enoxaparin*':ti,ab,kw OR lovenox:ti,ab,kw OR 'dalteparin'/exp OR 'dalteparin*':ti,ab,kw OR 'acenocoumarol'/exp OR 'acenocoumarol*':ti,ab,kw OR 'acenocumarol*':ti,ab,kw OR sintrom:ti,ab,kw OR 'phenprocoumon'/exp OR 'fenprocoumon':ti,ab,kw OR marcumar:ti,ab,kw OR marcoumar:ti,ab,kw OR phenprocoumarol:ti,ab,kw OR 'phenprocoumon':ti,ab,kw OR 'warfarin'/exp OR coumadin:ti,ab,kw OR 'warfarin*':ti,ab,kw |
953674 |
|
#3 |
#1 AND #2 NOT ('conference abstract'/it OR 'editorial'/it OR 'letter'/it OR 'note'/it) NOT (('animal'/exp OR 'animal experiment'/exp OR 'animal model'/exp OR 'nonhuman'/exp) NOT 'human'/exp) AND [2014-2024]/py |
882 |
|
#4 |
'meta analysis'/exp OR 'meta analysis (topic)'/exp OR metaanaly*:ti,ab OR 'meta analy*':ti,ab OR metanaly*:ti,ab OR 'systematic review'/de OR 'cochrane database of systematic reviews'/jt OR prisma:ti,ab OR prospero:ti,ab OR (((systemati* OR scoping OR umbrella OR 'structured literature') NEAR/3 (review* OR overview*)):ti,ab) OR ((systemic* NEAR/1 review*):ti,ab) OR (((systemati* OR literature OR database* OR 'data base*') NEAR/10 search*):ti,ab) OR (((structured OR comprehensive* OR systemic*) NEAR/3 search*):ti,ab) OR (((literature NEAR/3 review*):ti,ab) AND (search*:ti,ab OR database*:ti,ab OR 'data base*':ti,ab)) OR (('data extraction':ti,ab OR 'data source*':ti,ab) AND 'study selection':ti,ab) OR ('search strategy':ti,ab AND 'selection criteria':ti,ab) OR ('data source*':ti,ab AND 'data synthesis':ti,ab) OR medline:ab OR pubmed:ab OR embase:ab OR cochrane:ab OR (((critical OR rapid) NEAR/2 (review* OR overview* OR synthes*)):ti) OR ((((critical* OR rapid*) NEAR/3 (review* OR overview* OR synthes*)):ab) AND (search*:ab OR database*:ab OR 'data base*':ab)) OR metasynthes*:ti,ab OR 'meta synthes*':ti,ab |
1043857 |
|
#5 |
'clinical trial'/exp OR 'randomization'/exp OR 'single blind procedure'/exp OR 'double blind procedure'/exp OR 'crossover procedure'/exp OR 'placebo'/exp OR 'prospective study'/exp OR rct:ab,ti OR random*:ab,ti OR 'single blind':ab,ti OR 'randomised controlled trial':ab,ti OR 'randomized controlled trial'/exp OR placebo*:ab,ti |
4065855 |
|
#6 |
'major clinical study'/de OR 'clinical study'/de OR 'case control study'/de OR 'family study'/de OR 'longitudinal study'/de OR 'retrospective study'/de OR 'prospective study'/de OR 'comparative study'/de OR 'cohort analysis'/de OR ((cohort NEAR/1 (study OR studies)):ab,ti) OR (('case control' NEAR/1 (study OR studies)):ab,ti) OR (('follow up' NEAR/1 (study OR studies)):ab,ti) OR (observational NEAR/1 (study OR studies)) OR ((epidemiologic NEAR/1 (study OR studies)):ab,ti) OR (('cross sectional' NEAR/1 (study OR studies)):ab,ti) |
8310051 |
|
#7 |
'case control study'/de OR 'comparative study'/exp OR 'control group'/de OR 'controlled study'/de OR 'controlled clinical trial'/de OR 'crossover procedure'/de OR 'double blind procedure'/de OR 'phase 2 clinical trial'/de OR 'phase 3 clinical trial'/de OR 'phase 4 clinical trial'/de OR 'pretest posttest design'/de OR 'pretest posttest control group design'/de OR 'quasi experimental study'/de OR 'single blind procedure'/de OR 'triple blind procedure'/de OR (((control OR controlled) NEAR/6 trial):ti,ab,kw) OR (((control OR controlled) NEAR/6 (study OR studies)):ti,ab,kw) OR (((control OR controlled) NEAR/1 active):ti,ab,kw) OR 'open label*':ti,ab,kw OR (((double OR two OR three OR multi OR trial) NEAR/1 (arm OR arms)):ti,ab,kw) OR ((allocat* NEAR/10 (arm OR arms)):ti,ab,kw) OR placebo*:ti,ab,kw OR 'sham-control*':ti,ab,kw OR (((single OR double OR triple OR assessor) NEAR/1 (blind* OR masked)):ti,ab,kw) OR nonrandom*:ti,ab,kw OR 'non-random*':ti,ab,kw OR 'quasi-experiment*':ti,ab,kw OR crossover:ti,ab,kw OR 'cross over':ti,ab,kw OR 'parallel group*':ti,ab,kw OR 'factorial trial':ti,ab,kw OR ((phase NEAR/5 (study OR trial)):ti,ab,kw) OR ((case* NEAR/6 (matched OR control*)):ti,ab,kw) OR ((match* NEAR/6 (pair OR pairs OR cohort* OR control* OR group* OR healthy OR age OR sex OR gender OR patient* OR subject* OR participant*)):ti,ab,kw) OR ((propensity NEAR/6 (scor* OR match*)):ti,ab,kw) OR versus:ti OR vs:ti OR compar*:ti OR ((compar* NEAR/1 study):ti,ab,kw) OR (('major clinical study'/de OR 'clinical study'/de OR 'cohort analysis'/de OR 'observational study'/de OR 'cross-sectional study'/de OR 'multicenter study'/de OR 'correlational study'/de OR 'follow up'/de OR cohort*:ti,ab,kw OR 'follow up':ti,ab,kw OR followup:ti,ab,kw OR longitudinal*:ti,ab,kw OR prospective*:ti,ab,kw OR retrospective*:ti,ab,kw OR observational*:ti,ab,kw OR 'cross sectional*':ti,ab,kw OR cross?ectional*:ti,ab,kw OR multicent*:ti,ab,kw OR 'multi-cent*':ti,ab,kw OR consecutive*:ti,ab,kw) AND (group:ti,ab,kw OR groups:ti,ab,kw OR subgroup*:ti,ab,kw OR versus:ti,ab,kw OR vs:ti,ab,kw OR compar*:ti,ab,kw OR 'odds ratio*':ab OR 'relative odds':ab OR 'risk ratio*':ab OR 'relative risk*':ab OR 'rate ratio':ab OR aor:ab OR arr:ab OR rrr:ab OR ((('or' OR 'rr') NEAR/6 ci):ab))) |
15226357 |
|
#8 |
#3 AND #4 - SR |
39 |
|
#9 |
#3 AND #5 NOT #8 - RCT |
79 |
|
#10 |
#3 AND (#6 OR #7) NOT (#8 OR #9) - observationeel |
333 |
|
#11 |
#8 OR #9 OR #10 |
451 |
Ovid/Medline
|
# |
Searches |
Results |
|
1 |
exp Fontan Procedure/ or (fontan or (total adj3 (cavopulmonary or 'cavo pulmonary') adj3 (anastomos* or connection* or shunt*)) or tcpc or 'extracardiac conduit' or ecc or 'lateral tunnel' or 'single ventricle').ti,ab,kf. |
17616 |
|
2 |
exp Anticoagulants/ or exp Platelet Aggregation Inhibitors/ or thromboprophyla*.ti,ab,kf. or (thrombo* adj3 (prophylaxis or prophylactic or prevention)).ti,ab,kf. or (anti coagulant* or anticoagulant* or anticoagulat* or anti coagulat* or antithrombotic* or anti thrombotic* or antithrombocytic* or anti thrombocytic* or 'antiplatelet agent*' or 'antiplatelet drug*' or 'platelet aggregation inhibitor*' or 'platelet inhibitor*' or platelet antagonist* or 'thrombocyte aggregation inhibiting agent*' or 'thrombocyte aggregation inhibitor*' or doac*).ti,ab,kf. or apixaban'.ti,ab,kf. or exp Rivaroxaban/ or Rivaroxaban.ti,ab,kf. or xarelto.ti,ab,kf. or 'edoxaban'.ti,ab,kf. or 'endoxaban'.ti,ab,kf. or Vitamin K/ai or (antivitamin k or anti vitamin k or vitamin k antagonist* or vka or choay).ti,ab,kf. or exp Heparin/ or exp Heparin, Low-Molecular-Weight/ or (heparin* or traxyparin* or lmwh).ti,ab,kf. or 'alcacyl'.ti,ab,kf. or 'calcium carbasalate'.ti,ab,kf. or 'carbasalate calcium'.ti,ab,kf. or 'acetylsalicylic acid calcium'.ti,ab,kf. or 'ascal'.ti,ab,kf. or 'calcium acetyl salicylate'.ti,ab,kf. or 'calcium acetylsalicylate'.ti,ab,kf. or 'disprin'.ti,ab,kf. or exp Aspirin/ or 'acetyl* salicylic acid'.ti,ab,kf. or 'acetylsalicylic acid'.ti,ab,kf. or 'aspirin*'.ti,ab,kf. or 'acetylsalicylic acid lysine'.ti,ab,kf. or 'aspegic'.ti,ab,kf. or 'kardegic'.ti,ab,kf. or 'lysine acetyl salicylate'.ti,ab,kf. or 'lysine acetylsalicylate'.ti,ab,kf. or 'lysine acetylsalicylic acid'.ti,ab,kf. or exp Clopidogrel/ or 'clopidogrel'.ti,ab,kf. or 'pcr 4099'.ti,ab,kf. or 'pcr4099'.ti,ab,kf. or 'plavix'.ti,ab,kf. or exp Nadroparin/ or 'fraxiparin*'.ti,ab,kf. or 'nadroparin*'.ti,ab,kf. or exp Tinzaparin/ or 'logiparin'.ti,ab,kf. or 'tinzaparin'.ti,ab,kf. or exp Enoxaparin/ or 'clexane'.ti,ab,kf. or 'enoxaparin*'.ti,ab,kf. or lovenox.ti,ab,kf. or exp Dalteparin/ or 'dalteparin*'.ti,ab,kf. or exp Acenocoumarol/ or exp Acenocoumarol/ or 'acenocumarol*'.ti,ab,kf. or sintrom.ti,ab,kf. or exp Phenprocoumon/ or 'fenprocoumon'.ti,ab,kf. or marcumar.ti,ab,kf. or marcoumar.ti,ab,kf. or phenprocoumarol.ti,ab,kf. or 'phenprocoumon'.ti,ab,kf. or exp Warfarin/ or coumadin.ti,ab,kf. or 'warfarin*'.ti,ab,kf. |
502404 |
|
3 |
(1 and 2) not (comment/ or editorial/ or letter/) not ((exp animals/ or exp models, animal/) not humans/) |
641 |
|
4 |
limit 3 to yr="2014 -Current" |
290 |
|
5 |
meta-analysis/ or meta-analysis as topic/ or (metaanaly* or meta-analy* or metanaly*).ti,ab,kf. or systematic review/ or cochrane.jw. or (prisma or prospero).ti,ab,kf. or ((systemati* or scoping or umbrella or "structured literature") adj3 (review* or overview*)).ti,ab,kf. or (systemic* adj1 review*).ti,ab,kf. or ((systemati* or literature or database* or data-base*) adj10 search*).ti,ab,kf. or ((structured or comprehensive* or systemic*) adj3 search*).ti,ab,kf. or ((literature adj3 review*) and (search* or database* or data-base*)).ti,ab,kf. or (("data extraction" or "data source*") and "study selection").ti,ab,kf. or ("search strategy" and "selection criteria").ti,ab,kf. or ("data source*" and "data synthesis").ti,ab,kf. or (medline or pubmed or embase or cochrane).ab. or ((critical or rapid) adj2 (review* or overview* or synthes*)).ti. or (((critical* or rapid*) adj3 (review* or overview* or synthes*)) and (search* or database* or data-base*)).ab. or (metasynthes* or meta-synthes*).ti,ab,kf. |
758627 |
|
6 |
exp clinical trial/ or randomized controlled trial/ or exp clinical trials as topic/ or randomized controlled trials as topic/ or Random Allocation/ or Double-Blind Method/ or Single-Blind Method/ or (clinical trial, phase i or clinical trial, phase ii or clinical trial, phase iii or clinical trial, phase iv or controlled clinical trial or randomized controlled trial or multicenter study or clinical trial).pt. or random*.ti,ab. or (clinic* adj trial*).tw. or ((singl* or doubl* or treb* or tripl*) adj (blind$3 or mask$3)).tw. or Placebos/ or placebo*.tw. |
2748336 |
|
7 |
Epidemiologic studies/ or case control studies/ or exp cohort studies/ or Controlled Before-After Studies/ or Case control.tw. or cohort.tw. or Cohort analy$.tw. or (Follow up adj (study or studies)).tw. or (observational adj (study or studies)).tw. or Longitudinal.tw. or Retrospective*.tw. or prospective*.tw. or consecutive*.tw. or Cross sectional.tw. or Cross-sectional studies/ or historically controlled study/ or interrupted time series analysis/ [Onder exp cohort studies vallen ook longitudinale, prospectieve en retrospectieve studies] |
4771243 |
|
8 |
Case-control Studies/ or clinical trial, phase ii/ or clinical trial, phase iii/ or clinical trial, phase iv/ or comparative study/ or control groups/ or controlled before-after studies/ or controlled clinical trial/ or double-blind method/ or historically controlled study/ or matched-pair analysis/ or single-blind method/ or (((control or controlled) adj6 (study or studies or trial)) or (compar* adj (study or studies)) or ((control or controlled) adj1 active) or "open label*" or ((double or two or three or multi or trial) adj (arm or arms)) or (allocat* adj10 (arm or arms)) or placebo* or "sham-control*" or ((single or double or triple or assessor) adj1 (blind* or masked)) or nonrandom* or "non-random*" or "quasi-experiment*" or "parallel group*" or "factorial trial" or "pretest posttest" or (phase adj5 (study or trial)) or (case* adj6 (matched or control*)) or (match* adj6 (pair or pairs or cohort* or control* or group* or healthy or age or sex or gender or patient* or subject* or participant*)) or (propensity adj6 (scor* or match*))).ti,ab,kf. or (confounding adj6 adjust*).ti,ab. or (versus or vs or compar*).ti. or ((exp cohort studies/ or epidemiologic studies/ or multicenter study/ or observational study/ or seroepidemiologic studies/ or (cohort* or 'follow up' or followup or longitudinal* or prospective* or retrospective* or observational* or multicent* or 'multi-cent*' or consecutive*).ti,ab,kf.) and ((group or groups or subgroup* or versus or vs or compar*).ti,ab,kf. or ('odds ratio*' or 'relative odds' or 'risk ratio*' or 'relative risk*' or aor or arr or rrr).ab. or (("OR" or "RR") adj6 CI).ab.)) |
5732104 |
|
9 |
4 and 5 - SR |
23 |
|
10 |
(4 and 6) not 9 - RCT |
36 |
|
11 |
(4 and (7 or 8)) not (9 or 10) - observationeel |
109 |
|
12 |
9 or 10 or 11 |
168 |