Eerstelijnsbehandeling BRAF-V600E/K gemuteerd irresectabel of gemetastaseerd stadium III/IV
Uitgangsvraag
Wat is de plaats van systemische therapie in de eerste lijns-behandeling van patiënten met een BRAF-V600E/K gemuteerd irresectabel of gemetastaseerd stadium III/IV melanoom?
Aanbeveling
Overweeg behandeling in studieverband.
Individualiseer de behandelbeslissing rekening houdend met het behandeldoel (kortetermijnvoordeel versus langetermijnvoordeel) en klinische kenmerken [lactaatdehydrogenase (LDH), betrokken organen waaronder de hersenen, algehele conditie (performance status), tumorlast, snelheid van ziekteprogressie], comorbiditeiten, bijwerkingen en patiëntvoorkeuren.
Overweeg immuuntherapie bij patiënten die dit in de eerste maanden kunnen verdragen, ongeacht de BRAF-status, omdat PD-1 blokkade, eventueel gecombineerd met ipilimumab, langdurige ziektecontrole kan bieden, zelfs na het beëindigen van de behandeling. Bovendien is aangetoond dat ipilimumab/nivolumab in deze situatie superieur is aan dabrafenib/trametinib.
Baseer de keuze tussen monotherapie en combinatiebehandeling op klinische parameters. Combinatiebehandeling is meer voorbehouden voor patiënten met hersenmetastasen en/of verhoogd serum LDH.
Overweeg bij patiënten met een BRAFV600 gemuteerd melanoom bij wie immuuntherapie gedurende de eerste paar maanden vanwege snelle progressie en/of bedreiging van een belangrijk orgaan of functie, doelgerichte therapie (BRAF + MEK-inhibitie) in de eerstelijn.
Kies bij doelgerichte behandeling altijd voor de combinatie van BRAF- en MEK-remmers; gebruik BRAF-inhibitor alleen als monotherapie bij een absolute contra-indicatie voor MEK-remmers. Gelijktijdig combineren van doelgerichte behandeling (BRAF + MEK remmers) met immuuntherapie wordt niet aanbevolen buiten studieverband.
Overwegingen
RELATIVITY-047 rapporteerde het effect van relatlimab plus nivolumab versus nivolumab op progression free survival, adverse events en kwaliteit van leven bij patiënten met unresectable of gemetastaseerd stadium III/IV melanoom (Tawbi, 2022). Het absolute verschil in mediane progression free survival was 5.5 maanden met een HR van 0.75 (95% CI 0.62-0.92). De klinische relevantie van dit verschil kan niet worden beoordeeld op basis van de PASKWIL criteria (mediane overall survival in de controle groep > 12 maanden). De bewijskracht van deze studie is laag, dit heeft te maken met het risico op bias (door de rol van de sponsor in deze studie) en door imprecisie.
Relatlimab plus nivolumab resulteert waarschijnlijk niet of nauwelijks in een vermindering of toename van adverse events of kwaliteit van leven vergeleken met nivolumab.
Aanvullend op de oorspronkelijke publicatie rapporteerden Long (2023) en Tawbi (2024) de eerste gegevens over overall survival en objective response rate voor RELATIVITY-047. De mediane overall survival was 51,0 maanden met relatlimab plus nivolumab versus 34,1 maanden met nivolumab (HR 0,80; 95% CI 0,66–0,99). Ook de objective response rate was hoger (43,7% vs. 33,7%). Er traden geen nieuwe adverse events op, al was het aantal graad 3–4 therapiegerelateerde bijwerkingen hoger bij combinatiebehandeling (22% vs. 12%).
CHECKMATE-067 rapporteerde het effect van nivolumab plus ipilimumab versus nivolumab versus ipilimumab op overall survival bij patiënten met niet-receerbaar of gemetastaseerd stadium III/IV melanoom (Larkin 2015, Wolchock 2017, Hodi 2018, Larkin 2019, Wolchok 2022). Er werd daarbij een klinisch relevant voordeel gevonden voor het gebruik van nivolumab plus ipilimumab. Het absolute verschil in mediane overall survival tussen behandeling met nivolumab plus ipilimumab versus behandeling met alleen ipilimumab was 52.2 maanden met een hazard ratio van 0.52 (95% CI 0.43-0.63). De bewijskracht hiervan is hoog. Het absolute verschil in mediane overall survival tussen behandeling met nivolumab plus ipilimumab versus behandeling met alleen nivolumab was 35.2 maanden met een hazard ratio van 0.84 (95% CI 0.67 to 1.04). De bewijskracht hiervan is redelijk tot laag, dit heeft te maken met imprecisie, omdat de confidence interval de grens voor klinische besluitvorming omvat en het risico op bias (doordat de studie niet gepowered was voor de vergelijking van deze middelen).
De studie rapporteerde ook het effect op progression free survival en adverse events. Een langere mediane progression free survival werd gevonden voor patiënten die behandeld warden met nivolumab plus ipilimumab, met een verschil van 8.6 maanden (HR 0.42; CI 0.35-0.51) met patiënten die behandeld warden met alleen ipilimumab en een verschil van 4.6 maanden (HR 0.79; CI 0.65 to 0.97) met patiënten die behandeld warden met alleen nivolumab. Behandeling met nivolumab plus ipilimumab resulteerde in een hoger percentage behandelingsgerelateerde adverse events grade ≥3 in vergelijking met behandeling met alleen nivolumab of alleen ipilimumab.
CA184-024 rapporteerde het effect van ipilimumab plus dacarbazine versus dacarbazine op overall survival bij patiënten met unresectable of gemetastaseerd stadium III/IV melanoom (Maio, 2015; Robert, 2011). Er werd daarbij geen klinisch relevant voordeel gevonden voor het gebruik van ipilimumab plus dacarbazine. Het absolute verschil in mediane overall survival tussen behandeling met ipilimumab plus dacarbazine versus behandeling met alleen dacarbazine was 2.12 maanden met een hazard ratio van 0.69 (95% CI 0.57 to 0.84). De bewijskracht van deze studie is zeer laag. Dit heeft te maken met het risico op bias (onduidelijke behandelings allocatie en doordat meer patiënten in de interventie groep stopten vanwege behandelingsgerelateerde adverse events) en door imprecisie omdat de confidence interval de grens voor klinische besluitvorming omvat.
De studie rapporteerde ook het effect op progression free survival en adverse events. Er werd geen klinisch relevant verschil gevonden tussen de studiegroepen voor de uitkomst progression free survival. Het ontstaan van adverse events. Behandeling ipilimumab plus dacarbazine resulteerde in een hoger percentage behandelingsgerelateerde adverse events grade ≥3 in vergelijking met behandeling met alleen dacarbazine.
KEYNOTE-006 rapporteerde het effect van pembrolizumab (iedere 2 of 3 weken) in vergelijking met ipilimumab (iedere 3 weken) op overall survival bij patiënten met unresectable of gemetastaseerd stadium III/IV melanoom (Robert, 2019; Carlino, 2018; Schachter, 2017; Robert, 2015). Er werd daarbij geen klinisch relevant voordeel gevonden voor het gebruik van pembrolizumab volgens de PASWKIL-criteria. Het absolute verschil in mediane overall survival tussen behandeling met pembrolizumab versus behandeling met ipilimumab was 16.8 maanden met een hazard ratio van 0.73 (95% CI 0.61–0.88). De bewijskracht van deze studie is zeer laag. Dit heeft te maken met het risico op bias (open-label studie design; meer patiënten stopten in de controle groep; de rol van de sponsor) en door imprecisie, omdat de confidence interval de grens voor klinische besluitvorming omvat.
De studie rapporteerde ook het effect op progression free survival en adverse events. Voor de uitkomst progression free survival werd een klinisch relevant voordeel gevonden voor behandeling met pembrolizumab. Het absolute verschil in mediane progressievrije overleving was 5.0 maanden (HR 0.57; 95% CI 0.48–0.67). Er werd geen klinisch relevant verschil gevonden tussen de studiegroepen voor de uitkomst adverse events.
MASTERKEY-265 rapporteerde het effect van T-VEC plus pembrolizumab in vergelijking met placebo plus pembrolizumab op overall survival bij patiënten met unresectable of gemetastaseerd stadium III/IV melanoom (Chesney, 2023). Er werd daarbij geen klinisch relevant voordeel gevonden voor het gebruik van T-VEC plus pembrolizumab (HR 0.96; 95% CI 0.76 to 1.22). De bewijskracht van deze studie is zeer laag. Dit heeft te maken met het risico op bias (meer patiënten stopten in de controle groep; studie werd vroegtijdig gestopt) en door imprecisie.
De studie rapporteerde ook het effect op progression free survival en adverse events. Er werd geen klinisch relevant verschil gevonden tussen de studiegroepen voor de uitkomsten progression free survival en adverse events.
OPTiM rapporteerde het effect van T-VEC in vergelijking met GM-CSF op overall survival bij patiënten met niet-receerbaar of gemetastaseerd stadium III/IV melanoom (Andtbacka, 2019; Andtbacka, 2015). Er werd daarbij geen klinisch relevant voordeel gevonden voor het gebruik van T-VEC. Het absolute verschil in mediane overall survival tussen behandeling met T-VEC versus behandeling met GM-CSF was 4.4 maanden met een hazard ratio 0.79 (95% CI 0.62–1.00). De bewijskracht van deze studie is zeer laag. Dit heeft te maken met het risico op bias (open-label studie design; meer patiënten stopten in de interventie groep; de rol van de sponsor) en door imprecisie, omdat het betrouwbaarheidsinterval de grens voor klinische besluitvorming omvat.
De studie rapporteerde ook het effect op adverse events. Er werd geen klinisch relevant verschil gevonden tussen de studiegroepen voor de uitkomst adverse events.
CoBRIM rapporteerde het effect van cobimetinib plus vemurafenib versus vemurafenib en placebo op de mediane OS. Behandeling met cobimetinib plus vemurafenib resulteerde in een langere mediane OS dan behandeling met vemurafenib en placebo. Het absolute verschil tussen de cobimetinib plus vemurafenib groep (22,5 maanden) en de vemurafenib en placebo groep (17,4 maanden) was 5,1 maanden met een HR van 0,70 (95% BI: 0,55 – 0,89). Dit verschil werd volgens de PASKWIL-criteria niet als klinisch relevant beschouwd. De bewijskracht hiervan is redelijk, dit heeft te maken met de imprecisie, omdat de confidence interval de grens voor klinische besluitvorming omvat.
De studie rapporteerde ook het effect op progression free survival en adverse events. Er werd geen klinisch relevant verschil gevonden tussen de studiegroepen voor de uitkomsten progression free survival en adverse events.
COMBI-v rapporteerde niet het effect van dabrafenib plus trametinib versus vemurafenib op de mediane OS. Echter, het absolute verschil in mediane 3-jaarsoverleving tussen dabrafenib plus trametinib (45%) en vemurafenib (32%) was 13%, met een hogere 3-jaarsoverleving in de dabrafenib en trametinib groep. Dit verschil werd volgens de PASKWIL-criteria als klinisch relevant beschouwd. De bewijskracht hiervan is redelijk, dit heeft te maken met het risico op bias.
De studie rapporteerde ook het effect op progression free survival en adverse events. Voor de uitkomst progression free survival werd een klinisch relevant voordeel gevonden voor behandeling met cobimetinib plus vemurafenib. Het absolute verschil in mediane progressievrije overleving was 5.4 maanden (HR 0.51; 95% CI 0.39–0.67). Er werd geen klinisch relevant verschil gevonden tussen de studiegroepen voor de uitkomst adverse events.
COLUMBUS rapporteerde het effect van encorafenib plus binimetinib versus encorafenib versus vemurafenib op de mediane OS. Behandeling met encorafenib plus binimetinib resulteerde in de langste mediane OS. Het absolute verschil tussen encorafenib plus binimetinib (33,6 maanden) en encorafenib (23,5 maanden) was 10,1 maanden met een HR van 0,81 (95% BI: 0,61 tot 1,06). Dit verschil werd volgens de PASKWIL-criteria niet als klinisch relevant beschouwd. Het absolute verschil tussen encorafenib plus binimetinib (33,6 maanden) en vemurafenib (16,9 maanden) was 16,7 maanden met een HR van 0,61 (95% BI: 0,47 tot 0,79). Dit verschil werd volgens de PASKWIL-criteria als klinisch relevant beschouwd. Het absolute verschil tussen encorafenib (23,5 maanden) plus vemurafenib (16,9 maanden) was 6,6 maanden met een HR van 0,76 (95% BI: 0,58 tot 0,98). Dit verschil werd volgens de PASKWIL-criteria niet als klinisch relevant beschouwd. De bewijskracht hiervan is redelijk, dit heeft te maken met de imprecisie, omdat de confidence interval de grens voor klinische besluitvorming omvat.
De studie rapporteerde ook het effect op progression free survival en adverse events. Voor de uitkomst progression free survival werd een klinisch relevant voordeel gevonden voor behandeling met encorafenib plus binimetinib vergeleken met vemurafenib. Het absolute verschil in mediane progressievrije overleving was 7.6 maanden (HR en CI niet gegeven). Er werd geen klinisch relevant verschil gevonden tussen de studiegroepen voor de uitkomst adverse events.
COMBI-d rapporteerde het effect van dabrafenib plus trametinib versus dabrafenib op de mediane OS. Behandeling met dabrafenib plus trametinib resulteerde in een langere mediane OS dan behandeling met alleen dabrafenib. Het absolute verschil tussen de dabrafenib plus trametinib groep (25,1 maanden) en de dabrafenib groep (18,7 maanden) was 6,4 maanden met een HR van 0,71 (95% BI: 0,55 tot 0,92). Dit verschil werd volgens de PASKWIL-criteria niet als klinisch relevant beschouwd. De bewijskracht hiervan is laag, dit heeft te maken met risk of bias en imprecisie, omdat de confidence interval de grens voor klinische besluitvorming omvat.
De studie rapporteerde ook het effect op progression free survival en adverse events. Er werd geen klinisch relevant verschil gevonden tussen de studiegroepen voor de uitkomsten progression free survival en adverse events.
DREAMseq rapporteerde het effect van nivolumab plus ipilimumab versus dabrafenib plus trametinib op overall survival bij patiënten met irresectabele of gemetastaseerd stadium III/IV melanoom (Atkins 2022). Een langere survival werd gevonden bij het gebruik van nivolumab plus ipilimumab, maar de klinische relevantie van dit verschil kan niet worden beoordeeld op basis omdat er geen mediane survival is gerapporteerd. Hierdoor is de bewijskracht redelijk.
De studie rapporteerde ook het effect op progression free survival en adverse events. Een langere mediane progression free survival werd gevonden voor patiënten in de groep met nivolumab plus ipilimumab , met een absoluut verschil van 3.3 maanden met patiënten in de nivolumab plus ipilimumab groep. De klinische relevantie van dit verschil kan niet worden beoordeeld op basis van de PASKWIL criteria (geen HR gerapporteerd). Er werd geen klinisch relevant verschil gevonden tussen de studiegroepen voor het ontstaan van adverse events. Behandeling met nivolumab plus ipilimumab resulteerde in een hoger percentage behandeling gerelateerde adverse events grade >3 in vergelijking met behandeling met dabrafenib plus trametinib.
IMspire150 rapporteerde het effect van atezoluzumab plus vemurafenib plus cobimetinib versus vemurafenib plus cobimetinib op overall survival bij patiënten met irresectabele of gemetastaseerd stadium III/IV melanoom (Ascierto 2023). Het absolute verschil in mediane overall survival tussen behandeling met atezoluzumab en vemurafenib en cobimetinib versus behandeling met vemurafenib en cobimetinib was 13.2 maanden met een hazard ratio van 0.84 (95% CI 0.66–1.06), maar dit verschil was niet klinisch relevant. De bewijskracht hiervan is redelijk, dit heeft te maken met imprecisie, omdat de confidence interval de grens voor klinische besluitvorming omvat.
De studie rapporteerde ook het effect op progression free survival en adverse events. Een langere mediane progression free survival werd gevonden voor patiënten die behandeld warden met behandeling met atezoluzumab en vemurafenib en cobimetinib versus behandeling met vemurafenib en cobimetinib, met een absoluut verschil van 4.5 maanden (HR 0.79 (95%CI 0.64-0.97), dit was niet klinisch relevant.
Behandeling met atezoluzumab en vemurafenib en cobimetinib resulteerde in een hoger percentage behandeling gerelateerde adverse events grade ≥3 in vergelijking met behandeling met alleen vemurafenib en cobimetinib, dit verschil was niet klinisch relevant.
Kwaliteit van bewijs
Relatlimab plus nivolumab versus nivolumab
De overall kwaliteit van bewijs is laag. Dit betekent dat we onzeker zijn over het gevonden geschatte effect van de belangrijke uitkomstmaten (cruciale uitkomstmaat overall survival werd niet gerapporteerd).
Nivolumab plus ipilimumab versus nivolumab versus ipilimumab
Nivolumab plus ipilimumab versus nivolumab
De overall kwaliteit van bewijs is laag. Dit betekent dat we onzeker zijn over het gevonden geschatte effect van de cruciale uitkomstmaat overall survival.
Nivolumab plus ipilimumab versus ipilimumab
De overall kwaliteit van bewijs is hoog. Dit betekent dat we zeer zeker zijn over het gevonden geschatte effect van de cruciale uitkomstmaat overall survival.
Nivolumab versus ipilimumab
De overall kwaliteit van bewijs is redelijk. Dit betekent dat we redelijk zeker zijn over het gevonden geschatte effect van de cruciale uitkomstmaat overall survival.
Ipilimumab plus dacarbazine versus dacarbazine
De overall kwaliteit van bewijs is laag. Dit betekent dat we onzeker zijn over het gevonden geschatte effect van de cruciale uitkomstmaat overall survival.
Pembrolizumab every 2 weeks or every 3 weeks versus ipilimumab
De overall kwaliteit van bewijs is zeer laag. Dit betekent dat we zeer onzeker zijn over het gevonden geschatte effect van de cruciale uitkomstmaat overall survival.
T-VEC plus pembrolizumab versus placebo plus pembrolizumab
De overall kwaliteit van bewijs is zeer laag. Dit betekent dat we zeer onzeker zijn over het gevonden geschatte effect van de cruciale uitkomstmaat overall survival.
T-VEC plus pembrolizumab versus GM-CSF
De overall kwaliteit van bewijs is zeer laag. Dit betekent dat we zeer onzeker zijn over het gevonden geschatte effect van de cruciale uitkomstmaat overall survival.
Cobimetinib plus vemurafenib versus vemurafenib plus placebo
De overall kwaliteit van bewijs is redelijk. Dit betekent dat we redelijk zeker zijn over het gevonden geschatte effect van de cruciale uitkomstmaat overall survival.
Dabrafenib plus trametinib versus vemurafenib
De overall kwaliteit van bewijs is redelijk. Dit betekent dat we redelijk zeker zijn over het gevonden geschatte effect van de cruciale uitkomstmaat overall survival.
Encorafenib plus binimetinib versus encorafenib versus vemurafenib
Encorafenib plus binimetinib versus encorafenib
De overall kwaliteit van bewijs is redelijk. Dit betekent dat we redelijk zeker zijn over het gevonden geschatte effect van de cruciale uitkomstmaat overall survival.
Encorafenib plus binimetinib versus vemurafenib
De overall kwaliteit van bewijs is laag. Dit betekent dat we onzeker zijn over het gevonden geschatte effect van de cruciale uitkomstmaat overall survival.
Encorafenib plus vemurafenib
De overall kwaliteit van bewijs is laag. Dit betekent dat we onzeker zijn over het gevonden geschatte effect van de cruciale uitkomstmaat overall survival.
Dabrafenib plus trametinib versus dabrafenib
De overall kwaliteit van bewijs is laag. Dit betekent dat we onzeker zijn over het gevonden geschatte effect van de cruciale uitkomstmaat overall survival.
Nivolumab plus ipilimumab versus dabrafenib plus trametinib.
De overall kwaliteit van bewijs is redelijk. Dit betekent dat we redelijk zeker zijn over het gevonden geschatte effect van de cruciale uitkomstmaat overall survival.
Atezoluzumab plus vemurafenib and cobimetinib versus vemurafenib pluscobimetinib.
De overall kwaliteit van bewijs is redelijk. Dit betekent dat we redelijk zeker zijn over het gevonden geschatte effect van de cruciale uitkomstmaat overall survival.
Ondanks de vooruitgang in de behandeling van irresectabel of gemetastaseerd melanoom, blijven veel vragen onbeantwoord en voor een belangrijke deel van de patiënten blijft de prognose slecht. Inclusie van irresectabel of gemetastaseerd stadium III/IV melanoom patienten in klinische studies blijft daarom de hoogste prioriteit in alle settings.
Waarden en voorkeuren van patiënten (en eventueel hun naasten/verzorgers)
Bij het kiezen van een behandelstrategie is het van belang om de waarden en voorkeuren van de patiënt centraal te stellen en de beslissing te individualiseren op basis van zowel het gewenste behandeldoel (kortetermijnvoordeel versus langetermijnvoordeel) als klinische kenmerken zoals lactaatdehydrogenase (LDH)-niveau, betrokken organen, performance status, tumorlast, de snelheid van ziekteprogressie en de bijwerkingen van de behandelingen.
Professioneel perspectief
Immuuntherapie (PD-1-blokkade of PD-1-blokkade gecombineerd met ipilimumab) heeft de voorkeur, omdat het na het stoppen langdurige ziektecontrole kan bieden. Bij patiënten met een BRAFV600- gemuteerd melanoom heeft immuuntherapie in deze situatie ook de voorkeur boven BRAF-/MEK-remmers omdat aangetoond is dat deze volgorde een betere overleving geeft.
De keuze tussen monotherapie en combinatiebehandeling wordt mede bepaald door klinische factoren; bijvoorbeeld, patiënten met asymptomatische hersenmetastasen en/of een verhoogd LDH kunnen meer baat hebben bij combinatiebehandeling. In fase 2 studies (ABC studie en Checkmate 204) werd bij patiënten met asymptomatische hersenmetastasen een hogere responskans en betere overleving vastgesteld met ipilimumab + nivolumab (Long, 2019).
Voor patiënten met een BRAFV600-gemuteerd melanoom waarbij immuuntherapie in de eerste maanden niet mogelijk is, zoals bij snel progressieve metastasen die vitale organen of functies bedreigen of gebruik van dexamethason bij symptomatische hersenmetastasen wordt aangeraden om in de eerstelijn doelgerichte therapie (BRAF + MEK-inhibitie) te overwegen. In dit geval zou vervolgens na 8 tot 12 weken behandeling met BRAF+MEK-inhibitie een switch naar ipilimumab + nivolumab kunnen overwogen worden (conform de SECOMBIT (arm C) en EBIN (arm B) studie) (Ascierto, 2023; Robert, 2024 (meeting abstract: 2024 ASCO Annual Metting II).
Kostenaspecten
Vanwege geheime prijsafspraken, kan de exacte impact op het geneesmiddelenbudget niet worden vastgesteld, maar het staat vast dat deze impact hoog is. Het huidig prijsniveau wordt echter acceptabel geacht in verhouding tot de effectiviteit van de behandeling. Een lagere prijs van de behandelingen zou desondanks in alle opzichten zeer wenselijk en naar mening van de werkgroep zelfs noodzakelijk zijn, mede met het oog op de komende ontwikkelingen en het betaalbaar houden en borgen van een goede kwaliteit van de zorg in de nabije toekomst.
Haalbaarheid/aanvaardbaarheid
Bij de behandeling van patiënten met een irresectabel of gemetastaseerd stadium III/IV melanoom is het van belang niet alleen te kijken naar klinische effectiviteit en patiëntvoorkeuren, maar ook naar de haalbaarheid en aanvaardbaarheid van de aanbevolen behandelopties. Deelname aan klinische studies kan voor sommige patiënten een haalbare optie zijn, mits er toegang is tot geschikte onderzoeksfaciliteiten en de patiënt bereid is de mogelijk intensieve studieverplichtingen te dragen.
Rationale van de aanbevelingen
De werkgroep is van mening dat deelname aan klinische studies in de behandeling van irresectabel of gemetastaseerd stadium III/IV melanoom de hoogste prioriteit heeft, gezien de sombere prognose voor een groot deel van de patiënten en de noodzaak om effectiviteit en aanvaardbaarheid van behandelingen verder te optimaliseren. Hierbij is het essentieel om patiëntwaarden en individuele klinische kenmerken leidend te laten zijn, zodat behandelbeslissingen aansluiten bij zowel haalbaarheid als de persoonlijke voorkeuren van de patiënt.
Onderbouwing
Relatlimab plus nivolumab versus nivolumab
Progression free survival
|
Low GRADE |
Relatlimab plus nivolumab may result in a small increase in progression free survival compared to nivolumab in patients with BRAF-V600E/K mutation with unresectable or metastatic stadium III/IV melanoma receiving first line systemic therapy.
Source: Tawbi, 2022 |
Adverse events
|
Low GRADE |
Relatlimab plus nivolumab may result in little to no difference in adverse events compared to nivolumab in patients with BRAF-V600E/K mutated unresectable or metastatic stadium III/IV melanoma receiving first line systemic therapy.
Source: Tawbi, 2022 |
Quality of Life
|
Low GRADE |
Relatlimab plus nivolumab may result in little to no difference in quality of life compared to nivolumab in patients with BRAF-V600E/K mutation with unresectable or metastatic stadium III/IV melanoma receiving first line systemic therapy.
Source: Tawbi, 2022 |
Nivolumab plus ipilimumab versus nivolumab versus ipilimumab
Overall survival
|
Low GRADE |
Nivolumab plus ipilimumab may result in little to no difference in overall survival compared to treatment with nivolumab only in patients with BRAF-V600E/K mutation with unresectable or metastatic stadium III/IV melanoma receiving first line systemic therapy.
Sources: Larkin, 2015; Wolchock, 2017; Hodi, 2018; Larkin, 2019; Wolchok, 2022 |
|
High GRADE |
Nivolumab plus ipilimumab results in a large increase in overall survival compared to treatment with ipilimumab only in patients with BRAF-V600E/K mutation with unresectable or metastatic stadium III/IV melanoma receiving first line systemic therapy.
Sources: Larkin, 2015; Wolchock, 2017; Hodi, 2018; Larkin, 2019; Wolchok, 2022 |
|
Moderate GRADE |
Nivolumab plus ipilimumab likely results in a large increase in overall survival compared to treatment with ipilimumab only in patients with BRAF-V600E/K mutation with unresectable or metastatic stadium III/IV melanoma receiving first line systemic therapy.
Sources: Larkin, 2015; Wolchock, 2017; Hodi, 2018; Larkin, 2019; Wolchok, 2022 |
Progression free survival
|
Low GRADE |
Nivolumab plus ipilimumab may result in little to no difference in progression free survival compared to treatment with nivolumab only in patients with BRAF-V600E/K mutation with unresectable or metastatic stadium III/IV melanoma receiving first line systemic therapy.
Sources: Larkin, 2015; Wolchock, 2017; Hodi, 2018; Larkin, 2019; Wolchok, 2022 |
|
High GRADE |
Nivolumab plus ipilimumab results in a large increase in progression free survival compared to treatment with ipilimumab only in patients with BRAF-V600E/K mutation with unresectable or metastatic stadium III/IV melanoma receiving first line systemic therapy.
Sources: Larkin, 2015; Wolchock, 2017; Hodi, 2018; Larkin, 2019; Wolchok, 2022 |
|
High GRADE |
Nivolumab versus ipilimumab result in an increase in progression free survival compared to treatment with ipilimumab only in patients with BRAF-V600E/K mutation with unresectable or metastatic stadium III/IV melanoma receiving first line systemic therapy.
Sources: Larkin, 2015; Wolchock, 2017; Hodi, 2018; Larkin, 2019; Wolchok, 2022 |
Adverse events
|
Low GRADE |
Nivolumab plus ipilimumab may result in an increase of adverse events compared to treatment with nivolumab only in patients with BRAF-V600E/K mutation with unresectable or metastatic stadium III/IV melanoma receiving first line systemic therapy.
Sources: Larkin, 2015; Wolchock, 2017; Hodi, 2018; Larkin, 2019; Wolchok, 2022 |
|
Moderate GRADE |
Nivolumab plus ipilimumab likely result in an increase of adverse events compared to treatment with ipilimumab only in patients with BRAF-V600E/K mutation with unresectable or metastatic stadium III/IV melanoma receiving first line systemic therapy.
Sources: Larkin, 2015; Wolchock, 2017; Hodi, 2018; Larkin, 2019; Wolchok, 2022 |
|
Low GRADE |
Nivolumab may result in little to no difference in adverse events compared to treatment with ipilimumab in patients with BRAF-V600E/K mutation with unresectable or metastatic stadium III/IV melanoma receiving first line systemic therapy.
Sources: Larkin, 2015; Wolchock, 2017; Hodi, 2018; Larkin, 2019; Wolchok, 2022 |
Ipilimumab plus dacarbazine versus dacarbazine
Overall survival
|
Very low GRADE |
The evidence is very uncertain about the effect of ipilimumab plus dacarbazine on overall survival compared to treatment with dacarbazine only in patients with BRAF-V600E/K mutation with unresectable or metastatic stadium III/IV melanoma receiving first line systemic therapy.
Sources: Maio, 2015; Robert, 2011 |
Progression free survival
|
Very low GRADE |
The evidence is very uncertain about the effect of ipilimumab plus dacarbazine on progression free survival compared to treatment with dacarbazine only in patients with BRAF-V600E/K mutation with unresectable or metastatic stadium III/IV melanoma receiving first line systemic therapy.
Sources: Maio, 2015; Robert, 2011 |
Adverse events
|
Very low GRADE |
The evidence is very uncertain about the effect of ipilimumab plus dacarbazine on the incidence of adverse events compared to treatment with dacarbazine only in patients with BRAF-V600E/K mutation with unresectable or metastatic stadium III/IV melanoma receiving first line systemic therapy.
Sources: Maio, 2015; Robert, 2011 |
Pembrolizumab every 2 weeks or every 3 weeks compared to ipilimumab
Overall survival
|
Very low GRADE |
The evidence is very uncertain about the effect of pembrolizumab every 2 weeks or every 3 weeks on overall survival compared to treatment with ipilimumab in patients with BRAF-V600E/K mutation with unresectable or metastatic stadium III/IV melanoma receiving first line systemic therapy.
Sources: Robert, 2019; Carlino, 2018; Schachter, 2017; Robert, 2015 |
Progression free survival
|
Very low GRADE |
The evidence is very uncertain about the effect of pembrolizumab every 2 weeks or every 3 weeks on progression free survival compared to treatment with ipilimumab in patients with BRAF-V600E/K mutation with unresectable or metastatic stadium III/IV melanoma receiving first line systemic therapy.
Sources: Robert, 2019; Carlino, 2018; Schachter, 2017; Robert, 2015 |
Adverse events
|
Very low GRADE |
The evidence is very uncertain about the effect of pembrolizumab every 2 weeks or every 3 weeks on adverse events compared to treatment with ipilimumab in patients with BRAF-V600E/K mutation with unresectable or metastatic stadium III/IV melanoma receiving first line systemic therapy.
Sources: Robert, 2019; Carlino, 2018; Schachter, 2017; Robert, 2015 |
T-VEC plus pembrolizumab versus placebo plus pembrolizumab
Overall survival
|
Very low GRADE |
The evidence is very uncertain about the effect of T-VEC plus pembrolizumab on overall survival compared to treatment with placebo plus pembrolizumab in patients with BRAF-V600E/K mutation with unresectable or metatsatic stadium III/IV melanoma receiving first line systemic therapy.
Source: Chesney, 2023 |
Progression free survival
|
Very low GRADE |
The evidence is very uncertain about the effect of T-VEC plus pembrolizumab on progression free survival compared to treatment with placebo plus pembrolizumab in patients with BRAF-V600E/K mutation with unresectable or metastatic stadium III/IV melanoma receiving first line systemic therapy.
Source: Chesney, 2023 |
Adverse events
|
Very low GRADE |
The evidence is very uncertain about the effect of T-VEC plus pembrolizumab on adverse events compared to treatment with placebo plus pembrolizumab in patients with BRAF-V600E/K mutation with unresectable or metastatic stadium III/IV melanoma receiving first line systemic therapy.
Source: Chesney, 2023 |
T-VEC versus GM-CSF
Overall survival
|
Very low GRADE |
The evidence is very uncertain about the effect of T-VEC on overall survival compared to treatment with GM-CSF in patients with BRAF-V600E/K mutation with unresectable or metastatic stadium III/IV melanoma receiving first line systemic therapy.
Sources: Andtbacka, 2019; Andtbacka, 2015 |
Adverse events
|
Very low GRADE |
The evidence is very uncertain about the effect of T-VEC on adverse eventscompared to treatment with GM-CSF in patients with BRAF-V600E/K mutation with unresectable or metastatic stadium III/IV melanoma receiving first line systemic therapy.
Sources: Andtbacka, 2019; Andtbacka, 2015 |
Cobimetinib plus vemurafenib versus vemurafenib and placebo
Overall survival
|
Moderate GRADE |
Cobimetinib plus vemurafenib likely results in an increase in overall survival when compared with vemurafenib and placebo in patients with BRAF-V600E/K mutation with unresectable or metastatic stadium III/IV melanoma receiving first line systemic therapy.
Sources: Larkin, 2014; Ascierto, 2016; Ascierto, 2021 |
Progression free survival
|
High GRADE |
Combimetinib plus vemurafenib results in an increase in progression free survival when compared with vemurafenib and placebo in patients with BRAF-V600E/K mutation with unresectable or metastatic stadium III/IV melanoma receiving first line systemic therapy.
Sources: Larkin, 2014; Ascierto, 2016; Ascierto, 2021 |
Adverse events
|
High GRADE |
Combimetinib plus vemurafenib results in little to no difference in adverse events when compared with vemurafenib and placebo in patients with BRAF-V600E/K mutation with unresectable or metastatic stadium III/IV melanoma receiving first line systemic therapy.
Sources: Larkin, 2014; Ascierto, 2016; Ascierto, 2021 |
Dabrafenib plus trametinib versus vemurafenib
Overall survival
|
Moderate GRADE |
Dabrafenib plus trametinib likely result in an increase in overall survival when compared with vemurafenib in patients with BRAF-V600E/K mutation with unresectable or metastatic stadium III/IV melanoma receiving first line systemic therapy.
Sources: Robert, 2015; Robert, 2016 |
Progression free survival
|
Moderate GRADE |
Dabrafenib plus trametinib likely result in an increase in progression free survival when compared with vemurafenib and placebo in patients with BRAF-V600E/K mutation with unresectable or metastatic stadium III/IV melanoma receiving first line systemic therapy.
Sources: Robert, 2015; Robert, 2016 |
Adverse events
|
Moderate GRADE |
Dabrafenib plus trametinib likely results in little to no difference in adverse events when compared with vemurafenib in patients with BRAF-V600E/K mutation with unresectable or metastatic stadium III/IV melanoma receiving first line systemic therapy.
Sources: Robert, 2015; Robert, 2016 |
Encorafenib plus binimetinib versus encorafenib versus vemurafenib
Encorafenib and binimetinib versus encorafenib
Overall survival
|
Moderate GRADE |
Encorafenib plus binimetinib likely result in little to no difference in overall survival when compared with encorafenib in patients with BRAF-V600E/K mutation with unresectable or metastatic stadium III/IV melanoma receiving first line systemic therapy.
Sources: Dummer, 2018; Dummer, 2018-2; Ascierto, 2020 |
Adverse events
|
Low GRADE |
Encorafenib plus binimetinib may result in little to no difference in adverse events when compared with encorafenib in patients with BRAF-V600E/K mutation with unresectable or metastatic stadium III/IV melanoma receiving first line systemic therapy.
Sources: Dummer, 2018; Dummer, 2018-2; Ascierto, 2020 |
Encorafenib plus binimetinib versus vemurafenib
Overall survival
|
Moderate GRADE |
Encorafenib plus binimetinib likely result in an increase in overall survival when compared with vemurafenib in patients with BRAF-V600E/K mutation with unresectable or metastatic stadium III/IV melanoma receiving first line systemic therapy.
Sources: Dummer, 2018; Dummer, 2018-2; Ascierto, 2020 |
Progression free survival
|
Moderate GRADE |
Encorafenib plus binimetinib likely result in an increase in progression free survival when compared with vemurafenib in patients with BRAF-V600E/K mutation with unresectable or metastatic stadium III/IV melanoma receiving first line systemic therapy.
Sources: Dummer, 2018; Dummer, 2018-2; Ascierto, 2020 |
Adverse events
|
Low GRADE |
Encorafenib plus binimetinib may result in little to no difference in adverse events when compared with vemurafenib in patients with BRAF-V600E/K mutation with unresectable or metastatic stadium III/IV melanoma receiving first line systemic therapy.
Sources: Dummer, 2018; Dummer, 2018-2; Ascierto, 2020 |
Encorafenib versus vemurafenib
Overall survival
|
Moderate GRADE |
Encorafenib likely results in little to no difference in overall survival when compared with vemurafenib in patients with BRAF-V600E/K mutation with unresectable or metastatic stadium III/IV melanoma receiving first line systemic therapy.
Sources: Dummer, 2018; Dummer, 2018-2; Ascierto, 2020 |
Progression free survival
|
Low GRADE |
Encorafenib may result in little to no difference in overall survival when compared with vemurafenib in patients with BRAF-V600E/K mutation with unresectable or metastatic stadium III/IV melanoma receiving first line systemic therapy.
Sources: Dummer, 2018; Dummer, 2018-2; Ascierto, 2020 |
Adverse events
|
Low GRADE |
Encorafenib may result in little to no difference in adverse events when compared with vemurafenib in patients with BRAF-V600E/K mutation with unresectable or metastatic stadium III/IV melanoma receiving first line systemic therapy.
Sources: Dummer, 2018; Dummer, 2018-2; Ascierto, 2020 |
Dabrafenib plus trametinib versus dabrafenib
Overall survival
|
Low GRADE |
Dabrafenib plus trametinib may result in little to no difference in overall survival when compared with dabrafenib in patients with BRAF-V600E/K mutation with unresectable or metastatic stadium III/IV melanoma receiving first line systemic therapy.
Sources: Long, 2014; Long, 2015; Long, 2017 |
Progression free survival
|
Moderate GRADE |
Dabrafenib plus trametinib likely result in little to no difference in progression free survival when compared with dabrafenib in patients with BRAF-V600E/K mutation with unresectable or metastatic stadium III/IV melanoma receiving first line systemic therapy.
Sources: Long, 2014; Long, 2015; Long, 2017 |
Adverse events
|
Low GRADE |
Dabrafenib plus trametinib may result in little to no difference in adverse events when compared with dabrafenib in patients with BRAF-V600E/K mutation with unresectable or metastatic stadium III/IV melanoma receiving first line systemic therapy.
Sources: Long, 2014; Long, 2015; Long, 2017 |
Dabrafenib and trametinib versus nivolumab and ipilimumab
Overall survival
|
Moderate GRADE |
Nivolumab plus ipilimumab likely result in a large increase in overall survival compared to dabrafenib and trametinib in patients with BRAF-V600E/K mutation with unresectable or metastatic stadium III/IV melanoma receiving first line systemic therapy.
Source: Atkins, 2022 |
Progression free survival
|
Moderate GRADE |
Nivolumab plus ipilimumab likely result in a large increase in progression free survival compared to dabrafenib and trametinib TKI in patients with BRAF-V600E/K mutation with unresectable or metastatic stadium III/IV melanoma receiving first line systemic therapy.
Source: Atkins, 2022 |
Adverse events
|
Low GRADE |
Nivolumab and ipilimumab may result in little to no difference in adverse events compared to dabrafenib and trametinib TKI in patients with BRAF-V600E/K mutation with unresectable or metastatic stadium III/IV melanoma receiving first line systemic therapy.
Source: Atkins, 2022 |
Atezolozumab plus vemurafenib plus cobimetinib versus vemurafenib plus cobimetinib
Overall survival
|
Moderate GRADE |
Atezolizumab plus vemurafenib plus cobimetinib likely result in little to no difference in overall survival compared to vemurafenib and cobimetinib TKI in patients with BRAF-V600E/K mutation with unresectable or metastatic stadium III/IV melanoma receiving first line systemic therapy.
Source: Ascierto, 2023 |
Progression free survival
|
Moderate GRADE |
Atezolizumab plus vemurafenib plus cobimetinib likely result in little to no difference in progression free survival compared to vemurafenib and cobimetinib TKI in patients with BRAF-V600E/K mutation with unresectable or metastatic stadium III/IV melanoma receiving first line systemic therapy.
Source: Ascierto, 2023 |
Adverse events
|
Low GRADE |
Atezolizumab plus vemurafenib plus cobimetinib may result in little to no difference in adverse events compared to vemurafenib and cobimetinib TKI in patients with BRAF-V600E/K mutation with unresectable or metastatic stadium III/IV melanoma receiving first line systemic therapy.
Source: Ascierto, 2023 |
Twelve randomized controlled trials that studied clinical outcomes of first line systemic therapy in patients with unresectable or metastatic stadium III/IV melanoma with a BRAF-V600E/K mutation were included in the literature analysis.
Description of studies
The study characteristics of the included trials are summarized in Table 1 in the main module module Systematische therapie.
Tawbi (2022) – RELATIVITY-047 described a phase 2-3 global, double-blind, randomized study, which was conducted in 111 sites in North America, Central America, South America, Europe, Australia, and New Zealand. They evaluated the efficacy and safety of the combination of relatlimab, a LAG-3–blocking antibody, and nivolumab in patients with previously intreated metastatic or unresectable melanoma. A total of 714 patients were randomized to receive a relatlimab and nivolumab combination (160 mg of relatlimab and 480 mg of nivolumab in a fixed-dose combination administered in a single 60-minute intravenous infusion every 4 weeks) versus nivolumab alone (480 mg of nivolumab administered in a single 60-minute intravenous infusion every 4 weeks). The median age was 63 (20-94) years in the relatlimab + nivolumab group and 62.0 (21–90) years in the control group. In the relatlimab + nivolumab group 59% was male, compared to 57% in the nivolumab alone group. 136 of the 355 (38.3%) patients that were randomized in the relatlimab + nivolumab group, and 139 of the 359 (38.7%) patients that were randomized in the nivolumab alone group, had a BRAF mutation. The following relevant outcomes were reported progression free survival (PFS), adverse events (AEs), and quality of life (QoL). In this literature analysis the median PFS is analysed.
Larkin (2015), Larkin (2019), Wolchock (2017), Wolchok (2022), Hodi (2018) - CHECKMATE 067 is a phase 3, randomized, double-blind study, which is conducted in 137 centres in Australia, Europe, Israel, New Zealand, and North America. They evaluated the efficacy and safety of the use of Nivolumab and Ipilimumab as combination therapy in patients with untreated unresectable stage III or stage IV melanoma. A total of 945 patients were randomized to receive nivolumab 1 mg/kg plus ipilimumab 3 mg/kg once every 3 weeks followed by nivolumab 3 mg/kg once every 2 weeks (n=314, of which 101 patients (32.2%) had a BRAF mutation), or nivolumab 3 mg/kg once every 2 weeks (n=316, of which 100 patients (31.6%) had a BRAF mutation), or ipilimumab 3 mg/kg once every 3 weeks (n=315, of which 97 patients (30.8%) had a BRAF mutation). The median age, of both patients with and without BRAF mutation, was 59 (18-88) years in the combination group, 59 (25-90) in the nivolumab group and 61 (18-89) in the Ipilimumab group. In the combination group, 66% was male, compared to 64% in the nivolumab alone group and 64% in the Ipilimumab group. The following relevant outcomes were reported, overall survival (OS), PFS, and number of patients with serious AEs. The study was not powered for a formal statistical comparison between the nivolumab group and the nivolumab-plus-ipilimumab group. In this literature analysis the median outcomes for OS and PFS are analysed and the last endpoint for OS (6.5-year OS rates) are described.
Robert (2011), Maio (2015) - CA184-024 is a multinational, randomized, double-blind, phase 3 study. This trial compared ipilimumab plus dacarbazine with dacarbazine and placebo in patients with previously untreated stage III (unresectable) or stage IV melanoma with measurable lesions. Tumors were not routinely assessed for the presence of the BRAF V600E mutation and therefore no sub analyses was possible in this study. Patients were randomized to receive ipilimumab at a dose of 10 mg per kg plus dacarbazine at a dose of 850 mg per square meter (n=250) or dacarbazine at a dose of 850 mg per square meter plus placebo (n=252). The mean age was 57.5 years in the intervention group and 56.4 years in the control group. In the intervention group 60.8% and in the control group 59.1% were male. Robert (2011) reported on OS, PFS, and AEs after a follow up time of 54 months between the start of the study (first visit of first patient) and end of the study. Maio (2015) reported updated results after a median survival follow-up of 11 months (range, 0.4 to 71.9 months) for the intervention group and 8.9 months (range, 0.1 to 73.2 months) for the control group. In this literature analysis the median outcomes for OS and PFS are analysed and the last endpoint for OS (5-year OS rates) are described.
Robert (2019), Carlino (2018), Schachter (2017), Robert (2015) - KEYNOTE-006 is an international, randomized, open-label phase 3 study performed in 16 countries. In this trial treatment with pembrolizumab versus ipilimumab was studied, to compare PD-1 inhibition with CTLA-4 blockade in patients with unresectable stage III/IV melanoma. Patients were randomized to pembrolizumab at a dose of 10 mg/kg of body weight every 2 weeks (n= 279); pembrolizumab at a dose of 10 mg/kg every 3 weeks (n=277); or ipilimumab at a dose of 3 mg/kg every 3 weeks (n=278). The mean age was 61 years in the pembrolizumab every 2 weeks group, 63 years in the pembrolizumab every 3 weeks group and 62 years in the ipilimumab group. The percentage of males was 57.7 %, 62.8%, and 58.3% for the three groups, respectively. Robert (2015) reported on OS, PFS, and AEs after a median follow-up of 7.9 months. Schachter (2017) reported updated results after a median follow-up of 22.9 months. Carlino (2018) reported updated outcomes by line of therapy and programmed death ligand 1 expression after a median follow-up of 33.9 months. Robert (2019) reported updated results of OS, PFS, and AES after a median follow-up of 57.7 months. In this literature analysis the median outcomes for OS and PFS are analysed and the last endpoints for OS and PFS (2-year OS rates and 2-year PFS rates) are described.
Chesney (2023) - MASTERKEY-265 is a multicenter, double-blind, placebo controlled, randomized phase 3 study in 21 countries. This trial evaluated the efficacy and safety of T-VEC-pembrolizumab versus placebo-pembrolizumab in patients with stage IIIB-IV M1c unresectable melanoma. Patients were randomized to a combination of T-VEC plus pembrolizumab 200 mg once every 3 weeks (n=346) or placebo plus pembrolizumab 200 mg once every 3 weeks (n=346). The median age was 64 years in both study groups. The percentage of males was 57.5% in the T-VEC-pembrolizumab group and 63.3% placebo-pembrolizumab group. Chesney (2023) reported OS, PFS, and AES, after a median follow-up of 25.6 months for the primary PFS analysis, 31.0 months for the second OS interim analysis, and 35.6 months for the final analysis. In this literature analysis the median outcomes for OS and PFS are analysed.
Andtbacka (2019), Andtbacka (2015) – OPTiM is a randomized open-label phase 3 trial at 64 sites in the United States, the United Kingdom, Canada, and South Africa. This trial evaluated outcomes after treatment with talimogene laherparepvec (T-VEC) compared with granulocyte macrophage colony-stimulating factor (GM-CSF) in patients with unresectable, stage IIIB/C/IV melanoma with ≥1 lesion that was suitable for direct or ultrasound-guided injection. Patients were randomized to T-VEC (at the approved dose) (n=295 (68%)) of subcutaneous recombinant GM-CSF (n=141 (32%)). The median age was 63 years in the T-VEC group and 64 years in the GM-CSF group. The percentage of males was 59% in the T-VEC group and 55% in the GM-CSF group. Of 204 of the 295 (69%) in the T-VEC group, and 95 of the 141 (67%) in the GM-CSF group, the BRAF mutation status was unknown. For 138 of the 295 (47%) patients in the T-VEC group, and 65 of the 141 (46%) in the GM-CSF group, this was a first line therapy. OS and AEs were reported after a median follow-up of 49 months in the final analysis of OS. In this literature analysis the median OS is analysed.
Larkin (2014), Ascierto (2016), Ascierto (2021) - CoBRIM described an international, multicentre, double-blind, randomized phase 3 study, which was conducted in 135 sites in the United States, Canada, Australia, New Zealand, Europe, Russia, Turkey, and Israel. The authors evaluated efficacy and safety of using cobimetinib combined with vemurafenib, versus vemurafenib and placebo, in previously untreated patients with advanced BRAF-mutated melanoma. A total of 495 patients were randomized in a 1:1 ratio to receive vemurafenib orally (at a dose of 960 mg twice daily) together with either placebo (control group) or cobimetinib (at a dose of 60 mg once daily for 21 days, followed by 7 days off) (combination group). The median age was 56 (23-88) years in the combination group and 55 (25-85) years in the control group. In the combination group 59% was male, compared to 56% in the control group. The following relevant outcomes were reported, OS, PFS and number of patients with serious AEs. In this literature analysis, the median outcomes for OS were analysed and the last endpoint (5-year OS and 3-year PFS) for the outcomes OS and PFS are described.
Robert (2015), Robert (2016) - COMBI-v described an open-label, randomized, phase 3 study performed at 193 centres worldwide, where they evaluated the efficacy and safety of using combination therapy with dabrafenib plus trametinib versus vemurafenib monotherapy in patients with previously untreated patients with unresectable stage IIIC or IV melanoma with BRAF V600E or V600K mutations. A total of 704 patients were randomized in a 1:1 ratio to receive either a combination of dabrafenib (150 mg orally twice daily) and trametinib (2 mg orally once daily) or vemurafenib (960 mg orally twice daily). The median age was 55 (18–91) years in the combination group and 54 (18–88) years in the control group. In the combination group 59% was male, compared to 51% in the control group. The following relevant outcomes were reported, OS, PFS, and number of patients with serious AEs. In this literature analysis the median outcomes for OS and PFS are analysed and the last endpoints (3-year OS and 3-year PFS) of the outcomes are described.
Dummer (2018), Dummer (2018-2), Ascierto (2020) - COLUMBUS described a two-part, randomised, open-label, phase 3 study, performed at 162 sites in 28 countries. They evaluated the efficacy and safety of encorafenib plus binimetinib versus encorafenib alone and versus vemurafenib alone in patients with histologically confirmed, locally advanced, unresectable, or metastatic cutaneous melanoma, or unknown primary melanoma. In part 1 of the study, 577 patients were randomly assigned (1:1:1) to receive oral encorafenib 450 mg once daily plus oral binimetinib 45 mg twice daily (encorafenib plus binimetinib group), oral encorafenib 300 mg once daily encorafenib group), or oral vemurafenib 960 mg twice daily (vemurafenib group). The median age was 57 (20–89; 48–66) years in the combination group, 54 (23–88; 46–63) years in the encorafenib control group and 56 (21–82; 45–65) years in the vemurafenib control group. In the combination group 60% was male, compared to 60% and 56% in the two control groups, respectively. The following relevant outcomes were reported, OS, PFS and number of patients with serious AEs. In this literature analysis the median outcomes for OS and PFS are analysed and the last endpoint (3-year OS) for the outcome OS is described.
Long (2014), Long (2015), Long (2017) – COMBI-d is a double-blind, randomized, phase 3 study without crossover that is carried out at 113 centres worldwide. This trial compared the combination of dabrafenib and trametinib with dabrafenib alone as first-line therapy in patients with metastatic melanoma with BRAF V600E or V600K mutations. Participants were randomized (1:1) to receive a combination of oral dabrafenib (150 mg twice daily) and oral trametinib (2 mg once daily) (n=211), or oral dabrafenib (150 mg twice daily) and placebo (n=212). The median age (range) was 55.0 (22–89) in the dabrafenib and trametinib group and 56.5 (22–86) in the dabrafenib alone group. In the dabrafenib and trametinib group and the dabrafenib alone group 53% and 54% were male respectively. Long (2014) reported on PFS, OS, and number of patients with AEs. At that time, the median duration of follow-up was 9 months (range, 0 to 16). Long (2015) reported updated results of PFS, OS, and AEs after a median follow-up of 20 months (range 0–30) for the dabrafenib and trametinib group and 16 months (range 0–32 months) for the dabrafenib only group. Long (2017) reported an updated 3-year landmark analysis of PFS, OS, and AEs. In this literature analysis the median outcomes for OS and PFS are analysed and the last endpoint (3-year OS, 3-year PFS) of the outcomes are described.
Atkins (2022) – DREAMseq is a two-arm, two-step, open-label, randomized phase 3 trial. This trial compared the combination of nivolumab/ipilimumab with the combination dabrafenib/trametinib in patients with histologically confirmed, unresectable stage III or IV melanoma containing a BRAFV600E/K mutation. Patients were randomized (1:1) to receive step 1 with either nivolumab/ipilimumab (Arm A, n=133) or dabrafenib/trametinib (Arm B, n=132). At disease progression patients were enrolled in step 2 to receive the alternate therapy (Arm C (n=27) and D (=46)). Patients in arms A and D received nivolumab 1 mg/kg and ipilimumab 3 mg/kg once every 3 weeks for four doses followed by nivolumab 240 mg intravenously once every 2 weeks for up to 72 weeks. Patients in arms B and C received dabrafenib 150 mg twice a day and trametinib 2 mg orally once daily. The median age (range) was 61 (25-85) in arm A, and 61 (30-84) in arm B. In arm A 81% were male and in arm B 86% were male. The following outcomes are reported PFS, OS, and AEs. Median follow-up was 27.7 months (IQR, 41.9-11.9). In this literature analysis the median outcomes for PFS is analysed and the last endpoints (3-year OS and 2-year PFS) of the outcomes are described. The median OS was not reported.
Ascierto (2023) - IMspire150 is a multicentre, double blind, placebo-controlled, randomised, phase 3 study performed in 108 academic and community hospitals in 20 countries. This trial studied the combination of atezolizumab with vemurafenib and cobimetinib in patients with untreated, histologically confirmed stage IV or unresectable stage IIIc melanoma, with BRAFV600 mutation-positive tumours. Patients were randomized (1:1) to receive atezolizumab, vemurafenib, and cobimetinib (atezolizumab group, n=256) or placebo, vemurafenib, and cobimetinib (control group, n=258). In cycle 1 all patients received oral cobimetinib 60 mg once daily plus oral vemurafenib 960 mg twice daily for 21 days then vemurafenib 720 mg twice daily (atezolizumab group) or 960 mg twice daily (control group) for 7 days. From cycle 2 onwards atezolizumab or placebo was added. Patients in the atezolizumab group received intravenous atezolizumab 840 mg (day 1 and 15), once-daily cobimetinib 60 mg (21 days on and 7 days off), and twice-daily vemurafenib 720 mg. Patients in the control group received intravenous placebo (day 1 and 15), once-daily cobimetinib 60 mg (21 days on and 7 days off), and twice-daily vemurafenib 960 mg. The median age (range) was 54.0 years (44.8-64.0 years) in the atezolizumab group, and 53.5 years (43.0-63.8 years) in the control group. In the atezolizumab group 59% was male and in the control group 58% was male. The following outcomes are reported PFS, OS, and AEs. Median follow-up was 29.1 months (IQR 10.1–45.4) in the atezolizumab group and 22.8 months (10.6–44.1) in the control group. OS was not fully met. In this literature analysis the median outcomes for OS and PFS are analysed and the last endpoints (2-year OS and 1.5-year PFS) of the outcomes are described.
Results
Overall survival (OS) – Critical outcome measure
Eleven of the twelve included studies reported on OS.
Checkmate-067 reported the effect of nivolumab plus ipilimumab versus nivolumab versus ipilimumab on OS in patients with BRAF-mutant tumors. The study was not powered for a formal comparison between the nivolumab plus ipilimumab and the nivolumab treatment groups. The 6.5-year OS rates in patients with BRAF-mutant tumors were 57% for the nivolumab plus ipilimumab group, 43% for the nivolumab group, and 6% for the ipilimumab group. The median OS in the nivolumab plus ipilimumab group was not reported, and therefore no clinical relevance could be assessed according to the PASKWIL criteria. The absolute difference in median OS between the group treated with nivolumab (45.5 months) versus those treated with ipilimumab (24.6 months) was 20.9 months with a HR of 0.63 (95% CI 0.44 to 0.90) favoring treatment with nivolumab. This difference was considered clinically relevant according to the PASKWIL criteria.
CA184-024 reported the effect of ipilimumab plus dacarbazine versus dacarbazine plus placebo on OS. The 5-year OS rates were 18.2% for the ipilimumab plus dacarbazine group and 8.8% for the dacarbazine group. Treatment with ipilimumab plus dacarbazine resulted in a longer median OS compared to treatment with dacarbazine only. The absolute difference in median OS between the group treated with ipilimumab plus dacarbazine (11.2 months) versus those treated with dacarbazine (9.1 months) was 2.1 months with a HR of 0.69 (95% CI 0.57 to 0.84) favoring treatment with ipilimumab plus dacarbazine. This difference was not considered clinically relevant according to the PASKWIL criteria.
KEYNOTE-006 reported the effect of pembrolizumab every 2 weeks or pembrolizumab every 3 weeks compared to ipilimumab every 3 weeks on OS. The 2-year OS rates were 55% in the pembrolizumab-every-2-weeks group, 55% in the pembrolizumab-every-3-weeks group, and 43% in the ipilimumab-every-3-weeks group. Treatment with pembrolizumab resulted in a longer median OS than treatment with ipilimumab. The absolute difference between the combined pembrolizumab groups (32.7 months) and the ipilimumab group (15.9 months) was 16.8 months with a HR of 0.73 (95% CI 0.61–0.88). This difference was not considered clinically relevant according to the PASKWIL criteria.
MASTERKEY-265 reported the effect of a combination of T-VEC plus pembrolizumab versus placebo plus pembrolizumab on OS. Treatment with T-VEC-pembrolizumab did not result in a longer OS compared with treatment with placebo-pembrolizumab. The median OS was not estimable in the T-VEC plus pembrolizumab group and 49.2 months (40.57 to not estimable) in the pembrolizumab group with a HR of 0.96 (95% CI 0.76 to 1.22; P =0 .74). This difference was not considered clinically relevant according to the PASKWIL criteria.
OPTiM reported overall survival outcomes for the intention-to-treat (ITT) population, without specific subgroup analyses for first-line treatment or BRAF mutation status. Treatment with T-VEC resulted in a longer median OS than treatment with GM-CSF. The median follow-up in the final OS analysis was 49 months. Median OS weas 23.3 months (CI, 19.5-29.6) and 18.9 months (95% CI, 16.0-23.7) in the TVEC and GM-CSF arm, respectively (HR, 0.79; 95% CI, 0.62-1.00). This difference was not considered clinically relevant according to the PASKWIL criteria.
CoBRIM reported the effect of cobimetinib combined with vemurafenib versus vemurafenib and placebo on median OS. Treatment with cobimetinib combined with vemurafenib resulted in a longer median OS than treatment with vemurafenib and placebo. The absolute difference between the cobimetinib combined with vemurafenib group (22.5 months) and the vemurafenib and placebo group (17.4 months) was 5.1 months with a HR of 0.70 (95% 0.55 – 0.89). This difference was not considered clinically relevant according to the PASKWIL criteria.
COMBI-v did not reported the effect of dabrafenib and trametinib versus vemurafenib on median OS. However, the absolute difference in median 3-year survival between dabrafenib and trametinib (45%) versus vemurafenib (32%) was 13%, with a higher 3-year survival in the dabrafenib and trametinib group. This difference was considered clinically relevant according to the PASKWIL criteria.
COLUMBUS reported the effect of encorafenib and binimetinib versus encorafenib versus vemurafenib on median OS. Treatment with encorafenib and binimetinib resulted in the longest median OS. The absolute difference between encorafenib and binimetinib (33.6 months) and encorafenib (23.5 months) was 10.1 months with a HR of 0.81 (95% 0.61 to 1.06). This difference was not considered clinically relevant according to the PASKWIL criteria. The absolute difference between encorafenib and binimetinib (33.6 months) and vemurafenib (16.9 months) was 16.7 months with a HR of 0.61 (95% 0.47 to 0.79). This difference was considered clinically relevant according to the PASKWIL criteria. The absolute difference between encorafenib (23.5 months) and vemurafenib (16.9 months) was 6.6 months with a HR of 0.76 (95% CI 0.58 to 0.98).
This difference was not considered clinically relevant according to the PASKWIL criteria.
COMBI-d reported the effect of dabrafenib and trametinib versus dabrafenib on median OS. Treatment with dabrafenib and trametinib resulted in a longer median OS than treatment with dabrafenib alone. The absolute difference between the dabrafenib and trametinib group (25.1 months) and dabrafenib group (18.7 months) was 6.4 months with a HR of 0.71 (95% 0.55 to 0.92). This difference was not considered clinically relevant according to the PASKWIL criteria.
In the DREAMseq trial, combination treatment with nivolumab and ipilimumab versus dabrafenib and trametinib resulted in a longer OS. The absolute difference between the intervention and control group was 23.4% after 3 years of follow-up. According to the PASKWIL criteria we could not assess clinical relevance (median OS in the control group not reported).
In the IMspire150 trial, combination treatment with atezoluzumab and vemurafenib and cobimetinib versus vemurafenib and cobimetinib did not show a significant survival benefit.The absolute difference between the intervention and control group was 9% after 2 years of follow-up. The median OS was 39 months (29.9–not estimable) in the intervention group, versus 25.8 months (22.0-34.6 months) in the control group, with a HR of 0.84 (95% CI 0.66–1.06). This difference was not considered clinically relevant according to the PASKWIL criteria.
Figure 1. Forest plot of median overall survival for first line systemic therapy versus placebo, other systemic therapy, or best supportive care in patients with a BRAF-V600E/K mutation with unresectable or metastatic stadium III/IV melanoma
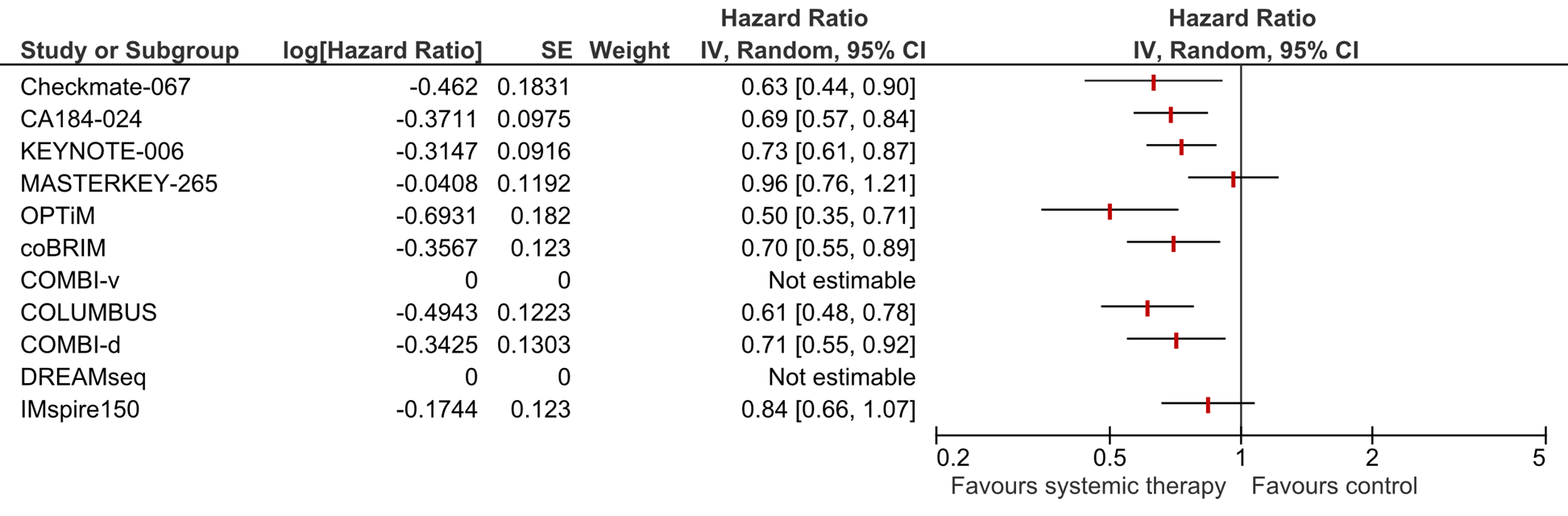
a For Checkmate-067 the HR for nivolumab versus ipilimumab is shown.
b For KEYNOTE-006 the HR for combined pembrolizumab versus ipilimumab is shown.
c For COLOMBUS the HR for encorafenib and binimetinib versus vemurafenib is shown.
d COMBI-v and DREAMseq did not report the HR for median OS and is therefore not shown in this figure.
Progression free survival – important outcome measure
Eleven of the twelve included studies reported on PFS.
RELATIVITY-047 reported the effect of relatlimab and nivolumab versus nivolumab on PFS. In the subgroup of patients with BRAF mutations, the median PFS was 10.1 months (95% CI, 4.6 to 23.1) in the relatlimab and nivolumab group and 4.6 months (95% CI, 3.0 to 6.5) in the nivolumab group. The absolute difference between the group treated with relatlimab and nivolumab and the group treated with nivolumab was 5.5 months with a HR of 0.74 (95%CI 0.54-1.03) favoring treatment with relatlimab and nivolumab. According to the PASKWIL criteria we could not assess clinical relevance (median OS in the control group not reported).
Checkmate-067 reported the effect of nivolumab plus ipilimumab versus nivolumab versus ipilimumab on PFS in patients with a BRAF mutant. Treatment with nivolumab plus ipilimumab resulted in a longer median PFS compared to treatment with nivolumab only. The absolute difference in median PFS between the group treated with nivolumab plus ipilimumab (16.8 months) versus those treated with nivolumab (5.6 months) was 11.2 months with a HR of 0.62 (0.44 to 0.89) favoring treatment with nivolumab plus ipilimumab. This difference was considered clinically relevant according to the PASKWIL criteria. This outcome was observed, but the study was not designed or powered to detect it initially. The absolute difference in median PFS between the group treated with nivolumab plus ipilimumab (16.8 months) versus those treated with ipilimumab (3.4 months) was 13.4 months with a HR of 0.44 (0.31 to 0.62) favoring treatment with nivolumab plus ipilimumab. This difference was considered clinically relevant according to the PASKWIL criteria. The absolute difference in median PFS between the group treated with nivolumab versus those treated with ipilimumab was 2.2 months with a HR of 0.71 (0.51 to 0.98) favoring treatment with nivolumab. This difference was not considered clinically relevant according to the PASKWIL criteria.
CA184-024 reported the effect of ipilimumab plus dacarbazine versus dacarbazine plus placebo on PFS. The median PFS was comparable between the ipilimumab-dacarbazine group and the dacarbazine-only group. After the first assessment of progression at week 12 (after the true median), the Kaplan-Meier curves separated and there was a 24% reduction in the risk of progression in the ipilimumab plus dacarbazine group compared to the dacarbazine only group with a HR of 0.76 (95% CI 0.63–0.93). According to the PASKWIL criteria we could not assess clinical relevance (median OS in the control group was < 12 months).
KEYNOTE-006 reported the effect of pembrolizumab every 2 weeks or pembrolizumab every 3 weeks compared to ipilimumab every 3 weeks on PFS. The 2-year PFS rates were 31% in the pembrolizumab-every-2-weeks group, 28% in the pembrolizumab-every-3-weeks group, and 14% in the ipilimumab-every-3-weeks group. Treatment with pembrolizumab resulted in a longer median PFS than treatment with ipilimumab. The absolute difference between the combined pembrolizumab groups and the ipilimumab group was 5.0 months with a HR of 0.57 (95% CI 0.48–0.67). This difference was considered clinically relevant according to the PASKWIL criteria.
MASTERKEY-265 reported the effect of a combination of T-VEC plus pembrolizumab versus placebo plus pembrolizumab on PFS. Treatment with T-VEC-pembrolizumab resulted in a longer PFS compared with treatment with placebo-pembrolizumab. The absolute difference between the combined pembrolizumab groups and the ipilimumab group was 5.8 months with a HR of 0.86 (95% CI, 0.71 to 1.04). This difference was not considered clinically relevant according to the PASKWIL criteria.
CoBRIM reported the effect of cobimetinib combined with vemurafenib versus vemurafenib and placebo on PFS. Treatment with cobimetinib combined with vemurafenib resulted in a longer median PFS than treatment with vemurafenib and placebo. The absolute difference between the cobimetinib combined with vemurafenib group (12.6 months) and the vemurafenib and placebo group (7.2 months) was 5.4 months with a HR of 0.51 (95% 0.39 to 0.67). This difference was considered clinically relevant according to the PASKWIL criteria.
COMBI-v reported the effect of dabrafenib and trametinib versus vemurafenib on PFS. Treatment with dabrafenib and trametinib resulted in a longer PFS than treatment with vemurafenib. The absolute difference between the dabrafenib and trametinib group (11.4 months) and the vemurafenib group (7.3 months) was 4 months with a HR of 0.56 (95% 0.46 to 0.69). This difference was considered clinically relevant according to the PASKWIL criteria.
COLUMBUS reported the effect of encorafenib and binimetinib versus encorafenib versus vemurafenib on PFS. Treatment with encorafenib and binimetinib resulted in the longest PFS. The absolute difference between the encorafenib and binimetinib group (14.9 months) and the encorafenib group (9.6 months) was 5.3 months. According to the PASKWIL criteria, clinical relevance could not be assessed as the HR and 95% CI were not provided. The absolute difference between the encorafenib and binimetinib group (14.9 months) and the vemurafenib group (7.3 months) was 7.6 months, with an HR of 0.51 (95% CI: 0.39 to 0.67). This difference was considered clinically relevant according to the PASKWIL criteria. The absolute difference between the encorafenib group (9.6 months) and the vemurafenib group (7.3 months) was 2.3 months. The PASKWIL-criteria could not be assessed (HR and 95% CI not provided).
COMBI-d reported the effect of dabrafenib and trametinib versus dabrafenib on PFS. Treatment with dabrafenib and trametinib resulted in a longer PFS than treatment with dabrafenib alone. The absolute difference in PFS between the dabrafenib and trametinib group (11.0 months) and dabrafenib group (8.8 months) was 2.2 months with a HR of 0.67 (95% 0.53 to 0.84). This difference was not considered clinically relevant according to the PASKWIL criteria.
DREAMseq reported a longer PFS when using combination treatment with nivolumab and ipilimumab versus dabrafenib and trametinib. The absolute difference between the intervention and control group was 22.7% after 2 years of follow-up. The median PFS was 11.8 months (95%CI, 5.9 - 33.5) versus 8.5 months (95%CI 6.5 to 11.3). No HR was reported for the median PFS.
IMspire150 reported a longer PFS when using combination treatment with atezoluzumab and vemurafenib and cobimetinib versus vemurafenib and cobimetinib. The absolute difference between the intervention and control group was 12% after 1.5 years of follow-up. The median PFS was 15.1 months (95%CI, 11.4-18.4) versus 10.6 months (95%CI 9.3 to 12.7), with a HR of 0.79 (95%CI 0.64 to 0.97), favouring the intervention group. This difference was considered not clinically relevant according to the PASKWIL criteria.
Figure 2. Forest plot of median progression free survival for first line systemic therapy versus placebo, other systemic therapy, or best supportive care in patients with a BRAF-V600E/K mutation with unresectable or metastatic stadium III/IV melanoma
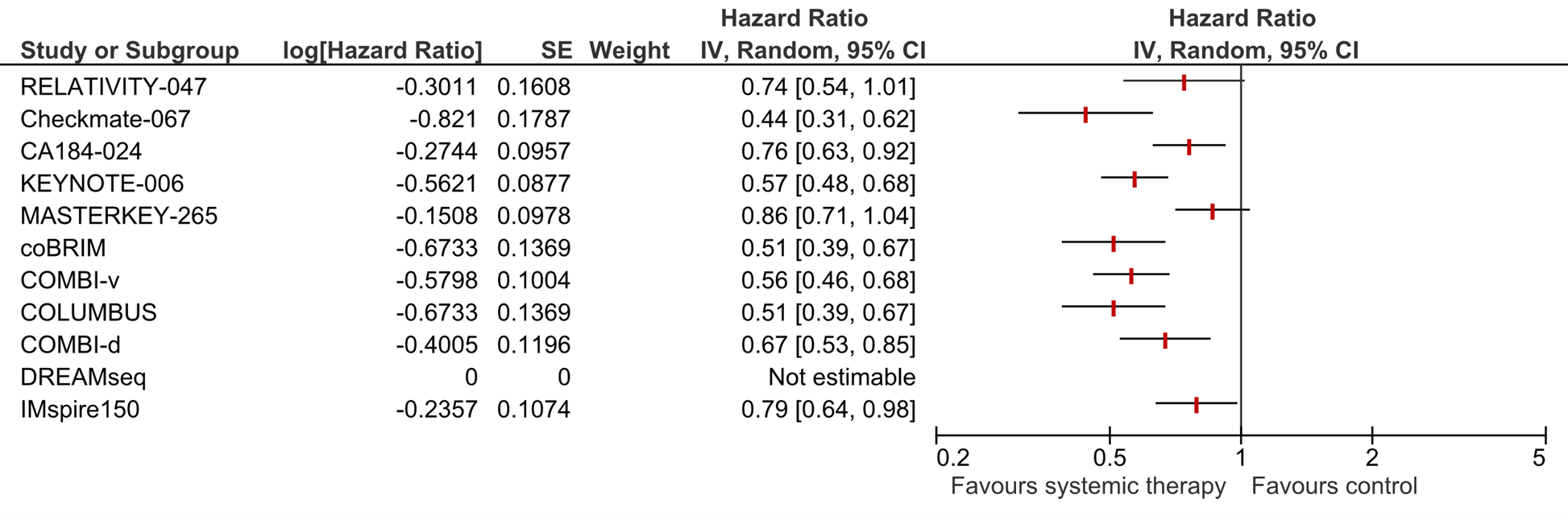
a For Checkmate-067 the HR for nivolumab versus ipilimumab is shown.
b For KEYNOTE-006 the HR for combined pembrolizumab versus ipilimumab is shown.
c For COLOMBUS the HR for encorafenib and binimetinib versus vemurafenib is shown.
d DREAMseq did not report the HR for median OS and is therefore not shown in this figure.
Treatment related adverse events (AEs) grade ≥ 3 - Important outcome
Twelve studies reported on AEs.
RELATIVITY-047 reported the effect of relatlimab and nivolumab versus nivolumab on AEs. No sub group analyses were used while analyzing this outcome. Treatment with relatlimab and nivolumab resulted in a higher percentage of treatment related AEs grade ≥3 compared to treatment with nivolumab only. The risk difference is 0.07 (95%CI -0.00, 0.14; NNH= 14) favoring treatment with nivolumab. This difference is not considered clinically relevant according to the PASKWIL criteria.
Checkmate-067 reported the effect of nivolumab plus ipilimumab versus nivolumab versus ipilimumab on AEs without using sub analyses. Treatment with nivolumab plus ipilimumab resulted in a higher percentage of treatment related AEs grade ≥3 compared to treatment with nivolumab or ipilimumab only. The risk difference between nivolumab plus ipilimumab and nivolumab is 0.36 (95%CI 0.29, 0.43; NNH= 2.8) favoring treatment with nivolumab. This difference is considered clinically relevant. The risk difference between nivolumab plus ipilimumab and ipilimumab is 0.32 (95% CI 0.25, 0.39; NNH= 3.1) favoring treatment with ipilimumab. This difference is considered clinically relevant. The risk difference between nivolumab and ipilimumab is 0.04 (95% CI -0.03, 0.11; NNH= 25) favoring treatment with nivolumab. This difference is not considered clinically relevant according to the PASKWIL criteria.
CA184-024 reported the effect of ipilimumab plus dacarbazine versus dacarbazine plus placebo on AEs. Treatment with ipilimumab plus dacarbazine resulted in a higher percentage of treatment related AEs grade ≥3 compared to treatment with dacarbazine only. The risk difference between ipilimumab plus dacarbazine and dacarbazine is 0.34 (95% CI 0.27, 0.40; NNH= 2.9) favoring treatment with dacarbazine. This difference is considered clinically relevant according to the PASKWIL criteria.
KEYNOTE-006 reported the effect of pembrolizumab every 2 weeks or pembrolizumab every 3 weeks compared to ipilimumab every 3 weeks on AEs. Treatment with pembrolizumab (pooled groups) resulted in a lower percentage of treatment related AEs grade ≥3 compared to treatment with ipilimumab. The risk difference between pembrolizumab (pooled groups) and ipilimumab is -0.01 (95% CI -0.06, 0.05; NNH=100) favoring treatment with pembrolizumab. This difference is not considered clinically relevant according to the PASKWIL criteria.
MASTERKEY-265 reported the effect of a combination of T-VEC plus pembrolizumab versus placebo plus pembrolizumab on AEs. Treatment with T-VEC-pembrolizumab resulted in a higher percentage of treatment related AEs grade ≥3 compared to treatment with placebo-pembrolizumab. The risk difference between T-VEC plus pembrolizumab and placebo-pembrolizumab is 0.05 (95% CI -0.01, 0.10; NNH=20) favoring treatment with placebo plus pembrolizumab. This difference is not considered clinically relevant according to the PASKWIL criteria.
OPTiM reported the effect of T-VEC versus GM-CSF on AEs. Treatment with T-VEC resulted in a higher percentage of treatment related AEs grade ≥3 compared to treatment with GM-CSF. The risk difference between T-VEC and GM-CSF is 0.07 (95% CI 0.02, 0.12; NNH=14) favoring treatment with GM-CSF. This difference is not considered clinically relevant according to the PASKWIL criteria.
CoBRIM reported the effect of cobimetinib combined with vemurafenib versus vemurafenib and placebo on AEs. Treatment with cobimetinib combined with vemurafenib resulted in a higher percentage of treatment related AEs grade ≥3 compared to treatment with vemurafenib and placebo. The risk difference between the cobimetinib combined with vemurafenib group (78%) and the vemurafenib and placebo group (63%) was 15%. This difference was not considered clinically relevant according to the PASKWIL criteria.
COMBI-v reported the effect of dabrafenib and trametinib versus vemurafenib on AEs. Treatment with dabrafenib and trametinib resulted in less AEs than treatment with vemurafenib. The risk difference between the dabrafenib and trametinib group (48%) and the vemurafenib group (57%) was 9%. This difference was not considered clinically relevant according to the PASKWIL criteria.
COLUMBUS reported the effect of encorafenib and binimetinib versus encorafenib versus vemurafenib on AEs. There was no risk difference in the percentage of AEs between encorafenib and binimetinib (68%) and encorafenib (68%). The risk difference in AEs between encorafenib and binimetinib (68%) and vemurafenib (66%) was 2%. This difference was not considered clinically relevant according to the PASKWIL criteria. The risk difference between encorafenib (68%) and vemurafenib (66%) was also 2%. This difference was not considered clinically relevant according to the PASKWIL criteria.
COMBI-d reported the effect of dabrafenib and trametinib versus dabrafenib on AEs. The risk difference in percentage of AEs between the dabrafenib and trametinib group (48%) and dabrafenib group (50%) was 2%. This difference was not considered clinically relevant according to the PASKWIL criteria.
DREAMseq reported the severe adverse events grade ≥3 after treatment with nivolumab and ipilimumab versus dabrafenib and trametinib. The risk difference is 0.06 (95%CI -0.06, 0.18, NNH=17), favoring treatment with nivolumab and ipilimumab. This difference is not considered clinically relevant according to the PASKWIL criteria.
IMspire150 reported the severe adverse events grade ≥3 after treatment with atezoluzumab and vemurafenib and cobimetinib versus vemurafenib and cobimetinib. The risk difference is -0.09 (95%CI -0.17, -0.01, NNH=11), favoring treatment with atezoluzumab and vemurafenib and cobimetinib. This difference is not considered clinically relevant according to the PASKWIL criteria.
Figure 3. Forest plot of treatment related adverse events grade ≥3 after first line systemic therapy versus placebo, other systemic therapy, or best supportive care in patients with a BRAF-V600E/K mutation with unresectable or metastatic stadium III/IV melanoma
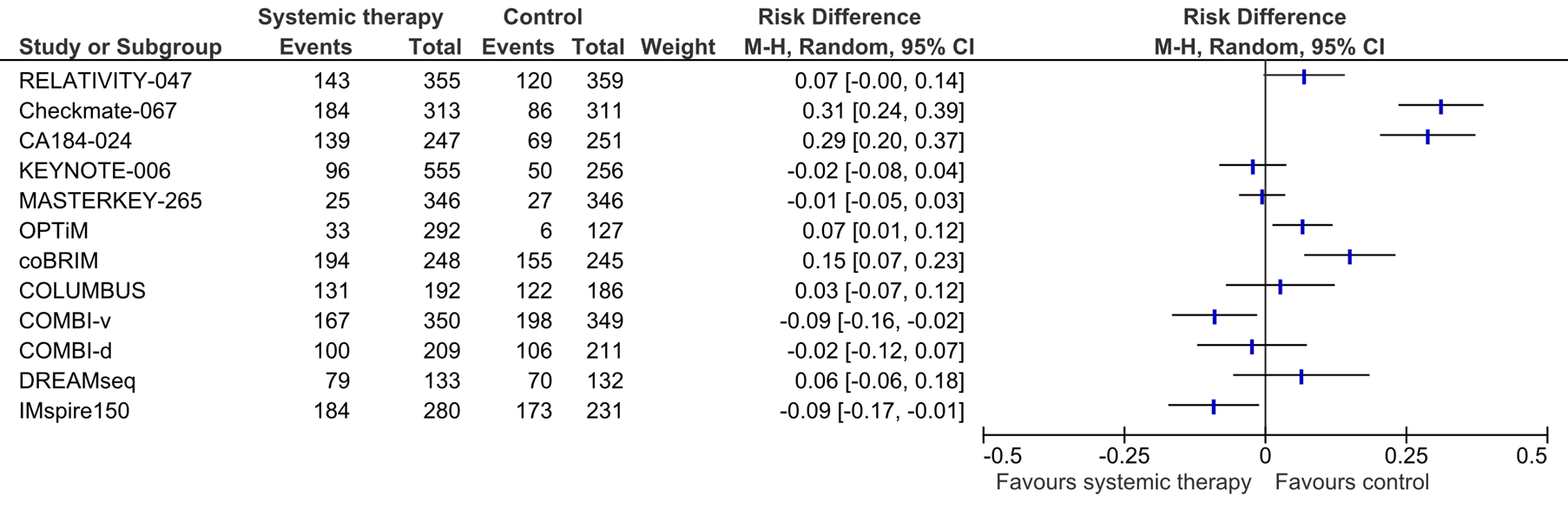
a For Checkmate-067, the risk difference for nivolumab versus ipilimumab is shown.
b For KEYNOTE-006, the the risk difference for combined pembrolizumab versus ipilimumab is shown.
c For COLOMBUS, the the risk difference for encorafenib and binimetinib versus vemurafenib is shown.
Quality of life (QoL) - Important outcome
One study reported the effect of first line systemic therapy on QoL.
RELATIVITY-047 reported the effect of relatlimab and nivolumab versus nivolumab on QoL. In this study, the changes from baseline in FACT-M total score and EQ-5D-3L health utility index were analysed without using subgroup analyses based on BRAF mutation status. The authors considered minimal clinically important differences of 5 in the FACT-M total score and 0.08 in the EQ-5D-3L health utility index to be clinically meaningful. The least-squares mean changes from baseline over time in the FACT-M total score ranged between 1 and -4 and did not exceed the minimal clinically important differences. The least-squares mean changes from baseline over time in the EQ-5D-3L utility index ranged between -0.025 and -0.08 and did not exceed the minimal clinically important differences.
Level of evidence of the literature
There are four levels of evidence: high, moderate, low, and very low. RCTs start at a high level of evidence.
Relatlimab plus nivolumab versus nivolumab
The level of evidence regarding the outcome measure progression free survival was downgraded by two levels because of study limitations (risk of bias) and because we could not asses clinical relevance according to the PASKWIL criteria (imprecision). Therefore, the level of evidence was graded as low.
The level of evidence regarding the outcome measure adverse events was downgraded by two levels because of study limitations (risk of bias) and the lack of a subgroup analysis (indirectness). Therefore, the level of evidence was graded as low.
The level of evidence regarding the outcome measure quality of life was downgraded by two levels because of study limitations (risk of bias) and the lack of a subgroup analyses (indirectness). Therefore, the level of evidence was graded as low.
Nivolumab plus ipilimumab versus nivolumab versus ipilimumab
Nivolumab plus ipilimumab versus nivolumab
The level of evidence regarding the outcome measure overall survival was downgraded by two levels because of study limitations (risk of bias) and because the confidence interval encloses the threshold for a clinically relevant effect and no clinically relevant effect (imprecision). Therefore, the level of evidence was graded as low.
The level of evidence regarding the outcome measure progression free survival was downgraded by two levels because of study limitations (risk of bias), and because the confidence interval encloses the threshold for a clinically relevant effect and no clinically relevant effect (imprecision). Therefore, the level of evidence was graded as low.
The level of evidence regarding the outcome measure adverse events was downgraded by two levels because of study limitations (risk of bias), and the lack of a subgroup analysis (indirectness). Therefore, the level of evidence was graded as low.
Nivolumab plus ipilimumab versus ipilimumab
The level of evidence regarding the outcome measure overall survival was graded as high.
The level of evidence regarding the outcome measure progression free survival the level of evidence was graded as high.
The level of evidence regarding the outcome measure adverse events was downgraded by one level due to the lack of a subgroup analysis (indirectness). Therefore, the level of evidence was graded as moderate
Nivolumab versus ipilimumab
The level of evidence regarding the outcome measure overall survival was downgraded by one level because the confidence interval encloses the threshold for a clinically relevant effect and no clinically relevant effect (imprecision). Therefore, the level of evidence was graded as moderate.
The level of evidence regarding the outcome measure progression free survival was graded as high.
The level of evidence regarding the outcome measure adverse events was downgraded by two levels because the optimal information size is not met (imprecision), and the lack of a subgroup analysis (indirectness). Therefore, the level of evidence was graded as low.
Ipilimumab plus dacarbazine versus dacarbazine
The level of evidence regarding the outcome measure overall survival was downgraded by four levels because of study limitations (risk of bias -2), the confidence interval encloses the threshold for a clinically relevant effect and no clinically relevant effect (imprecision), and the lack of a subgroup analysis (indirectness). Therefore, the level of evidence was graded as very low.
The level of evidence regarding the outcome measure progression free survival was downgraded by four levels because of study limitations (risk of bias -2), the confidence interval encloses the threshold for a clinically relevant effect and no clinically relevant effect (imprecision), and the lack of a subgroup analysis (indirectness). Therefore, the level of evidence was graded as very low.
The level of evidence regarding the outcome measure adverse events was downgraded by three levels because of study limitations (risk of bias -2) and the lack of a subgroup analysis (indirectness). Therefore, the level of evidence was graded as very low.
Pembrolizumab every 2 weeks or every 3 weeks compared to ipilimumab
The level of evidence regarding the outcome measure overall survival was downgraded by four levels because of study limitations (risk of bias -2); the confidence interval encloses the threshold for a clinically relevant effect and no clinically relevant effect (imprecision), and the lack of a subgroup analysis (indirectness). Therefore, the level of evidence was graded as very low.
The level of evidence regarding the outcome measure progression free survival was downgraded by three levels because of study limitations (risk of bias -2), and the lack of a subgroup analysis (indirectness). Therefore, the level of evidence was graded as very low.
The level of evidence regarding the outcome measure adverse events was downgraded by four levels because of study limitations (risk of bias -2), because the optimal information size is not met (imprecision), and the lack of a subgroup analysis (indirectness). Therefore, the level of evidence was graded as very low.
T-VEC plus pembrolizumab versus placebo plus pembrolizumab
The level of evidence regarding the outcome measure overall survival was downgraded by four levels because of study limitations (risk of bias -2), the optimal information size is not met (imprecision), and the lack of a subgroup analysis (indirectness). Therefore, the level of evidence was graded as very low.
The level of evidence regarding the outcome measure progression free survival was downgraded by four levels because of study limitations (risk of bias -2), the optimal information size is not met (imprecision), and the lack of a subgroup analysis (indirectness). Therefore, the level of evidence was graded as very low.
The level of evidence regarding the outcome measure adverse events was downgraded by four levels because of study limitations (risk of bias -2), the optimal information size is not met (imprecision), and the lack of a subgroup analysis (indirectness). Therefore, the level of evidence was graded as very low.
T-VEC versus GM-CSF
The level of evidence regarding the outcome measure overall survival was downgraded by four levels because of study limitations (risk of bias -2), the confidence interval encloses the threshold for a clinically relevant effect and no clinically relevant effect (imprecision), and the lack of a subgroup analysis (indirectness). Therefore, the level of evidence was graded as very low.
The level of evidence regarding the outcome measure adverse events was downgraded by four levels because of study limitations (risk of bias -2), was downgraded by one level because the optimal information size is not met (imprecision), and the lack of a subgroup analysis (indirectness). Therefore, the level of evidence was graded as very low.
Cobimetinib plus vemurafenib versus vemurafenib and placebo
The level of evidence regarding the outcome measure overall survival was downgraded by one level because the confidence interval encloses the threshold for a clinically relevant effect (imprecision -1). Therefore, the level of evidence was graded as moderate.
The level of evidence regarding the outcome measure progression free survival was graded as high.
The level of evidence regarding the outcome measure adverse events was graded as high.
Dabrafenib plus trametinib versus vemurafenib
The level of evidence regarding the outcome measure overall survival was downgraded by one level because of study limitations (risk of bias -1). Therefore, the level of evidence was graded as moderate.
The level of evidence regarding the outcome measure progression free survival was downgraded by one level because of study limitations (risk of bias -1). Therefore, the level of evidence was graded as moderate.
The level of evidence regarding the outcome measure adverse events was downgraded by two level because of study limitations (risk of bias -2). Therefore, the level of evidence was graded as low.
Encorafenib plus binimetinib versus encorafenib versus vemurafenib
Encorafenib and binimetinib versus encorafenib
The level of evidence regarding the outcome measure overall survival was downgraded by two levels because of study limitations (risk of bias -1) and because the confidence interval encloses the threshold for a clinically relevant effect (imprecision -1). Therefore, the level of evidence was graded as low.
The level of evidence regarding the outcome measure progression free survival was downgraded by two level because of study limitations (risk of bias -1), and no clinically relevant effect could be established (imprecision -1). Therefore, the level of evidence was graded as low.
The level of evidence regarding the outcome measure adverse events was downgraded by two level because of study limitations (risk of bias -2). Therefore, the level of evidence was graded as low.
Encorafenib plus binimetinib versus vemurafenib
The level of evidence regarding the outcome measure overall survival was downgraded by two levels because of study limitations (risk of bias -1) and because the confidence interval encloses the threshold for a clinically relevant effect (imprecision -1). Therefore, the level of evidence was graded as low.
The level of evidence regarding the outcome measure progression free survival was downgraded by one level because of study limitations (risk of bias -1). Therefore, the level of evidence was graded as moderate.
The level of evidence regarding the outcome measure adverse events was downgraded by two level because of study limitations (risk of bias -2). Therefore, the level of evidence was graded as low.
Encorafenib plus vemurafenib
The level of evidence regarding the outcome measure overall survival was downgraded by two levels because of study limitations (risk of bias -1) and because the confidence interval encloses the threshold for a clinically relevant effect (imprecision -1). Therefore, the level of evidence was graded as low.
The level of evidence regarding the outcome measure progression free survival was downgraded by two level because of study limitations (risk of bias -1), and no clinically relevant effect could be established (imprecision -1). Therefore, the level of evidence was graded as low.
The level of evidence regarding the outcome measure adverse events was downgraded by two level because of study limitations (risk of bias -2). Therefore, the level of evidence was graded as low.
Dabrafenib plus trametinib versus dabrafenib
The level of evidence regarding the outcome measure overall survival was downgraded by one level because of study limitations (risk of bias -1) and because the confidence interval encloses the threshold for a clinically relevant effect (imprecision -1). Therefore, the level of evidence was graded as low.
The level of evidence regarding the outcome measure progression free survival was downgraded by one level because of study limitations (risk of bias -1). Therefore, the level of evidence was graded as moderate.
The level of evidence regarding the outcome measure adverse events was downgraded by two level because of study limitations (risk of bias -2). Therefore, the level of evidence was graded as low.
Nivolumab plus ipilimumab versus dabrafenib plus trametinib.
The level of evidence regarding the outcome measure overall survival was downgraded by two levels because we could not asses clinical relevance according to the PASKWIL criteria (imprecision -1). Therefore, the level of evidence was graded as moderate.
The level of evidence regarding the outcome measure progression free survival was downgraded by two levels because we could not asses clinical relevance according to the PASKWIL criteria (imprecision -1). Therefore, the level of evidence was graded as moderate.
The level of evidence regarding the outcome measure adverse events was downgraded by two levels because of study limitations (risk of bias -2). Therefore, the level of evidence was graded as low.
Atezolizumab plus vemurafenib plus cobimetinib versus vemurafenib plus cobimetinib.
The level of evidence regarding the outcome measure overall survival was downgraded by one level because the confidence interval encloses the threshold for a clinically relevant effect and no clinically relevant effect (imprecision -1). Therefore, the level of evidence was graded as moderate.
The level of evidence regarding the outcome measure progression free survival was downgraded by one level because the confidence interval encloses the threshold for a clinically relevant effect and no clinically relevant effect (imprecision -1). Therefore, the level of evidence was graded as moderate.
The level of evidence regarding the outcome measure adverse events was downgraded by two levels because of study limitations (risk of bias -2). Therefore, the level of evidence was graded as low.
The search and selection methods can be found in the main module Systematische therapie.
- 1 - Andtbacka RH, Kaufman HL, Collichio F, Amatruda T, Senzer N, Chesney J, Delman KA, Spitler LE, Puzanov I, Agarwala SS, Milhem M, Cranmer L, Curti B, Lewis K, Ross M, Guthrie T, Linette GP, Daniels GA, Harrington K, Middleton MR, Miller WH Jr, Zager JS, Ye Y, Yao B, Li A, Doleman S, VanderWalde A, Gansert J, Coffin RS. Talimogene Laherparepvec Improves Durable Response Rate in Patients With Advanced Melanoma. J Clin Oncol. 2015 Sep 1;33(25):2780-8. doi: 10.1200/JCO.2014.58.3377. Epub 2015 May 26. PMID: 26014293.
- 2 - Andtbacka RHI, Collichio F, Harrington KJ, Middleton MR, Downey G, Ӧhrling K, Kaufman HL. Final analyses of OPTiM: a randomized phase III trial of talimogene laherparepvec versus granulocyte-macrophage colony-stimulating factor in unresectable stage III-IV melanoma. J Immunother Cancer. 2019 Jun 6;7(1):145. doi: 10.1186/s40425-019-0623-z. PMID: 31171039; PMCID: PMC6554874.
- 3 - Ascierto PA, Dréno B, Larkin J, Ribas A, Liszkay G, Maio M, Mandalà M, Demidov L, Stroyakovskiy D, Thomas L, de la Cruz-Merino L, Atkinson V, Dutriaux C, Garbe C, Hsu J, Jones S, Li H, McKenna E, Voulgari A, McArthur GA. 5-Year Outcomes with Cobimetinib plus Vemurafenib in BRAFV600 Mutation-Positive Advanced Melanoma: Extended Follow-up of the coBRIM Study. Clin Cancer Res. 2021 Oct 1;27(19):5225-5235. doi: 10.1158/1078-0432.CCR-21-0809. PMID: 34158360; PMCID: PMC9401485.
- 4 - Ascierto PA, Dummer R, Gogas HJ, Flaherty KT, Arance A, Mandala M, Liszkay G, Garbe C, Schadendorf D, Krajsova I, Gutzmer R, de Groot JWB, Loquai C, Gollerkeri A, Pickard MD, Robert C. Update on tolerability and overall survival in COLUMBUS: landmark analysis of a randomised phase 3 trial of encorafenib plus binimetinib vs vemurafenib or encorafenib in patients with BRAF V600-mutant melanoma. Eur J Cancer. 2020 Feb;126:33-44. doi: 10.1016/j.ejca.2019.11.016. Epub 2020 Jan 2. PMID: 31901705.
- 5 - Ascierto PA, Mandalà M, Ferrucci PF, Guidoboni M, Rutkowski P, Ferraresi V, Arance A, Guida M, Maiello E, Gogas H, Richtig E, Fierro MT, Lebbè C, Helgadottir H, Queirolo P, Spagnolo F, Tucci M, Del Vecchio M, Gonzales Cao M, Minisini AM, De Placido S, Sanmamed MF, Mallardo D, Curvietto M, Melero I, Palmieri G, Grimaldi AM, Giannarelli D, Dummer R, Chiarion Sileni V. Sequencing of Ipilimumab Plus Nivolumab and Encorafenib Plus Binimetinib for Untreated BRAF-Mutated Metastatic Melanoma (SECOMBIT): A Randomized, Three-Arm, Open-Label Phase II Trial. J Clin Oncol. 2023 Jan 10;41(2):212-221. doi: 10.1200/JCO.21.02961. Epub 2022 Sep 1. PMID: 36049147.
- 6 - Ascierto PA, McArthur GA, Dréno B, Atkinson V, Liszkay G, Di Giacomo AM, Mandalà M, Demidov L, Stroyakovskiy D, Thomas L, de la Cruz-Merino L, Dutriaux C, Garbe C, Yan Y, Wongchenko M, Chang I, Hsu JJ, Koralek DO, Rooney I, Ribas A, Larkin J. Cobimetinib combined with vemurafenib in advanced BRAF(V600)-mutant melanoma (coBRIM): updated efficacy results from a randomised, double-blind, phase 3 trial. Lancet Oncol. 2016 Sep;17(9):1248-60. doi: 10.1016/S1470-2045(16)30122-X. Epub 2016 Jul 30. PMID: 27480103.
- 7 - Ascierto PA, Stroyakovskiy D, Gogas H, Robert C, Lewis K, Protsenko S, Pereira RP, Eigentler T, Rutkowski P, Demidov L, Zhukova N, Schachter J, Yan Y, Caro I, Hertig C, Xue C, Kusters L, McArthur GA, Gutzmer R. Overall survival with first-line atezolizumab in combination with vemurafenib and cobimetinib in BRAFV600 mutation-positive advanced melanoma (IMspire150): second interim analysis of a multicentre, randomised, phase 3 study. Lancet Oncol. 2023 Jan;24(1):33-44. doi: 10.1016/S1470-2045(22)00687-8. Epub 2022 Nov 29. PMID: 36460017.
- 8 - Atkins MB, Lee SJ, Chmielowski B, Tarhini AA, Cohen GI, Truong TG, Moon HH, Davar D, O'Rourke M, Stephenson JJ, Curti BD, Urba WJ, Brell JM, Funchain P, Kendra KL, Ikeguchi AP, Jaslowski A, Bane CL, Taylor MA, Bajaj M, Conry RM, Ellis RJ, Logan TF, Laudi N, Sosman JA, Crockett DG, Pecora AL, Okazaki IJ, Reganti S, Chandra S, Guild S, Chen HX, Streicher HZ, Wolchok JD, Ribas A, Kirkwood JM. Combination Dabrafenib and Trametinib Versus Combination Nivolumab and Ipilimumab for Patients With Advanced BRAF-Mutant Melanoma: The DREAMseq Trial-ECOG-ACRIN EA6134. J Clin Oncol. 2023 Jan 10;41(2):186-197. doi: 10.1200/JCO.22.01763. Epub 2022 Sep 27. PMID: 36166727; PMCID: PMC9839305.
- 9 - Carlino MS, Long GV, Schadendorf D, Robert C, Ribas A, Richtig E, Nyakas M, Caglevic C, Tarhini A, Blank C, Hoeller C, Bar-Sela G, Barrow C, Wolter P, Zhou H, Emancipator K, Jensen EH, Ebbinghaus S, Ibrahim N, Daud A. Outcomes by line of therapy and programmed death ligand 1 expression in patients with advanced melanoma treated with pembrolizumab or ipilimumab in KEYNOTE-006: A randomised clinical trial. Eur J Cancer. 2018 Sep;101:236-243. doi: 10.1016/j.ejca.2018.06.034. Epub 2018 Aug 7. PMID: 30096704.
- 10 - Chesney JA, Ribas A, Long GV, Kirkwood JM, Dummer R, Puzanov I, Hoeller C, Gajewski TF, Gutzmer R, Rutkowski P, Demidov L, Arenberger P, Shin SJ, Ferrucci PF, Haydon A, Hyngstrom J, van Thienen JV, Haferkamp S, Guilera JM, Rapoport BL, VanderWalde A, Diede SJ, Anderson JR, Treichel S, Chan EL, Bhatta S, Gansert J, Hodi FS, Gogas H. Randomized, Double-Blind, Placebo-Controlled, Global Phase III Trial of Talimogene Laherparepvec Combined With Pembrolizumab for Advanced Melanoma. J Clin Oncol. 2023 Jan 20;41(3):528-540. doi: 10.1200/JCO.22.00343. Epub 2022 Aug 23. PMID: 35998300; PMCID: PMC9870217.
- 11 - Dummer R, Ascierto PA, Gogas HJ, Arance A, Mandala M, Liszkay G, Garbe C, Schadendorf D, Krajsova I, Gutzmer R, Chiarion Sileni V, Dutriaux C, de Groot JWB, Yamazaki N, Loquai C, Moutouh-de Parseval LA, Pickard MD, Sandor V, Robert C, Flaherty KT. Overall survival in patients with BRAF-mutant melanoma receiving encorafenib plus binimetinib versus vemurafenib or encorafenib (COLUMBUS): a multicentre, open-label, randomised, phase 3 trial. Lancet Oncol. 2018 Oct;19(10):1315-1327. doi: 10.1016/S1470-2045(18)30497-2. Epub 2018 Sep 12. Erratum in: Lancet Oncol. 2018 Oct;19(10):e509. doi: 10.1016/S1470-2045(18)30705-8. PMID: 30219628.
- 12 - Dummer R, Ascierto PA, Gogas HJ, Arance A, Mandala M, Liszkay G, Garbe C, Schadendorf D, Krajsova I, Gutzmer R, Chiarion-Sileni V, Dutriaux C, de Groot JWB, Yamazaki N, Loquai C, Moutouh-de Parseval LA, Pickard MD, Sandor V, Robert C, Flaherty KT. Encorafenib plus binimetinib versus vemurafenib or encorafenib in patients with BRAF-mutant melanoma (COLUMBUS): a multicentre, open-label, randomised phase 3 trial. Lancet Oncol. 2018 May;19(5):603-615. doi: 10.1016/S1470-2045(18)30142-6. Epub 2018 Mar 21. PMID: 29573941.
- 13 - Hodi FS, Chiarion-Sileni V, Gonzalez R, Grob JJ, Rutkowski P, Cowey CL, Lao CD, Schadendorf D, Wagstaff J, Dummer R, Ferrucci PF, Smylie M, Hill A, Hogg D, Marquez-Rodas I, Jiang J, Rizzo J, Larkin J, Wolchok JD. Nivolumab plus ipilimumab or nivolumab alone versus ipilimumab alone in advanced melanoma (CheckMate 067): 4-year outcomes of a multicentre, randomised, phase 3 trial. Lancet Oncol. 2018 Nov;19(11):1480-1492. doi: 10.1016/S1470-2045(18)30700-9. Epub 2018 Oct 22. Erratum in: Lancet Oncol. 2018 Dec;19(12):e668. doi: 10.1016/S1470-2045(18)30841-6. Erratum in: Lancet Oncol. 2018 Nov;19(11):e581. doi: 10.1016/S1470-2045(18)30775-7. PMID: 30361170.
- 14 - Larkin J, Ascierto PA, Dréno B, Atkinson V, Liszkay G, Maio M, Mandalà M, Demidov L, Stroyakovskiy D, Thomas L, de la Cruz-Merino L, Dutriaux C, Garbe C, Sovak MA, Chang I, Choong N, Hack SP, McArthur GA, Ribas A. Combined vemurafenib and cobimetinib in BRAF-mutated melanoma. N Engl J Med. 2014 Nov 13;371(20):1867-76. doi: 10.1056/NEJMoa1408868. Epub 2014 Sep 29. PMID: 25265494.
- 15 - Larkin J, Chiarion-Sileni V, Gonzalez R, Grob JJ, Cowey CL, Lao CD, Schadendorf D, Dummer R, Smylie M, Rutkowski P, Ferrucci PF, Hill A, Wagstaff J, Carlino MS, Haanen JB, Maio M, Marquez-Rodas I, McArthur GA, Ascierto PA, Long GV, Callahan MK, Postow MA, Grossmann K, Sznol M, Dreno B, Bastholt L, Yang A, Rollin LM, Horak C, Hodi FS, Wolchok JD. Combined Nivolumab and Ipilimumab or Monotherapy in Untreated Melanoma. N Engl J Med. 2015 Jul 2;373(1):23-34. doi: 10.1056/NEJMoa1504030. Epub 2015 May 31. Erratum in: N Engl J Med. 2018 Nov 29;379(22):2185. doi: 10.1056/NEJMx180040. PMID: 26027431; PMCID: PMC5698905.
- 16 - Larkin J, Chiarion-Sileni V, Gonzalez R, Grob JJ, Rutkowski P, Lao CD, Cowey CL, Schadendorf D, Wagstaff J, Dummer R, Ferrucci PF, Smylie M, Hogg D, Hill A, Márquez-Rodas I, Haanen J, Guidoboni M, Maio M, Schöffski P, Carlino MS, Lebbé C, McArthur G, Ascierto PA, Daniels GA, Long GV, Bastholt L, Rizzo JI, Balogh A, Moshyk A, Hodi FS, Wolchok JD. Five-Year Survival with Combined Nivolumab and Ipilimumab in Advanced Melanoma. N Engl J Med. 2019 Oct 17;381(16):1535-1546. doi: 10.1056/NEJMoa1910836. Epub 2019 Sep 28. PMID: 31562797.
- 17 - Long GV, Flaherty KT, Stroyakovskiy D, Gogas H, Levchenko E, de Braud F, Larkin J, Garbe C, Jouary T, Hauschild A, Chiarion-Sileni V, Lebbe C, Mandalà M, Millward M, Arance A, Bondarenko I, Haanen JBAG, Hansson J, Utikal J, Ferraresi V, Mohr P, Probachai V, Schadendorf D, Nathan P, Robert C, Ribas A, Davies MA, Lane SR, Legos JJ, Mookerjee B, Grob JJ. Dabrafenib plus trametinib versus dabrafenib monotherapy in patients with metastatic BRAF V600E/K-mutant melanoma: long-term survival and safety analysis of a phase 3 study. Ann Oncol. 2017 Jul 1;28(7):1631-1639. doi: 10.1093/annonc/mdx176. Erratum in: Ann Oncol. 2019 Nov 1;30(11):1848. doi: 10.1093/annonc/mdz221. PMID: 28475671; PMCID: PMC5834102.
- 18 - Long GV, Stroyakovskiy D, Gogas H, Levchenko E, de Braud F, Larkin J, Garbe C, Jouary T, Hauschild A, Grob JJ, Chiarion-Sileni V, Lebbe C, Mandalà M, Millward M, Arance A, Bondarenko I, Haanen JB, Hansson J, Utikal J, Ferraresi V, Kovalenko N, Mohr P, Probachai V, Schadendorf D, Nathan P, Robert C, Ribas A, DeMarini DJ, Irani JG, Swann S, Legos JJ, Jin F, Mookerjee B, Flaherty K. Dabrafenib and trametinib versus dabrafenib and placebo for Val600 BRAF-mutant melanoma: a multicentre, double-blind, phase 3 randomised controlled trial. Lancet. 2015 Aug 1;386(9992):444-51. doi: 10.1016/S0140-6736(15)60898-4. Epub 2015 May 31. PMID: 26037941.
- 19 - Long GV, Stroyakovskiy D, Gogas H, Levchenko E, de Braud F, Larkin J, Garbe C, Jouary T, Hauschild A, Grob JJ, Chiarion Sileni V, Lebbe C, Mandalà M, Millward M, Arance A, Bondarenko I, Haanen JB, Hansson J, Utikal J, Ferraresi V, Kovalenko N, Mohr P, Probachai V, Schadendorf D, Nathan P, Robert C, Ribas A, DeMarini DJ, Irani JG, Casey M, Ouellet D, Martin AM, Le N, Patel K, Flaherty K. Combined BRAF and MEK inhibition versus BRAF inhibition alone in melanoma. N Engl J Med. 2014 Nov 13;371(20):1877-88. doi: 10.1056/NEJMoa1406037. Epub 2014 Sep 29. PMID: 25265492.
- 20 - Long GV, Trefzer U, Davies MA, Kefford RF, Ascierto PA, Chapman PB, Puzanov I, Hauschild A, Robert C, Algazi A, Mortier L, Tawbi H, Wilhelm T, Zimmer L, Switzky J, Swann S, Martin AM, Guckert M, Goodman V, Streit M, Kirkwood JM, Schadendorf D. Dabrafenib in patients with Val600Glu or Val600Lys BRAF-mutant melanoma metastatic to the brain (BREAK-MB): a multicentre, open-label, phase 2 trial. Lancet Oncol. 2012 Nov;13(11):1087-95. doi: 10.1016/S1470-2045(12)70431-X. Epub 2012 Oct 8. PMID: 23051966.
- 21 - Maio M, Grob JJ, Aamdal S, Bondarenko I, Robert C, Thomas L, Garbe C, Chiarion-Sileni V, Testori A, Chen TT, Tschaika M, Wolchok JD. Five-year survival rates for treatment-naive patients with advanced melanoma who received ipilimumab plus dacarbazine in a phase III trial. J Clin Oncol. 2015 Apr 1;33(10):1191-6. doi: 10.1200/JCO.2014.56.6018. Epub 2015 Feb 23. PMID: 25713437; PMCID: PMC5795709.
- 22 - Robert C, Karaszewska B, Schachter J, Rutkowski P, Mackiewicz A, Stroiakovski D, Lichinitser M, Dummer R, Grange F, Mortier L, Chiarion-Sileni V, Drucis K, Krajsova I, Hauschild A, Lorigan P, Wolter P, Long GV, Flaherty K, Nathan P, Ribas A, Martin AM, Sun P, Crist W, Legos J, Rubin SD, Little SM, Schadendorf D. Improved overall survival in melanoma with combined dabrafenib and trametinib. N Engl J Med. 2015 Jan 1;372(1):30-9. doi: 10.1056/NEJMoa1412690. Epub 2014 Nov 16. PMID: 25399551.
- 23 - Robert C, Ribas A, Schachter J, Arance A, Grob JJ, Mortier L, Daud A, Carlino MS, McNeil CM, Lotem M, Larkin JMG, Lorigan P, Neyns B, Blank CU, Petrella TM, Hamid O, Su SC, Krepler C, Ibrahim N, Long GV. Pembrolizumab versus ipilimumab in advanced melanoma (KEYNOTE-006): post-hoc 5-year results from an open-label, multicentre, randomised, controlled, phase 3 study. Lancet Oncol. 2019 Sep;20(9):1239-1251. doi: 10.1016/S1470-2045(19)30388-2. Epub 2019 Jul 22. PMID: 31345627.
- 24 - Robert C, Schachter J, Long GV, Arance A, Grob JJ, Mortier L, Daud A, Carlino MS, McNeil C, Lotem M, Larkin J, Lorigan P, Neyns B, Blank CU, Hamid O, Mateus C, Shapira-Frommer R, Kosh M, Zhou H, Ibrahim N, Ebbinghaus S, Ribas A; KEYNOTE-006 investigators. Pembrolizumab versus Ipilimumab in Advanced Melanoma. N Engl J Med. 2015 Jun 25;372(26):2521-32. doi: 10.1056/NEJMoa1503093. Epub 2015 Apr 19. PMID: 25891173.
- 25 - Robert C, Thomas L, Bondarenko I, O'Day S, Weber J, Garbe C, Lebbe C, Baurain JF, Testori A, Grob JJ, Davidson N, Richards J, Maio M, Hauschild A, Miller WH Jr, Gascon P, Lotem M, Harmankaya K, Ibrahim R, Francis S, Chen TT, Humphrey R, Hoos A, Wolchok JD. Ipilimumab plus dacarbazine for previously untreated metastatic melanoma. N Engl J Med. 2011 Jun 30;364(26):2517-26. doi: 10.1056/NEJMoa1104621. Epub 2011 Jun 5. PMID: 21639810.
- 26 - Schachter J, Ribas A, Long GV, Arance A, Grob JJ, Mortier L, Daud A, Carlino MS, McNeil C, Lotem M, Larkin J, Lorigan P, Neyns B, Blank C, Petrella TM, Hamid O, Zhou H, Ebbinghaus S, Ibrahim N, Robert C. Pembrolizumab versus ipilimumab for advanced melanoma: final overall survival results of a multicentre, randomised, open-label phase 3 study (KEYNOTE-006). Lancet. 2017 Oct 21;390(10105):1853-1862. doi: 10.1016/S0140-6736(17)31601-X. Epub 2017 Aug 16. PMID: 28822576.
- 27 - Tawbi HA, Forsyth PA, Algazi A, Hamid O, Hodi FS, Moschos SJ, Khushalani NI, Lewis K, Lao CD, Postow MA, Atkins MB, Ernstoff MS, Reardon DA, Puzanov I, Kudchadkar RR, Thomas RP, Tarhini A, Pavlick AC, Jiang J, Avila A, Demelo S, Margolin K. Combined Nivolumab and Ipilimumab in Melanoma Metastatic to the Brain. N Engl J Med. 2018 Aug 23;379(8):722-730. doi: 10.1056/NEJMoa1805453. PMID: 30134131; PMCID: PMC8011001.
- 28 - Tawbi HA, Schadendorf D, Lipson EJ, Ascierto PA, Matamala L, Castillo Gutiérrez E, Rutkowski P, Gogas HJ, Lao CD, De Menezes JJ, Dalle S, Arance A, Grob JJ, Srivastava S, Abaskharoun M, Hamilton M, Keidel S, Simonsen KL, Sobiesk AM, Li B, Hodi FS, Long GV; RELATIVITY-047 Investigators. Relatlimab and Nivolumab versus Nivolumab in Untreated Advanced Melanoma. N Engl J Med. 2022 Jan 6;386(1):24-34. doi: 10.1056/NEJMoa2109970. PMID: 34986285; PMCID: PMC9844513.
- 29 - Wolchok JD, Chiarion-Sileni V, Gonzalez R, Grob JJ, Rutkowski P, Lao CD, Cowey CL, Schadendorf D, Wagstaff J, Dummer R, Ferrucci PF, Smylie M, Butler MO, Hill A, Márquez-Rodas I, Haanen JBAG, Guidoboni M, Maio M, Schöffski P, Carlino MS, Lebbé C, McArthur G, Ascierto PA, Daniels GA, Long GV, Bas T, Ritchings C, Larkin J, Hodi FS. Long-Term Outcomes With Nivolumab Plus Ipilimumab or Nivolumab Alone Versus Ipilimumab in Patients With Advanced Melanoma. J Clin Oncol. 2022 Jan 10;40(2):127-137. doi: 10.1200/JCO.21.02229. Epub 2021 Nov 24. PMID: 34818112; PMCID: PMC8718224.
- 30 - Wolchok JD, Chiarion-Sileni V, Gonzalez R, Rutkowski P, Grob JJ, Cowey CL, Lao CD, Wagstaff J, Schadendorf D, Ferrucci PF, Smylie M, Dummer R, Hill A, Hogg D, Haanen J, Carlino MS, Bechter O, Maio M, Marquez-Rodas I, Guidoboni M, McArthur G, Lebbé C, Ascierto PA, Long GV, Cebon J, Sosman J, Postow MA, Callahan MK, Walker D, Rollin L, Bhore R, Hodi FS, Larkin J. Overall Survival with Combined Nivolumab and Ipilimumab in Advanced Melanoma. N Engl J Med. 2017 Oct 5;377(14):1345-1356. doi: 10.1056/NEJMoa1709684. Epub 2017 Sep 11. Erratum in: N Engl J Med. 2018 Nov 29;379(22):2185. doi: 10.1056/NEJMx180040. PMID: 28889792; PMCID: PMC5706778.
|
Tawbi 2022, RELATIVITY-047 |
Type of study: phase 2-3, global, double-blind, randomized controlled trial
Setting and country: Multicentre, 111 sites in North America, Central America, South America, Europe, Australia, and New Zealand.
Funding and conflicts of interest: Funded by Bristol-Meyers Squibb. The funder participated in data collection and medical writing support.
Detailed declarations of interests are provided in the article. |
Inclusion criteria: -Aged >= 12 years -Measurable disease according to Response Evaluation Criteria in Solid Tumors (RECIST), version 1.1 -Expression of LAG-3 and programmed death ligand 1 (PD-L1) that could be evaluated in tumor tissue -Patients who had received previous adjuvant or neoadjuvant therapies containing a PD-1, CTLA-4, BRAF, or MEK inhibitor (or a combination of BRAF and MEK inhibitors) were eligible if the therapy was completed at least 6 months before the date of recurrence -Patients who received previous treatment with interferon were eligible if the last dose was received at least 6 weeks before randomization Exclusion criteria: -Active untreated brain or leptomeningeal metastases -Uveal melanoma -A history of serious autoimmune disease.
N total at baseline: 714 Intervention: 355 Control: 359
Important prognostic factors2:
Median age (IQR) I: 63 (20-94) C: 62.0 (21–90)
Sex: I: 59.2% M C: 57.4% M
ECOG performance status: I: 66.5% 0 C: 67.4% 0
No BRAF mutation: I: 219 (61.7) C: 220 (61.3)
LAG-3 expression >=1% I: 268 (75.5) C: 269 (74.9)
PD-L1 expression >=1% I: 146 (41.4) C: 147 (40.9)
Groups comparable at baseline? Yes. |
Describe intervention (treatment/procedure/test):
160 mg of relatlimab and 480 mg of nivolumab in a fixed-dose combination administered in a single 60-minute intravenous infusion every 4 weeks |
Describe control (treatment/procedure/test):
480 mg of nivolumab administered in a single 60-minute intravenous infusion every 4 weeks |
Median follow-up: 13.2 months
Loss-to-follow-up: Intervention: 237 (66.8%) discontinued treatment. 129 disease progression 63 AE related to study drug 19 request to discontinue 12 AE unrelated to study drug
Control: 233 (64.9%) discontinued treatment. 165 disease progression 32 AE related to study drug 12 request to discontinue 14 AE unrelated to study drug
|
Outcome measures and effect size (include 95%CI and p-value if available):
Overall survival: Not reported.
Median progression-free survival: I: 10.1 months (95%CI, 6.4 to 15.7)
Adverse events: I: 345 (97.2%) C: 339 (94.4%) Treatment related grade 3 and 4: I: 143 (40.3%) C: 120 (33.4%)
Quality of life: No substantial differences in health-related quality of life were noted between the treatment groups. |
Authors’ conclusion: The inhibition of two immune checkpoints, LAG-3 and PD-1, provided a greater benefit with regard to progression-free survival than inhibition of PD-1 alone in patients with previously untreated metastatic or unresectable melanoma. Relatlimab and nivolumab in combination showed no new safety signals.
-Outcome overall survival: At the final analysis of progression-free survival, the data monitoring committee conducted a prespecified interim analysis of overall survival, which at that time point had not reached significance.
-A subgroup analysis was performed for BRAF positive mutants. The benefit of treatment with relatlimab–nivolumab was observed regardless of patients’ BRAF mutation status. In the subgroup of patients with BRAF mutations, the median progression-free survival was 10.1 months (95% CI, 4.6 to 23.1) in the relatlimab–nivolumab group and 4.6 months (95% CI, 3.0 to 6.5) in the nivolumab group (hazard ratio for progression or death, 0.74 [95% CI, 0.54 to 1.03]); in the subgroup of patients with wild-type BRAF, the median progression-free survival was 10.1 months (95% CI, 5.9 to 17.0) with relatlimab–nivolumab and 4.6 months (95% CI, 2.9 to 6.6) with nivolumab (hazard ratio, 0.76 [95% CI, 0.59 to 0.98]). |
|
Larkin 2015, Wolchock 2017, Hodi 2018, Larkin 2019, Wolchok 2022
CheckMate 067 |
Type of study: multicentre, randomized, double-blind, phase 3 study.
Setting and country: Multicentre, 137 centres in Australia, Europe, Israel, New Zealand, and North America.
Funding and conflicts of interest: Funded by Bristol-Meyers Squibb.
Detailed declarations of interests are provided in the article. |
Inclusion criteria: -histologically confirmed stage III (unresectable) or stage IV melanoma -no prior systemic treatment for advanced disease. -age >= 18 years -ECOG performance-status score of 0-1 -measurable disease as assessed by means of computed tomography or magnetic resonance imaging according to Response Evaluation Criteria in Solid Tumors (RECIST), version 1.1 -availability of tissue collected from metastatic or unresectable tumors (archival or recently biopsied samples) for the assessment of PD-L1 status -known BRAF V600 mutation status
Exclusion criteria: -ECOG score of 2 or higher -presence of active brain metastases -ocular melanoma -autoimmune disease.
N total at baseline: 945 Nivolumab: 316 Nivolumab + ipilimumab: 314 Ipilimumab: 315
Important prognostic factors2:
Median age (IQR) nivo: 59 (25-90) nivo+ipi: 59 (18-88) ipi: 61 (18-89)
Sex: nivo: 202 (63.9%) M nivo+ipi: 206 (65.6%) M ipi: 202 (64.1%) M
ECOG performance status 0: nivo: 238 (75.3%) nivo+ipi: 230 (73.2%) ipi: 224 (71.1%)
No BRAF mutation: nivo: 216 (68.4%) nivo+ipi: 213 (67.8%) ipi: 218 (69.2%)
Increased serum lactate dehydrogenase <ULN: Nivo: 112 (35.4%) nivo+ipi: 114 (36.3%) ipi: 115 (36.5%)
Brain metastases nivo: 8 (2.5%) nivo+ipi: 11 (3.5%) ipi: 15 (4.8%)
PD-L1 status positive: nivo: 80 (25.3%) nivo+ipi: 68 (21.7%) ipi: 75 (23.8%)
Groups comparable at baseline? Yes
|
Describe intervention (treatment/procedure/test):
Nivo + ipi: 1 mg of nivolumab per kilogram every 3 weeks plus 3 mg of ipilimumab per kilogram every 3 weeks for 4 doses, followed by 3 mg of nivolumab per kilogram every 2 weeks for cycle 3 and beyond. Administered by means of intravenous infusion. |
Describe control (treatment/procedure/test):
Nivo: 3 mg of nivolumab per kilogram of body weight every 2 weeks (plus ipilimumab-matched placebo).
Ipi: 3 mg of ipilimumab per kilogram every 3 weeks for 4 doses (plus nivolumab-matched placebo).
Administered by means of intravenous infusion. |
Larkin 2015 Median follow-up: Clinical data cutoff February 17, 2015. Range 12.2-12.5 months
Loss-to-follow-up: Nivo: 3 (0.95%) 1 no longer met study criteria 1 withdrew consent 1 request to discontinue
Nivo + ipi: 1 (0.32%) 1 no longer met study criteria
Ipi: 4 (1.27%) 2 no longer met study criteria 1 withdrew consent 1 disease progression
Wolchok 2017 Median follow-up: Clinical data cutoff May 24, 2017. Nivo: 35.7 months Nivo+ipi: 38.0 months Ipi: 18.6 months
Loss-to-follow-up: Nivo: 265 (84.7%) 174 disease progression 42 study drug toxicity 1 death 8 adverse events 24 patient request 1 lost to follow-up 12 maximum clinical benefit 1 poor/noncompliance 2 other
Nivo + ipi: 288 (92.0%) 90 disease progression 131 study drug toxicity 4 deaths 18 adverse events 24 patient request 3 withdrew consent 12 maximum clinical benefit 1 poor/noncompliance 1 no longer met study criteria 4 other
Ipi: 303 (97.4%) 224 disease progression 52 study drug toxicity 1 death 6 adverse events 13 patient request 1 withdrew consent 3 maximum clinical benefit 1 poor/noncompliance 2 other
Hodi 2018 Median follow-up (IQR): Clinical data cutoff May 10, 2018. Nivo: 36.0 months (10.5-51.4 months) Nivo+ipi: 46.9 months (10.9-51.8 months) Ipi: 18.6 months (7.6-49.5 months)
Loss-to-follow-up:
Nivo: 280 177 disease progression 44 study drug toxicity 1 death 8 adverse events 29 patient request 1 lost to follow-up 16 achieved maximum clinical benefit 1 poor or non-compliance 3 other
Nivo + ipi: 295 90 disease progression 134 study drug toxicity 4 deaths 19 adverse events 27 patient request 3 withdrew consent 13 achieved maximum clinical benefit 1 poor or non-compliance 1 no longer met study criteria 3 other
Ipi: 311 224 disease progression 52 study drug toxicity 1 death 6 adverse events 13 patient request 1 withdrew consent 4 achieved maximum clinical benefit 1 poor or non-compliance 6 administrative reason* 3 other
Larkin 2019 Median follow-up: Clinical data cutoff July 2, 2019. Nivo: 54.6 months Nivo+ipi: 36.0 months Ipi: 18.6 months
Loss-to-follow-up: Nivo: 289 (92.3%) 179 disease progression 45 study drug toxicity 1 death 8 adverse events 33 patient request 1 lost to follow-up 18 maximum clinical benefit 1 poor/non-compliance 3 other
Nivo + ipi: 301 (96.2%) 90 disease progression 139 study drug toxicity 4 death 18 adverse events 27 patient request 3 withdrew consent 15 maximum clinical Benefit 1 poor/non-compliance 1 no longer meets study criteria 3 other
Ipi: 311 (100%) 224 disease progression 52 study drug toxicity 1 death 6 adverse events 13 patient request 1 withdrew consent 4 maximum clinical benefit 1 poor/non-compliance 6 administrative reasons 3 other
Wolchok 2022 Median follow-up: Clinical data cutoff October 19, 2020. Nivo: 57.5 months Nivo+ipi: 36.0 months Ipi: 18.6 months
Loss-to-follow-up: Nivo: 305 (97.4%) 180 disease progression 49 study drug toxicity 1 death 8 adverse events 39 patient request 2 withdrew consent 1 lost to follow-up 21 maximum clinical benefit 1 poor/non-compliance 3 other
Nivo + ipi: 306 (97.8%) 91 disease progression 139 study drug toxicity 4 death 18 adverse events 30 patient request 3 withdrew consent 16 maximum clinical Benefit 1 poor/non-compliance 1 no longer meets study criteria 3 other
Ipi: 311 (100%) 224 disease progression 52 study drug toxicity 1 death 6 adverse events 13 patient request 1 withdrew consent 4 maximum clinical benefit 1 poor/non-compliance 6 administrative reasons 3 other |
Outcome measures and effect size (include 95%CI and p-value if available):
Larkin 2015 Median progression-free survival: Nivo: 6.9 months (95% CI 4.3-9.5) Nivo + ipi: 11.5 months (95% CI 8.9-16.7) Ipi: 2.9 months (95% CI 2/8-3.4) HR nivo+ipi vs ipi 0.42; 99.5% CI, 0.31 to 0.57 HR nivo+ipi vs nivo 0.74 (95% CI, 0.60 to 0.92) HR nivo vs ipi HR, 0.57; 99.5% CI, 0.43 to 0.76.
Adverse events: Nivo: 257 (82.1%) Nivo+ipi: 299 (95.5%) Ipi: 268 (86.2%)
Treatment related grade 3 and 4: Nivo: 51 (16.3%) Nivo+ipi: 172 (55.0%) Ipi: 85 (27.3%)
Quality of life: Not reported.
Wolchock 2017 Median progression-free survival: Nivo: 6.9 months (95% CI, 5.1 to 9.7) Nivo + ipi: 11.5 months (95% CI 8.9-19.3) Ipi: 2.9 months (95% CI 2.8-3.2) HR nivo+ipi vs ipi 0.43 (95% CI, 0.35 to 0.52) HR nivo+ipi vs nivo 0.78 (95% CI, 0.64 to 0.96) HR nivo vs ipi HR, 0.55 (95% CI, 0.45 to 0.66)
Overall survival at 2 years: Nivo: 59% Nivo+ipi: 64% Ipi: 45%
Overall survival at 3 years: Nivo: 52% Nivo+ipi: 58% Ipi: 34%
Median overall survival: Nivo: 37.6 months; 95% CI, 29.1 to not reached Nivo+ipi: not reached Ipi: 19.9 months; 95% CI, 16.9 to 24.6
Adverse events: Nivo: 270 (86) Nivo+ipi: 300 (96) Ipi: 268 (86)
Treatment related grade 3 and 4: Nivo: 67 (21) Nivo+ipi: 184 (59) Ipi: 86 (28)
Quality of life: Not reported.
Hodi 2018: Median progression-free survival: Nivo: 6.9 months (95% CI, 5.1 to 10.2) Nivo + ipi: 11.5 months (95% CI 8.7–19.3) Ipi: 2.9 months (95% CI 2.8-3.2) HR nivo+ipi vs ipi 0.42 (95% CI, 0.35 to 0.51) HR nivo+ipi vs nivo 0.79 (95% CI, 0.65 to 0.97) HR nivo vs ipi HR, 0.53 (95% CI, 0.44 to 0.64)
Median overall survival Nivo: 36·9 months (28·3–not reached) Nivo+ipi: not reached (95% CI 38·2–not reached) Ipi: 19·9 months (16·9–24·6)
Overall survival at 4 years: Nivo: 46% (41−52) Nivo+ipi: 53% (95% CI 47−58) Ipi: 30% (25−35)
Adverse events: Nivo: 270 (86) Nivo+ipi: 300 (96) Ipi: 268 (86)
Treatment related grade 3 and 4: Nivo: 70 (22%) Nivo+ipi: 185 (59%) Ipi: 86 (28%)
Quality of life: Not reported.
Larkin 2019 Median progression-free survival: Nivo: 6.9 months (95% CI, 5.1 to 10.2) Nivo + ipi: 11.5 months (95% CI, 8.7 to 19.3) Ipi: 2.9 months (95% CI 2.8-3.2)
Median overall survival Nivo: 36.9 months (95% CI, 28.2 to 58.7) Nivo+ipi: 60.0 months (median not reached; 95% confidence interval [CI], 38.2 to not reached) Ipi: 19.9 months (95% CI, 16.8 to 24.6)
Overall survival at 5 years: Nivo: 44% Nivo+ipi: 52% Ipi: 26%
Adverse events: Nivo: 271 (87) Nivo+ipi: 300 (96) Ipi: 268 (86)
Treatment related grade 3 and 4: Nivo: 73 (23%) Nivo+ipi: 186 (59%) Ipi: 86 (28%)
Quality of life: Not reported. Wolchok 2022
Median progression-free survival: Nivo: 6.9 months (95% CI, 5.1 to 10.2) Nivo + ipi: 11.5 months (95% CI, 8.7 to 19.3) Ipi: 2.9 months (95% CI 2.8-3.2)
Median overall survival Nivo: 36.9 months (95% CI, 28.2 to 58.7) Nivo+ipi: 72.1 months (38.2 to not reached) Ipi: 19.9 months (95% CI, 16.8 to 24.6)
Overall survival at 6.5 years: Nivo: 42% Nivo+ipi: 49% Ipi: 23%
Adverse events: Nivo: 271 (87) Nivo+ipi: 300 (96) Ipi: 268 (86)
Treatment related grade 3 and 4: Nivo: 73 (23%) Nivo+ipi: 186 (59%) Ipi: 86 (28%)
Quality of life: Not reported.
|
-Subgroup analyses performed on PD-L1 status, BRAF mutation status, and metastasis stage. |
|
Maio, 2015 Robert, 2011
CA184-024
NCT00324155 |
Multinational, randomized, double-blind, phase 3 study.
Patient enrolment between: August 8, 2006, and January 22, 2008.
Funding and conflicts of interest:
Disclosure forms provided by the authors are available with the full text of this article. |
Inclusion criteria:
Exclusion criteria:
Mean age, years I: 57.5 C: 56.4
Male, n (%) I: 152 (60.8%) C: 149 (59.1%)
ECOG PS: 0 – I: 177 (70.8%) 0 – C: 179 (71.0%) 1 – I: 73 (29.2%) 1 – C: 73 (29.0%)
Groups were comparable at baseline. |
Ipilimumab (dose: 10 mg per kg) plus dacarbazine (850 mg per square meter)
n=250 |
Dacarbazine (850 mg per square meter) plus placebo.
n=252 |
Maio, 2015 Remained on ipilimumab treatment as of the last database lock: n=7
Alive at a minimum follow-up of 5 years: I: n=40 C: n=20
Median survival follow-up: I: 11 months (range, 0.4 to 71.9 months) C: 8.9 months (range, 0.1 to 73.2 months)
Robert, 2011 Follow-up time between the start of the study (first visit of first patient) and end of the study (last visit of last patient): 54 months
Follow-up time between the time the last patient underwent randomization (the first visit of the last patient) and the end of the study: 36.6 months.
A survival analysis was performed after 414 deaths occurred, 37 months after the last patient was enrolled.
Discontinuation due to treatment related AEs, n (%): I: 89/247 (36%) C: 10/251 (4%)
|
Maio, 2015 Median OS, months (95% CI) I: 11.2 (9.5 to 13.8) C: 9.1 (7.8 to 10.5) HR, 0.69; 95% CI, 0.57 to 0.84
1-Year OS, months (95% CI): I: 47.6% (41.2 to 53.7) C: 36.4% (30.4 to 42.4)
2-Year OS, months (95% CI): I: 28.9% (23.3 to 34.7) C: 17.8% (13.3 to 22.8)
3-Year OS, months (95% CI): I: 21.3% (16.3 to 26.6) C: 12.1% (8.4 to 16.5)
4-Year OS, months (95% CI): I: 19.1% (14.4 to 24.3) C: 9.7% (6.4 to 13.7)
5-Year OS, months (95% CI): I: 18.2% (13.6 to 23.4) C: 8.8% (5.7 to 12.8) (P=0.002)
irAEs with onset during ipilimumab maintenance therapy in patients who survived ≥ 5 years: n=7.
reported grade 3 to 4 irAEs (rash, pruritus) occurred in the same patient.
Grade 5 irAEs: None.
Robert, 2011 Deaths I: 196/250 (78.4%) C: 218/252 (86.5%)
Median OS, months (95% CI) I: 11.2 (9.4 to 13.6) C: 9.1 (7.8 to 10.5) HR for death 0.72; P<0.001.
1-Year OS, months (95% CI): I: 47.3% C: 36.3%
2-Year OS, months (95% CI): I: 28.5% C: 17.9%
3-Year OS, months (95% CI): I: 20.8% C: 12.2%
Disease progression, n: I: 203 C: 223 HR for progression, 0.76; P = 0.006.
AEs grade 3 or 4: I: 56.3% C: 27.5% P<0.001
Immunemediated adverse reactions grade ≥ 3 I: 38.1% C: 4.4%
Drug-related deaths, n: I: 0 C: 1 (gastrointestinal Hemorrhage)
For more information on AEs see results section of the article. |
randomization and received at least one dose of the assigned study drug (498 patients).
Authors conclusions: Robert 2011: Ipilimumab (at a dose of 10 mg per kilogram) in combination with dacarbazine, as compared with dacarbazine plus placebo, improved overall survival in patients with previously untreated metastatic melanoma. The types of adverse events were consistent with those seen in prior studies of ipilimumab; however, the rates of elevated liver-function values were higher and the rates of gastrointestinal events were lower than expected on the basis of prior studies.
Maio, 2015: The additional survival benefit of ipilimumab plus dacarbazine is maintained with twice as many patients alive at 5 years compared with those who initially received placebo plus dacarbazine. These results demonstrate a durable survival benefit with ipilimumab in advanced melanoma.
|
|
Robert, 2019 Carlino, 2018 Robert, 2015
KEYNOTE-006
NCT01866319 |
International, randomized, open-label phase 3 study.
In 16 countries.
Patient enrolment: From September 18, 2013, to March 3, 2014.
Funding and conflicts of interest:
contributed to:
Disclosure forms provided by the authors are available with the full text of this article. |
Inclusion criteria:
Exclusion criteria:
Mean age, years Ia: 61 (18–89) Ib: 63 (22–89) C: 62 (18–88)
Male, n (%) Ia: 161 (57.7) Ic: 174 (62.8) C: 162 (58.3)
ECOG PS: 0 – Ia: 196 (70.3) 0 – Ib: 189 (68.2) 0 – C: 188 (67.6) 1 – Ia: 83 (29.7) 1 – Ib: 88 (31.8) 1 – C: 90 (32.4)
PD-L1-positive tumours: 80.6%
Groups were comparable at baseline. |
Ia: pembrolizumab at a dose of 10 mg per kilogram of body weight every 2 weeks n= 279
Ib: n=277
|
C: dose of 3 mg per kilogram every 3 weeks n=278
|
Robert, 2019: Median follow-up for survival: 57.7 months (IQR 56.7–59.2).
Carlino, 2018: Median follow-up: 33.9 months.
Schachter, 2017: Median follow-up: 22.9 months
Discontinued treatment: Ia: 147 progressive disease 29 adverse events 2 deaths 2 complete responses 21 other
Ib: 139 progressive disease 45 adverse events 1 death 5 complete responses 23 other
C: 46 progressive disease 35 adverse events 5 deaths 24 other
Withdrew consent and did not receive treatment: Ia: n=1 C: n=22
Robert, 2015: Median follow-up at data cutoff (with 502 events reported), months: 7.9 (range: 6.1 to 11.5)
March 3, 2015: Follow-up for OS: Minimum follow-up: 12 months with 289 deaths occurred
Mean duration of exposure, days: Ia: 164 Ib: 151 C: 50
Rate discontinuation of a study drug because of treatment related AEs: Ia: 4.0%, Ib: 6.9%, C: 9.4%,
|
Robert, 2019: Median OS: C: 15.9 months (13.3–22.0) HR: 0.73 (95% CI 0.61–0.88, p=0·00049). Median PFS: C: 3.4 months (2.9–4.2) HR 0.57, 95% CI 0.48–0.67, p<0.0001.
Grade 3–4 treatment-related AEs: C: 50 (20%)
Treatment-related sepsis. C: n=1
Schachter, 2017: Death: n=383
Median OS: Ib: not reached (23.5 months–not reached) C: 16.0 months (range 13.5–22.0) HR pembro every 2 weeks vs ipi: 0.68, 95% CI 0·53–0·87; p=0·0009 HR pembro every 3 weeks vs ipi: 0.68, 0·53–0·86; p=0·0008.
2 year OS rate: Ia: 55% (95% CI 49–61) Ib: 55% (95% CI 49–61) C: 43% (95% CI 37–49)
PFS events: n= 566 I (pooled groups): 364 (65%) C: 202 (35%)
Median PFS, months: Ia: 5.6 months (range 3.4–8.2) Ib: 4.1 months (range 2.9–7.2) C: 2.8 months (range 2.8–2.9) HR for both Pembro schedules vs ipi: 0.61; 95% CI 0·50–0·75; p<0·0001 HR for Ia vs Ib: 0.95; 95% CI 0·77–1·17; p=0·62).
2-year PFS rate: Ia: 31% Ib: 28% C: 14%
Treatment related AEs grade 3 to 5: Ia: 47 (17%) of 278 Ib: 46 (17%) of 277 C: 50 (20%) of 256
Robert, 2015: Median overall survival was not reached in any study group.
1-Year OS: Ia: 74.1% Ib: 68.4% C: 58.2%
Median PFS, months (95% CI): Ia: 5.5 (95% CI, 3.4 to 6.9) Ib: 4.1 (95% CI, 2.9 to 6.9) C: 2.8 (95% CI, 2.8 to 2.9)
6-month PFS, months: Ia: 47.3% Ib: 46.4% C: 26.5%
Treatment related AEs grade 3 to 5: Ia: 13.3% Ib: 10.1% C: 19.9%
Drug-related deaths, n: Ia: 0 Ib: 0 C: 1
For more information on AEs see results section of the article. |
Co-primary endpoints were OS and PFS. Robert, 2019:
Carlino, 2018:
Schachter, 2017:
Robert, 2015:
Authors conclusions: Robert, 2019: Pembrolizumab continued to show superiority over ipilimumab after almost 5 years of follow-up. These results provide further support for use of pembrolizumab in patients with advanced melanoma.
Carlino, 2018: with advanced melanoma, regardless of first- or second-line therapy or PD-L1 status.
Schachter, 2017: Substantiating the results of the interim analyses of KEYNOTE-006, pembrolizumab continued to provide superior overall survival versus ipilimumab, with no difference between pembrolizumab dosing schedules. These conclusions further support the use of pembrolizumab as a standard of care for advanced melanoma.
Robert, 2015: The anti–PD-1 antibody pembrolizumab prolonged progression-free survival and overall survival and had less high-grade toxicity than did ipilimumab in patients with advanced melanoma.
|
|
Chesney, 2023 MASTERKEY-265 NCT02263508 |
A multicenter, double-blind, placebo controlled, randomized phase III study in 21 countries.
Patient enrolment between: March 17, 2016, through April 26, 2018.
Funding and conflicts of interest: - Supported by Amgen Inc and Merck Sharp & Dohme LLC, a subsidiary of Merck & Co, Inc, Rahway, NJ. The sponsors contributed to:
- Authors’ disclosures of potential conflict of interest is provided at the end of the full text article. |
Main Inclusion criteria:
Exclusion criteria:
For more information on in-/exclusion see the article.
Median age, years (range) I: 64 (26-92) C: 64 (19-94)
Male, n (%) I: 199 (57.5) C: 219 (63.3)
ECOG PS: 0 – I: 259 (74.9) 0 – C: 249 (72.0) 1 – I: 87 (25.1) 1 – C: 97 (28.0)
Groups were comparable at baseline. |
I: A combination of T-VEC plus pembrolizumab (T-VEC-pembrolizumab) n=346
T-VEC was administered at ≤ 4 x 106 plaque-forming unit followed by ≤ 4 x 108 PFU 3 weeks later and once every 2 weeks until dose 5 and once every 3 weeks thereafter.
Pembrolizumab was administered intravenously 200 mg once every 3 weeks. |
C: Placebo plus pembrolizumab (placebo-pembrolizumab n=346
Pembrolizumab was administered intravenously 200 mg once every 3 weeks. |
Median follow-up in months:
Discontinued study: I: n = 152 Death n = 131 Withdrawal of consent n = 15 Lost to follow-up n=6
C: n = 170 Death n = 142 Withdrawal of consent n = 22 Lost to follow-up n = 6
April 2020, all patients discontinued study treatments. The final analysis was performed early given the futility noted in the second interim analysis and included an additional follow-up of 6 months. |
Deaths at planned second interim OS analysis: I: 136 (39.3%) C: 146 (42.2%)
Median OS, months (95% CI): I: Not estimable C: 49.2 (40.57 to not estimable) HR of 0.96 (95% CI, 0.76 to 1.22; P = .74)
The primary analysis of PFS was to be performed after 407 PFS events occurred.
Median PFS (95% CI) (months): I: 14.3 (10.25 to 22.11) C: 8.5 (5.72 to 13.54) Stratified log-rank: HR, 0.86 (95% CI, 0.71 to 1.04), P=.13
Treatment related AEs grade 3 or 4: I: 70 (20.3%) C: 54 (15.7%)
Fatal AEs: I: 45 (13.1%) C: 42 (12.2%)
Treatment related fatal AEs, n: I: 4 (1.2%) C: 1 (0.3%)
Immune-related AEs: I: 27.5% C: 24.8%
For more information on AEs see results section of the article
At final analysis:
No new safety signals were observed. |
Sep 29, 2020, for the second interim OS analysis; Mar 26, 2021, the final analysis.
subsequent anticancer therapy
Authors conclusions: This randomized, double-blinded, placebo-controlled, multicenter, international phase III trial did not show improved PFS or OS for the combination of T-VEC plus pembrolizumab compared with placebo plus pembrolizumab for immunotherapy- naïve patients with advanced melanoma in the frontline setting. There were no new safety concerns with the addition of T-VEC to pembrolizumab, and the safety profile of the combination was consistent with the known safety profile of each drug. |
|
Andtbacka, 2019; Andtbacka, 2015 OPTiM |
A randomized open-label phase III trial at 64 sites in the United States, the United Kingdom, Canada, and South Africa.
Patient enrolment between: 2009 and 2011
Funding and conflicts of interest: - Funded by BioVex, who were subsequently acquired by Amgen Inc. during the OPTiM trial. The sponsor contributed to:
- A competing interests statement is provided at the end of the full text article. |
Main inclusion criteria:
Exclusion criteria:
For more information on in-/exclusion see the article.
Median age, years (range) I: 63 (22 to 94) C: 64 (26 to 91)
Male, n (%) I: 173 (59%) C: 77 (55%)
ECOG PS: 0 – I: 209 (71%) 0 – C: 97 (69%) 1 – I: 82 (28%) 1 – C: 32 (23%) Unknown: I: 4 (1%) C: 12 (9%)
Groups were comparable at baseline.
|
I: intratumoral Talimogene laherparepvec (T-VEC) (at the approved dose) n= 295 (68%)
|
C: subcutaneous recombinant granulocyte-macrophage colony-stimulating factor (GM-CSF) n= 141 (32%) |
Median follow-up in the final analysis of OS: 49 months.
Median duration of treatment in weeks (range): I: 23.1 (0.1–176.7) C: 10.0 (0.6–120.0)
Andtbacka, 2015: Discontinued T-VEC: n=291 Disease progression: n=191 PR or CR for ≥ 6 continuous months: n=42 Maximum allowed dose without PR/CR: n=26 Adverse event: n=11 Consent withdrawn: n=10 Physician decision: n=6 Death: n=5
Discontinued GM-CSF: n=127 Disease progression: n=95 PR or CR for ≥ 6 continuous months: n=0 Maximum allowed dose without PR/CR: n=9 Adverse event: n=3 Consent withdrawn: n=12 Physician decision: n=5 Death: n=3 |
Intent-to treat population (stage IIIB–IVM1c melanoma): Median OS, months (95% CI): I: 23.3 (19.5–29.6) C: 18.9 (16.0–23.7) unstratified HR for death, 0.79 (95% CI,0.62–1.00); P = 0.0494).
Estimated 5-year survival I: 33.4% C: Not estimable
Stage IIIB–IVM1a disease Effect of T-VEC on OS vs GM-CSF:
Effect of T-VEC on OS vs ITT population including stage IVM1b/c disease:
Estimated 5-year survival with T-VEC:
Treatment related AEs grade 3/4: I: 33 (11.3%) C: 6 (4.7%)
Immune-related AEs: I: 24/295 C: ?
Immune-related AEs grade 3: n=4
Immune-related AEs grade 4: None reported
Treatment-related deaths, n: I: 0 C: 0
For more information on AEs see results section of the article |
assessment. Key secondary end points: OS and overall response rate
Authors conclusions: Andtbacka, 2019: In conclusion, as well as demonstrating a longer-term effect on survival, this analysis confirms that T-VEC resulted in high CR rates, most notably in patients with early metastatic melanoma (stage IIIB–IVM1a). Once achieved, CRs were durable and associated with prolonged survival. The favorable clinical outcomes observed in some patients treated with T-VEC, along with its good safety profile, support continued efforts to further define its future role in melanoma as a combination partner with immunotherapy.
Andtbacka, 2015: T-VEC is the first oncolytic immunotherapy to demonstrate therapeutic benefit against melanoma in a phase III clinical trial. T-VEC was well tolerated and resulted in a higher DRR (P<0.001) and longer median OS (P=0.051), particularly in untreated patients or those with stage IIIB, IIIC, or IVM1a disease. T-VEC represents a novel potential therapy for patients with metastatic melanoma. |
|
Larkin 2014, Ascierto 2016, Ascierto 2021, CoBRIM |
Type of study: multicentre, randomized, controlled, double-blind, phase 3 study.
Setting and country: Multicentre, 135 sites in the United States, Canada, Australia, New Zealand, Europe, Russia, Turkey, and Israel.
Funding and conflicts of interest: Funded by Bristol-Meyers Squibb. The funder participated in data collection and medical writing support.
Detailed declarations of interests are provided in the article. |
Inclusion criteria: advanced stage IIIC or stage IV melanoma with a BRAF V600 mutation detected with the use of a real-time polymerase-chain-reaction assay (Cobas 4800 BRAF V600 Mutation Test, Roche Molecular Systems) -Aged >= 18 years -Had measurable disease, according to the Response Evaluation Criteria in Solid Tumors (RECIST), version 1.1, as assessed by means of computed tomography -ECOG performance status 0 or 1. -Had adequate hematologic, hepatic, renal, and cardiac function
Exclusion criteria: ?
N total at baseline: 495 Intervention: 247 Control: 248
Important prognostic factors2:
Median age (IQR) I: 56 (23-88) C: 55 (25-85)
Sex: I: 59% M C: 56% M
ECOG performance status: I: 76% 0 C: 67% 0
BRAF mutation V600E: I: 69% C: 70%
Brain metastases: I: <1% C: 1%
Increased lactate dehydrogenase levels <ULN: I: 46% C: 43%
Groups comparable at baseline? Yes.
|
Describe intervention (treatment/procedure/test):
Oral administration of vemurafenib (at a dose of 960 mg twice daily) together with cobimetinib (at a dose of 60 mg once daily for 21 days, followed by 7 days off) (combination group). |
Describe control (treatment/procedure/test):
Oral administration of vemurafenib (at a dose of 960 mg twice daily) together with placebo (control group) |
Larkin 2014: Clinical data cutoff July 10, 2014 Median follow-up 7.3 months (range, 0.5 to 16.5).
Loss-to-follow-up: Intervention: 1 104 discontinued vemurafenib 107 discontinued cobimetinib 102 discontinued vemurafenib and cobimetinib
Control: 3 139 discontinued vemurafenib 140 discontinued placebo 138 discontinued vemurafenib and placebo
Ascierto 2016 Clinical data cutoff Aug 28, 2015 Median follow-up 18·5 months (IQR 8·5–23·5).
Loss-to-follow-up: Intervention: 136 114 died 2 lost to follow-up 17 patient decision to withdraw 3 physician decision to withdraw
Control: 164 141 died 6 lost to follow-up 17 patient decision to withdraw
Ascierto 2021 Clinical data cutoff July 21, 2019. Median follow-up I: 21.2 months (10.4–59.0) C: 16.6 months (7.3–42.5)
Loss-to-follow-up: Intervention: 100 discontinued treatment 167 died 8 lost to follow-up 20 patient decision to withdraw 1 physician decision 4 other
Control: 247 discontinued treatment 157 died 4 lost to follow-up 21 patient decision to Withdraw 4 physician decision 2 other
|
Outcome measures and effect size (include 95%CI and p-value if available):
Larkin 2014 Median overall survival at 9 months: C: 73% (95% CI, 65 to 80) HR 0.65; 95% CI, 0.42 to 1.00
Median progression-free survival: I: 9.9 months (95% CI, 9.0 - not reached), C: 6.2 months (95% CI, 5.6-7.4) HR 0.51 (95% CI, 0.39 to 0.68
Adverse events: I: 96% C: 96% Treatment related grade 3 and 4: I: 63% C: 58%
Quality of life: Not reported.
Ascierto 2016 Median overall survival: I: 22·3 months (95% CI 20·3–not estimable) C: 17·4 months (95% CI 15·0–19·8) HR 0·70 [95% CI 0·55–0·90]
Median 1 year survival: I: 74·5% (95% CI 68·9–80·2) C: 63·8% (57·6–70·0) Median 2 year survival: I: 48·3% (41·4–55·2) C: 38·0% (31·3–44·7)
Median progression-free survival: I: 12·3 months (9·5–13·4) HR 0·59 (0·47–0·73)
Adverse events: I: 99% C: 98% Treatment related grade 3 and 4: I: 75% C: 61%
Quality of life: Not reported.
Ascierto 2021 Median overall survival: I: 22.5 months (95% CI, 20.3–28.8) C: 17.4 months (95% CI, 15.0–19.8) HR 0·70 [95% CI 0·55–0·90]
Median 3 year survival: I: 38% (95% CI, 32–45) C: 31% (95% CI, 25–37) Median 4 year survival: I: 34% (95% CI, 28–40) C: 29% (95% CI, 23–35) Median 5 year survival: I: 31% (95% CI, 25–37) C: 26% (95% CI, 20–32)
Median progression-free survival: I: 12.6 months (95% CI, 9.5–14.8)
Adverse events: I: 99% C: 98% Treatment related grade 3 and 4: I: 78% C: 63%
Quality of life: Not reported. |
Author’s conclusion: The OS results confirmed the long-term OS benefit in patients treated with cobimetinib plus vemurafenib compared with placebo plus vemurafenib in patients with previously untreated, BRAFV600 mutation–positive advanced melanoma. The greatest benefit was observed in patients who achieved a complete response and in those with normal LDHlevels and low tumor burden at baseline. |
|
Robert 2015, Robert 2016, COMBI-v
|
Type of study: open-label, randomized, phase 3 study
Setting and country: Multicentre, 193 centries.
Funding and conflicts of interest: The study was funded by the sponsor, GlaxoSmithKline, and also provided editorial assistance.
Detailed declarations of interests are provided in the article. |
Inclusion criteria: -the presence of BRAF V600E or V600K mutations was centrally determined with the investigational use of the THxID BRAF assay (bioMérieux) -measureable disease, according to the Response Evaluation Criteria in Solid Tumors (RECIST), version 1.1,15 -An Eastern Cooperative Oncology Group (ECOG) performance status of 0 or 1 -Patients who had undergone treatment for brain metastases with no increase in lesion size for at least 12 weeks were eligible
Exclusion criteria: -See supplement.
N total at baseline: 703 I: 352 C: 352
Important prognostic factors2:
Median age (IQR) I: 55 (18–91) C: 54 (18–88)
Sex: I: 208 (59) C: 180 (51)
ECOG performance status: I: 248 (71) C: 248 (70)
BRAF mutation V600E: I: 312(90) C: 317 (90)
Groups comparable at baseline? Yes. |
Describe intervention (treatment/procedure/test):
A combination of dabrafenib (150 mg orally twice daily) and trametinib (2 mg orally once daily)
|
Describe control (treatment/procedure/test):
Vemurafenib (960 mg orally twice daily). |
Robert 2015 Clinical data cutoff April 17, 2014 Median follow-up 11 (I), 10 (C) months
Loss-to-follow-up: I: 16 4 lost to follow-up 2 investigator discretion 10 withdrew consent
C: 28 9 lost to follow-up 1 investigator discretion 18 withdrew consent
Robert 2016 Clinical data cutoff July 2016 Median follow-up 27 (I), 26 (C) months
Loss-to-follow-up: NR
|
Robert 2015 Median overall survival: I: not reached C: 17.2 months
Median 1 year survival: I: 72% (95% CI, 67 to 77) C: 65% (95% CI, 59 to 70)
Median progression-free survival: I: 11.4 months C: 7.3 months HR, 0.56; 95% CI, 0.46 to 0.69
Adverse events: I: 343 (98) C: 345 (99)
Grade 3-4: I: 167 (48) C: 198 (57)
Quality of life: Not reported.
Robert 2016 Median overall survival: NR
Median 3 year survival: I: 45% [95% CI, 39-50] C: 32% [95% CI, 27-37]
Median 3 year progression-free survival: I: 25% [95% CI, 20-30] C: 11% [95% CI, 7-16]
Adverse events: Similar to Robert 2015
Quality of life: Not reported.
|
Author’s conclusion: In conclusion, the combination of dabrafenib plus trametinib was superior to vemurafenib monotherapy with regard to all efficacy end points, including overall survival, with no additional overall toxicity.
Robert 2016 is only a published conference abstract, limited information available. |
|
Long, 2014 COMBI-d NCT01584648 |
A double-blind, randomized, phase 3 study without crossover, at 113 centres worldwide.
Patient enrolment between: May 2012 through January 2013
Funding and conflicts of interest:
Disclosure forms provided by the authors are available with the full text of this article at NEJM.org. |
Inclusion criteria:
treated and stable for at least 12 weeks were eligible to participate.
Exclusion criteria:
Additional inclusion and exclusion criteria are provided in the Supplementary Appendix, available with the full text of this article at NEJM.org.
Median age, years I: 55.0 (22–89) C: 56.5 (22–86)
Male, n (%) I: 111 (53) C: 114 (54)
ECOG PS: 0 – I: 155/210 (74) 0 – C: 150/211 (71) 1 – I: 55/210 (26) Groups were comparable at baseline. |
A combination of oral dabrafenib (150 mg twice daily) and oral trametinib (2 mg once daily)
n=211 |
Oral dabrafenib (150 mg twice daily) and placebo.
n=212 |
Long 2017: At data cut-off, 15 Feb 2016, follow-up among patients who were alive: ≥36 months from time of randomization.
Remained on randomized treatment: I: 40 (19%) C: 6 (3%)
Median time on treatment: I: 11.8 (range, 0.4–43.7) C: 8.3 (range, 0.1–45.3) months
>12 months of treatment: I: 49% C: 38%
Long, 2015: Median follow-up: I: 20.0 months (range 0–30) C: 16.0 months (range 0–32)
|
Long, 2017: 3-year OS (95% CI): I: 44% At data cutoff:
2-year PFS: 3-year PFS: ORR: AEs grade 3 or 4: For more information on AEs see results section of the article. Long, 2015: • 334 deaths
HR of 0·71 (95% CI 0·55–0·92; p=0·0107 Median OS, months I: 25.1 (95% CI 19·2–not reached) C: 18.7 (15·2–23·7) HR, 0.71; 95% CI, 0.55–0.92
1-year OS: I: 74% (67–79) C: 68% (61–74)
2-year OS: I: 51% (44–58) C: 42% (35–49)
At data cutoff: 301 progressed
HR 0·67, 95% CI 0·53–0·84, p=0·0004
Median PFS in months I: 11.0 (8·0–13·9) C: 8.8 (5·9–9·3) HR, 0.67; 95% CI, 0.53–0.84
ORR: I: 144 (69%; 62–75) C: 112 (53%; 46–60) Difference: 15% (6–25)§ 0·0014 AEs grade 3: I: 66 (32%) C: 63 (30%) |
Authors conclusions: Long 2017: These data demonstrate that durable (3 years) survival is achievable with dabrafenib plus trametinib in patients with BRAF V600-mutant metastatic melanoma and support long-term first-line use of the combination in this setting.
Long, 2015: The improvement in overall survival establishes the combination of dabrafenib and trametinib as the standard targeted treatment for BRAF Val600 mutation-positive melanoma. Studies assessing dabrafenib and trametinib in combination with immunotherapies are ongoing.
Long, 2014: A combination of dabrafenib and trametinib, as compared with dabrafenib alone, improved the rate of progression-free survival in previously untreated patients who had metastatic melanoma with BRAF V600E or V600K mutations.
|
|
Dummer 2018, Dummer 2018-2, Ascierto 2020, COLUMBUS |
Type of study: multicentre, two-part, randomised, open-label, phase 3 study.
Setting and country: Multicentre, 162 hospitals in 28 countriest.
Funding and conflicts of interest: Funded by Array BioPharma, Novartis. The sponsors had a role in data collection, analysis, and interpretation.
Detailed declarations of interests are provided in the article. |
Inclusion criteria: -A histologically confirmed diagnosis of locally advanced, unresectable, or metastatic cutaneous melanoma or unknown primary melanoma classified as American Joint Committee on Cancer (AJCC) stage IIIB, IIIC, or IV -Treatment naive or had progressed on or after previous first-line immunotherapy -Had a BRAFV600E or BRAFV600K mutation or both in tumour tissue as ascertained by central genetic mutation analysis with the bioMérieux THxID BRAF diagnostic test (bioMérieux, Marcy l’Etoile, France) before enrolment -ECOG performance status of 0 or 1 -Adequate bone marrow, organ function, and laboratory parameters -At least one measurable lesion, according to guidelines based on Response Evaluation Criteria in Solid Tumors (RECIST), version 1.1.
Exclusion criteria: -An untreated CNS lesions; uveal or mucosal melanoma -A history of leptomeningeal metastases -Gilbert’s syndrome -history, current evidence, Or risk of retinal vein occlusion -Previous BRAF inhibitor or MEK inhibitor treatment -Previous use of systemic chemotherapy -Extensive radiotherapy as evaluated by local investigators, or an investigational agent other than previous immunotherapy for locally advanced, unresectable, or metastatic melanoma (immunotherapy must have ended ≥6 weeks before randomisation).
N total at baseline: 577 encorafenib & binimetinib (A): 192 encorafenib (B): 194 vemurafenib (C): 191
Important prognostic factors2:
Median age (IQR) A: 57 (20–89; 48–66) B: 54 (23–88; 46–63) C: 56 (21–82; 45–65)
Sex: A: 115 (60%) M B: 108 (56%) M C: 111 (58%) M
ECOG performance status: A: 136 (71%) 0 B: 140 (72%) 0 C: 140 (73%) 0
BRAF mutation V600E: A: 170 (89%) B: 173 (89%) C: 168 (88%)
Groups comparable at baseline? Yes.
|
Describe intervention (treatment/procedure/test):
Oral encorafenib 450 mg once daily plus oral binimetinib 45 mg twice daily (encorafenib plus binimetinib group) |
Describe control (treatment/procedure/test):
Encorafenib 300 mg once daily orally, or vemurafenib 960 mg twice daily orally |
Dummer 2018 Clinical data cutoff May 19, 2016 Median follow-up 16·6 months (14·8–16·9)
Loss to follow-up: A: 124 discontinued treatment 83 had progressive disease 16 had adverse events 8 physician decision 7 patient or guardian decision 7 died 2 protocol deviation 1 lost to follow-up
B: 146 discontinued 87 had progressive disease 24 had adverse events 19 physician decision 13 patient or guardian decision 1 died 1 protocol deviation 1 lost to follow-up
C: 159 discontinued 101 had progressive disease 26 had adverse events 13 physician decision 15 patient or guardian decision 4 died
Dummer 2018-2 Clinical data cutoff Nov 7, 2017 Median follow-up 36·8 months (95% CI 35·9–37·5)
Loss-to-follow-up: A: 149 discontinued 99 progressive disease 20 adverse events 9 physician decision 11 patient or guardian decision 8 died 1 protocol deviation 1 lost to follow-up
B: 168 discontinued 100 progressive disease 25 adverse events 24 physician decision 17 patient or guardian decision 1 died 1 protocol deviation 0 lost to follow-up
C: 173 discontinued 109 progressive disease 25 adverse events 17 physician decision 17 patient or guardian decision 4 died 1 new therapy for study indication 0 protocol deviation 0 lost to follow-up
Ascierto 2020 Clinical data cutoff Nov 2018 Median follow-up 48.8 months
Loss-to-follow-up: A: 156 discontinued 104 progressive disease 20 adverse events 21 physician decision/ patient or guardian decision 9 died 2 other
B: 172 discontinued 101 progressive disease 24 adverse events 35 physician decision/patient or guardian decision 1 died 1 other
C: 177 discontinued 111 progressive disease 26 adverse events 35 physician decision/patient or guardian decision 4 died 1 other |
Dummer 2018 Median progression-free survival: A: 14·9 months (95% CI 11·0–18.5) B: 9·6 months (7·4–14·8)
Adverse events: A: 66 (34%) B: 65 (34%) C: 69 (37%) Grade 3-4 adverse events: A: 111 (58%) B: 127 (66%) C: 118 (63%)
Quality of life: Not reported.
Dummer 2018-2 Median overall survival: A: 33·6 months (95% CI 24·4–39·2) B: 23·5 months (19·6–33·6) C: 16·9 months (14·0–24·5) A vs C: HR 0·61 [95% CI 0·47–0·79) A vs B: HR 0·81 [95% CI 0·61–1·06] B vs C: HR 0·76 (95% CI 0·58–0·98)
Median 1 year survival: A: 75·5% (95% CI 68·8–81·0) B: 74·6% (67·6–80·3) C: 63·1% (55·7–69·6)
Median 2 year survival: A: 57·6% (95% CI 50·3–64·3) B: 49·1% (41·5–56·2) C: 43·2% (35·9–50·2)
Median progression-free survival: A: 14·9 months (95% CI 11·0–20·2) B: 9·6 months (7·4–14·8)
Adverse events: Similar to Dummer 2018
Quality of life: Not reported.
Ascierto 2020 Median overall survival: A: 33.6 months (95% CI, 24.4-39.2), B: 23.5 months (95% CI, 19.6-33.6) C: 16.9 months (95% CI, 14.0-24.5 HR 0.61 (95%CI 0.48-0.79)
Median 3 year survival: A: 47% B: 41% C: 31%
Median progression-free survival: A: 14.9 months (95% CI, 11.0- 20.2) B: 9.6 months (95% CI, 7.4-14.8) C: 7.3 months (95% CI, 5.6-7.9) HR (0.51, 95%CI 0.39-0.67)
Grade 3/4 adverse events: A: 68% B: 68% C: 66%
Quality of life: Not reported.
|
Author’s conclusion: In conclusion, patients treated with encorafenib plus binimetinib had longer PFS and OS than those treated with vemurafenib, with landmark analyses showing consistent improved OS and PFS for COMBO450 vs VEM for each year. Safety results were consistent with the known tolerability profile of COMBO450, and the toxicity burden was reduced over time. These data reinforce encorafenib plus binimetinib as an important treatment option for patients with BRAF-mutant melanoma. |
|
Atkins, 2022 The DREAMseq Trial
NCT02224781. |
A two-arm, two-step, open-label, randomized phase 3 trial.
Patient enrolment between: July 13, 2015, and July 16, 2021.
Funding and conflicts of interest:
UG1CA189809, UG1CA189822, UG1CA189829, UG1CA189830, UG1CA189863, UG1CA189953, UG1CA189957, UG1CA189997, UG1CA233184, UG1CA233193, UG1CA239769, UG1CA233234, UG1CA233290, UG1CA233320, UG1CA233270, UG1CA233330, UG1CA233331, and UG1CA239758. Authors’ disclosures of potential conflicts of interest are provided with the full text of this article. |
Inclusion criteria Step 1:
To enroll onto step 2:
Exclusion criteria:
Full eligibility criteria and toxicity management guidelines are provided in the Supplements (Table S1) describing the components of each protocol amendment.
Median age (range) A: 61 (25-85) B: 61 (30-84)
Male, n (%) A: 81 (60.9) B: 86 (65.2)
ECOG PS: 0 – A: 90 (67.8) 0 – B: 89 (67.4) 1 – A: 43 (32.2) 1 – B: 43 (32.6)
Arm A and B were balanced for most characteristics. More patients on arm B had BRAFV600K-mutant tumors than those on arm A (25.2% v 12.1%) |
Arm A: nivolumab/ipilimumab n=133
At disease progression patients were enrolled in step 2 to receive the alternate therapy: Arm C: dabrafenib/ trametinib n=27
|
Arm B: dabrafenib/trametinib n=132
At disease progression patients were enrolled in step 2 to receive the alternate therapy: Arm D: nivolumab/ ipilimumab n=46
A: 59.5% (79) B: 53.1% (70) C: 53.8% D: 50.0%
|
Median follow-up time: 27.7 months (IQR, 41.9-11.9 = 30 months).
At the time of the 4th DSMC interim analysis, 176 patients had 2-year follow-up data (arm A=87; arm B=89; 59% information).
Median duration of treatment, weeks: A: 8.9 (range, 1-86.9) B: 28.5 (range, 3.7-192.1).
Reasons for ending treatment Arm A: (n=130)
Arm B (n=132):
Arm C (n=26)
Arm D (n=46):
|
Death: A: n=38 B: n=62
2-Year OS (95% CI): A: 71.8% (62.5 to 79.1) B: 51.5% (41.7 to 60.4) P = .010, log-rank
Deaths at the 4th DSMC interim analysis: Arm A/C = 32 Arm B/D = 42
3-Year OS (95% CI): A: 66.2% (56.0 to 74.6) B: 42.8% (32.9 to 52.4)
OS in BRAFV600E: A: 71.4 (60.9 to 79.5) B: 43.9 (37.5 to 60.2) P=.020
OS in BRAFV600K A: 80.3 (50.1 to 93.2) B: 53.2 (32.9 to 69.9) P=.075
Median PFS, months (95% CI) A: 11.8 (5.9 to 33.5) B: 8.5 months (6.5 to 11.3) P = .054, log-rank.
2-Year PFS (95% CI): A: 41.9% (31.2 to 52.3) B: 19.2% (12.1 to 27.5)
Median PFS for step 2, months (95% CI): C: 9.9 (8.3 to 20.8) D: 2.9 (2.6 to 8.9)
ORR (95% CI): Step 1: A (n=113): 46.0% (36.6 to 55.6) B (n=114): 43.0% (33.8 to 52.6) Fisher’s exact test P value 5 .690 Step 2: C (n=23): 47.8% (26.8 to 69.4) D (n=29): 29.6% (12.7 to 47.2)
Grade ≥3 treatment related AEs: A: 59.5% B: 53.1% C: 53.8% D: 50.0% Treatment-related AEs on arms A and D were primarily immune-related and for arms B and C were primarily fevers, leukopenia, and hyponatremia.
Treatment related Deaths: A: n=2 (myocarditis and colitis) B: n=1 (cerebral vascular event) C: n=1 (thromboembolic event). |
The protocol-specified comparison of 2-year OS rates by the Mantel-Haenszel chi-square test did not cross the efficacy boundary (P = .163); however, 2-year OS rates from a Kaplan-Meier analysis indicated a significant difference and crossed the O’Brien Fleming boundary at 59% information time. Furthermore, the 95% repeated CI around the 2-year OS difference remained positive (95% CI, 2.6% to 37.9%). Therefore, the DSMC deemed this difference in OS to be clinically meaningful and recommended that the study be closed to accrual and patients currently on arm B be given the option to switch to arm D without the need for disease progression.
Authors conclusions: Combination nivolumab/ipilimumab followed by BRAF and MEK inhibitor therapy, if necessary, should be the preferred treatment sequence for a large majority of patients. |
|
Ascierto, 2023 IMspire150
NCT02908672. |
A multicentre, doubleblind, placebo-controlled, randomised, phase 3 study done in 108 academic and community hospitals in 20 countries.
Patient enrolment between: Jan 13, 2017, and April 26, 2018.
Funding and conflicts of interest:
Authors’ declaration of interests are provided at the end of the full text of this article. |
Main inclusion criteria
Main exclusion criteria:
therapies included concomitant use of approved or experimental cancer treatment, investigational therapy, prophylactic antiemetics, antidiarrhoea medication, haematopoietic growth factors, antiarrhythmic drugs, medications with a risk of torsades de pointes, and acetaminophen.
Additional details on in/exclusion criteria are available in the protocol (appendix).
Median age (range) A: 54.0 years (44.8-64.0 years) B: 53.5 years (43.0-63.8 years)
Male, n (%) A: B:
|
Atezolizumab group n= 256 (50%)
Cycle 1: All patients received oral cobimetinib 60 mg once daily plus oral vemurafenib 960 mg twice daily for 21 days then vemurafenib 720 mg twice daily (atezolizumab group) or 960 mg twice daily (control group) for 7 days.
From cycle 2 onwards atezolizumab was added: Patients received intravenous atezolizumab 840 mg (day 1 and 15), once-daily cobimetinib 60 mg (21 days on and 7 days off), and twice-daily vemurafenib 720 mg. |
Control group n= 258 (50%)
Cycle 1: All patients received oral cobimetinib 60 mg once daily plus oral vemurafenib 960 mg twice daily for 21 days then vemurafenib 720 mg twice daily (atezolizumab group) or 960 mg twice daily (control group) for 7 days.
From cycle 2 onwards placebo was added: Patients received intravenous placebo (day 1 and 15), once-daily cobimetinib 60 mg (21 days on and 7 days off), and twice-daily vemurafenib 960 mg. |
Median follow-up, months: I: 29·1 (IQR 10·1–45·4) C: 22·8 (10·6–44·1)
Continued treatment at time of analysis: I: 37 (14%) C: 37 (14%)
Median treatment, months: I: 9·2 (IQR 3·3–23·0) C: 8·9 (4·2–18·8)
Reasons for ending treatment I: Cycle 1: (256 received allocated intervention):
Cycle 2: (n=230 received allocated intervention)
C: Cycle 1 (n=255 received allocated intervention):
Cycle 2 (n=231received allocated intervention):
1 symptom deterioration |
Death: n=273 I: n=126 C: n=147
The secondary efficacy endpoint of overall survival was not met.
Median OS, months (95% CI): I: 39·0 (29·9–not estimable) C: 25·8 (22·0–34·6) HR 0·84 (95% CI 0·66–1·060, p=0·14.
Post-hoc landmark 12-month OS (95% CI): I: 76% (71–81) C: 76% (71–82)
Prespecified landmark OS at 24 months (95% CI): I: 62% (55–68) C: 53% (47–60)
With additional follow-up median PFS, months (95% CI): I: 15·1 (11·4–18·4) C: 10·6 (9·3–12·7) HR 0·79 (95% CI 0·64–0·97), p=0·022.
PFS events: I: n=168 C: 198
Median PFS, months (95% CI): I: 15·1 (11·4–18·4) C: 10·6 (9·3–12·7)
Post-hoc landmark PFS (95% CI): At 6 months: I: 73% (67–78) C: 74% (69–80); At 12 months: I: 54% (48–60) C: 46% (39–52); At 18 months: I: 44% (37–50) C: 32% (26–38)
ORR : I: 170/255 (67%; 61–72) C: 160/246 (65%; 59–71)
Serious AEs: I: 112 (48%)/231 C: 117 (42%)/280
Atezolizumab-associated SAEs: n=57 (25%)
Placebo associated SAEs: n=35 (13%)
Cobimetinib-associated SAEs: I: 53 (23%) C: 61 (22%)
Vemurafenib-related SAEs: I: 58 (25%) C: 71 (25%)
Grade 5 AEs I: n=8 (3%) C: n=6 (2%) For more information on AEs see results section of the article. |
compared with the control group (difference of ≥2%) had some AEs increased. For more information see the article.
|
Beoordelingsdatum en geldigheid
Publicatiedatum : 09-10-2025
Beoordeeld op geldigheid : 01-08-2025
De richtlijnmodules zijn geautoriseerd door de: Nederlandse Vereniging voor Heelkunde, Nederlandse Vereniging voor Dermatologie en Venereologie, Nederlandse Internisten Vereniging, Nederlandse Vereniging voor Nucleaire Geneeskunde, Nederlandse Vereniging voor Radiologie, Nederlandse Vereniging voor Pathologie, Verpleegkundigen & Verzorgenden Nederland.
Algemene gegevens
De ontwikkeling/herziening van deze richtlijnmodule werd ondersteund door het Kennisinstituut van de Federatie Medisch Specialisten (www.demedischspecialist.nl/kennisinstituut) en werd gefinancierd uit de Stichting Kwaliteitsgelden Medisch Specialisten (SKMS). De financier heeft geen enkele invloed gehad op de inhoud van de richtlijnmodule.
Samenstelling werkgroep
Voor het ontwikkelen van de richtlijnmodule is in 2017 een multidisciplinaire werkgroep ingesteld, bestaande uit vertegenwoordigers van alle relevante specialismen (zie hiervoor de Samenstelling van de werkgroep) die betrokken zijn bij de zorg voor patiënten met Melanoom.
Huidige samenstelling van de werkgroep:
- Dr. A.A.M. (Astrid) van der Veldt, voorzitter, internist-oncoloog, werkzaam in het Erasmus Medisch Centrum, NIV
- Dr. M.J.B. (Maureen) Aarts, internist-oncoloog, werkzaam in het Maastricht Universitair Medisch Centrum, NIV
- Prof. dr. A.J.M. (Fons) van den Eertwegh, internist-oncoloog, werkzaam in het Amsterdam Universitair Medisch Centrum, NIV
- Dr. M. (Hilde) Jalving, internist-oncoloog, werkzaam in het Universitair Medisch Centrum Groningen, NIV
- Dr. S. (Sofie) Wilgenhof, internist-oncoloog, werkzaam in het Antoni van Leeuwenhoek, NIV
- Dr. J.J. (Han) Bonenkamp, chirurgisch oncoloog, werkzaam in het Radboudumc, NVvH
- Dr. D.J. (Dirk) Grünhagen, chirurgisch oncoloog, werkzaam in het Erasmus Medisch Centrum, NVvH
- Dr. A.B. (Anne Brecht) Francken, chirurgisch oncoloog, werkzaam in het Isala, NVvH
- Dr. E.I. (Elsemieke) Plasmeijer, dermatoloog, werkzaam in het Antoni van Leeuwenhoek, NVDV
- Dr. R. (Remco) van Doorn, dermatoloog, werkzaam in het Leids Universitair Medisch Centrum, NVDV
- Dr. Q.G. (Quido) de Lussanet de la Sablonière, nucleair radioloog, werkzaam in het Erasmus Medisch Centrum, NVvR
- Dr. E.H.J.G. (Erik) Aarntzen, nucleair geneeskundige, werkzaam in het Universitair Medisch Centrum Groningen, NVNG
- Drs. B.A. (Beatrijs) Seinstra, radioloog, werkzaam in het Antoni van Leeuwenhoek, NVvR
- H.C. (Hanna) van der Pol, MSC, verpleegkundig specialist melanoom, werkzaam in het Antoni van Leeuwenhoek, V&VN
- Dr. T.P. (Thomas) Potjer, klinisch geneticus, werkzaam in het Leids Universitair Medisch Centrum, VKGN
- Dr. W.A.M. (Willeke) Blokx, patholoog, werkzaam in het Universitair Medisch Centrum Utrecht, NVVP
- Dr. A.M.L. (Anne) Jansen, klinisch moleculair bioloog in de pathologie, werkzaam in het Universitair Medisch Centrum Utrecht, NVVP
- K. (Koen) van Elst, Stichting Melanoom
Met speciale dank aan:
- Dr. B. Leeneman, Universitair docent, werkzaam in het Erasmus Medisch Centrum (bijdrage aan de doelmatigheidsmodule)
Met ondersteuning van:
- Dr. D. (Dagmar) Nieboer, senior adviseur, Kennisinstituut van de Federatie Medisch Specialisten
- Dr. R.J.S. (Rayna) Anijs, adviseur, Kennisinstituut van de Federatie Medisch Specialisten
- Dr. L. (Lisanne) Verbruggen, adviseur, Kennisinstituut van de Federatie Medisch Specialisten
- Drs. F. (Fieke) Pepping, junior adviseur, Kennisinstituut van de Federatie Medisch Specialisten
Voormalig betrokken werkgroepleden:
- Drs. J.G.M. (Anne) van den Hoek, radiotherapeut, werkzaam in het Universitair Medisch Centrum Groningen, NVRO (tot en met juni 2024)
- Drs. B. (Bernies) van der Hiel, nucleair geneeskundige, werkzaam in het werkzaam in het Antoni van Leeuwenhoek, NVNG (tot en met mei 2024)
- Drs. A. (Annemarie) Bruining, radioloog, werkzaam in het Nederlands Kanker Instituut, NVVR (tot en met mei 2024)
Belangenverklaringen
De Code ter voorkoming van oneigenlijke beïnvloeding door belangenverstrengeling is gevolgd. Alle werkgroepleden hebben schriftelijk verklaard of zij in de laatste drie jaar directe financiële belangen (betrekking bij een commercieel bedrijf, persoonlijke financiële belangen, onderzoeksfinanciering) of indirecte belangen (persoonlijke relaties, reputatiemanagement) hebben gehad. Gedurende de ontwikkeling of herziening van een module worden wijzigingen in belangen aan de voorzitter doorgegeven. De belangenverklaring wordt opnieuw bevestigd tijdens de commentaarfase.
Een overzicht van de belangen van werkgroepleden en het oordeel over het omgaan met eventuele belangen vindt u in onderstaande tabel. De ondertekende belangenverklaringen zijn op te vragen bij het secretariaat van het Kennisinstituut van de Federatie Medisch Specialisten.
|
Werkgroeplid |
Functie |
Nevenfuncties |
Gemelde belangen |
Ondernomen actie |
|
Veldt, van der (voorzitter) |
Internist-oncoloog, afdeling Interne Oncologie (0,8 fte) en afdeling Radiologie & Nucleaire Geneeskunde (0,2 fte) Erasmus MC, Rotterdam |
Adviesraden: BMS, MSD, Merck, Novartis, Pfizer, Eisai, Sanofi, Pierre-Fabre en Ipsen. Betaald aan het instituut (Erasmus MC) |
A.A.M. van der Veldt is principal investigator van meerdere studies van firma's (BMS, Exelexis, Novartis, Roche) en investigator-initiated studies (o.a. Safe Stop Trials) die financieel worden ondersteund door onder andere de zorgverzekeraars en non-profit organisaties Participatie aan NADINA-trial
|
Actie ondernomen bij modules over systemische behandelingen Zie hiervoor ‘Werkwijze en toelichting belangen richtlijn Melanoom’
|
|
Aarts |
Internist-oncoloog Maastricht Universitair Medisch Centrum |
* Bestuurslid WIN-O (Werkgroep Immunotherapie Nederland voor Oncologie) melanoom en nierkanker (onbetaald)
|
* Pfizer - Tyrosine Inhibitor effect op trombocyten - Co-promotor Participatie aan NADINA-trial
|
Actie ondernomen bij modules over systemische behandelingen Zie hiervoor ‘Werkwijze en toelichting belangen richtlijn Melanoom’
|
|
Eertwegh, van den |
Medisch-oncoloog, afdeling medische oncologie, Cancer Center Amsterdam, Amsterdam UMC, Vrije Universiteit Amsterdam Voorzitter DMTR (Dutch Melanoma Treatment registry)(vacatiegeld aan Amsterdam UMC)
|
Adviesraad (betaald aan Amsterdam UMC): Bristol-Myers Squibb, MSD Oncology, Ipsen, Pierre Fabre, Janssen Cilag BV |
* Sanofi - Prostaat studies (cabazipet en RECAB) - Projectleider * TEVA - prostaat studie (RECAB) - Projectleider * Bristol-Myers Squibb - Onco-kompas - Geen projectleider Huidig:" * Idera - INTRIM melanoom studie - Projectleider * Roche - REPOSIT melanoom studie - Projecteider * Novartis, Pierre Fabre, MSD, BMS en ziektekosten - DMTR - Geen projectleider
PI NADINA-trial |
Actie ondernomen bij modules over systemische behandelingen Zie hiervoor ‘Werkwijze en toelichting belangen richtlijn Melanoom’
|
|
Jalving |
Internist-oncoloog, UMCG Groningen |
Adviesraden: Bristol-Myers Squibb, AstraZenica,Pierre Fabre (betaald aan instituut (UMCG)
|
* KWF - TAMIC: Dichloroacetate in patients with metastatic melanoma prior to treatment with immune-checkpoint inhibition - Projectleider |
Actie ondernomen bij modules over systemische behandelingen Zie hiervoor ‘Werkwijze en toelichting belangen richtlijn Melanoom’
|
|
Wilgenhof |
Internist-oncoloog in het Antoni van Leeuwenziekenhuis |
Adviesraden: Eisai, Bristol-Myers Squibb, Pierre Fabre, Novartis, Pfizer en lpsen (betaald aan instituut (AVL)); educatief symposium: MSD en Bristol-Myers Squibb (betaald aan insituut (AVL)) |
* EU Horizon 2020 (no 875052) - CAPABLE: Pilot study of the eHealth application Cancer Patients Better Life Experience - Geen Projectleider * EU (101104801) - CARE-1: Optimizing Treatmensts for Metastatic Renal Carcinoma - Geen Projectleider
Studies: principal investigator: CA224020 studie (Clinicalîrials.gov number, NCT01968109) R3767-ONC-2011 studie (NCT05352672) E2139 (NCT05270044) vorinostat studie (NCT02836548)
subinvestigator: TIL studie (Clinicalîrials.gov number, NCT02278887) NADINA studie (Clinicalîrials.gov number, NCT04949113) safe stop studie safe stop ipi-nivo (NCT05652673) E1325 (NCT02362594) NIVEC studie (NCT04330430) NKTR-214 + nivolumab (NCT03635983) EBIN (NCT03235245) DONIMI (NCT04133948) MASTERKEY-115 (NCT04068181) IOB-013 (NCT05155254) |
Actie ondernomen bij modules over systemische behandelingen Zie hiervoor ‘Werkwijze en toelichting belangen richtlijn Melanoom’
|
|
Bonenkamp |
Chirurgisch oncoloog, RadboudUMC Nijmegen |
* Lid DB TFG Melanoom (onbetaald) |
Geen |
Geen actie |
|
Grünhagen |
Chirurg, Erasmus MC |
Lid bestuur WIN-O melanoom, onbetaald |
Deelname NADINA-trial |
Geen actie |
|
Francken |
Chirurgisch oncoloog, Isala |
* Voorzitter werkgroep audit NVvH |
Geen |
Geen actie |
|
Plasmeijer |
Dermatoloog, AVL |
* Bestuurslid Win-O: onbetaald |
Geen |
Geen actie |
|
Doorn, van |
Dermatoloog, Leids Universitair Medisch Centrum |
Onbetaald lid van enkele besturen (European Society for Dermatological Research, Nederlandse Vereniging voor Experimentele Dermatologie) |
Stichting KiKA - Therapeutic targeting of congenital melanocytic naevus and childhood melanoma using FOXO4 anti-senescence peptides - Projectleider |
Geen actie |
|
Lussanet de la Sablonière |
Nucleair- en Abdomen radioloog, Erasmus Medisch Centrum, Rotterdam |
Geen |
Geen |
Geen actie |
|
Aarntzen |
Nucleair geneeskundige, UMC Groningen (0,8 fte) * UMCGroningen, nucleair geneeskundige (0,8 fte) * Radboudumc, post-doc onderzoeker (0,1 fte) * Eberhard Karls University, Tuebingen, Duitsland (0,5 fte) |
* Post-doc onderzoeker Radboudumc, betaald (0,1 fte) * Post-doc onderzoeker Eberhard Karls University Tuebingen (Duitsland), betaald (0,05 fte)
|
* EU Innovatieve Health Initiative (IHI) - IMAGIO - IMAGING and advanced guidance for workflow optimalization in interventionaal oncology - Projectleider * Bergh in het Zadel/ Radboud oncologie Fonds - 'Breek de barrière: 'een nieuwe lokale en gerichte behandelmethode voor alvleesklierkanker' - Projectleider * ImaginAB Inc - iPREDICT, Trial: A phase IIB, Open Label, Study of 89Zr-crefmirlimab berdoxam PET/CT in Subjects with Selected Advanced or Metastetic Malignancies to ? - Geen projectleider site PI * Bergh in het Zadel/Radboud oncologie Fonds - Inzet AI voor betere overleving niet-kleincellig longkanker - Projectleider * ImaginAB Inc - (89Zr)Df-lAB22MC anti-CD8 minibody PET/CT-imaging to assess the in vivo distribution of CD8+ T-cells in COVID-19 patienst (NCT04874818) - Projectleider * KWF - Imaging tumor-infiltrating CD8+ T-cells in non-small cell lung cancer patienst upon neo-adjuvant treatment with Durvalumab - Projectleider |
Geen actie |
|
Seinstra |
Radioloog - Antoni van Leeuwenhoek Ziekenhuis |
Betrokken bij NADINA-trial als mede-auteur |
Geen |
Geen actie |
|
Van der Pol |
Verpleegkkundig specialist, Antoni van Leeuwenhoekziekenhuis, Amsterdam |
Geen |
Geen |
Geen actie |
|
Potjer |
Klinisch Geneticus, LUMC |
Cluster expertisegroep Maligniteiten van de huid |
Geen |
Geen actie |
|
Blokx |
Klinisch patholoog, UMC Utrecht |
Geen |
Geen |
Geen actie |
|
Jansen |
Klinisch Moleculair Bioloog in de Pathologie, UMC Utrecht |
Bestuurslid Stichting PALGA (vacatiegelden) |
Geen |
Geen actie |
|
Leeneman |
Universitair docent, Erasmus Universiteit Rotterdam |
Geen |
* ZIN - Ontwikkeling van ziektemodel voor melanoom - Geen projectleider * ZIN - Actualisatie van ziektemodel voor melanoom - Projectleider |
Geen actie |
|
Van der Elst |
Manager Quality and Improvement, Contour Advanced Systems B.V. |
Bestuurslid Stichting Melanoom |
Geen |
Geen actie |
Werkwijze en toelichting belangen richtlijn Melanoom
De Code ter voorkoming van oneigenlijke beïnvloeding door belangenverstrengeling is gevolgd. Alle werkgroepleden hebben schriftelijk verklaard of zij in de laatste drie jaar directe financiële belangen (betrekking bij een commercieel bedrijf, persoonlijke financiële belangen, onderzoeksfinanciering) of indirecte belangen (persoonlijke relaties, reputatiemanagement) hebben gehad. Gedurende de ontwikkeling of herziening van een module worden wijzigingen in belangen aan de voorzitter doorgegeven. De belangenverklaring wordt opnieuw bevestigd tijdens de commentaarfase.
De NIV heeft vastgesteld dat het niet mogelijk was werkgroepleden af te vaardigen met voldoende expertise zonder potentiële belangenverstrengeling. Het gaat daarbij met name om werkgroepleden die deelnemen aan adviesraden/kennisuitwisselingsbijeenkomsten met de farmaceutische industrie of deel nemen of hebben genomen als onderzoeker van een klinische studie. Gedurende de ontwikkeling van de [richtlijn / modules] heeft daarom afstemming plaatsgevonden tussen de werkgroepvoorzitter, de belangencommissie van het Kennisinstituut van de Federatie Medisch Specialisten en de NIV over passende acties naar aanleiding van de gemelde belangen.
Restricties voor de modules over onderwerpen (medicamenteuze behandeling) waar de adviesraden betrekking op hebben:
- Werkgroeplid werkt niet als enige inhoudsdeskundige aan de module;
- Werkgroeplid werkt tenminste samen met een werkgroeplid met een vergelijkbare expertise in alle fasen (zoeken, studieselectie, data-extractie, evidence synthese, Evidence-to-decision, aanbevelingen formuleren) van het ontwikkelproces. Indien nodig worden werkgroepleden toegevoegd aan de werkgroep;
- In alle fasen van het ontwikkelproces is een onafhankelijk methodoloog betrokken;
- Overwegingen en aanbevelingen worden besproken en vastgesteld tijdens een werkgroepvergadering onder leiding van een onafhankelijk voorzitter (zonder gemelde belangen).
Aansluitend op de reguliere commentaarronde bij de achterban van de bij de richtlijn betrokken wetenschappelijke verenigingen, hebben (een aantal) leden van richtlijn- en kwaliteitscommissie van de NIV en een methodoloog van het Kennisinstituut die niet betrokken waren bij ontwikkeling van de modules, aanvullend beoordeeld of de aanbevelingen logischerwijs aansluiten bij het gevonden bewijs en de overwegingen, om de onafhankelijkheid van de richtlijn te waarborgen.
Wellicht ten overvloede willen wij erop wijzen dat medisch specialistische richtlijnen niet worden vastgesteld door de betreffende richtlijnwerkgroep maar door de besturen/ledenvergadering van de betrokken verenigingen.
Een overzicht van de belangen van werkgroepleden en het oordeel over het omgaan met eventuele belangen vindt u in onderstaande tabel. De ondertekende belangenverklaringen zijn op te vragen bij het secretariaat van het Kennisinstituut van de Federatie Medisch Specialisten.
Inbreng patiëntenperspectief
Bij elke module is het patiëntperspectief meegenomen door de werkgroep. Er werd aandacht besteed aan het patiënten perspectief door een afgevaardigde van een patiëntenvereniging, de Stichting Melanoom, in de werkgroep te laten participeren.
NHG-standaard Verdachte huidafwijkingen
Voor huisartsen is de NHG-Standaard Verdachte huidafwijkingen leidend. Deze sluit aan op de richtlijn Melanoom.
Kwalitatieve raming van mogelijke financiële gevolgen in het kader van de Wkkgz
Bij de richtlijnmodule is conform de Wet kwaliteit, klachten en geschillen zorg (Wkkgz) een kwalitatieve raming uitgevoerd om te beoordelen of de aanbevelingen mogelijk leiden tot substantiële financiële gevolgen. Bij het uitvoeren van deze beoordeling is de richtlijnmodule op verschillende domeinen getoetst (zie het stroomschema op de Richtlijnendatabase).
|
Module |
Uitkomst raming |
Toelichting |
|
Systemische therapie - Eerstelijnsbehandeling BRAF-V600E/K gemuteerd irresectabel of gemetastaseerde stadium III/IV |
Geen substantiële financiële gevolgen |
Hoewel uit de toetsing volgt dat de aanbeveling(en) breed toepasbaar zijn (5.000-40.000 patiënten), volgt ook uit de toetsing dat het geen nieuwe manier van zorgverlening of andere organisatie van zorgverlening betreft. Er worden daarom geen substantiële financiële gevolgen verwacht. |
Werkwijze
AGREE
Deze richtlijnmodule is opgesteld conform de eisen vermeld in het rapport Medisch Specialistische Richtlijnen 2.0 van de adviescommissie Richtlijnen van de Raad Kwaliteit. Dit rapport is gebaseerd op het AGREE II instrument (Appraisal of Guidelines for Research & Evaluation II; Brouwers, 2010).
Knelpuntenanalyse en uitgangsvragen
De werkgroep beoordeelde de aanbeveling(en) uit de eerdere richtlijnmodules op noodzaak tot revisie.
Uitkomstmaten
Na het opstellen van de zoekvraag behorende bij de uitgangsvraag inventariseerde de werkgroep welke uitkomstmaten voor de patiënt relevant zijn, waarbij zowel naar gewenste als ongewenste effecten werd gekeken. Hierbij werd een maximum van acht uitkomstmaten gehanteerd. De werkgroep waardeerde deze uitkomstmaten volgens hun relatieve belang bij de besluitvorming rondom aanbevelingen, als cruciaal (kritiek voor de besluitvorming), belangrijk (maar niet cruciaal) en onbelangrijk. Tevens definieerde de werkgroep tenminste voor de cruciale uitkomstmaten welke verschillen zij klinisch (patiënt) relevant vonden.
Methode literatuursamenvatting
Een uitgebreide beschrijving van de strategie voor zoeken en selecteren van literatuur is te vinden onder ‘Zoeken en selecteren’ onder Onderbouwing. Indien mogelijk werd de data uit verschillende studies gepoold in een random-effects model. Review Manager 5.4 werd gebruikt voor de statistische analyses. De beoordeling van de kracht van het wetenschappelijke bewijs wordt hieronder toegelicht.
Beoordelen van de kracht van het wetenschappelijke bewijs
De kracht van het wetenschappelijke bewijs werd bepaald volgens de GRADE-methode. GRADE staat voor ‘Grading Recommendations Assessment, Development and Evaluation’ (zie http://www.gradeworkinggroup.org/). De basisprincipes van de GRADE-methodiek zijn: het benoemen en prioriteren van de klinisch (patiënt) relevante uitkomstmaten, een systematische review per uitkomstmaat, en een beoordeling van de bewijskracht per uitkomstmaat op basis van de acht GRADE-domeinen (domeinen voor downgraden: risk of bias, inconsistentie, indirectheid, imprecisie, en publicatiebias; domeinen voor upgraden: dosis-effect relatie, groot effect, en residuele plausibele confounding).
GRADE onderscheidt vier gradaties voor de kwaliteit van het wetenschappelijk bewijs: hoog, redelijk, laag en zeer laag. Deze gradaties verwijzen naar de mate van zekerheid die er bestaat over de literatuurconclusie, in het bijzonder de mate van zekerheid dat de literatuurconclusie de aanbeveling adequaat ondersteunt (Schünemann, 2013; Hultcrantz, 2017).
|
GRADE |
Definitie |
|
Hoog |
|
|
Redelijk |
|
|
Laag |
|
|
Zeer laag |
|
Bij het beoordelen (graderen) van de kracht van het wetenschappelijk bewijs in richtlijnen volgens de GRADE-methodiek spelen grenzen voor klinische besluitvorming een belangrijke rol (Hultcrantz, 2017). Dit zijn de grenzen die bij overschrijding aanleiding zouden geven tot een aanpassing van de aanbeveling. Om de grenzen voor klinische besluitvorming te bepalen moeten alle relevante uitkomstmaten en overwegingen worden meegewogen. De grenzen voor klinische besluitvorming zijn daarmee niet één op één vergelijkbaar met het minimaal klinisch relevant verschil (Minimal Clinically Important Difference, MCID). Met name in situaties waarin een interventie geen belangrijke nadelen heeft en de kosten relatief laag zijn, kan de grens voor klinische besluitvorming met betrekking tot de effectiviteit van de interventie bij een lagere waarde (dichter bij het nuleffect) liggen dan de MCID (Hultcrantz, 2017).
Overwegingen (van bewijs naar aanbeveling)
Om te komen tot een aanbeveling zijn naast (de kwaliteit van) het wetenschappelijke bewijs ook andere aspecten belangrijk en worden meegewogen, zoals aanvullende argumenten uit bijvoorbeeld de biomechanica of fysiologie, waarden en voorkeuren van patiënten, kosten (middelenbeslag), aanvaardbaarheid, haalbaarheid en implementatie. Deze aspecten zijn systematisch vermeld en beoordeeld (gewogen) onder het kopje ‘Overwegingen’ en kunnen (mede) gebaseerd zijn op expert opinion. Hierbij is gebruik gemaakt van een gestructureerd format gebaseerd op het evidence-to-decision framework van de internationale GRADE Working Group (Alonso-Coello, 2016a; Alonso-Coello 2016b). Dit evidence-to-decision framework is een integraal onderdeel van de GRADE methodiek.
Formuleren van aanbevelingen
De aanbevelingen geven antwoord op de uitgangsvraag en zijn gebaseerd op het beschikbare wetenschappelijke bewijs en de belangrijkste overwegingen, en een weging van de gunstige en ongunstige effecten van de relevante interventies. De kracht van het wetenschappelijk bewijs en het gewicht dat door de werkgroep wordt toegekend aan de overwegingen, bepalen samen de sterkte van de aanbeveling. Conform de GRADE-methodiek sluit een lage bewijskracht van conclusies in de systematische literatuuranalyse een sterke aanbeveling niet a priori uit, en zijn bij een hoge bewijskracht ook zwakke aanbevelingen mogelijk (Agoritsas, 2017; Neumann, 2016). De sterkte van de aanbeveling wordt altijd bepaald door weging van alle relevante argumenten tezamen. De werkgroep heeft bij elke aanbeveling opgenomen hoe zij tot de richting en sterkte van de aanbeveling zijn gekomen.
In de GRADE-methodiek wordt onderscheid gemaakt tussen sterke en zwakke (of conditionele) aanbevelingen. De sterkte van een aanbeveling verwijst naar de mate van zekerheid dat de voordelen van de interventie opwegen tegen de nadelen (of vice versa), gezien over het hele spectrum van patiënten waarvoor de aanbeveling is bedoeld. De sterkte van een aanbeveling heeft duidelijke implicaties voor patiënten, behandelaars en beleidsmakers (zie onderstaande tabel). Een aanbeveling is geen dictaat, zelfs een sterke aanbeveling gebaseerd op bewijs van hoge kwaliteit (GRADE gradering HOOG) zal niet altijd van toepassing zijn, onder alle mogelijke omstandigheden en voor elke individuele patiënt.
|
Implicaties van sterke en zwakke aanbevelingen voor verschillende richtlijngebruikers |
||
|
|
||
|
|
Sterke aanbeveling |
Zwakke (conditionele) aanbeveling |
|
Voor patiënten |
De meeste patiënten zouden de aanbevolen interventie of aanpak kiezen en slechts een klein aantal niet. |
Een aanzienlijk deel van de patiënten zouden de aanbevolen interventie of aanpak kiezen, maar veel patiënten ook niet. |
|
Voor behandelaars |
De meeste patiënten zouden de aanbevolen interventie of aanpak moeten ontvangen. |
Er zijn meerdere geschikte interventies of aanpakken. De patiënt moet worden ondersteund bij de keuze voor de interventie of aanpak die het beste aansluit bij zijn of haar waarden en voorkeuren. |
|
Voor beleidsmakers |
De aanbevolen interventie of aanpak kan worden gezien als standaardbeleid. |
Beleidsbepaling vereist uitvoerige discussie met betrokkenheid van veel stakeholders. Er is een grotere kans op lokale beleidsverschillen. |
Organisatie van zorg
In de knelpuntenanalyse en bij de ontwikkeling van de richtlijnmodule is expliciet aandacht geweest voor de organisatie van zorg: alle aspecten die randvoorwaardelijk zijn voor het verlenen van zorg (zoals coördinatie, communicatie, (financiële) middelen, mankracht en infrastructuur). Randvoorwaarden die relevant zijn voor het beantwoorden van deze specifieke uitgangsvraag zijn genoemd bij de overwegingen. Meer algemene, overkoepelende, of bijkomende aspecten van de organisatie van zorg worden behandeld in de module Organisatie van zorg.
Commentaar- en autorisatiefase
De conceptrichtlijnmodule werd aan de betrokken (wetenschappelijke) verenigingen en (patiënt) organisaties voorgelegd ter commentaar. De commentaren werden verzameld en besproken met de werkgroep. Naar aanleiding van de commentaren werd de conceptrichtlijnmodule aangepast en definitief vastgesteld door de werkgroep. De definitieve richtlijnmodule werd aan de deelnemende (wetenschappelijke) verenigingen en (patiënt) organisaties voorgelegd voor autorisatie en door hen geautoriseerd dan wel geaccordeerd.
Literatuur
Agoritsas T, Merglen A, Heen AF, Kristiansen A, Neumann I, Brito JP, Brignardello-Petersen R, Alexander PE, Rind DM, Vandvik PO, Guyatt GH. UpToDate adherence to GRADE criteria for strong recommendations: an analytical survey. BMJ Open. 2017 Nov 16;7(11):e018593. doi: 10.1136/bmjopen-2017-018593. PubMed PMID: 29150475; PubMed Central PMCID: PMC5701989.
Alonso-Coello P, Schünemann HJ, Moberg J, Brignardello-Petersen R, Akl EA, Davoli M, Treweek S, Mustafa RA, Rada G, Rosenbaum S, Morelli A, Guyatt GH, Oxman AD; GRADE Working Group. GRADE Evidence to Decision (EtD) frameworks: a systematic and transparent approach to making well informed healthcare choices. 1: Introduction. BMJ. 2016 Jun 28;353:i2016. doi: 10.1136/bmj.i2016. PubMed PMID: 27353417.
Alonso-Coello P, Oxman AD, Moberg J, Brignardello-Petersen R, Akl EA, Davoli M, Treweek S, Mustafa RA, Vandvik PO, Meerpohl J, Guyatt GH, Schünemann HJ; GRADE Working Group. GRADE Evidence to Decision (EtD) frameworks: a systematic and transparent approach to making well informed healthcare choices. 2: Clinical practice guidelines. BMJ. 2016 Jun 30;353:i2089. doi: 10.1136/bmj.i2089. PubMed PMID: 27365494.
Brouwers MC, Kho ME, Browman GP, Burgers JS, Cluzeau F, Feder G, Fervers B, Graham ID, Grimshaw J, Hanna SE, Littlejohns P, Makarski J, Zitzelsberger L; AGREE Next Steps Consortium. AGREE II: advancing guideline development, reporting and evaluation in health care. CMAJ. 2010 Dec 14;182(18):E839-42. doi: 10.1503/cmaj.090449. Epub 2010 Jul 5. Review. PubMed PMID: 20603348; PubMed Central PMCID: PMC3001530.
Hultcrantz M, Rind D, Akl EA, Treweek S, Mustafa RA, Iorio A, Alper BS, Meerpohl JJ, Murad MH, Ansari MT, Katikireddi SV, Östlund P, Tranæus S, Christensen R, Gartlehner G, Brozek J, Izcovich A, Schünemann H, Guyatt G. The GRADE Working Group clarifies the construct of certainty of evidence. J Clin Epidemiol. 2017 Jul;87:4-13. doi: 10.1016/j.jclinepi.2017.05.006. Epub 2017 May 18. PubMed PMID: 28529184; PubMed Central PMCID: PMC6542664.
Medisch Specialistische Richtlijnen 2.0 (2012). Adviescommissie Richtlijnen van de Raad Kwalitieit. http://richtlijnendatabase.nl/over_deze_site/over_richtlijnontwikkeling.html
Neumann I, Santesso N, Akl EA, Rind DM, Vandvik PO, Alonso-Coello P, Agoritsas T, Mustafa RA, Alexander PE, Schünemann H, Guyatt GH. A guide for health professionals to interpret and use recommendations in guidelines developed with the GRADE approach. J Clin Epidemiol. 2016 Apr;72:45-55. doi: 10.1016/j.jclinepi.2015.11.017. Epub 2016 Jan 6. Review. PubMed PMID: 26772609.
Schünemann H, Brożek J, Guyatt G, et al. GRADE handbook for grading quality of evidence and strength of recommendations. Updated October 2013. The GRADE Working Group, 2013. Available from http://gdt.guidelinedevelopment.org/central_prod/_design/client/handbook/handbook.html.
