Supplementen
Uitgangsvraag
Wat is de rol van supplementen en anti-oxidanten bij de behandeling van mannen met subfertiliteit?
Aanbeveling
Aanbeveling-1
Informeer patiënten met idiopathische mannelijke subfertiliteit over nadelige effecten van oxidatieve stress op semen parameters en fertiliteit. Bespreek desgevraagd dat het gebruik van supplementen mogelijk de semenkwaliteit kan verbeteren, maar dat er geen overtuigend bewijs is dat een specifiek supplement, dosering of gebruiksduur daadwerkelijk effectief is om de kans op zwangerschap te verhogen.
Aanbeveling-2
Wees terughoudend met het gebruik van supplement-doseringen boven de aanbevolen dagelijkse hoeveelheid of in onbekende combinaties.
Overwegingen
Balans tussen gewenste en ongewenste effecten
Het gebruik van supplementen om mannelijke subfertiliteit te herstellen is aantrekkelijk vanwege de eenvoud en het niet invasieve karakter van de interventie.
In de samengevoegde uitkomsten van geïncludeerde studies wordt een subtiele toename van levend geboren kinderen gezien bij gebruik van supplementen RR 1.39 (95% CI 1.02 tot 1.91). Deze bevinding wordt ondersteund door de resultaten van studies waar de kans op een doorgaande zwangerschap als uitkomst maat wordt gebruikt RR 1.47 (95% CI 1.23 tot 1.77). Tegengesteld hieraan is echter een waargenomen toename in de kans op een miskraam van 1.41 (95% CI 0.67 tot 2.96).
De geïncludeerde studies onderzoeken een diversiteit aan supplementen en doseringen waarbij veelvuldig verschillende supplementen worden gecombineerd, maar allen met als onderliggende hypothese dat zij de oxidatieve stress compenseren waardoor er een afname is in ROS en daarmee een toegenomen kans op zwangerschap.
In vrijwel alle studies worden semen parameters gebruikt als primaire uitkomstmaat. De individuele parameters; concentratie, volume, motiliteit en morfologie laten vrijwel uniform een tendens tot verbetering zien ten opzichte van de placebogroep.
Tenslotte wordt in verschillende studies gekeken naar directe of indirecte parameters van oxidatieve stress inclusief DNA-fragmentatie, meestal na een interventie periode van drie maanden. De uitkomsten zijn opnieuw uniform in een tendens tot verbetering tov placebo.
Er zijn geen gerandomiseerde studies die de meerwaarde van het gebruik van supplementen vergelijken met verandering van (andere) lifestyle factoren. Verschillende lifestyle factoren zijn bekende exogene veroorzakers van verhoogde ROS zoals alcoholconsumptie, roken, slechte voeding en obesitas waardoor het onduidelijk is wat de toegevoegde waarde van supplement gebruik is als aanvulling op verbetering van lifestyle.
Er zijn aanwijzingen dat het gebruik van supplementen niet geheel zonder risico’s is, een te hoge dosering kan in zeldzame gevallen leiden tot een supplement intoxicatie (Charen, 2020; Miller, 2005). Diverse voedingssupplementen voor mannelijke vruchtbaarheid, die zonder recept worden verkocht, bevatten doses die de aanbevolen dagelijkse dosering overschrijden (de Ligny, 2023).
Kwaliteit van bewijs
De kwaliteit van de evidence wordt geclassificeerd als laag tot zeer laag voor alle vergelijkingen op basis van het risico op bias. Verder komen de volgende zwaktes van vrijwel alle studies naar voren: de inclusie in veel studies is gebaseerd op een eenmalig afwijkende semen analyse. De follow up periode is in veel studies relatief kort. De onderzoekspopulatie is klein. De gewenste uitkomstmaat, de kans op een levend geboren kind, is slechts in een kleine groep van studies beschikbaar. In veel studies worden combinaties van supplementen gebruikt in wisselende dosering.
Door deze heterogeniteit is het vrijwel onmogelijk om een specifiek middel of preparaat te adviseren boven een ander.
Waarden en voorkeuren van patiënten (en eventueel hun partner)
Het gebruik van antioxidanten kan milde gastro-intestinale klachten geven in vergelijking met placebo. De drop out rate in studies varieerde waardoor hierover geen betrouwbaar oordeel kan worden gegeven.
Gewenste effecten
Er is geen onderzoek gedaan naar overige gewenste effecten behoudens de primaire uitkomst maten. Omdat een onvervulde kinderwens een grote invloed heeft op de kwaliteit van leven, en de vrouwelijke partner doorgaans aanzienlijk ongemak ervaart bij ART, kan zelfs een beperkte verbetering in de uitkomst, zeker wanneer de mannelijke partner hieraan kan bijdragen, van groot belang zijn voor het paar.
Kostenaspecten:
Er zijn geen kosten-effectiviteitsstudies verricht. De kosten van middelen met antioxidanten variëren. Op basis van de geanalyseerde studies is geen voorkeur aan te wijzen richting een bepaald preparaat of samenstelling. Hoewel de kosten per individu beperkt zijn, zou een standaard advies om supplementen te gaan gebruiken, door de omvang van de populatie het cumulatief hoge kosten met zich meebrengen, vooralsnog zonder duidelijk klinisch voordeel.
Aanvaardbaarheid
De interventie lijkt aanvaardbaar voor de betrokkenen. Er zijn geen ethische bezwaren.
Duurzaamheid
Afhankelijk van het formaat van de implementatie zal dit een meer of minder grote impact hebben op het milieu. Of dit vergeleken met het niet gebruik ook een toename is ten opzichte van het niet gebruiken van supplementen is niet onderzocht.
Rationale van aanbeveling-1: weging van argumenten voor en tegen de interventies
De aanbeveling is gebaseerd op studies met een lage kwaliteit van bewijs. Vrijwel geen van de studies had de impact op zwangerschapskansen als uitkomst parameter. Met zeer laag tot laag niveau van zekerheid lijkt het dat (combinaties van) supplementen oxidatieve stress verminderen, met kleine, klinisch beperkte verbeteringen in semenparameters. Er is geen overtuigend bewijs dat een specifiek supplement, dosering of gebruiksduur ondersteunt.
Eindoordeel:
Zwakke aanbeveling neutraal voor het gebruik van supplementen en antioxidanten.
Rationale van aanbeveling-2: weging van argumenten voor en tegen de interventies
Er zijn geen dose-finding studies gevonden die een maximaal veilige of effectieve dosering aantonen van supplementen bij mannen met subfertiliteit. Er zijn echter wel aanwijzingen dat er mogelijk negatieve gezondheidseffecten kunnen zijn bij overmatig gebruik. Omdat het om middelen gaat waar geen voorschrift voor nodig is, bestaat het riscio op overdosering en is gekozen voor een zwakke aanbeveling tegen het gebruik van hoog gedoseerde supplementen.
Eindoordeel:
Zwakke aanbeveling tegen het gebruik van hoog gedoseerde supplementen.
Onderbouwing
Oxidative stress (OS) has found to play a role in 25% to 87% of male factor subfertility. OS negatively affects all sperm functions through reduced sperm DNA integrity and thereby hampers the fertilization process and reproductive outcomes. One hypothesis is that an increase in reactive oxygen species (ROS) and nitrogen species, alter the balance of the redox status of both the steroidogenic and germline cell populations, leading to a dysregulation of the hypothalamic-pituitary-testicular axis and a reduction in sperm quality. Supplements may positively influence these conditions by inhibiting or delaying the oxidation of biologically relevant molecules, either by directly scavenging free radicals or by chelation of redox metals. There is some evidence that dietary supplements can improve male fertility, but the results of the studies are inconsistent. The results of these studies often concern sperm quality or specific aspects of sperm quality and not the chance of pregnancy or a healthy child. In practice, there are many supplements available, the substantiation and effectiveness of which are not always clear. Also, deficiencies are not routinely tested.
Summary of Findings table: Supplements as treatment for male subfertility?
Population: Men with subfertility
Intervention: Supplements
Comparison: No supplements or placebo
|
Outcome |
Study results and measurements |
Absolute effect estimates |
Certainty of the Evidence (Quality of evidence) |
Summary |
||
|
Placebo |
Supplement |
|||||
|
Live birth rate (critical) |
Risk ratio: 1.39 (95% CI 1.02 to 1.91)
Based on data of 1546 participants in 13 studies |
142 per 1000 |
197 per 1000 |
Very low Due to serious risk of bias, serious imprecision and suspicion of publication bias1 |
The evidence is very uncertain about the effect of supplements on live birth rate when compared with no supplements or placebo in men with subfertility. A |
|
|
Difference: 55 more per 1000 (95%CI 3 more - 129 more) |
||||||
|
Pregnancy rate, clinical (critical) |
Risk ratio: 1.47 (95% CI 1.23 to 1.77)
Based on data of 2140 participants in 23 studies |
143 per 1000 |
210 per 1000 |
Low Due to serious risk of bias and serious inconsistency2 |
Supplements may increase pregnancy rate when compared with no supplements or placebo in men with subfertility. B |
|
|
Difference: 67 more per 1000 (95%CI 33 more - 110 more) |
||||||
|
Miscarriage rate (important) |
Risk ratio: 1.41 (95% CI 0.67 to 2.96)
Based on data of 772 participants in 7 studies |
44 per 1000 |
62 per 1000 |
Very low Due to serious risk of bias and very serious imprecision3 |
The evidence is very uncertain about the effect of supplements on miscarriage rate when compared with no supplements or placebo in men with subfertility. C |
|
|
Difference: 18 more per 1000 (95%CI 15 fewer - 86 more) |
||||||
|
Semen para-meters (important) |
Concentration (mln/mL) |
Higher is better Follow-up 3 months
Based on data of 2974 participants in 41 studies |
Mean difference: 5.50 mln/mL higher (95% CI 3.12 higher - 7.88 higher) |
Low Due to serious risk of bias and serious inconsistency4 |
Supplements may result in a higher semen concentration when compared with no supplements or placebo in men with subfertility. D |
|
|
Total motility (%) |
Higher is better Follow-up 3 months
Based on data of 2303 participants in 31 studies |
Mean difference: 8.10% higher (95% CI 3.08 higher - 13.11 higher) |
Low Due to serious risk of bias and serious inconsistency5 |
Supplements may result in a higher total motility when compared with no supplements or placebo in men with subfertility. E |
||
|
Progressive motility (%) |
Higher is better Follow-up 3 months
Based on data of 2466 participants in 32 studies |
Mean difference: 6.96% higher (95% CI 3.89 higher - 10.02 higher) |
Low Due to serious risk of bias and serious inconsistency6 |
Supplements may result in a higher progressive motility when compared with no supplements or placebo in men with subfertility. F |
||
|
Normal morphology (%) |
Higher is better Follow-up 3 months
Based on data of 822 participants in 9 studies |
Mean difference: 0.58% higher (95% CI 0.03 higher - 1.13 higher) |
Low Due to serious risk of bias and serious inconsistency7 |
Supplements may result in little to no difference in percentage of normal semen morphology when compared with no supplements or placebo in men with subfertility. G |
||
|
Volume (mL) |
High is better Follow-up 3 months
Based on data of 816 participants in 7 studies |
Mean difference: 0.41mL higher (95% CI 0.13 lower - 0.95 higher) |
Low Due to serious risk of bias and serious inconsistency8 |
Supplements may result in little to no difference in semen volume when compared with no supplements or placebo in men with subfertility. H |
||
|
Total fertilization failure (important) |
Based on data of 0 participants in 0 studies |
- |
No GRADE |
No evidence was found regarding the effect of supplements on total fertilization failure when compared with no supplements or placebo in men with subfertility. |
||
|
DNA fragmentation (important) |
Lower is better Follow-up 3 months
Based on data of 1792 participants in 18 studies |
Standardized mean difference: 0.29% lower (95% CI 0.54 lower - 0.04% lower) |
Low Due to serious risk of bias and serious inconsistency9 |
Supplements may result in little to no difference in DNA fragmentation when compared with no supplements or placebo in men with subfertility. I |
||
|
Parameters indicating oxidative stress in a semen sample (important) |
Lower is better Follow-up 3 months
Based on data of 338 participants in 6 studies |
Results are presented in various different ways. |
Very low Due to serious risk of bias and very serious inconsistency 10 |
The evidence is very uncertain about the effect of supplements on oxidative stress when compared with no supplements or placebo in men with subfertility. J |
||
1. Risk of bias: serious. Due to multiple limitations in study design (scored by the authors of the Cochrane review).
Imprecision: serious. Due to overlap of the upper limit of the 95% confidence interval with the minimal clinically important difference.
Publication bias: strong suspicion based on funnel plot (scored by the authors of the Cochrane review).
2. Risk of bias: serious. Due to multiple limitations in study design (scored by the authors of the Cochrane review).
Inconsistency: serious. Due to conflicting results.
3. Risk of bias: serious. Due to multiple limitations in study design (scored by the authors of the Cochrane review).
Imprecision: very serious. Due to low number of events and overlap of both limits of the 95% confidence interval with the minimal clinically important difference.
4. Risk of bias: serious. Due to multiple limitations in study design.
Inconsistency: serious. Due to conflicting results.
5. Risk of bias: serious. Due to multiple limitations in study design.
Inconsistency: serious. Due to conflicting results.
6. Risk of bias: serious. Due to multiple limitations in study design.
Inconsistency: serious. Due to conflicting results.
7. Risk of bias: serious. Due to multiple limitations in study design.
Inconsistency: serious. Due to conflicting results.
8. Risk of bias: serious. Due to multiple limitations in study design.
Inconsistency: serious. Due to conflicting results.
9. Risk of bias: serious. Due to multiple limitations in study design.
Inconsistency: serious. Due to conflicting results.
10. Risk of bias: serious. Due to multiple limitations in study design.
Inconsistency: very serious. Due to conflicting results and many ways of measurements.
References:
A. Balercia 2005, Balercia 2009, Blomberg Jensen 2018, Gamidov 2019, Joseph 2020, Kessopoulou 1995, Korshunov 2018, Kumalic 2020, Lahimer 2023, Omu 1998, Steiner 2020, Suleiman 1996, Tremellen 2007.
B. Attallah 2013, Azizollahi 2013, Balercia 2005, Balercia 2009, Barekat 2016, Busetto 2018, D’argent 2021, Gamidov 2019, Habibi 2022, Joseph 2020, Kessopoulou 1995, Kizilay 2019, Kopets 2020, Korshunov 2018, Kumalic 2020, Lahimer 2023, Omu 1998, Popova 2019, Steiner 2020, Suleiman 1996, Tsounapi 2018, Tremellen 2007, Zavaczki 2003.
C. D’argent 2021, Joseph 2020, Korshunov 2018, Omu 1998, Steiner 2020, Suleiman 1996, Tremellen 2007.
D. Abbasi 2020, Abbasi 2021, Amini 2020, Attallah 2013, Azizollahi 2013, Bahmyari 2021, Balercia 2005, Barekat 2016, Boonyarangkul 2015, Cavallini 2004, Conquer 2000, Cyrus 2015, D’argent 2021, Dadgar 2023, Dimitriadis 2010, Ener 2016, Gamidov 2017, Gamidov 2019, Gonzalez-Ravina 2018, Gopinath 2013, Greco 2005, Habibi 2022, Haghighian 2015, Helli 2023, Joseph 2020, Kopets 2020, Kumalic 2020, Martinez-Soto 2010, Mehni 2014, Morgante 2010, Nadjarzadeh 2011, Nouri 2019, Peivandi 2010, Popova 2019, Raigani 2014, Rolf 1999, Sabeti 2021, Scott 1998, Steiner 2020, Stenqvist 2018, Zavaczki 2003.
E. Abbasi 2020, Abbasi 2021, Attallah 2013, Azizollahi 2013, Bahmyari 2021, Balercia 2005, Barekat 2016, Conquer 2000, D’argent 2021, Dimitriadis 2010, Ener 2016, Gopinath 2013, Greco 2005, Habibi 2022, Hodeeb 2023, Kumalic 2020, Lenzi 2003, Martinez-Soto 2010, Morgante 2010, Nadjarzadeh 2011, Nouri 2019, Omu 2008, Patki 2023, Peivandi 2010, Raigani 2014, Sabeti 2021, Scott 1998, Sigman 2006, Steiner 2020, Stenqvist 2018, Zavaczki 2003.
F. Abbasi 2020, Amini 2020, Attallah 2013, Azizollahi 2013, Bahmyari 2021, Boonyarangkul 2015, Cavallini 2004, Cyrus 2015, D’argent 2021, Dadgar 2023, Dawson 1990, Gamidov 2017, Gamidov 2019, Gonzalez-Ravina 2018, Habibi 2022, Haghighian 2015, Hajb 2023, Hodeeb 2023, Joseph 2020, Kopets 2020, Kumalic 2020, Martinez-Soto 2010, Mehni 2014, Micic 2019, Morgante 2010, Nadjarzadeh 2011, Nouri 2019, Peivandi 2010, Popova 2019, Rolf 1999, Stenqvist 2018
G. Abbasi 2021, D’argent 2021, D’argent 2023, Habibi 2022, Hajb 2023, Helli 2023, Hodeeb 2023, Patki 2023, Sabeti 2021.
H. Abbasi 2021, Habibi 2022, Helli 2023, Hodeeb 2023, Lahimer 2023, Patki 2023, Sabeti 2021.
I. Abbasi 2020, Abbasi 2021, Barekat 2016, D’argent 2021, Dadgar 2023, Gamidov 2017, Gonzalez-Ravina 2018, Greco 2005, Habibi 2022, Kumalic 2020, Lahimer 2023, Martinez-Soto 2010, Micic 2019, Patki 2023, Raigani 2014, Steiner 2020, Stenqvist 2018.
J. Abbasi 2021, Dadgar 2023, Haidari 2021, Helli 2023, Hodeeb 2023, Sabeti 2021.
The selected Cochrane review with meta-analysis (de Ligny, 2022) included 90 studies covering data of 10,303 participants. Inclusion criteria of the review were (P) subfertile men of a couple referred to a fertility clinic, (I) any type or dose of oral antioxidant supplement that can be obtained without prescription and is not regulated as a pharmaceutical drug, (C) placebo, no treatment, another type or dose of oral antioxidant, (O), live birth rate, clinical pregnancy rate, adverse events, total sperm motility, progressive sperm motility, sperm concentration, and (design) RCTs or cross-over trials (then only the first-phase data were included). The systematic review included studies from inception up until February 2021. Important study characteristics of the 90 studies included in the Cochrane review can be found in the review itself (from page 82 and onwards).
In the review, all participants were aged 18 to 65 years and most had either oligospermia or asthenospermia. Five studies included men with an increased level of DNA fragmentation or oxidative stress. In two studies men with a varicocele were enrolled, 10 studies enrolled men post-varicocelectomy and one study enrolled men with chronic prostatitis. Various kinds and combinations of antioxidants were used in the studies. Twelve studies compared one antioxidant with another antioxidant and were thus excluded from this summary of literature. Funding sources were stated by 36 studies. Eight of these studies stated that funding was from a commercial source, and the remaining 28 obtained funding through non-commercial avenues or university grants. Nine studies specifically reported no funding. Forty-five studies did not mention any funding sources.
The other 13 studies were randomized controlled trials (RCTs) published after the search data of the Cochrane review. In all studies, adult men with sub-/infertility referred to a fertility clinic were included. Eleven studies investigated any kind of antioxidant and two a probiotic supplement (Abbasi, 2021; Helli, 2022). All studies were placebo controlled and investigated pregnancy outcomes, semen outcomes and/or parameters indicating oxidative stress. Two out of 13 RCTs were sponsored by industry.
A wide variety of antioxidants were used in the included studies: arginine, carnitines (L-carnitine, L-acetyl carnitine, L-carnitine plus L-acetyl carnitine),carotenoids (β-carotene), coenzyme Q10 (CoQ10), carob seed powder, cysteines(ethylcysteine and N-acetylcysteine (NAC)), folic acid, maca (Lepidium meyenii Walpers), magnesium, melatonin, pollen extract, polyunsaturated fatty acids (PUFAs) (alpha-lipoic-acidand docosahexaenoic acid (DHA)), probiotics (Lactobacillus casei, Lactobacillus rhamnosus, Lactobacillus bulgaricus, Lactobacillus acidophilus, Bifidobacterium breve, Bifidobacterium longum, fructooligosaccharides and Streptococcus thermophiles), resveratrol, selenium, vitamin B,vitamin C, vitamin D with calcium, vitamin E and zinc. Combined antioxidants were used in 28 individual studies.
Results
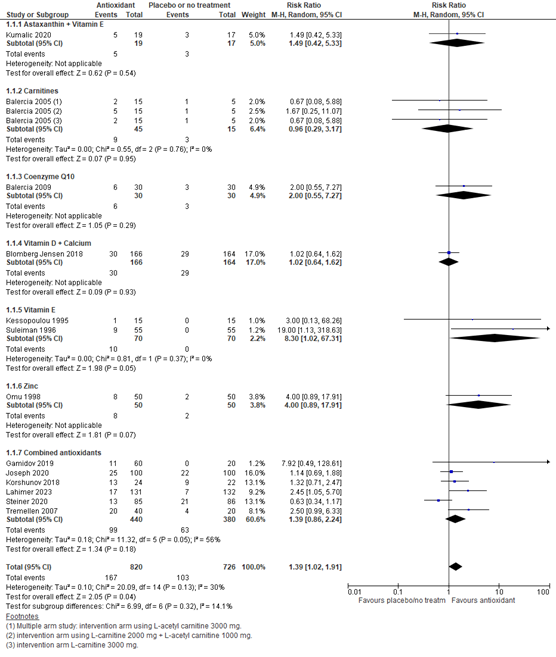
1. Live birth rate (critical)
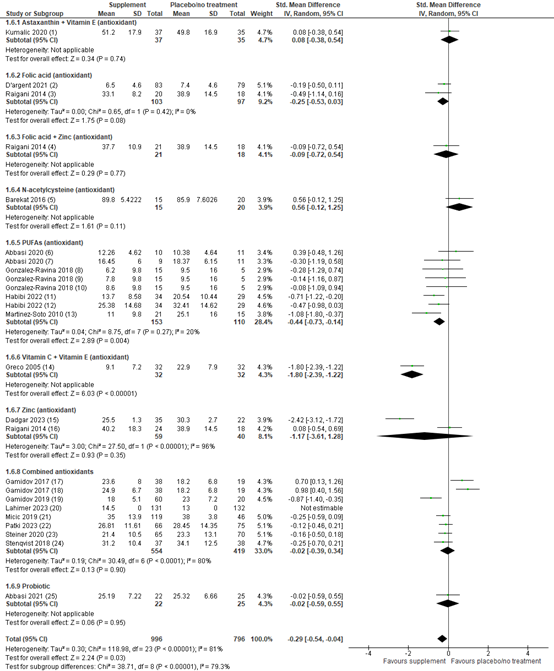
Live birth was reported in 13 studies with a total of 1546 participants. In all studies, the investigated supplement was an antioxidant. Overall, the risk ratio for live birth with antioxidants or placebo was 1.39 (95% CI 1.02 to 1.91), in favor of antioxidants (figure 1). This result is considered clinically relevant.
A sensitivity analysis for the different kinds of antioxidants was performed (figure 1). For astaxanthin plus vitamin E, the risk ratio was 1.49 (95% CI 0.42 to 5.33) in favor of the antioxidant, although based on one study. For carnitines, the risk ratio was 0.96 (95% CI 0.29 to 3.17) in favor of the antioxidant, although based on one study. For coenzyme Q10, the risk ratio was 2.00 (95% CI 0.55 to 7.27) in favor of the antioxidant, although based on one study. For vitamin D plus calcium, the risk ratio was 1.02 (95% CI 0.64 to 1.62) in favor of the antioxidant, although based on one study. For vitamin E, the risk ratio was 8.30 (95% CI 1.02 to 67.31) in favor of the antioxidant. For zinc, the risk ratio was 4.00 (95% CI 0.89 to 17.91) in favor of the antioxidant, although based on one study. For combined antioxidants, the risk ratio was 1.39 (95% CI 0.86 to 2.24) in favor of the antioxidant.
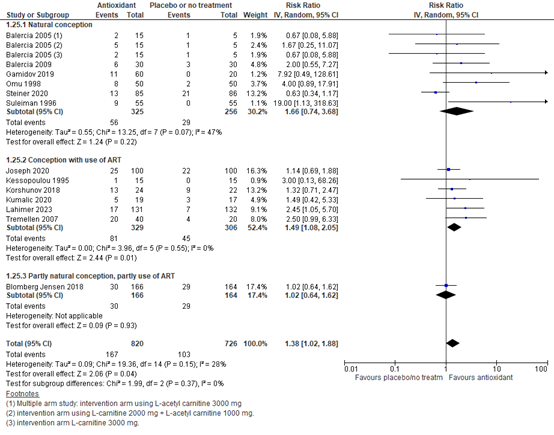
Another sensitivity analysis was performed for natural conception versus conception with the use of artificial reproduction techniques (e.g., IUI, IVF and/or ICSI) (figure 2). The risk ratio for natural conception with antioxidants versus placebo or no treatment was 1.66 (95% CI 0.74 to 3.68) in favor of antioxidants. For conception with the use of ART, the risk ratio was 1.49 (95% CI 1.08 to 2.05), in favor of antioxidants. These are both considered clinically relevant.
Figure 1. Forest plot of live birth comparing the use of antioxidants with placebo or no treatment with a sensitivity analysis for type of antioxidant
(1) Multiple arm study: intervention arm using L-acetyl carnitine 3000 mg. (2) intervention arm using L-carnitine 2000 mg + L-acetyl carnitine 1000 mg. (3) intervention arm L-carnitine 3000 mg
Figure 2. Forest plot of live birth comparing the use of antioxidants with placebo or no treatment with a sensitivity analysis for method of conception
(1) Multiple arm study: intervention arm using L-acetyl carnitine 3000 mg. (2) intervention arm using L-carnitine 2000 mg + L-acetyl carnitine 1000 mg. (3) intervention arm L-carnitine 3000 mg
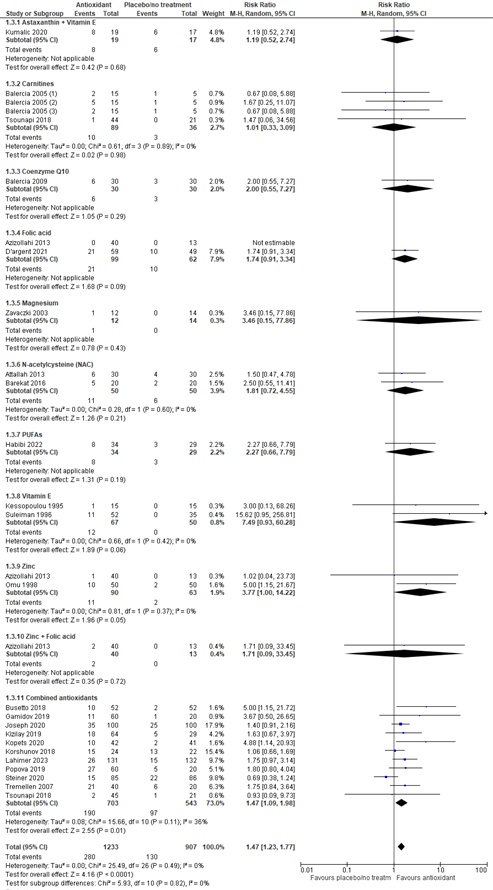
2. Clinical pregnancy rate (critical)
Pregnancy rate was reported in 23 studies covering data of 2140 participants. In all these studies pregnancy was defined as a pregnancy confirmed by ultrasound. The investigated supplement was an antioxidant in all studies. The risk ratio for pregnancy was 1.47 (95% CI 1.23 to 1.77), favoring antioxidants (figure 3). This finding is considered clinically relevant.
A sensitivity analysis for the different kinds of antioxidants was performed (figure 3). For astaxanthin plus vitamin E, the risk ratio was 1.19 (95% CI 0.52 to 2.74) in favor of the antioxidant, although based on one study. For carnitines, the risk ratio was 1.01 (95% CI 0.33 to 3.09) in favor of the antioxidant. For coenzyme Q10, the risk ratio was 2.00 (95% CI 0.55 to 7.27) in favor of the antioxidant, although based on one study. For folic acid, the risk ratio was 1.74 (95% CI 0.91 to 3.34) in favor of the antioxidant. For magnesium, the risk ratio was 3.46 (95% CI 0.15 to 77.86) in favor of the antioxidant, although based on one study. For N-acetylcysteine, the risk ratio was 1.81 (95% CI 0.72 to 4.55) in favor of the antioxidant. For polyunsaturated fatty acids (i.e., alpha lipoic acid), the risk ratio was 2.72 (95% CI 0.66 to 7.79) in favor of the antioxidant, although based on one study. For vitamin E, the risk ratio was 7.49 (95% CI 0.93 to 60.28) in favor of the antioxidant. For zinc, the risk ratio was 3.77 (95% CI 1.00 to 14.22) in favor of the antioxidant. For zinc plus folic acid, the risk ratio was 1.71 (95% CI 0.09 to 33.45) in favor of the antioxidant, although based on one study. For combined antioxidants, the risk ratio was 1.47 (95% CI 1.09 to 1.98) in favor of the antioxidant.
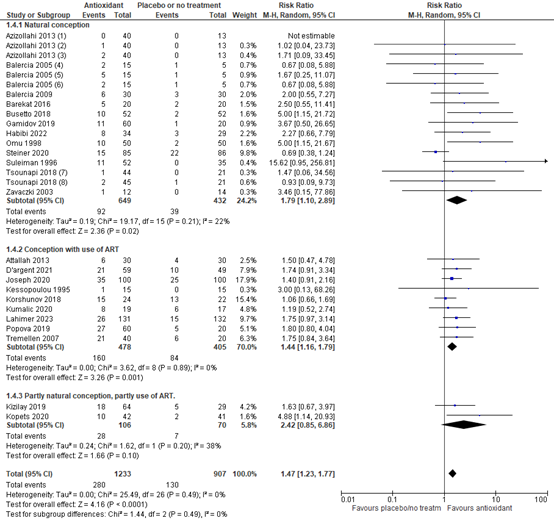
Another sensitivity analysis was performed for natural conception versus conception with the use of artificial reproduction techniques (e.g., IUI, IVF and/or ICSI) (figure 4). The risk ratio for natural conception with antioxidants versus placebo or no treatment was 1.79 (95% CI 1.10 to 2.89) in favor of antioxidants. For conception with the use of ART, the risk ratio was 1.44 (95% CI 1.16 to 1.79), in favor of antioxidants. These are both considered clinically relevant.
Figure 3. Forest plot of clinical pregnancy rate comparing the use of antioxidants with placebo or no treatment with a sensitivity analysis for type of antioxidant
(1) Multiple arm study: intervention arm using L-acetyl carnitine 3000 mg. (2) intervention arm using L-carnitine 2000 mg + L-acetyl carnitine 1000 mg. (3) intervention arm L-carnitine 3000 mg
Figure 4. Forest plot of clinical pregnancy rate comparing the use of antioxidants with placebo or no treatment with a sensitivity analysis for method of conception
(1) Multiple arm study: intervention arm using folic acid 5mg. (2) intervention arm using zinc 66mg. (3) intervention arm folic acid 5mg + zinc 66mg. (4) Multiple arm study: intervention arm using L-acetyl carnitine 3000 mg. (5) intervention arm using L-carnitine 2000 mg + L-acetyl carnitine 1000 mg. (6) intervention arm L-carnitine 3000 mg. (7) Multiple arm study: intervention arm using L-carnitine 1000mg. (8) intervention arm using Profertil
3. Miscarriage rate (important)
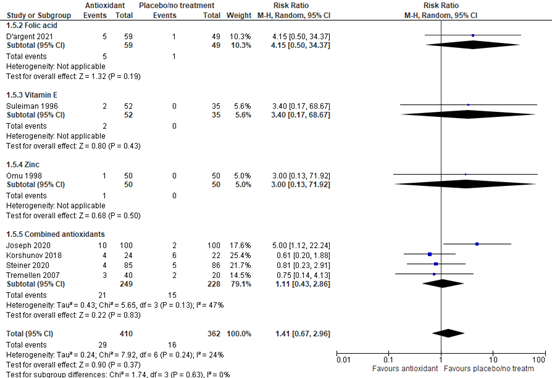
The rate of miscarriage was reported in seven studies with data of 772 participants. The investigated supplement was an antioxidant in all studies. Overall, the risk ratio for a miscarriage was 1.41 (95% CI 0.67 to 2.96) in favor of placebo (figure 5).
A sensitivity analysis for the different kinds of antioxidants was performed (figure 5). For folic acid, the risk ratio was 4.15 (95% CI 0.50 to 34.37) in favor of placebo, although based on one study. For vitamin E, the risk ratio was 3.40 (95% CI 0.17 to 68.67) in favor of placebo, although based on one study. For zinc, the risk ratio was 3.00 (95% CI 0.13 to 71.92) in favor of placebo, although based on one study. For combined antioxidants, the risk ratio was 1.11 (95% CI 0.43 to 2.86) in favor of placebo.
Figure 5. Forest plot of miscarriage rate comparing the use of antioxidants with placebo or no treatment with a sensitivity analysis for type of antioxidant
4. Total fertilization failure (important)
Total fertilization failure was not reported in any of the studies.
5. Semen parameters (important)
A variety of semen parameters were reported: semen concentration, semen count, total motility, progressive motility, percentage (ab)normal morphology, vitality and semen volume. The outcomes of concentration, total motility, progressive motility, morphology and volume are reported below.
5.1 Concentration
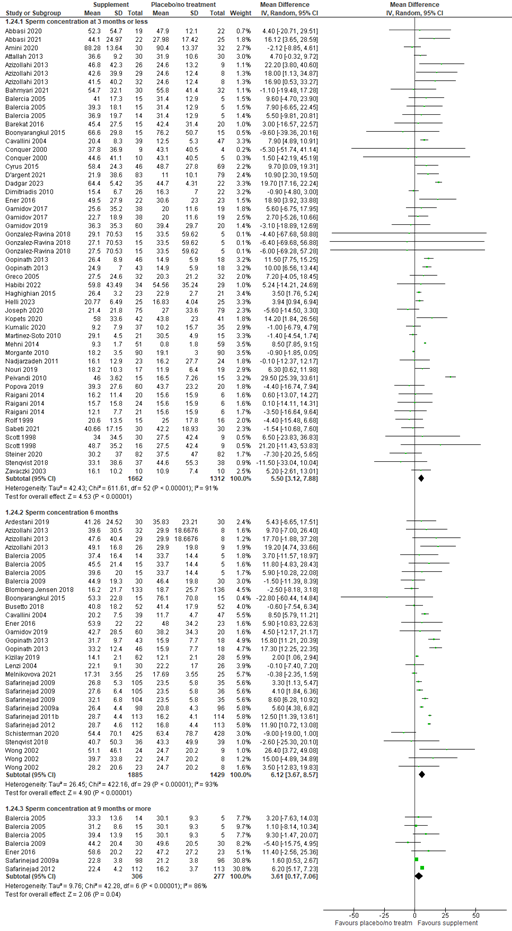
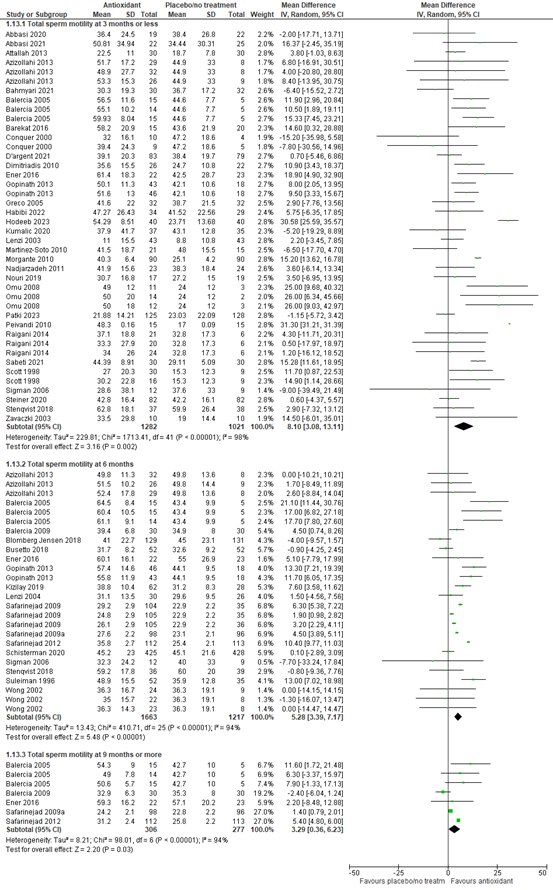
Sperm concentration (million/mL) was reported in 54 studies. Sperm concentration was measured at different timepoints after start of the intervention across the studies. At three months or less after start of the intervention, data of 41 studies with 2974 participants were available. The mean difference in sperm concentration between participants using supplements versus participants using placebo or no treatment was 5.50 million/ml (95% CI 3.12 to 7.88 million/ml), in favor of supplements. At six months, 21 studies with a total of 3314 participants could be included in the analysis. The mean difference in sperm concentration was 6.12 million/ml (95% CI 3.67 to 8.57), in favor of supplements. At nine months or more, five studies with 583 participants were included. The mean difference in sperm concentration was 3.61 million/ml (95% CI 0.17 to 7.06), in favor of supplements (figure 6).
Figure 6. Forest plot of semen concentration (million per milliliter) comparing the use of supplements with placebo or no treatment
Analysis split on time of measurement
5.2 Total motility
Total sperm motility (%) was reported in 42 studies. Total motility was measured at different timepoints after start of the intervention across the studies. At three months or less after start of the intervention, data of 31 studies with 2303 participants were available. The mean difference in total motility between participants using supplements versus participants using placebo or no treatment was 8.10% (95% CI 3.08 to 13.11), in favor of supplements. At six months, 17 studies with a total of 2880 participants could be included in the analysis. The mean difference in total motility was 5.28% (95% CI 3.39 to 7.17), in favor of supplements. At nine months or more, five studies with 583 participants were included. The mean difference in total motility was 3.29% (95% CI 0.36 to 6.23), in favor of supplements (figure 7).
Figure 7. Forest plot of total sperm motility (%) comparing the use of supplements with placebo or no treatment
Analysis split on time of measurement
5.3 Progressive motility
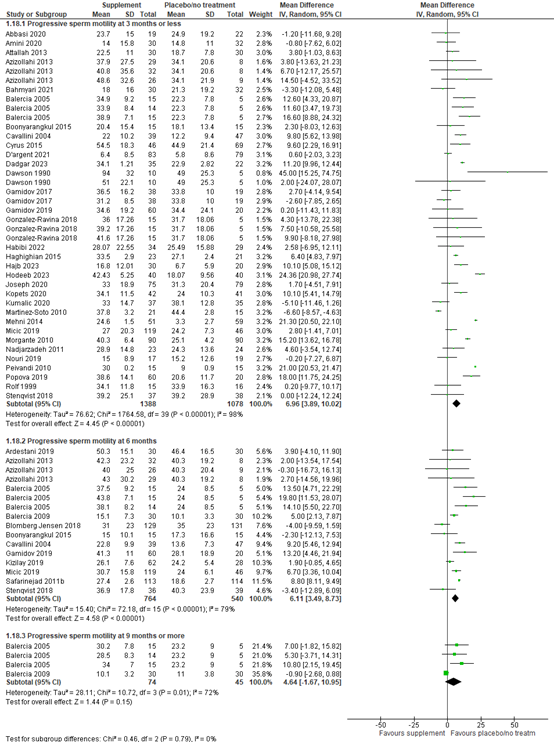
Progressive sperm motility (%) was reported in 37 studies. Progressive motility was measured at different timepoints after start of the intervention across the studies. At three months or less after start of the intervention, data of 32 studies with 2466 participants were available. The mean difference in progressive motility between participants using supplements versus participants using placebo or no treatment was 6.96% (95% CI 3.89 to 10.02%), in favor of supplements. At six months, 12 studies with a total of 1304 participants could be included in the analysis. The mean difference in progressive motility was 6.11% (95% CI 3.49 to 8.73%), in favor of supplements. At nine months or more, two studies with 119 participants were included. The mean difference in progressive motility was 4.64% (95% CI -1.67 to 10.95%), in favor of supplements (figure 8).
Figure 8. Forest plot of progressive sperm motility (%) comparing the use of supplements with placebo or no treatment
Analysis split on time of measurement
5.4 Morphology
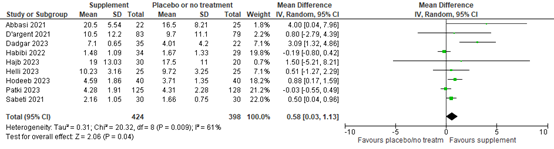
The percentage of sperm cells with normal morphology was not included as outcome in the Cochrane review, but was reported in nine of the studies published after publication of the Cochrane review with data of 822 participants. The mean difference in percentage of sperm cells with normal morphology was 0.58% (95% CI 0.03 to 1.13%) in favor of supplement (figure 9).
Figure 9. Forest plot of percentage of sperm cells with normal morphology comparing the use of supplements with placebo or no treatment
5.5 Volume
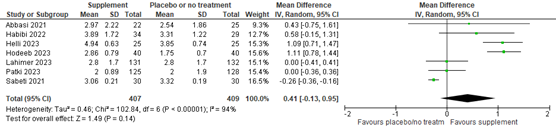
Sperm volume (mL) was not included as outcome in the Cochrane review, but was reported in seven of the studies published after publication of the Cochrane review with data of 775 participants. The mean difference in sperm volume was 0.41 (95% CI -0.13 to 0.95) in favor of supplement (figure 10).
Figure 10. Forest plot of semen volume in milliliter comparing the use of supplements with placebo or no treatment
6. DNA fragmentation (important)
DNA fragmentation was reported in 19 studies with a total of 2598 participants. Fifteen of these studies measured sperm DNA fragmentation three months or less after start of the intervention, three measured it three months or less and six months afterwards (Gamidov, 2019; Micic, 2019; Stenqvist, 2018) and one study measured it only six months afterwards (Schisterman, 2020). Thus, at the timepoint of three months or less after start of the intervention data of 18 studies with 1792 participants were available and at six months data of four studies with 1173 participants were available.
DNA fragmentation was measured in various ways, most often with the use of a Terminal deoxynucleotidyl transferase dUTP Nick End Labeling (TUNEL assay) or Sperm Chromatin Structure Analysi) (SCSA) or Toluidine Blue staining (TB).
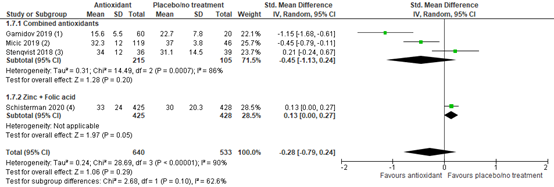
Three months or less after start of the intervention, the total standardized mean difference in percentage of DNA fragmentation between participants using supplements versus participants using placebo or no treatment was -0.29% (95% CI -0.54 to -0.04%), in favor of supplements (figure 11). In this figure, also a sensitivity analysis for type of antioxidant is presented. At six months after start of the intervention, the total standardized mean difference was -0.28% (95% CI -0.79 to 0.24%), in favor of supplements (figure 12).
Figure 11. Forest plot of DNA fragmentation comparing the use of supplements with placebo or no treatment at three months or less after start of the intervention with a sensitivity analysis for type of antioxidant
(1, 2, 5, 6, 8, 9, 10, 11, 13, 14, 19, 20) TUNEL assay. (3, 4, 16) TB staining. (7, 12, 22, 23, 24, 25) SCSA test. (15) Sperm DNA fragmentation assay. (17, 18) test unknown. (21) Sperm Chromatin Dispersion test (Halosperm)
Figure 12. Forest plot of DNA fragmentation comparing the use of antioxidants with placebo or no treatment at six months after start of the intervention with a sensitivity analysis for type of antioxidant
(1) TUNEL assay. (2) Sperm Chromatin Dispersion test (Halosperm). (3) SCSA test. (4) Comet assay
7. Parameters indicating oxidative stress in a semen sample (important)
Six of the studies published after the Cochrane review reported any parameter indicating oxidative stress in semen. The data of studies included in the Cochrane review can be found in the review itself. Various kind of parameters were reported. The available data are presented below.
|
Study |
Treatment |
No treatment |
||||
|
Mean |
SD |
Total (N) |
Mean |
SD |
Total (N) |
|
|
Sperm lipid peroxidation (%) |
||||||
|
Abbasi 2021 |
26 |
18.82 |
22 |
24.72 |
15.91 |
25 |
|
CMA3 (Chromomycin A3) positivity (%) |
||||||
|
Abbasi 2021 |
34.33 |
3.24 |
22 |
33.67 |
9.69 |
25 |
|
Sabeti 2021 |
36.76 |
5.96 |
30 |
37.13 |
5.11 |
30 |
|
TAC (total antioxidant capacity) (um/L) |
||||||
|
Dadgar 2023 |
1269 |
63.5 |
35 |
1091 |
63.5 |
22 |
|
Helli 2023 |
2.33 |
0.6 |
25 |
1.47 |
0.2 |
25 |
|
Hodeeb 2023 |
1.79 |
0.37 |
40 |
1.20 |
0.4 |
40 |
|
MDA (malonedialdehyde) (nmol/ml) |
||||||
|
Dadgar 2023 |
0.75 |
0,04 |
35 |
0.90 |
0,05 |
22 |
|
Helli 2023 |
0.69 |
0.07 |
25 |
0.91 |
0.13 |
25 |
|
ROS (reactive oxygen species) (RLU/s) |
||||||
|
Dadgar 2023 |
213.5 |
21.9 |
35 |
398.9 |
28.7 |
22 |
|
Haidari 2021 |
4.24 |
4.75 |
23 |
5.27 |
4.89 |
21 |
|
Glutathione S-transferase activity (nmol/min) |
||||||
|
Haidari 2021 |
7.992 |
0.01 |
23 |
6.75 |
1.61 |
21 |
A systematic review of the literature was performed to answer the following question:
What is the additional value of supplements and/or antioxidants in the treatment of males with subfertility?
Table 1. PICO
| Patients | Men with subfertility |
| Intervention | Supplements |
| Control | No supplements or placebo |
| Outcomes | Live birth rate, clinical pregnancy rate, miscarriage rate, total fertilization failure, semen parameters, DNA fragmentation, parameters indicating oxidative stress in a semen sample |
| Other selection criteria |
Study design: systematic reviews, randomized controlled trials and observational studies. From 2010 onwards |
Relevant outcome measures
The guideline panel considered live birth rate and clinical pregnancy rate as critical outcome measures for decision making; and miscarriage rate, total fertilization failure, semen parameters, DNA fragmentation, parameters indicating oxidative stress in a semen sample as important outcome measures for decision making.
The guideline panel defined a 5% difference (RR<0.95 or >1.05) for live birth rate and pregnancy rate as minimal clinically (patient) important difference. For other outcomes, the default boundaries as defined by GRADE were used: a 25% difference for dichotomous outcomes (RR< 0.8 or >1.25) and 0.5 SD for continuous outcomes as minimal clinically (patient) important difference.
Search and select (Methods)
The databases Medline (via OVID) and Embase (via Embase.com) were searched with relevant search terms from 2010 until April 3, 2024. The detailed search strategy is listed under the tab ‘Literature search strategy’. The systematic literature search resulted in 3922 hits. Due to the large number of studies, initially only systematic reviews were screened based on the PICO criteria. 42 systematic reviews were initially selected based on title and abstract screening. After reading the full text, 41 were excluded (see the exclusion table under the tab ‘Evidence tabellen’), and one Cochrane review was included. Then, a search was performed for randomized controlled trials from 2021 (search data of the review) until July 27, 2024. All studies were screened based on the PICO criteria. 70 RCTs were initially selected based on title and abstract screening. After reading the full text, 57 studies were excluded (see the exclusion table under the tab ‘Evidence tabellen’), and 13 studies were included. Thus, the summary of literature is based on 14 studies.
The results of a large Dutch multicenter placebo-controlled RCT on the effect of supplements on male subfertility have become available after evidence evaluation (Smits, 2020).
Results
Results of the 13 RCT’s are summarized in table 2. The assessment of the risk of bias is summarized in the risk of bias tables (under the tab ‘Evidence tabellen’).
Table 2. Characteristics of included RCT’s
|
Study |
Participants (number, age, other important characteristics) |
Comparison |
Outcome measures |
Comments |
Risk of bias* |
|
Individual studies (not included in the Cochrane review of de Ligny, 2022). |
|||||
|
Dadgar, 2023 |
N total: 140 of which 70 meet PICO - Intervention: 35 - Control: 35
Men aged 25-43 years with idiopathic infertility ((including oligo-asthenozoospermia, asthenozoospermia, astheno-teratozoospermia, oligoastheno- teratozoospermia) without other diseases. |
Intervention: 15mg zinc once daily for 3 months.
Control: placebo twice daily for 3 months.
Other groups received either pentoxifylline or pentoxifylline plus zinc.
|
Sperm concentration, progressive motility, percentage normal morphology, percentage DNA fragmentation, oxidative stress (malondialdehyde, total antioxidant capacity, reactive oxygen species).
|
Setting: hospital in Iran. |
High |
|
Hajb, 2023 |
N total: 50 - Intervention: 30 - Control: 20
Men with infertility: total count <15 million per milliliter, morphology <4% and progressive motility <40%. |
Intervention: 350mg date pollen extract + 250mg black seed pollen twice daily for 3 months.
Control: two tables of placebo twice daily for 3 months. |
Sperm volume, count, progressive motility, motility, percentage normal morphology.
|
Setting: hospital in Iran. |
High |
|
Hodeeb, 2023 |
N total: 80 - Intervention: 40 - Control: 40
Men with infertility (asthenozoospermia) and normal hormonal profile.
|
Intervention: 300mg alpha-lipoic acid twice daily for 90 days.
Control: placebo twice daily for 90 days. |
Sperm volume, count, progressive motility, motility, percentage normal morphology, percentage vitality, total antioxidant capacity, malondialdehyde.
|
Setting: university hospital in Egypt.
Title states ‘case-controlled study’. |
Low |
|
Lahimer, 2023 |
N total: 263 - Intervention: 131 - Control: 132
Men ≥20 years with idiopathic infertility (oligoastheno-teratozoospermia) and candidate for IUI, IVF and/or ICSI. |
Intervention: Fertilis* twice daily for 3 months.
Control: placebo twice daily for 3 months. * Fertilis antioxidant contains coenzyme Q10, folic acid, L-arginine, L-carnitine, L-glutathione, selenium, vitamin E and zinc. |
Sperm volume, count, motility, vitality, DNA fragmentation index, decondensation index, fertilization rate, pregnancy rate, live birth rate.
|
Setting: university hospital in Tunesia. |
Moderate |
|
Patki, 2023 |
N total: 300 - Intervention: 150 - Control: 150
Men aged 25-45 years with oligoastheno-teratozoospermia. |
Intervention: antioxidant blend* once daily for 3 months.
Control: placebo once daily for 3 months. * antioxidant blend contains coenzyme Q10, copper selenium, folic acid ginseng extract, iron, L-arginine, L-carnitine, L-glutathione, lycopene, manganese, selenium, vitamin A, B1, B6, B12, C, D, E and zinc. |
Sperm count, volume, motility, morphology, DNA fragmentation index. |
Setting: 10 hospitals in India. |
Moderate |
|
Pilehvari, 2023 |
N total: 60 - Intervention: 30 - Control: 30
Men <40 years with primary infertility (abnormality of at least one semen parameter). |
Intervention: 1.5g carob seed powder once daily for 12 weeks.
Control: placebo once daily for 12 weeks.
|
Sperm volume, count, progressive motility, morphology, viscosity.
|
Setting: hospital in Iran. |
Low |
|
Habibi, 2022 |
N total: 70 - Intervention: 35 - Control: 35
Men with or without impaired semen analysis with high sperm DNA damage. |
Intervention: 600mg alpha-lipoic acid once daily for 80 days.
Control: placebo once daily for 80 days.
|
Sperm concentration, motility, percentage abnormal morphology, DNA fragmentation, reactive oxygen species, clinical pregnancy rate. |
Setting: hospital in Iran. |
Low |
|
Helli, 2022 |
N total: 50 - Intervention: 25 - Control: 25
Men aged 20-45 years with idiopathic oligoastheno-teratozoospermia and normal hormonal profile. |
Intervention: 500mg probiotics* daily for 10 weeks.
Control: placebo daily for 10 weeks. * probiotic contains Lactobacillus casei, Lactobacillus rhamnosus, Lactobacillus bulgaricus, Lactobacillus acidophilus, Bifidobacterium breve, Bifidobacterium longum and Streptococcus thermophiles. |
Sperm volume, count, concentration, motility, percentage normal morphology, vitality, total antioxidant capacity, malondialdehyde.
|
Setting: university hospital in Iran. |
Moderate |
|
Abbasi, 2021 |
N total: 56 - Intervention: 28 - Control: 28
Men with oligozoospermia, teratozoospermia, and asthenozoospermia and no other diseases. |
Intervention: 500mg FamiLact* once daily for 80 days.
Control: placebo once daily for 80 days. * FamiLact contains Lactobacillus rhamnosus, Lactobacillus casei, Lactobacillus bulgaricus, Lactobacillus acidophilus, Bifidobacterium breve, Bifidobacterium longum, Streptococcus thermophilus and fructooligosaccharides. |
Sperm volume, concentration, motility, percentage normal morphology, DNA fragmentation index, protamine deficiency, lipid peroxidation. |
Setting: hospital in Iran. |
Low |
|
D’argent, 2021 |
N total: 162 - Intervention: 83 - Control: 79
Men 18-60 years with sperm concentration <15 million/mL, motility <50%, vitality <60% and normal morphology <4% and no other diseases. |
Intervention: 15mg folic acid once daily for 3 months.
Control: placebo once daily for 3 months.
|
Pregnancy rate, clinical pregnancy, sperm volume, motility, concentration, vitality, percentage normal morphology, DNA fragmentation index. |
Setting: six hospitals in France. |
Moderate |
|
Haidari, 2021 |
N total: 48 - Intervention: 24 - Control: 24
Men with idiopathic asthenozoospermia and no other diseases. |
Intervention: 600mg lipoic acid once daily for 12 weeks.
Control: placebo once daily for 12 weeks.
|
Reactive oxygen species, glutathione S-transferase activity. |
Setting: hospital in Iran. |
Low |
|
Melnikovova, 2021 |
N total: 50 - Intervention: 25 - Control: 25
Men aged 28-52 years with impaired semen quality and no other diseases. |
Intervention: 2800mg maca (Lepidium meyenii Walpers) once daily for 16 weeks.
Control: placebo once daily for 16 weeks.
|
Sperm count and concentration. |
Setting: hospital in Czech Republic. |
Moderate |
|
Sabeti, 2021 |
N total: 60 - Intervention: 30 - Control: 30
Men with asthenoteratozoospermia and no other diseases. |
Intervention: 400IU vitamin E + 200µg selenium once daily for 3 months.
Control: placebo once daily for 3 months.
|
Sperm volume, vitality, concentration, motility, percentage normal morphology, DNA fragmentation, markers for oxidative stress. |
Setting: hospital in Iran. |
Moderate |
- Abbasi B, Abbasi H, Niroumand H. Synbiotic (FamiLact) administration in idiopathic male infertility enhances sperm quality, DNA integrity, and chromatin status: A triple-blinded randomized clinical trial. Int J Reprod Biomed. 2021 Mar 21;19(3):235-244. doi: 10.18502/ijrm.v19i3.8571. PMID: 33842820; PMCID: PMC8023005.
- Charen E, Harbord N. Toxicity of Herbs, Vitamins, and Supplements. Adv Chronic Kidney Dis. 2020 Jan;27(1):67-71. doi: 10.1053/j.ackd.2019.08.003. PMID: 32147004.
- Dadgar Z, Shariatzadeh SMA, Mehranjani MS, Kheirolahi A. The therapeutic effect of co-administration of pentoxifylline and zinc in men with idiopathic infertility. Ir J Med Sci. 2023 Feb;192(1):431-439. doi: 10.1007/s11845-022-02931-0. Epub 2022 Feb 19. PMID: 35182288.
- d'Argent EM, Ravel C, Rousseau A, Morcel K, Massin N, Sussfeld J, Simon T, Antoine JM, Mandelbaume J, Daraï E, Kolanska K. High-Dose Supplementation of Folic Acid in Infertile Men Improves IVF-ICSI Outcomes: A Randomized Controlled Trial (FOLFIV Trial). J Clin Med. 2021 Apr 26;10(9):1876. doi: 10.3390/jcm10091876. PMID: 33925981; PMCID: PMC8123699.
- de Ligny WR, Fleischer K, Grens H, Braat DDM, de Bruin JP. The lack of evidence behind over-the-counter antioxidant supplements for male fertility patients: a scoping review. Hum Reprod Open. 2023 May 17;2023(3):hoad020. doi: 10.1093/hropen/hoad020. PMID: 37293243; PMCID: PMC10244220.
- de Ligny W, Smits RM, Mackenzie-Proctor R, Jordan V, Fleischer K, de Bruin JP, Showell MG. Antioxidants for male subfertility. Cochrane Database Syst Rev. 2022 May 4;5(5):CD007411. doi: 10.1002/14651858.CD007411.pub5. PMID: 35506389; PMCID: PMC9066298.
- Haidari F, Mohammadi-Asl J, Kavianpour M, Dadfar M, Haghighian HK. Effect of lipoic acid supplementation on gene expression and activity of glutathione S-transferase enzyme in infertile men. Hum Fertil (Camb). 2021 Oct;24(4):276-283. doi: 10.1080/14647273.2019.1639221. Epub 2019 Jul 11. PMID: 31291793.
- Hajb A, Salehpour Z, Aghaei R, Najafian A, Mahmoodi M, Latifi M, Fallahi S. The Effect of Palm Pollen and Black Seed Pollen on Male Sex Hormones and Sperm Quality: A Single-Blind, Placebo-Controlled Clinical Trial Study. Int J Fertil Steril. 2023 Jan 1;17(1):75-79. doi: 10.22074/ijfs.2022.548477.1259. PMID: 36617207; PMCID: PMC9807887.
- Helli B, Kavianpour M, Ghaedi E, Dadfar M, Haghighian HK. Probiotic effects on sperm parameters, oxidative stress index, inflammatory factors and sex hormones in infertile men. Hum Fertil (Camb). 2022 Jul;25(3):499-507. doi: 10.1080/14647273.2020.1824080. Epub 2020 Sep 27. PMID: 32985280.
- Hodeeb YM, El-Rewiny EM, Gaafar AM, Zayed AN, Hasan MS, Elsaie ML. The effect of alpha lipoic acid supplementation on sperm functions in idiopathic asthenozoospermic patients: a case-controlled study. Hum Fertil (Camb). 2023 Dec;26(5):949-957. doi: 10.1080/14647273.2021.2025271. Epub 2022 Jan 13. PMID: 35023797.
- Lahimer M, Gherissi O, Ben Salem N, Ben Mustapha H, Bach V, Khorsi-Cauet H, Khairi H, Ben Ali H, BenKhalifa M, Ajina M. Effect of Micronutrients and L-Carnitine as Antioxidant on Sperm Parameters, Genome Integrity, and ICSI Outcomes: Randomized, Double-Blind, and Placebo-Controlled Clinical Trial. Antioxidants (Basel). 2023 Oct 31;12(11):1937. doi: 10.3390/antiox12111937. PMID: 38001791; PMCID: PMC10669279.
- Miller ER 3rd, Pastor-Barriuso R, Dalal D, Riemersma RA, Appel LJ, Guallar E. Meta-analysis: high-dosage vitamin E supplementation may increase all-cause mortality. Ann Intern Med. 2005 Jan 4;142(1):37-46. doi: 10.7326/0003-4819-142-1-200501040-00110. Epub 2004 Nov 10. PMID: 15537682.
- Melnikovova I, Russo D, Fait T, Kolarova M, Tauchen J, Kushniruk N, Falabella R, Milella L, Fernández Cusimamani E. Evaluation of the effect of Lepidium meyenii Walpers in infertile patients: A randomized, double-blind, placebo-controlled trial. Phytother Res. 2021 Nov;35(11):6359-6368. doi: 10.1002/ptr.7287. Epub 2021 Sep 28. PMID: 34585449; PMCID: PMC9293450.
- Patki A, Shelatkar R, Singh M, Agarwal S, M V, Umbardand S, Reddy A, Kannan P, Gorthi S, Khastgir G, Kulshreshtha A, Ganu G. Impact of antioxidants in improving semen parameters like count, motility and DNA fragmentation in sub-fertile males: a randomized, double-blind, placebo-controlled clinical trial. Transl Clin Pharmacol. 2023 Mar;31(1):28-39. doi: 10.12793/tcp.2023.31.e6. Epub 2023 Mar 28. PMID: 37034126; PMCID: PMC10079508.
- Pilehvari S, Gharakhani Bahar T, Masoumi SZ, Kazemi F, Moradkhani S, Rastgoo Haghi A, Maleki P. Effect of Carob Supplement on Spermogram Parameters and Sexual Function of Infertile Men Referred to the Infertility Center, Hamadan, Iran, 2019: A Randomized Controlled Trial. J Family Reprod Health. 2023 Sep;17(3):142-150. doi: 10.18502/jfrh.v17i3.13537. PMID: 38716296; PMCID: PMC11070741.
- Sabeti P, Pourmasumi S, Fagheirelahee N. Effect of Selenium and Vitamin E on the Level of Sperm HSPA2+, Intracellular Superoxide Anion and Chromatin Integrity in Idiopathic Asthenoteratozoospermia: A Double-Blind, Randomized, Placebo- Controlled Trial. Urol J. 2021 Sep 13;18(5):549-555. doi: 10.22037/uj.v18i.6325. PMID: 34516655.
- Smits R, D'Hauwers K, IntHout J, Braat D, Fleischer K. Impact of a nutritional supplement (Impryl) on male fertility: study protocol of a multicentre, randomised, double-blind, placebo-controlled clinical trial (SUppleMent Male fERtility, SUMMER trial). BMJ Open. 2020 Jul 2;10(7):e035069. doi: 10.1136/bmjopen-2019-035069. PMID: 32616489; PMCID: PMC7333867.
Risk of Bias tables
Risk of bias table for systematic reviews
|
Study reference |
Appropriate and clearly focused question?1 |
Comprehensive and systematic literature search?2 |
Description of included and excluded studies?3 |
Description of relevant characteristics of included studies?4 |
Assessment of scientific quality of included studies?5 |
Enough similarities between studies to make combining them reasonable?6 |
Potential risk of publication bias taken into account?7 |
Potential conflicts of interest reported?8
|
|
De Ligny, 2022 |
Yes |
Yes |
Yes |
Yes |
Yes |
Yes |
Yes |
Yes |
1. Research question (PICO) and inclusion criteria should be appropriate (in relation to the research question to be answered in the clinical guideline) and predefined
2. Search period and strategy should be described; at least Medline searched
3. Potentially relevant studies that are excluded at final selection (after reading the full text) should be referenced with reasons
4. Characteristics of individual studies relevant to the research question (PICO) should be reported
5. Quality of individual studies should be assessed using a quality scoring tool or checklist (preferably QUADAS-2; COSMIN checklist for measuring instruments) and taken into account in the evidence synthesis
6. Clinical and statistical heterogeneity should be assessed; clinical: enough similarities in patient characteristics, diagnostic tests (strategy) to allow pooling? For pooled data: at least 5 studies available for pooling; assessment of statistical heterogeneity and, more importantly (see Note), assessment of the reasons for heterogeneity (if present)? Note: sensitivity and specificity depend on the situation in which the test is being used and the thresholds that have been set, and sensitivity and specificity are correlated; therefore, the use of heterogeneity statistics (p-values; I2) is problematic, and rather than testing whether heterogeneity is present, heterogeneity should be assessed by eye-balling (degree of overlap of confidence intervals in Forest plot), and the reasons for heterogeneity should be examined.
7. There is no clear evidence for publication bias in diagnostic studies, and an ongoing discussion on which statistical method should be used. Tests to identify publication bias are likely to give false-positive results, among available tests, Deeks’ test is most valid. Irrespective of the use of statistical methods, you may score “Yes” if the authors discuss the potential risk of publication bias.
8. Sources of support (including commercial co-authorship) should be reported in both the systematic review and the included studies. Note: To get a “yes,” source of funding or support must be indicated for the systematic review AND for each of the included studies.
Risk of bias table for intervention studies
|
Study reference (first author, year) |
Was the allocation sequence adequately generated? |
Was the allocation adequately concealed? |
Blinding: |
Was loss to follow-up (missing outcome data) infrequent? |
Are reports of the study free of selective outcome reporting? |
Was the study apparently free of other problems that could put it at a risk of bias? |
Overall risk of bias |
|
Dadgar, 2023 |
Unclear
Reason: Study design states ‘randomized trial’, but no further information is provided. |
Unclear
Reason: No information is provided. |
Unclear
Reason: Study design states ‘double-blind’, but no further information is provided. |
Definitely yes
Reason: 4 (11%) -0-2 (6%) -13 (37%) were lost to follow-up in the respective arms. |
Definitely no
Reason: Not all outcomes stated in the methods section are reported. |
Probably yes
Reason: Registered trial, no funding source reported. |
high risk of bias
Reason: Unclear randomization and blinding procedures. |
|
Hajb, 2023 |
Probably yes
Reason: In methods section is stated: ‘placement of people was done by simple randomization method’. |
Unclear
Reason: No information is provided. |
Unclear
Reason: Study design states ‘single-blind’, but no further information is provided. |
Definitely no
Reason: No loss to follow-up. |
Definitely no
Reason: Not all outcomes stated in the methods section are reported. |
Probably yes
Reason: Registered trial, no funding source reported. |
high risk of bias
Reason: Unclear randomization and blinding procedures. |
|
Hodeeb, 2023 |
Definitely yes
Reason: Computer-generated randomization sequence. |
Unclear
Reason: No information is provided. |
Definitely no
Reason: Blinding of clinician, healthcare providers and statistician, not of participants. |
Definitely no
Reason: No loss to follow-up |
Probably yes
Reason: All outcomes stated in the methods section are reported. |
Probably yes
Reason: No conflict of interest or funding sources. Study is stated as ‘randomized case-controlled study’. |
Low risk of bias
Reason: Participants not blinded, but this has likely no influence on outcomes (objective outcomes). |
|
Lahimer, 2023 |
Definitely yes
Reason: Computer-generated randomization sequence. |
Unclear
Reason: No information is provided. |
Definitely yes
Reason: Double-blind trial. |
Definitely no
Reason: No loss to follow-up |
Probably yes
Reason: All outcomes stated in the methods section are reported. |
Probably no
Reason: Funding provided by ‘MEDIS laboratories’ (not stated how they were involved). |
Some concerns
Reason: Industry sponsored trial. |
|
Patki, 2023 |
Definitely yes
Reason: Computer-generated randomization sequence. |
Unclear
Reason: No information is provided. |
Definitely yes
Reason: Double-blind trial. |
Probably yes
Reason: 25 (17%) and 22 (15%) were lost to follow-up in the respective arms. |
Probably yes
Reason: All outcomes stated in the methods section are reported. |
Probably no
Reason: Funding provided by ‘Meyer Organics’ (not stated how they were involved). |
Some concerns
Reason: Industry sponsored trial, high number of lost to follow-up. |
|
Pilehvari, 2023 |
Definitely yes
Reason: Mixed cards with label A or B. |
Definitely yes
Reason: Taking out cards. |
Definitely yes
Reason: Blinding of researchers and participants. |
Probably no
Reason: 3 and 7 were lost to follow-up in the respective arms. |
Probably yes
Reason: All outcomes stated in the methods section are reported. |
Probably yes
Reason: Registered trial, no funding source reported. |
Low risk of bias
Reason: Adequate randomization and blinding procedures. |
|
Habibi, 2022 |
Definitely yes
Reason: Computer-mediated random digits table with simple randomization. |
Definitely yes
Reason: Randomization performed by persons unrelated to the study. |
Definitely yes
Reason: Blinding of researchers, drug prescribers, statistician and participants. |
Probably no
Reason: 4 and 1 were lost to follow-up in the respective arms. |
Probably yes
Reason: All outcomes stated in the methods section are reported. |
Probably yes
Reason: Registered trial, no funding source reported. |
Low risk of bias
Reason: Adequate randomization and blinding procedures. |
|
Helli, 2022 |
Probably yes
Reason: In methods section is stated: ‘patients were randomized by a third person’. |
Probably yes
Reason: In methods section is stated: ‘patients were randomized by a third person’. |
Definitely yes
Reason: Blinding of researchers and participants. |
Probably no
Reason: 1 and 1 were lost to follow-up in the respective arms. |
Probably yes
Reason: All outcomes stated in the methods section are reported. |
Probably yes
Reason: Registered trial, no industrial funding source. |
Some concerns
Reason: Randomization procedure unclear. |
|
Abbasi, 2021 |
Definitely yes
Reason: Permutation block method with random sequence. |
Unclear
Reason: No information is provided. |
Definitely yes
Reason: Blinding of healthcare providers, data collectors, statistician and participants. |
Probably no
Reason: 3 and 4 were lost to follow-up in the respective arms. |
Probably yes
Reason: All outcomes stated in the methods section are reported. |
Probably yes
Reason: Registered trial, no funding source reported. |
Low risk of bias
Reason: Adequate randomization and blinding procedures. |
|
D’argent, 2021 |
Definitely yes
Reason: Web-based randomization system. |
Unclear
Reason: No information is provided. |
Unclear
Reason: Study design states ‘double-blind’, but no further information is provided. |
Probably no
Reason: 2 and 6 were lost to follow-up in the respective arms. |
Definitely no
Reason: Not all outcomes stated in the methods section are reported. |
Probably yes
Reason: Registered trial, no industrial funding source. |
Some concerns
Reason: Blinding procedure unclear. Selective outcome reporting |
|
Haidari, 2021 |
Definitely yes
Reason: Computer-generated randomization sequence by the method of permutated blocks. |
Probably yes
Reason: Randomization overseen by someone independent of the study. |
Definitely yes
Reason: Blinding of investigator, clinical prescriber and participants. |
Probably no
Reason: 1 and 3 were lost to follow-up in the respective arms. |
Probably yes
Reason: All outcomes stated in the methods section are reported. |
Probably yes
Reason: Registered trial, no industrial funding source. |
Low risk of bias
Reason: Adequate randomization and blinding procedures. |
|
Melnikovova, 2021 |
Unclear
Reason: ‘Randomization created by investigator.’ |
Definitely yes
Reason: Separated and sealed envelopes with order number. |
Definitely yes
Reason: Blinding of investigators and participants. |
Probably no
Reason: 6 and 5 were lost to follow-up in the respective arms. |
Probably yes
Reason: All outcomes stated in the methods section are reported. |
Probably yes
Reason: Non-registered trial, no industrial funding source. |
Some concerns
Reason: Randomization procedure unclear. |
|
Sabeti, 2021 |
Unclear
Reason: Study design states ‘randomized trial’, but no further information is provided. |
Unclear
Reason: No information is provided. |
Definitely yes
Reason: Blinding of investigators and participants. |
Definitely no
Reason: No lost to follow-up. |
Probably yes
Reason: All outcomes stated in the methods section are reported. |
Probably yes
Reason: Registered trial, no industrial funding source. |
Some concerns
Reason: Randomization procedure unclear. |
Table of Excluded studies after reading full text
|
Reference |
Reason for exclusion |
|
Adamczewska D, Słowikowska-Hilczer J, Walczak-Jędrzejowska R. The Association between Vitamin D and the Components of Male Fertility: A Systematic Review. Biomedicines. 2022 Dec 29;11(1):90. doi: 10.3390/biomedicines11010090. PMID: 36672602; PMCID: PMC9912266. |
Wrong population, no intervention. |
|
Agarwal A, Cannarella R, Saleh R, Harraz AM, Kandil H, Salvio G, Boitrelle F, Kuroda S, Farkouh A, Rambhatla A, Zini A, Colpi G, Gül M, Kavoussi P, Hamoda TAA, Ko E, Calik G, Toprak T, Pinggera GM, Park HJ, Ghayda RA, Minhas S, Busetto GM, Bakırcıoğlu ME, Kadioglu A, Chung E, Russo GI, Calogero AE, Ambar RF, Jayasena CN, Shah R. Impact of Antioxidant Therapy on Natural Pregnancy Outcomes and Semen Parameters in Infertile Men: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. World J Mens Health. 2023 Jan;41(1):14-48. doi: 10.5534/wjmh.220067. Epub 2022 Sep 7. PMID: 36102104; PMCID: PMC9826914. |
Meets PICO but not the most comprehensive review. |
|
Agarwal A, Leisegang K, Majzoub A, Henkel R, Finelli R, Panner Selvam MK, Tadros N, Parekh N, Ko EY, Cho CL, Arafa M, Alves MG, Oliveira PF, Alvarez JG, Shah R. Utility of Antioxidants in the Treatment of Male Infertility: Clinical Guidelines Based on a Systematic Review and Analysis of Evidence. World J Mens Health. 2021 Apr;39(2):233-290. doi: 10.5534/wjmh.200196. Epub 2021 Jan 15. PMID: 33474843; PMCID: PMC7994666. |
Wrong population. |
|
Ahmadi S, Bashiri R, Ghadiri-Anari A, Nadjarzadeh A. Antioxidant supplements and semen parameters: An evidence based review. Int J Reprod Biomed. 2016 Dec;14(12):729-736. PMID: 28066832; PMCID: PMC5203687. |
Wrong design. |
|
Arcaniolo D, Favilla V, Tiscione D, Pisano F, Bozzini G, Creta M, Gentile G, Menchini Fabris F, Pavan N, Veneziano IA, Cai T. Is there a place for nutritional supplements in the treatment of idiopathic male infertility? Arch Ital Urol Androl. 2014 Sep 30;86(3):164-70. doi: 10.4081/aiua.2014.3.164. PMID: 25308577. |
Wrong design. |
|
Banihani SA. Effect of ginger (Zingiber officinale) on semen quality. Andrologia. 2019 Jul;51(6):e13296. doi: 10.1111/and.13296. Epub 2019 Apr 22. PMID: 31012134. |
Wrong population. |
|
Barbonetti A, Tienforti D, Castellini C, Giulio FD, Muselli M, Pizzocaro A, Vena W, Baroni MG, Pivonello R, Isidori AM, Maggi M, Corona G. Effect of antioxidants on semen parameters in men with oligo-astheno-teratozoospermia: a network meta-analysis. Andrology. 2024 Mar;12(3):538-552. doi: 10.1111/andr.13498. Epub 2023 Jul 26. PMID: 37495550. |
Wrong comparison. |
|
Buhling K, Schumacher A, Eulenburg CZ, Laakmann E. Influence of oral vitamin and mineral supplementation on male infertility: a meta-analysis and systematic review. Reprod Biomed Online. 2019 Aug;39(2):269-279. doi: 10.1016/j.rbmo.2019.03.099. Epub 2019 Mar 16. PMID: 31160241. |
Meets PICO but not the most recent review. |
|
Busetto GM, Rodrigues BF, Virmani A, Checchia A, Ninivaggi A, Ricapito A, Barbieri G, Fischetti P, Falagario UG, Annese P, d'Altilia N, Mancini V, Ferro M, Crocetto F, Porreca A, Bettocchi C, Cormio L, Agarwal A, Carrieri G. Antioxidant treatment for oligoasthenoteratozoospermia and varicocele: a DBPC trial to evaluate the impact of age and body mass index. Asian J Androl. 2024 May 1;26(3):239-244. doi: 10.4103/aja202381. Epub 2024 Feb 2. PMID: 38305695; PMCID: PMC11156455. |
Secondary analysis of included study. |
|
Cannarella R, Calogero AE, Condorelli RA, Giacone F, Mongioi' LM, La Vignera S. Non-hormonal treatment for male infertility: the potential role of Serenoa repens, selenium and lycopene. Eur Rev Med Pharmacol Sci. 2019 Apr;23(7):3112-3120. doi: 10.26355/eurrev_201904_17595. PMID: 31002161. |
Wrong design. |
|
Corbett GA, Crosby DA, McAuliffe FM. Probiotic therapy in couples with infertility: A systematic review. Eur J Obstet Gynecol Reprod Biol. 2021 Jan;256:95-100. doi: 10.1016/j.ejogrb.2020.10.054. Epub 2020 Oct 27. PMID: 33188995. |
Wrong population and/or design. |
|
Dimitriadis F, Borgmann H, Struck JP, Salem J, Kuru TH. Antioxidant Supplementation on Male Fertility-A Systematic Review. Antioxidants (Basel). 2023 Mar 30;12(4):836. doi: 10.3390/antiox12040836. PMID: 37107211; PMCID: PMC10135082. |
Low quality: no data reported, no search date, no reason for exclusion of studies, no assessment of quality of studies. |
|
Dong L, Yang F, Li J, Li Y, Yu X, Zhang X. Effect of oral alpha-lipoic acid (ALA) on sperm parameters: a systematic review and meta-analysis. Basic Clin Androl. 2022 Dec 8;32(1):23. doi: 10.1186/s12610-022-00173-9. PMID: 36476425; PMCID: PMC9730623. |
Meets PICO but not the most comprehensive review. |
|
Durg S, Shivaram SB, Bavage S. Withania somnifera (Indian ginseng) in male infertility: An evidence-based systematic review and meta-analysis. Phytomedicine. 2018 Nov 15;50:247-256. doi: 10.1016/j.phymed.2017.11.011. Epub 2017 Nov 29. PMID: 30466985. |
Included observationals/ pilot studies only |
|
Fallahi S, Rajaei M, Malekzadeh K, Kalantar SM. Would Phoenix Dactyflera Pollen (palm seed) be considered as a treatment agent against Males' infertility? A systematic review. Electron Physician. 2015 Dec 20;7(8):1590-6. doi: 10.19082/1590. PMID: 26816585; PMCID: PMC4725411. |
No data provided. |
|
Hosseini B, Nourmohamadi M, Hajipour S, Taghizadeh M, Asemi Z, Keshavarz SA, Jafarnejad S. The Effect of Omega-3 Fatty Acids, EPA, and/or DHA on Male Infertility: A Systematic Review and Meta-analysis. J Diet Suppl. 2019;16(2):245-256. doi: 10.1080/19390211.2018.1431753. Epub 2018 Feb 16. PMID: 29451828. |
Meets PICO but not the most recent review. |
|
Imamovic Kumalic S, Pinter B. Review of clinical trials on effects of oral antioxidants on basic semen and other parameters in idiopathic oligoasthenoteratozoospermia. Biomed Res Int. 2014;2014:426951. doi: 10.1155/2014/426951. Epub 2014 Mar 31. PMID: 24800224; PMCID: PMC3988936. |
Wrong design. |
|
Jin X, Man C, Gong D, Fan Y. Adjuvant Treatment with Qilin Pill for Men with Oligoasthenospermia: A Meta-Analysis of Randomized Controlled Trials. Phytother Res. 2017 Sep;31(9):1291-1297. doi: 10.1002/ptr.5854. Epub 2017 Jun 21. PMID: 28635070. |
Wrong comparison. |
|
Khaw SC, Wong ZZ, Anderson R, Martins da Silva S. l-carnitine and l-acetylcarnitine supplementation for idiopathic male infertility. Reprod Fertil. 2020 Dec 23;1(1):67-81. doi: 10.1530/RAF-20-0037. PMID: 35128424; PMCID: PMC8812460. |
Meets PICO but not the most comprehensive or recent review. |
|
Li X, Zeng YM, Luo YD, He J, Luo BW, Lu XC, Zhu LL. Effects of folic acid and folic acid plus zinc supplements on the sperm characteristics and pregnancy outcomes of infertile men: A systematic review and meta-analysis. Heliyon. 2023 Jul 13;9(7):e18224. doi: 10.1016/j.heliyon.2023.e18224. PMID: 37539255; PMCID: PMC10395467. |
Meets PICO but not the most comprehensive review. |
|
Mahdavi R, Heshmati J, Namazi N. Effects of black seeds (Nigella sativa) on male infertility: A systematic review. March 2015. Journal of Herbal Medicine 5(3). |
No data provided. |
|
Majzoub A, Agarwal A. Antioxidant therapy in idiopathic oligoasthenoteratozoospermia. Indian J Urol. 2017 Jul-Sep;33(3):207-214. doi: 10.4103/iju.IJU_15_17. PMID: 28717270; PMCID: PMC5508431. |
Wrong design. |
|
Musavi H, Tabnak M, Sheini FA, Bezvan MH, Amidi F, Abbasi M. Effect of garlic (allium sativum) on male fertility: A systematic review. J Herbmed Pharmacol. 2018;7(4): 306-312. doi: 10.15171/jhp.2018.46 |
Wrong population. |
|
Nasimi Doost Azgomi R, Zomorrodi A, Nazemyieh H, Fazljou SMB, Sadeghi Bazargani H, Nejatbakhsh F, Moini Jazani A, Ahmadi AsrBadr Y. Effects of Withania somnifera on Reproductive System: A Systematic Review of the Available Evidence. Biomed Res Int. 2018 Jan 24;2018:4076430. doi: 10.1155/2018/4076430. Erratum in: Biomed Res Int. 2019 Nov 21;2019:7591541. doi: 10.1155/2019/7591541. PMID: 29670898; PMCID: PMC5833251. |
Wrong population. |
|
Noegroho BS, Siregar S, Tampubolon KAG. Antioxidant Supplementation on Sperm DNA Fragmentation and Sperm Parameters: A Systematic Review and Meta-Analysis. Turk J Urol. 2022 Sep;48(5):375-384. doi: 10.5152/tud.2022.22058. PMID: 36197144; PMCID: PMC9623341. |
Meets PICO but not the most comprehensive review. |
|
Oliveira LCSL, Costa EC, Martins FDG, Rocha ASD, Brasil GA. Probiotics supplementation in the treatment of male infertility: A Systematic Review. JBRA Assist Reprod. 2024 Jun 1;28(2):341-348. doi: 10.5935/1518-0557.20240013. PMID: 38530761; PMCID: PMC11152433. |
Meets PICO but not the most comprehensive review. |
|
Omar MI, Pal RP, Kelly BD, Bruins HM, Yuan Y, Diemer T, Krausz C, Tournaye H, Kopa Z, Jungwirth A, Minhas S. Benefits of Empiric Nutritional and Medical Therapy for Semen Parameters and Pregnancy and Live Birth Rates in Couples with Idiopathic Infertility: A Systematic Review and Meta-analysis. Eur Urol. 2019 Apr;75(4):615-625. doi: 10.1016/j.eururo.2018.12.022. Epub 2019 Jan 8. PMID: 30630643. |
Meets PICO but not the most recent review. |
|
Salas-Huetos A, Rosique-Esteban N, Becerra-Tomás N, Vizmanos B, Bulló M, Salas-Salvadó J. The Effect of Nutrients and Dietary Supplements on Sperm Quality Parameters: A Systematic Review and Meta-Analysis of Randomized Clinical Trials. Adv Nutr. 2018 Nov 1;9(6):833-848. doi: 10.1093/advances/nmy057. PMID: 30462179; PMCID: PMC6247182. |
Meets PICO but not the most recent review. |
|
Salvio G, Cutini M, Ciarloni A, Giovannini L, Perrone M, Balercia G. Coenzyme Q10 and Male Infertility: A Systematic Review. Antioxidants (Basel). 2021 May 30;10(6):874. doi: 10.3390/antiox10060874. PMID: 34070761; PMCID: PMC8226917. |
Wrong comparison. |
|
Shang XJ, Wang LL, Mo DS, Cai HC, Zheng DD, Zhou YZ. [Effect and safety of L-carnitine in the treatment of idiopathic oligoasthenozoospermia: a systemic review]. Zhonghua Nan Ke Xue. 2015 Jan;21(1):65-73. Chinese. PMID: 25707144. |
Full text in Chinese |
|
Sharma AP, Sharma G, Kumar R. Systematic Review and Meta-analysis on Effect of Carnitine, Coenzyme Q10 and Selenium on Pregnancy and Semen Parameters in Couples With Idiopathic Male Infertility. Urology. 2022 Mar;161:4-11. doi: 10.1016/j.urology.2021.10.041. Epub 2021 Dec 4. PMID: 34871624. |
Meets PICO but not the most comprehensive review. |
|
Tahvilzadeh M, Hajimahmoodi M, Toliyat T, Karimi M, Rahimi R. An evidence-based approach to medicinal plants for the treatment of sperm abnormalities in traditional Persian medicine. Andrologia. 2016 Oct;48(8):860-79. doi: 10.1111/and.12676. PMID: 27681644. |
Wrong design, wrong intervention. |
|
Tania C, Tobing ERPL, Tansol C, Prasetiyo PD, Wallad CK, Hariyanto TI. Vitamin D supplementation for improving sperm parameters in infertile men: A systematic review and meta-analysis of randomized clinical trials. Arab J Urol. 2023 Jan 10;21(4):204-212. doi: 10.1080/2090598X.2023.2165232. PMID: 38178948; PMCID: PMC10763592. |
Meets PICO but not the most comprehensive review. |
|
Tatar T, Akdevelioğlu Y. Effect of Pollen, Pit Powder, and Gemmule Extract of Date Palm on Male Infertility: A Systematic Review. J Am Coll Nutr. 2018 Feb;37(2):154-160. doi: 10.1080/07315724.2017.1364183. Epub 2017 Oct 31. PMID: 29087245. |
Wrong design of included studies. |
|
Wang M, Wang Q, Du Y, Jiang H, Zhang X. Vitamins combined with traditional Chinese medicine for male infertility: A systematic review and meta-analysis. Andrology. 2020 Sep;8(5):1038-1050. doi: 10.1111/andr.12787. Epub 2020 Mar 31. PMID: 32170803. |
Wrong comparison. |
|
Wang R, Wang S, Song Y, Zhou H, Pan Y, Liu L, Niu S, Liu X. Effect of vitamin E on Semen Quality Parameters: A Meta-Analysis of a Randomized Controlled Trial. Urol J. 2022 Nov 8;19(5):343-351. doi: 10.22037/uj.v19i.7160. PMID: 36029025. |
Meets PICO but not the most comprehensive review. |
|
Wei G, Zhou Z, Cui Y, Huang Y, Wan Z, Che X, Chai Y, Zhang Y. A Meta-Analysis of the Efficacy of L-Carnitine/L-Acetyl-Carnitine or N-Acetyl-Cysteine in Men With Idiopathic Asthenozoospermia. Am J Mens Health. 2021 Mar-Apr;15(2):15579883211011371. doi: 10.1177/15579883211011371. PMID: 33906513; PMCID: PMC8108089. |
Meets PICO but not the most comprehensive review. |
|
Yan TF, Qi JJ, Li LX, Li F. The Association between Serum Vitamin D Levels and Male Fertility: A Systematic Review and Meta-Analysis. Andrologia 2023(8):1-8 DOI: 10.1155/2023/9002938. |
No intervention. |
|
Zafar MI, Mills KE, Baird CD, Jiang H, Li H. Effectiveness of Nutritional Therapies in Male Factor Infertility Treatment: A Systematic Review and Network Meta-analysis. Drugs. 2023 Apr;83(6):531-546. doi: 10.1007/s40265-023-01853-0. Epub 2023 Mar 21. PMID: 36943634. |
Wrong comparison. |
|
Zhang X, Cui Y, Dong L, Sun M, Zhang Y. The efficacy of combined l-carnitine and l-acetyl carnitine in men with idiopathic oligoasthenoteratozoospermia: A systematic review and meta-analysis. Andrologia. 2020 Mar;52(2):e13470. doi: 10.1111/and.13470. Epub 2019 Nov 7. PMID: 31701550. |
Meets PICO but not the most comprehensive review. |
|
Zhou X, Shi H, Zhu S, Wang H, Sun S. Effects of vitamin E and vitamin C on male infertility: a meta-analysis. Int Urol Nephrol. 2022 Aug;54(8):1793-1805. doi: 10.1007/s11255-022-03237-x. Epub 2022 May 23. PMID: 35604582. |
Meets PICO but not the most comprehensive review. |
|
Zhou Z, Cui Y, Zhang X, Zhang Y. The role of N-acetyl-cysteine (NAC) orally daily on the sperm parameters and serum hormones in idiopathic infertile men: A systematic review and meta-analysis of randomised controlled trials. Andrologia. 2021 Mar;53(2):e13953. doi: 10.1111/and.13953. Epub 2021 Jan 6. PMID: 33405232. |
Meets PICO but not the most comprehensive review. |
|
Spivak L G, Gamidov S I, Al-Shukri S H, Morozov A O, Androsov A A, Popova A Y, Kupriyanov Yu A, Pushkar D Y. [Evaluation of long-term results of using the drug Fertiwell: impact on the likelihood of conception and pregnancy outcome]. Urologiia. 2023 Dec;(6):72-79. Russian. PMID: 38156687. |
Full text in Russian. |
|
Bozhedomov VA, Epanchintseva EA, Bozhe-Domova GE, Kamarina RA, Rokhlikov IM, Kamalov AA. [Hydrophilic and lipophilic nutrients for the treatment of male idiopathic infertility: a randomized, comparative, open-label, multicenter, prospective, controlled study]. Urologiia. 2021 Mar;(1):70-78. Russian. PMID: 33818939. |
Full text in Russian. |
|
Asadi M, Gholipour F, Rahavian A, Javanbakht M. Effects of probiotic supplementation on semen parameters after varicocelectomy: A randomized controlled trial. J Res Med Sci. 2023 Oct 26;28:74. doi: 10.4103/jrms.jrms_392_23. PMID: 38152072; PMCID: PMC10751512. |
Wrong intervention/ comparison. |
|
Pilehvari S, Masoumi SZ, Bahar TG, Kazemi F, Moradkhani S, Maleki P. Effect of Carob and Ginseng Supplements on Semen Analysis Parameters, Sexual Function, and Sex Hormones in Infertile Men: Double-Blind, Randomized Controlled Trial Study. Iran J Nurs Midwifery Res. 2024 Jan 9;29(1):113-119. doi: 10.4103/ijnmr.ijnmr_460_21. PMID: 38333335; PMCID: PMC10849286. |
No extractable data (also primary outcome is sexual functioning). |
|
Joseph T, Mascarenhas M, Karuppusami R, Karthikeyan M, Kunjummen AT, Kamath MS. Antioxidant pretreatment for male partner before ART for male factor subfertility: a randomized controlled trial. Hum Reprod Open. 2020 Nov 16;2020(4):hoaa050. doi: 10.1093/hropen/hoaa050. PMID: 33225077; PMCID: PMC7668397. |
Included in Cochrane review. |
|
Banks N, Sun F, Krawetz SA, Coward RM, Masson P, Smith JF, Trussell JC, Santoro N, Zhang H, Steiner AZ. Male vitamin D status and male factor infertility. Fertil Steril. 2021 Oct;116(4):973-979. doi: 10.1016/j.fertnstert.2021.06.035. Epub 2021 Jul 18. PMID: 34289935; PMCID: PMC8561776. |
Secondary analysis of MOXI trial, which is already included. |
|
Knudtson JF, Sun F, Coward RM, Hansen KR, Barnhart KT, Smith J, Legro RS, Diamond MP, Krawetz SA, Zhang H, Usadi R, Baker VL, Santoro N, Steiner AZ. The relationship of plasma antioxidant levels to semen parameters: the Males, Antioxidants, and Infertility (MOXI) randomized clinical trial. J Assist Reprod Genet. 2021 Nov;38(11):3005-3013. doi: 10.1007/s10815-021-02301-2. Epub 2021 Aug 28. PMID: 34455507; PMCID: PMC8609082. |
Secondary analysis of MOXI trial, which is already included. |
|
Bahmyari R, Ariafar A, Sayadi M, Hossieni S, Azima S. The Effect of Daily Intake of Selenium, Vitamin E and Folic Acid on Sperm Parameters in Males with Idiopathic Infertility: A Single-Blind Randomized Controlled Clinical Trial. Int J Fertil Steril. 2021 Jan;15(1):8-14. doi: 10.22074/ijfs.2021.6236. Epub 2021 Jan 19. PMID: 33497041; PMCID: PMC7838762. |
Included in Cochrane review. |
|
Fan Q, Chang FJ, Xue JG, Han ZY, Sun ZX, Chen Y. [Efficacy of Jujing Decoction combined with lipoic acid in the treatment of asthenospermia and teratospermia]. Zhonghua Nan Ke Xue. 2022 Feb;28(2):157-161. Chinese. PMID: 37462489. |
Full text in Chinese, wrong comparison. |
|
Jenkins T, Aston K, Carrell D, DeVilbiss E, Sjaarda L, Perkins N, Mills JL, Chen Z, Sparks A, Clemons T, Chaney K, Peterson CM, Emery B, Hotaling J, Johnstone E, Schisterman E, Mumford SL. The impact of zinc and folic acid supplementation on sperm DNA methylation: results from the folic acid and zinc supplementation randomized clinical trial (FAZST). Fertil Steril. 2022 Jan;117(1):75-85. doi: 10.1016/j.fertnstert.2021.09.009. Epub 2021 Oct 14. PMID: 34656303. |
Secondary analysis of MOXI trial, which is included in Cochrane review. |
|
Akbari H, Elyasi L, Khaleghi AA, Mohammadi M. The effect of zinc supplementation on improving sperm parameters in infertile diabetic men. J Obstet Gynaecol India. 2023 Aug;73(4):316-321. doi: 10.1007/s13224-023-01767-7. Epub 2023 May 25. PMID: 37701089; PMCID: PMC10492728. |
No extractable data. |
|
Safarinejad MR, Safarinejad S. Expression of Concern: Efficacy of Selenium and/or N-Acetyl-Cysteine for Improving Semen Parameters in Infertile Men: A Double-Blind, Placebo Controlled, Randomized Study. J Urol. 2023 Jan 10:101097JU0000000000003113. doi: 10.1097/JU.0000000000003113. Epub ahead of print. PMID: 36626340. |
Wrong design |
|
Liang M, Yang HL, Meng FW, Wang X, Shi BK. [Compound Amino Acid Capsule (8-11) combined with L-carnitine for the treatment of asthenospermia]. Zhonghua Nan Ke Xue. 2023 Mar;29(3):233-238. Chinese. PMID: 38597704. |
Full text in Chinese. |
|
Gharakhani Bahar T, Masoumi SZ, Pilehvari S, Kazemi F, Moradkhani S, Mahmoudi S. Effect of CoQ10 Supplement on Spermogram Parameters and Sexual Function of Infertile Men Referred to The Infertility Center of Fatemieh Hospital, Hamadan, Iran, 2019: A Randomized Controlled Trial Study. Int J Fertil Steril. 2023 Feb 1;17(2):99-106. doi: 10.22074/ijfs.2022.544330.1234. PMID: 36906826; PMCID: PMC10009510. |
No extractable data. |
Beoordelingsdatum en geldigheid
Publicatiedatum : 30-04-2026
Beoordeeld op geldigheid : 30-04-2026
Algemene gegevens
De ontwikkeling/herziening van deze richtlijnmodule werd ondersteund door het Kennisinstituut van de Federatie Medisch Specialisten (www.demedischspecialist.nl/kennisinstituut) en werd gefinancierd door de Stichting Kwaliteitsgelden Medisch Specialisten (SKMS). De financier heeft geen enkele invloed gehad op de inhoud van de richtlijnmodule.
Samenstelling werkgroep
Voor het ontwikkelen van de richtlijnmodule is in 2022 een multidisciplinaire werkgroep ingesteld, bestaande uit vertegenwoordigers van alle relevante specialismen (zie hiervoor de Samenstelling van de werkgroep) die betrokken zijn bij de zorg voor patiënten met mannelijke subfertiliteit.
Werkgroep
- Cantineau A.E.P. (Astrid), gynaecoloog, subspecialist voortplantingsgeneeskunde Universitair Medisch Centrum Groningen te Groningen, NVOG
- Dinkelman-Smit M. (Marij), uroloog-androloog, Erasmus Medisch Centrum te Rotterdam, NVU
- Verberg M.F.G. (Marieke), gynaecoloog, Medisch Spectrum Twente te Enschede, NVOG
- Heida K.Y. (Karst), gynaecoloog, Dijklander ziekenhuisbte Purmerend, NVOG
- van der Houwen L.E.E. (Lisette), gynaecoloog, Radboud universitair medisch centrum te Nijmegen, NVOG
- de Bruin J.P. (Jan Peter), gynaecoloog, Jeroen Bosch Ziekenhuis te ’s Hertogenbosch, NVOG
- van Breda H.M.K. (Jetske), uroloog-androloog, Universitair Medisch Centrum Utrecht te Utrecht, NVU
- van Roijen J.H. (Herman), uroloog-androloog, voorheen ETZ Tilburg – nu gepensioneerd
- D’Hauwers K.W.M. (Kathleen), uroloog-androloog, Radboud universitair medisch centrum te Nijmegen, NVU
- Meißner A. (Andreas), uroloog-androloog, Amsterdam Universitair Medisch Centrum te Amsterdam, NVU
- Bailleux B.B.E.P. (Bart), laboratoriumspecialist Klinische Chemie en Endocrinologie, Leids Universitair Medisch Centrum te Leiden, NVKC
- Kempers M.J.E. (Marlies), klinisch geneticus, Radboud universitair medisch centrum te Nijmegen, VKGN
- van der Kevie-Kersemaekers A.M.F. (Anne-Marie), laboratoriumspecialist klinische genetica, Universitair Medisch Centrum Utrecht te Utrecht, VKGL
- Westra D. (Dineke), laboratoriumspecialist klinische genetica, Radboud universitair medisch centrum te Nijmegen, VKGL
- Ramos L. (Liliana), klinisch embryoloog, Radboud universitair medisch centrum te Nijmegen, KLEM
- Grolle R.L. (Rosanne), fertiliteitsarts, Erasmus Medisch Centrum te Rotterdam, VVF
- Sinjorgo S. (Simone), medewerker medewerker patientenperspectief wetenschappelijk onderzoek en richtlijnen, Freya (vanaf december 2024)
- Vermeulen M. (Marloes), medewerker patientenperspectief wetenschappelijk onderzoek en richtlijnen, Freya (tot december 2024)
Met ondersteuning van
- Mostovaya I.M. (Irina), senior-adviseur, Kennisinstituut van de Federatie Medisch Specialisten
- van der Lee J.H. (Hanneke), senior-adviseur, Kennisinstituut van de Federatie Medisch Specialisten
- Middelhuis D. (Danique), adviseur, Kennisinstituut van de Federatie Medisch Specialisten
- Van Bommel M. (Majke), adviseur, Kennisinstituut van de Federatie Medisch Specialisten
- Schultink J.M. (Janneke), adviseur, Kennisinstituut van de Federatie Medisch Specialisten
- E. (Esther) van der Bijl, informatiespecialist, Kennisinstituut van de Federatie Medisch Specialisten
Belangenverklaringen
Een overzicht van de belangen van werkgroepleden en het oordeel over het omgaan met eventuele belangen vindt u in onderstaande tabel. De ondertekende belangenverklaringen zijn op te vragen bij het secretariaat van het Kennisinstituut van de Federatie Medisch Specialisten via secretariaat@kennisinstituut.nl.
Tabel Gemelde (neven)functies en belangen werkgroep
|
Naam |
Hoofdfunctie |
Nevenwerkzaam-heden |
Persoonlijke financiële belangen |
Persoonlijke relaties |
Extern gefinancierd onderzoek |
Intellectuele belangen en reputatie |
Overige belangen |
Datum |
Restrictie |
|
Astrid Cantineau (voorzitter) |
Gynaecoloog. UMCG, 1.0 FTE |
auteur Up to date Hyperthecosis |
Geen |
Geen |
Geen |
Voorzitter SIG andrologie NVOG aanvrager subsidie ZonMw over leefstijl en mannelijke subfertiliteit |
Geen |
13/11/2022 |
Geen restricties. |
|
Marij Dinkelman-Smit (vice voorzitter) |
Staflid Uroloog-Androloog Voorzitter Voortplantingscentrum Erasmus MC Plaatsvervangend afdelingshoofd Urologie Erasmus MC Erasmus MC Universitair Medisch Centrum |
Bestuurslid Nederlandse Vereniging voor Urologie (NVU) (onbetaald) Bestuurslid multidisciplenaire Special Interest Group (SIG) Andrologie, Nederlandse Verening voor Obstetrie en Gynaecologie (NVOG) (onbetaald) Bestuurslid European Association of Urology (EAU) section for Andrological Urology (ESAU) (onbetaald) Medical director European Academy of Andrology (EAA) training center Andrology Erasmus MC (onbetaald) Lid Commissie Kwaliteitvisitatie NVU (onbetaald) Medische adviesraad Klinefelter Vereniging (onbetaald) Medische adviesraad Stichting Lichen Sclerosus (onbetaald) |
Geen |
Geen |
Geen |
Geen |
Geen |
12/12/2022 |
Geen restricties. |
|
Marieke Verberg |
Gynaecoloog, Medisch Spectrum Twente 0.5 FTE betaalde functie Gynaecoloog en statutair directeur Fertiliteitskliniek Twente 0.5 FTE betaalde functie |
Geen relevante |
Geen |
Geen |
Geen |
Geen |
Geen |
19/11/2022 |
Geen restricties. |
|
Karst Heida |
Gynaecoloog bij het Dijklander ziekenhuis - betaald |
Geen |
Geen |
Geen |
Geen |
Geen |
Geen |
7/12/2022 |
Geen restricties. |
|
Lisette van der Houwen |
Gynaecoloog - fellow voortplantingsgeneeskunde Radboudumc |
Bestuurslid SIG endometriose NVOG algemeen lid SIG ART en SIG fertiliteitspreservatie van de NVOG |
Geen |
Geen |
Merck / Impect study - improving patient-centeredness in endometriosis care. / Ja |
Geen |
Geen |
02/12/2022 |
Geen restricties, extern gefinancierd onderzoek heeft geen betrekking op richtlijn onderwerp. |
|
Jan Peter de Bruin |
Gynaecoloog Jeroen Bosch Ziekenhuis, betaald |
Waarnemend gynaecoloog Radboudumc, betaald |
Medical Advisory Board Ferring BV - De medical advisory board komt op verzoek van Ferring ongeveer eens per jaar samen. Daarbij worden ontwikkelingen in het veld en relevant studie uitkomsten bediscussieerd. Ferring gebruikt de uitkomsten van deze besprekingen als input voor hun strategie. |
- |
Ja - Het gaat om eigen onderzoek op gebied voortplantingsgeneeksunde waarvoor mijn afdeling een unrestricted research grant kreeg. Dit onderzoek bestaat uit de volgende lijnen: • het ontwikkelen en evalueren van toepassingen voor online fertilteitszorg (geen raakvlak met uit te werken modules voor deze richtlijn). • het effect van gebruik van supplementen op de zwangerschapskans bij paren met mannelijke subfertiliteit (raakvlak met module over supplementen waar de Bruin bij betrokken is als locale hoofdonderzoeker, tweede auteur van de nog te publiceren paper) Gefinincierd door Goodlife pharma |
- |
- |
26/11/2022 |
Vanwege deelname aan een adviesraad aan de start van het traject is de richtlijn tijdens de commentaarfase ook aan de Commissie Kwaliteitsdocumenten van de NVOG voorgelegd. Verder is dit lid uitgesloten van besluitvorming bij onderwerpen die raken aan de adviesraad. |
|
Jetske van Breda |
Uroloog/ Androloog UMCUtrecht |
Geen |
Geen |
Geen |
Geen |
Geen |
Geen |
18/11/2022 |
Geen restricties. |
|
Kathleen D' Hauwers |
Uroloog, staflid Radboudumc: betaald |
Lid Cie Kwaliteit NVU: onbetaald Lid Cie Andrologie NVU: onbetaald Board memeber Klinefelter Vereniging Nederland: onbeteaald Cie Keuzehulp ED: vergoed |
Geen |
Geen |
Geen |
Inbrengen van expertise. Geen specifieke, persoonlijke baat bij één of ander: het gaat om de patiënt / klant. |
Geen |
28/09/2022 |
Geen restricties. |
|
Andreas Meißner |
Uroloog - Androloog Staflid Centrum voor Voortplantingsgeneeskunde, locatie AMC, en afdeling Urologie, locatie VUMC, van het Amsterdam UMC betaald |
Geen |
Geen |
Geen |
Geen |
Geen |
Geen |
24/11/2022 |
Geen restricties. |
|
Herman van Roijen |
Uroloog ETZ Tilburg |
Geen |
Geen |
Geen |
Geen |
Geen |
Geen |
13/11/2022 |
Geen restricties. |
|
Bart Ballieux |
Laboratoriumspecialist Klinische Chemie en Endocrinologie Afdeling KCL, LUMC, Leiden |
Geen |
geen persoonlijke financiele belangen |
Geen relevante persoonlijke relaties die van invloed kunnen zijn op mijn adviezen |
geen |
Geen intellectuele belangen bij mijn adviezen |
Geen overige belangen |
10/01/2023 |
Geen restricties. |
|
Marlies Kempers |
Klinisch Geneticus (Radboudumc) |
Voorzitter subcommissie Richtlijnen, onderdeel van Kwaliteitscommissie VKGN |
Geen |
Geen |
Aytu Pharma / Prevent studie (effect Enzastaurin bij vasculair EDS) Studie is nog niet gestart, financiering on hold / Tijdelijk, ter vervanging verlof projectleider |
Geen |
Geen |
10/11/2022 |
Geen restricties. De studie waar mw. Kempers bij betrokken was is nooit gestart. Inhoudelijk had deze studie niet met mannelijke infertiliteit te maken. |
|
Anne-Marie van der Kevie - Kersemaekers |
laboratoriumspecialist klinische genetica UMC Utrecht |
Vakdeskundige Raad van Accreditatie |
Geen |
Geen |
Geen |
Geen |
Geen |
28/03/2023 |
Geen restricties. |
|
Manon Oud |
Laboratoriumspecialist Klinische Genetica in opleiding, Radboudumc Betaalde functie (hoofdberoep) |
Geen |
Geen |
Geen |
Deelname aan onderzoeksproject als PhD student De novo mutations in male infertility (NWO VICI van prof. Joris Veltman) van 2015-2020 Travel Grant van de Catherine van Tussenbroek Foundation voor bezoek van 3 maanden aan Monash University (2019) |
Geen |
Geen |
8/11/2022 |
Geen restricties, niet actief geweest in de werkgroep. |
|
Dineke Westra |
Laboratoriumspecialist Klinische Genetica (1.0 FTE, betaald) Afd. Genetica, sectie Genoomdiagnostiek Radboudumc, Nijmegen Werkzaamheden: Genetische diagnostiek |
Technical Assessor ISO15189 Raad van Accreditatie betaald |
Geen |
Geen |
Geen |
Geen |
Geen |
16/02/2023 |
Geen restricties. |
|
Liliana Ramos |
Klinisch embryoloog Radboudumc |
Geen |
Geen |
Geen |
Geen |
Boegbeeldfunctie bij patiënten en beroepsorganisatie |
Geen |
10/08/2023 |
Geen restricties. |
|
Rosanne Grolle |
Fertiliteitsarts, Erasmus MC. Het begeleiden en behandelen van paren met een kinderwens. Functie is betaald. |
Lid van de symposiumcommissie van de VVF Onder andere het opstellen van het wetenschappelijk programma, het vragen van sprekers, het regelen van een symposiumlocatie en buffet, het versturen van de uitnodigingen en het aanvragen van accreditatie en sponsoring. Functie is onbetaald. |
Geen |
Geen |
Geen |
Geen |
Geen |
3/11/2022 |
Geen restricties. |
|
Marloes Vermeulen |
Freya, medewerker externe relaties |
Verloskundige 1e lijn. |
Geen |
Geen |
Geen |
Geen |
Geen |
27/09/2022 |
Geen restricties. |
|
José Knijnenburg |
Directeur bij Freya, vereniging voor mensen met vruchtbaarheidsproblemen - 32u p/wk Medewerker communicatie en marketing bij van der Pol-consulting b.v. - 8u p/wk |
Geen |
Dienstverband bij de (onafhankelijke) landelijke patiëntenvereniging voor mensen met vruchtbaarheidsproblemen. |
Geen |
ZonMW / Alife2 en T4Life / nee ZonMW / Antarctica2 / nee ZonMW / Cosy - Zin en zwanger / nee ZonMW / FOAM / nee ZonMW / Scratch-OFO / nee ZonMW / TOF (3 or 5) / nee ZonMW / H2Olie / nee ZonMW / H2OlieFlush / nee ZonMW / MYPP - myo-inositol bij PCOS / nee ZonMW / PSIDER Embryonic checkpoints - releasing the brakes on IVF /nee RadboudUMC/Goodlife / Summer - voedingssupplement man / nee ZonMW / Divine - dosiscalculator / nee Amsterdam UMC / ANDES / nee ZonMW / PSIDER - Hip gametes / nee ZonMW / COPIE - - endometriose en IVF / nee |
Belangenbehartiger bij patiëntenorganisatie |
Radboud/MUMC / Hmove sporten en afweersysteem bij miskramen / nee ZonMW / Medium2 kweekvloeistof IVF / nee ZonMW / Selectimo timelapse / nee MUMC+ / Murlm hhmk en ril / nee ZonMw / LUMO luteal phase support / nee ZonMw / PSIDER SteMBlast / nee ZonMw / SEGa veiligheid vp-technieken / nee ZonMw / Covid-19 Impact endometriose/fertiliteitspatiënten / nee ZonMw / PREMI / nee ZonMw / HyFosy-HSG stugie / nee ZonMw / REMI III / nee |
20/07/2023 |
Geen restricties, mevrouw was tijdelijke inval tijdens zwangerschapsverlof. In rol van patientvertegenwoordiger betrokken bij onderzoek. |
|
Simone Sinjorgo |
Parttime medewerker patientenperspectief en -participatie bij Freya (vereniging voor mensen met vruchtbaarheidsproblemen) - betaald 14 uur https://www.freya.nl/over-freya/wie-we-zijn/de-werknemers. Ter info: door mijn functie bij Freya ben ik betrokken bij diverse richtlijnontwikkeling en/of onderzoeken op het gebied van fertiliteit. In de vorm van toelichten, belichten en/of vertegenwoordigen van alleen het patiëntenperspectief tav het bepaalde onderwerp, zoals bij deze 3 richtlijnen OFO, OHSS en mannelijke subfertiliteit. |
Lichaamsgericht psychosociaal therapeut - zzp eigen praktijk in Dongen-Vaart - geregistreerd therapeut RBCZ incl. AGB-codes. www.praktijkdediamant.nl
- Psychosociaal docent Bijscholing Instituut voor therapeuten - betaling via praktijk https://bivt.nl/?s=lichaamsgericht+coachen
- Psychosociaal docent vrouwencoach opleiding - betaling via praktijk https://www.devrouwencoach.nl/vrouwencoach-opleiding/ |
Geen |
Geen |
Geen |
Geen |
Geen |
03/12/2024 |
Geen restricties. |
Inbreng patiëntenperspectief
De werkgroep besteedde aandacht aan het patiëntenperspectief door uitnodigen van Freya en de Patientenfederatie Nederland voor de invitational conference. Daarnaast zat namens Freya een afgevaardigde in de werkgroep. Het verslag van de invitational conference is besproken in de werkgroep. De verkregen input is meegenomen bij het opstellen van de uitgangsvragen, de keuze voor de uitkomstmaten en bij het opstellen van de overwegingen (zie kop waarden en voorkeuren van patiënten). De conceptrichtlijn is tevens voor commentaar voorgelegd aan Freya en de Patiëntenfederatie Nederland en de eventueel aangeleverde commentaren zijn bekeken en verwerkt.
Kwalitatieve raming van mogelijke financiële gevolgen in het kader van de Wkkgz
Bij de richtlijnmodule voerde de werkgroep conform de Wet kwaliteit, klachten en geschillen zorg (Wkkgz) een kwalitatieve raming uit om te beoordelen of de aanbevelingen mogelijk leiden tot substantiële financiële gevolgen. Bij het uitvoeren van deze beoordeling is de richtlijnmodule op verschillende domeinen getoetst (zie het stroomschema bij Werkwijze).
|
Module |
Uitkomst raming |
Toelichting |
|
Supplementen |
Geen financiele gevolgen. |
Uit de toetsing volgt dat de aanbevelingen niet breed toepasbaar zijn (<5.000 patiënten) en daarom naar verwachting geen substantiële financiële gevolgen zullen hebben voor de collectieve uitgaven. |
Werkwijze
Voor meer details over de gebruikte richtlijnmethodologie verwijzen wij u naar de Werkwijze. Relevante informatie voor de ontwikkeling/herziening van deze richtlijnmodule is hieronder weergegeven.
Zoekverantwoording
Zoekstrategie - 3 april 2024
Embase.com
|
No. |
Query |
Results |
|
#1 |
'male infertility'/exp OR 'male fertility'/exp OR (('subfertility'/de OR 'childlessness'/exp) AND 'male'/exp) OR (((male* OR men OR man) NEAR/3 (infertil* OR hypofertil* OR 'fertil*' OR subfertil* OR infecund* OR childless* OR reproduct*)):ti,ab,kw) OR (((spermatogen* OR spermiation OR spermiogen*) NEAR/3 failure):ti,ab,kw) OR ((spermatozoon NEAR/3 (abnormalit* OR anomal*)):ti,ab,kw) OR (((semen OR sperm) NEAR/3 (qualit* OR quantit* OR parameter*)):ti,ab,kw) OR asperm*:ti,ab,kw OR asthenazoosperm*:ti,ab,kw OR asthenosperm*:ti,ab,kw OR asthenozoosperm*:ti,ab,kw OR azoosperm*:ti,ab,kw OR cryptosperm*:ti,ab,kw OR cryptozoosperm*:ti,ab,kw OR hyposperm*:ti,ab,kw OR oligoasthenosperm*:ti,ab,kw OR oligosperm*:ti,ab,kw OR oligozoosperm*:ti,ab,kw OR 'retrograde ejaculat*':ti,ab,kw OR 'sco syndrome*':ti,ab,kw OR 'sertoli cell only syndrome*':ti,ab,kw OR teratozoosperm*:ti,ab,kw OR 'thermosensitive genic male steril*':ti,ab,kw OR globozoosperm*:ti,ab,kw OR macrozoosperm*:ti,ab,kw |
111051 |
|
#2 |
'supplementation'/exp OR 'diet supplementation'/exp OR 'iron therapy'/exp OR 'mineral supplementation'/exp OR 'nutritional support'/exp OR 'vitamin supplementation'/exp OR 'zinc'/exp OR 'selenium'/exp OR 'glutathione'/exp OR 'folic acid'/exp OR 'ubiquinone'/exp OR 'carnitine'/exp OR 'carotenoid'/exp OR 'astaxanthin'/exp OR 'lycopene'/exp OR 'menevit'/exp OR 'multivitamin'/exp OR 'beta carotene'/exp OR 'ascorbic acid'/exp OR 'acetylcysteine'/exp OR 'ethylcysteine'/exp OR 'alpha tocopherol'/exp OR 'fish oil'/exp OR 'omega amino acid'/exp OR 'fatty acid'/exp OR 'vegetable oil'/exp OR 'pycnogenol'/exp OR 'xanthophyll'/exp OR 'thioctic acid'/exp OR 'antioxidant'/exp OR 'vitamin'/exp OR 'vitamin b group'/exp OR 'arginine'/exp OR 'flavonoid'/exp OR 'riboflavin'/exp OR 'edible oil'/exp OR 'castor oil'/exp OR 'lyprinol'/exp OR 'olive oil'/exp OR 'safflower oil'/exp OR 'essential fatty acid'/exp OR 'arachidonic acid'/exp OR 'linoleic acid'/exp OR 'unsaturated fatty acid'/exp OR 'linolenic acid'/exp OR 'gamma linolenic acid'/exp OR 'omega 6 fatty acid'/exp OR 'polyunsaturated fatty acid'/exp OR 'cod liver oil'/exp OR 'omega 3 fatty acid'/exp OR 'inositol'/exp OR 'docosahexaenoic acid'/exp OR 'magnesium'/exp OR 'melatonin'/exp OR 'nutraceutical'/exp OR (((nutritional OR iron) NEAR/3 (therap* OR treatment* OR support*)):ti,ab,kw) OR (((plant* OR seed* OR vegetable* OR dietary OR edible OR ricinus OR cast?r OR ricin* OR olive* OR safflower OR fish OR tuna) NEAR/3 (oil* OR fat)):ti,ab,kw) OR 'supplement*':ti,ab,kw OR vitamin*:ti,ab,kw OR 'zinc':ti,ab,kw OR 'zincum':ti,ab,kw OR 'selenicum':ti,ab,kw OR 'selenium':ti,ab,kw OR 'glutathine':ti,ab,kw OR 'glutathiol':ti,ab,kw OR 'glutathion':ti,ab,kw OR 'glutathione':ti,ab,kw OR 'glutathioral':ti,ab,kw OR 'glutatiol':ti,ab,kw OR 'tathion':ti,ab,kw OR 'tationil':ti,ab,kw OR 'tiobione':ti,ab,kw OR 'acfol':ti,ab,kw OR 'acifolic':ti,ab,kw OR 'filicine':ti,ab,kw OR 'folacid*':ti,ab,kw OR 'folacin':ti,ab,kw OR 'folart':ti,ab,kw OR 'folate':ti,ab,kw OR 'folavit':ti,ab,kw OR 'foldine':ti,ab,kw OR 'foliamin':ti,ab,kw OR 'folicet':ti,ab,kw OR 'folicid':ti,ab,kw OR 'folina':ti,ab,kw OR 'folinsyre':ti,ab,kw OR 'folitab':ti,ab,kw OR 'folivit':ti,ab,kw OR 'folsan':ti,ab,kw OR 'folsav':ti,ab,kw OR 'folverlan':ti,ab,kw OR 'folvite':ti,ab,kw OR 'ingafol':ti,ab,kw OR 'lafol':ti,ab,kw OR 'lexpec':ti,ab,kw OR 'megafol':ti,ab,kw OR 'neocepri':ti,ab,kw OR 'rubiefol':ti,ab,kw OR 'sodium folate':ti,ab,kw OR 'speciafoldine':ti,ab,kw OR 'vifolin':ti,ab,kw OR 'ubiquinone':ti,ab,kw OR 'coenzyme q*':ti,ab,kw OR 'abedine':ti,ab,kw OR 'biocarn':ti,ab,kw OR 'biomux':ti,ab,kw OR 'cardiogen':ti,ab,kw OR 'carnicor':ti,ab,kw OR 'carnil':ti,ab,kw OR 'carnitene*':ti,ab,kw OR 'carnitin':ti,ab,kw OR 'carnitine*':ti,ab,kw OR 'carnitor':ti,ab,kw OR 'carnovis':ti,ab,kw OR 'cartin':ti,ab,kw OR 'entomin':ti,ab,kw OR 'lefcar':ti,ab,kw OR 'monocamin':ti,ab,kw OR 'nefrocarnit':ti,ab,kw OR 'novain':ti,ab,kw OR 'vitacarn':ti,ab,kw OR 'carotenoid*':ti,ab,kw OR 'carotinoid*':ti,ab,kw OR 'astaxanthin*':ti,ab,kw OR 'astaxanthine*':ti,ab,kw OR 'ovoester':ti,ab,kw OR 'lycopene*':ti,ab,kw OR 'menevit':ti,ab,kw OR 'multivitamin*':ti,ab,kw OR 'b-tene':ti,ab,kw OR 'beta carotene':ti,ab,kw OR 'beta carotin*':ti,ab,kw OR 'betacarotene*':ti,ab,kw OR 'betatene*':ti,ab,kw OR 'carotaben':ti,ab,kw OR 'lurotin':ti,ab,kw OR 'solatene':ti,ab,kw OR 'solvin':ti,ab,kw OR 'acidylina':ti,ab,kw OR 'adenex':ti,ab,kw OR 'agrumina':ti,ab,kw OR 'allercorb':ti,ab,kw OR 'allescorb':ti,ab,kw OR 'arcavit c':ti,ab,kw OR 'arcavite c':ti,ab,kw OR 'arkovital c':ti,ab,kw OR 'ascelat':ti,ab,kw OR 'ascofar':ti,ab,kw OR 'ascomed':ti,ab,kw OR 'asconvita':ti,ab,kw OR 'ascor':ti,ab,kw OR 'ascorbate':ti,ab,kw OR 'ascorbate sodium':ti,ab,kw OR 'ascorbic acid*':ti,ab,kw OR 'ascorbicap':ti,ab,kw OR 'ascorbicin':ti,ab,kw OR 'ascorbico':ti,ab,kw OR 'ascorbin':ti,ab,kw OR 'ascorbina':ti,ab,kw OR 'ascorbinic acid*':ti,ab,kw OR 'ascorbit':ti,ab,kw OR 'ascorbite':ti,ab,kw OR 'ascorbitol':ti,ab,kw OR 'ascorbivit':ti,ab,kw OR 'ascorbivite':ti,ab,kw OR 'ascorbone':ti,ab,kw OR 'ascorbutina':ti,ab,kw OR 'ascorbyl':ti,ab,kw OR 'ascorbyn':ti,ab,kw OR 'ascorcee':ti,ab,kw OR 'ascorgil':ti,ab,kw OR 'ascorin':ti,ab,kw OR 'ascormin':ti,ab,kw OR 'ascorteal':ti,ab,kw OR 'ascorval':ti,ab,kw OR 'ascorvel':ti,ab,kw OR 'ascorvit':ti,ab,kw OR 'ascorvite':ti,ab,kw OR 'ascorvitina':ti,ab,kw OR 'askorbin':ti,ab,kw OR 'austrovit c':ti,ab,kw OR 'austrovite c':ti,ab,kw OR 'bentavit c':ti,ab,kw OR 'bentavite c':ti,ab,kw OR 'cantan':ti,ab,kw OR 'cantaxin':ti,ab,kw OR 'cebetate':ti,ab,kw OR 'cebicure':ti,ab,kw OR 'cebiolon':ti,ab,kw OR 'cebion':ti,ab,kw OR 'cebione':ti,ab,kw OR 'cecap':ti,ab,kw OR 'cecon':ti,ab,kw OR 'cecone':ti,ab,kw OR 'cecorbin':ti,ab,kw OR 'cecorbine':ti,ab,kw OR 'cecorbyl':ti,ab,kw OR 'cecorbyle':ti,ab,kw OR 'cecrisina':ti,ab,kw OR 'cedon':ti,ab,kw OR 'cedone':ti,ab,kw OR 'cedoxon':ti,ab,kw OR 'cedoxone':ti,ab,kw OR 'ceevifil':ti,ab,kw OR 'cegiolan':ti,ab,kw OR 'celaskon':ti,ab,kw OR 'celaskone':ti,ab,kw OR 'celin':ti,ab,kw OR 'celine':ti,ab,kw OR 'cenetone':ti,ab,kw OR 'cenol':ti,ab,kw OR 'cenolate':ti,ab,kw OR 'cequinyl':ti,ab,kw OR 'cereon':ti,ab,kw OR 'cergona':ti,ab,kw OR 'cescorbat':ti,ab,kw OR 'cetamican':ti,ab,kw OR 'cetamid':ti,ab,kw OR 'cetamine':ti,ab,kw OR 'cetebe':ti,ab,kw OR 'ceterapion':ti,ab,kw OR 'ceterapione':ti,ab,kw OR 'cetrinets':ti,ab,kw OR 'cevalin':ti,ab,kw OR 'cevaline':ti,ab,kw OR 'cevatine':ti,ab,kw OR 'cevex':ti,ab,kw OR 'cevibram':ti,ab,kw OR 'cevigal':ti,ab,kw OR 'cevigen':ti,ab,kw OR 'cevigol':ti,ab,kw OR 'cevikap':ti,ab,kw OR 'cevilat':ti,ab,kw OR 'cevimin':ti,ab,kw OR 'cevimine':ti,ab,kw OR 'cevisol':ti,ab,kw OR 'cevit':ti,ab,kw OR 'cevita':ti,ab,kw OR 'cevitamin*':ti,ab,kw OR 'cevitan':ti,ab,kw OR 'cevite':ti,ab,kw OR 'cevitex':ti,ab,kw OR 'cevitil':ti,ab,kw OR 'cevitol':ti,ab,kw OR 'cewin':ti,ab,kw OR 'chewcee':ti,ab,kw OR 'ci drol':ti,ab,kw OR 'ciamin':ti,ab,kw OR 'ciergin':ti,ab,kw OR 'cifilina':ti,ab,kw OR 'cipca':ti,ab,kw OR 'cisir':ti,ab,kw OR 'citamino':ti,ab,kw OR 'cith':ti,ab,kw OR 'citoascorbina':ti,ab,kw OR 'citoxyl':ti,ab,kw OR 'citran':ti,ab,kw OR 'citravite':ti,ab,kw OR 'citritabs':ti,ab,kw OR 'citrovitamin*':ti,ab,kw OR 'civigor':ti,ab,kw OR 'civitin':ti,ab,kw OR 'civitine':ti,ab,kw OR 'concemin':ti,ab,kw OR 'cortalex':ti,ab,kw OR 'dagrascorbin':ti,ab,kw OR 'dagravit c':ti,ab,kw OR 'dancimin c':ti,ab,kw OR 'davitamon c':ti,ab,kw OR 'dayvital':ti,ab,kw OR 'delo c':ti,ab,kw OR 'difvitamin* c':ti,ab,kw OR 'dropice':ti,ab,kw OR 'dumovit c':ti,ab,kw OR 'dumovite c':ti,ab,kw OR 'esuron':ti,ab,kw OR 'esurvit':ti,ab,kw OR 'esurvite':ti,ab,kw OR 'flavettes':ti,ab,kw OR 'hicee':ti,ab,kw OR 'hybrin':ti,ab,kw OR 'ikacee':ti,ab,kw OR 'irocevit':ti,ab,kw OR 'irocevite':ti,ab,kw OR 'jarexin':ti,ab,kw OR 'jarexine':ti,ab,kw OR 'lacivit':ti,ab,kw OR 'lacivite':ti,ab,kw OR 'laroscorbine':ti,ab,kw OR 'lemascorb':ti,ab,kw OR 'levo ascorbate':ti,ab,kw OR 'levo ascorbic acid*':ti,ab,kw OR 'limcee':ti,ab,kw OR 'limo ce':ti,ab,kw OR 'liqui cee':ti,ab,kw OR 'myascorbin':ti,ab,kw OR 'natrascorb':ti,ab,kw OR 'novo ascorbic':ti,ab,kw OR 'nybadol':ti,ab,kw OR 'pascorbin':ti,ab,kw OR 'pharmascorbine':ti,ab,kw OR 'potassium ascorbate':ti,ab,kw OR 'proscorbin':ti,ab,kw OR 'proscorbine':ti,ab,kw OR 'redoxon':ti,ab,kw OR 'redoxon forte':ti,ab,kw OR 'ribena':ti,ab,kw OR 'scorbacid*':ti,ab,kw OR 'scorbettes':ti,ab,kw OR 'scorbex':ti,ab,kw OR 'scorbitol':ti,ab,kw OR 'scorbumine':ti,ab,kw OR 'secorbate':ti,ab,kw OR 'sevalin':ti,ab,kw OR 'sodascorbate':ti,ab,kw OR 'sodium ascorbate':ti,ab,kw OR 'sweetcee':ti,ab,kw OR 'testascorbic':ti,ab,kw OR 'upsavit':ti,ab,kw OR 'vicef':ti,ab,kw OR 'vicelat':ti,ab,kw OR 'vicetrin':ti,ab,kw OR 'viciman':ti,ab,kw OR 'vicin':ti,ab,kw OR 'vicitina':ti,ab,kw OR 'vicon':ti,ab,kw OR 'viforcit':ti,ab,kw OR 'viforcite':ti,ab,kw OR 'viscorin':ti,ab,kw OR 'viscorine':ti,ab,kw OR 'vitac':ti,ab,kw OR 'vitace':ti,ab,kw OR 'vitacee':ti,ab,kw OR 'vitaci':ti,ab,kw OR 'vitacimin':ti,ab,kw OR 'vitacimine':ti,ab,kw OR 'vitacin':ti,ab,kw OR 'vitacine':ti,ab,kw OR 'vitaplex c':ti,ab,kw OR 'vitapric':ti,ab,kw OR 'vitapur c':ti,ab,kw OR 'vitasan c':ti,ab,kw OR 'vitascorbin':ti,ab,kw OR 'vitascorbine':ti,ab,kw OR 'vitascorbol':ti,ab,kw OR 'vitelix c':ti,ab,kw OR 'vitocee':ti,ab,kw OR 'vorange':ti,ab,kw OR 'wandervit c':ti,ab,kw OR 'wandervite c':ti,ab,kw OR 'witamina c':ti,ab,kw OR 'xitix':ti,ab,kw OR 'xyloascorbic acid*':ti,ab,kw OR 'acerac':ti,ab,kw OR 'acetadote':ti,ab,kw OR 'acetain':ti,ab,kw OR 'acetyl cysteine':ti,ab,kw OR 'acetylcystein':ti,ab,kw OR 'acetylcysteine':ti,ab,kw OR 'acypront':ti,ab,kw OR 'airbron':ti,ab,kw OR 'alveolex':ti,ab,kw OR 'bromuc':ti,ab,kw OR 'bronchocil':ti,ab,kw OR 'brunac':ti,ab,kw OR 'cetilan':ti,ab,kw OR 'cetylev':ti,ab,kw OR 'drenaflen':ti,ab,kw OR 'ecomucyl':ti,ab,kw OR 'euronac':ti,ab,kw OR 'exomuc':ti,ab,kw OR 'fabrol':ti,ab,kw OR 'fluimicil':ti,ab,kw OR 'fluimucil':ti,ab,kw OR 'fluimukan':ti,ab,kw OR 'flumil':ti,ab,kw OR 'flumil infantil':ti,ab,kw OR 'fluprowit':ti,ab,kw OR 'flutafin':ti,ab,kw OR 'genac':ti,ab,kw OR 'granon':ti,ab,kw OR 'hidonac':ti,ab,kw OR 'ilube':ti,ab,kw OR 'inspir':ti,ab,kw OR 'lappe':ti,ab,kw OR 'libramucil':ti,ab,kw OR 'lysomucil':ti,ab,kw OR 'lysox':ti,ab,kw OR 'menaxol':ti,ab,kw OR 'mercapturic acid*':ti,ab,kw OR 'mucocil':ti,ab,kw OR 'mucodrill':ti,ab,kw OR 'mucofillin':ti,ab,kw OR 'mucolator':ti,ab,kw OR 'mucomiste':ti,ab,kw OR 'mucomyst':ti,ab,kw OR 'mucomystendo':ti,ab,kw OR 'mucopect':ti,ab,kw OR 'mucoserin':ti,ab,kw OR 'mucosil':ti,ab,kw OR 'mucosof':ti,ab,kw OR 'mucosolvin':ti,ab,kw OR 'mucosten':ti,ab,kw OR 'mucoza':ti,ab,kw OR 'mukolit':ti,ab,kw OR 'muteran':ti,ab,kw OR 'parvolex':ti,ab,kw OR 'pulmosal':ti,ab,kw OR 'reolin':ti,ab,kw OR 'respaire':ti,ab,kw OR 'rinofluimucil':ti,ab,kw OR 'sigamucil':ti,ab,kw OR 'solmucol':ti,ab,kw OR 'spatam':ti,ab,kw OR 'sputoprompt':ti,ab,kw OR 'stecin':ti,ab,kw OR 'tirocular':ti,ab,kw OR 'tixair':ti,ab,kw OR 'zifluvis':ti,ab,kw OR 'ethylcysteine':ti,ab,kw OR 'alpha tocopherol*':ti,ab,kw OR 'alpha tocopheryl acetate':ti,ab,kw OR 'alpha tocopheryl acetic acid*':ti,ab,kw OR 'aquasol e':ti,ab,kw OR 'austrovit e':ti,ab,kw OR 'covitol':ti,ab,kw OR 'dagravit e':ti,ab,kw OR 'dalfatol':ti,ab,kw OR 'davitamon e':ti,ab,kw OR 'dermorelle':ti,ab,kw OR 'detulin':ti,ab,kw OR 'dumovit e':ti,ab,kw OR 'e ferol':ti,ab,kw OR 'e perle':ti,ab,kw OR 'e perte':ti,ab,kw OR 'e recordati':ti,ab,kw OR 'e toplex':ti,ab,kw OR 'e vicotrat':ti,ab,kw OR 'e vimin':ti,ab,kw OR 'e vita':ti,ab,kw OR 'e viterbin':ti,ab,kw OR 'ecoferol':ti,ab,kw OR 'efer':ti,ab,kw OR 'eferol':ti,ab,kw OR 'enoulan forte':ti,ab,kw OR 'ephynal':ti,ab,kw OR 'eplonat':ti,ab,kw OR 'eprolin':ti,ab,kw OR 'erevit':ti,ab,kw OR 'esol':ti,ab,kw OR 'esorb':ti,ab,kw OR 'eterapion':ti,ab,kw OR 'eviabit':ti,ab,kw OR 'evigen':ti,ab,kw OR 'eviol':ti,ab,kw OR 'evion':ti,ab,kw OR 'evit':ti,ab,kw OR 'evitol':ti,ab,kw OR 'godabion e':ti,ab,kw OR 'gonavit':ti,ab,kw OR 'hanobak':ti,ab,kw OR 'ido e':ti,ab,kw OR 'juvela':ti,ab,kw OR 'juvele':ti,ab,kw OR 'livingpherol':ti,ab,kw OR 'metaperex':ti,ab,kw OR 'mulsal e':ti,ab,kw OR 'natopherol':ti,ab,kw OR 'optovit e':ti,ab,kw OR 'phytoferol':ti,ab,kw OR 'pletocol':ti,ab,kw OR 'socopherol':ti,ab,kw OR 'spondyvit':ti,ab,kw OR 'toco 500':ti,ab,kw OR 'tocoferolo bioglan':ti,ab,kw OR 'tocomine':ti,ab,kw OR 'tocopherex':ti,ab,kw OR 'tocopherol acetate':ti,ab,kw OR 'tocopherol gumpro':ti,ab,kw OR 'tocopheryl acetate':ti,ab,kw OR 'tocophrine':ti,ab,kw OR 'tocovigor':ti,ab,kw OR 'tocovital':ti,ab,kw OR 'toferol':ti,ab,kw OR 'topherol':ti,ab,kw OR 'vibolex e':ti,ab,kw OR 'vidom e':ti,ab,kw OR 'viea':ti,ab,kw OR 'vietal':ti,ab,kw OR 'viprimol':ti,ab,kw OR 'viteolin':ti,ab,kw OR 'viteoline':ti,ab,kw OR 'wandervit e':ti,ab,kw OR 'ameu':ti,ab,kw OR 'efamed':ti,ab,kw OR 'epax':ti,ab,kw OR 'feniko':ti,ab,kw OR 'himega':ti,ab,kw OR 'k 85':ti,ab,kw OR 'lachs 550':ti,ab,kw OR 'lipitac':ti,ab,kw OR 'maxepa':ti,ab,kw OR 'olemar':ti,ab,kw OR 'omegaven':ti,ab,kw OR 'optimepa':ti,ab,kw OR 'pikasol':ti,ab,kw OR 'promega':ti,ab,kw OR 'super epa':ti,ab,kw OR 'superepa':ti,ab,kw OR 'omega*':ti,ab,kw OR 'fat* acid*':ti,ab,kw OR 'arginin*':ti,ab,kw OR 'arginyl residue':ti,ab,kw OR 'argivene':ti,ab,kw OR 'bioarginina':ti,ab,kw OR 'r gene 10':ti,ab,kw OR 'flavonoid*':ti,ab,kw OR 'beflavin*':ti,ab,kw OR 'beflavine*':ti,ab,kw OR 'flavaxin':ti,ab,kw OR 'hyrye':ti,ab,kw OR 'lactoflavin*':ti,ab,kw OR 'ovoflavin':ti,ab,kw OR 'pabriflan':ti,ab,kw OR 'riboflavin*':ti,ab,kw OR 'ribovel':ti,ab,kw OR 'leucocianidol':ti,ab,kw OR 'leucocyanidin*':ti,ab,kw OR 'leucocyanidol':ti,ab,kw OR 'pycnogenol':ti,ab,kw OR 'pygnoforton':ti,ab,kw OR 'pyknogenol':ti,ab,kw OR 'lutein*':ti,ab,kw OR 'xanthophyl*':ti,ab,kw OR 'xantofyl*':ti,ab,kw OR 'xantophyl*':ti,ab,kw OR 'berlithione':ti,ab,kw OR 'biletan':ti,ab,kw OR 'heparlipon':ti,ab,kw OR 'lipoate':ti,ab,kw OR 'lipoic acid*':ti,ab,kw OR 'liponic acid*':ti,ab,kw OR 'neurothioct':ti,ab,kw OR 'protogen':ti,ab,kw OR 'thioctacid*':ti,ab,kw OR 'thioctan':ti,ab,kw OR 'thioctic acid*':ti,ab,kw OR 'thiogamma':ti,ab,kw OR 'thiotocid':ti,ab,kw OR 'tioctan':ti,ab,kw OR 'tioctic acid*':ti,ab,kw OR 'tioctidasi':ti,ab,kw OR 'antioxidant*':ti,ab,kw OR 'antioxidat*':ti,ab,kw OR 'antoxidant*':ti,ab,kw OR 'radical scavenger*':ti,ab,kw OR 'neoloid':ti,ab,kw OR 'lyprinol':ti,ab,kw OR 'arachidonate*':ti,ab,kw OR 'arachidonate sodium':ti,ab,kw OR 'arachidonic acid*':ti,ab,kw OR 'sodium arachidonate*':ti,ab,kw OR 'linoic acid*':ti,ab,kw OR 'linolate*':ti,ab,kw OR 'linoleate*':ti,ab,kw OR 'linoleic acid*':ti,ab,kw OR 'linolic acid*':ti,ab,kw OR 'sodium linoleate*':ti,ab,kw OR 'ufa':ti,ab,kw OR 'unsaturated fat*':ti,ab,kw OR 'unsaturated lipid*':ti,ab,kw OR 'linolenate*':ti,ab,kw OR 'linolenic acid*':ti,ab,kw OR 'efagel':ti,ab,kw OR 'polyunsaturat* fat*':ti,ab,kw OR 'cod* liver oil*':ti,ab,kw OR 'eicosapen':ti,ab,kw OR 'epaisdin':ti,ab,kw OR 'epanova':ti,ab,kw OR 'omega 3':ti,ab,kw OR 'omega forte':ti,ab,kw OR 'sakana':ti,ab,kw OR 'betitol':ti,ab,kw OR 'cyclohexanehexol':ti,ab,kw OR 'cyclohexitol':ti,ab,kw OR 'dambose':ti,ab,kw OR 'hexol':ti,ab,kw OR 'inosil':ti,ab,kw OR 'inosimesol':ti,ab,kw OR 'inosit*':ti,ab,kw OR 'meat sugar':ti,ab,kw OR 'mesitol':ti,ab,kw OR 'meso inosit*':ti,ab,kw OR 'mesoinosil':ti,ab,kw OR 'mesoinosit*':ti,ab,kw OR 'mesol':ti,ab,kw OR 'mesovil':ti,ab,kw OR 'muscle sugar':ti,ab,kw OR 'myoinosil':ti,ab,kw OR 'myoinosit*':ti,ab,kw OR 'nucite':ti,ab,kw OR 'phaseomannite':ti,ab,kw OR 'phaseomannitol':ti,ab,kw OR 'scyllite':ti,ab,kw OR 'tonozil':ti,ab,kw OR 'dhasco':ti,ab,kw OR 'docosahexaenoate':ti,ab,kw OR 'docosahexaenoic acid*':ti,ab,kw OR 'docosahexenoic acid*':ti,ab,kw OR 'magnesium':ti,ab,kw OR 'romag':ti,ab,kw OR 'adaflex':ti,ab,kw OR 'aritonin':ti,ab,kw OR 'ceyestaeusom':ti,ab,kw OR 'ceyesto':ti,ab,kw OR 'circadin':ti,ab,kw OR 'civasta':ti,ab,kw OR 'eusom':ti,ab,kw OR 'lestinora':ti,ab,kw OR 'mallozen':ti,ab,kw OR 'mecastrin':ti,ab,kw OR 'melabiorytm':ti,ab,kw OR 'melatal':ti,ab,kw OR 'melatan':ti,ab,kw OR 'melatol':ti,ab,kw OR 'melatonin*':ti,ab,kw OR 'melatonite':ti,ab,kw OR 'mellaras':ti,ab,kw OR 'mellozzan':ti,ab,kw OR 'melovine':ti,ab,kw OR 'mucomel':ti,ab,kw OR 'noxarem':ti,ab,kw OR 'orlogin':ti,ab,kw OR 'sental':ti,ab,kw OR 'slenyto':ti,ab,kw OR 'sloremina':ti,ab,kw OR 'syncrodin':ti,ab,kw OR 'waferest':ti,ab,kw OR 'nutr?ceutical*':ti,ab,kw |
3787181 |
|
#3 |
#1 AND #2 |
18819 |
|
#4 |
#3 AND [2010-2024]/py NOT ('conference abstract'/it OR 'editorial'/it OR 'letter'/it OR 'note'/it) NOT (('animal'/exp OR 'animal experiment'/exp OR 'animal model'/exp OR 'nonhuman'/exp) NOT 'human'/exp) |
4988 |
|
#5 |
'meta analysis'/exp OR 'meta analysis (topic)'/exp OR metaanaly*:ti,ab OR 'meta analy*':ti,ab OR metanaly*:ti,ab OR 'systematic review'/de OR 'cochrane database of systematic reviews'/jt OR prisma:ti,ab OR prospero:ti,ab OR (((systemati* OR scoping OR umbrella OR 'structured literature') NEAR/3 (review* OR overview*)):ti,ab) OR ((systemic* NEAR/1 review*):ti,ab) OR (((systemati* OR literature OR database* OR 'data base*') NEAR/10 search*):ti,ab) OR (((structured OR comprehensive* OR systemic*) NEAR/3 search*):ti,ab) OR (((literature NEAR/3 review*):ti,ab) AND (search*:ti,ab OR database*:ti,ab OR 'data base*':ti,ab)) OR (('data extraction':ti,ab OR 'data source*':ti,ab) AND 'study selection':ti,ab) OR ('search strategy':ti,ab AND 'selection criteria':ti,ab) OR ('data source*':ti,ab AND 'data synthesis':ti,ab) OR medline:ab OR pubmed:ab OR embase:ab OR cochrane:ab OR (((critical OR rapid) NEAR/2 (review* OR overview* OR synthes*)):ti) OR ((((critical* OR rapid*) NEAR/3 (review* OR overview* OR synthes*)):ab) AND (search*:ab OR database*:ab OR 'data base*':ab)) OR metasynthes*:ti,ab OR 'meta synthes*':ti,ab |
1016264 |
|
#6 |
'clinical trial'/exp OR 'randomization'/exp OR 'single blind procedure'/exp OR 'double blind procedure'/exp OR 'crossover procedure'/exp OR 'placebo'/exp OR 'prospective study'/exp OR rct:ab,ti OR random*:ab,ti OR 'single blind':ab,ti OR 'randomised controlled trial':ab,ti OR 'randomized controlled trial'/exp OR placebo*:ab,ti |
4005117 |
|
#7 |
'major clinical study'/de OR 'clinical study'/de OR 'case control study'/de OR 'family study'/de OR 'longitudinal study'/de OR 'retrospective study'/de OR 'prospective study'/de OR 'comparative study'/de OR 'cohort analysis'/de OR ((cohort NEAR/1 (study OR studies)):ab,ti) OR (('case control' NEAR/1 (study OR studies)):ab,ti) OR (('follow up' NEAR/1 (study OR studies)):ab,ti) OR (observational NEAR/1 (study OR studies)) OR ((epidemiologic NEAR/1 (study OR studies)):ab,ti) OR (('cross sectional' NEAR/1 (study OR studies)):ab,ti) |
8157287 |
|
#8 |
'case control study'/de OR 'comparative study'/exp OR 'control group'/de OR 'controlled study'/de OR 'controlled clinical trial'/de OR 'crossover procedure'/de OR 'double blind procedure'/de OR 'phase 2 clinical trial'/de OR 'phase 3 clinical trial'/de OR 'phase 4 clinical trial'/de OR 'pretest posttest design'/de OR 'pretest posttest control group design'/de OR 'quasi experimental study'/de OR 'single blind procedure'/de OR 'triple blind procedure'/de OR (((control OR controlled) NEAR/6 trial):ti,ab,kw) OR (((control OR controlled) NEAR/6 (study OR studies)):ti,ab,kw) OR (((control OR controlled) NEAR/1 active):ti,ab,kw) OR 'open label*':ti,ab,kw OR (((double OR two OR three OR multi OR trial) NEAR/1 (arm OR arms)):ti,ab,kw) OR ((allocat* NEAR/10 (arm OR arms)):ti,ab,kw) OR placebo*:ti,ab,kw OR 'sham-control*':ti,ab,kw OR (((single OR double OR triple OR assessor) NEAR/1 (blind* OR masked)):ti,ab,kw) OR nonrandom*:ti,ab,kw OR 'non-random*':ti,ab,kw OR 'quasi-experiment*':ti,ab,kw OR crossover:ti,ab,kw OR 'cross over':ti,ab,kw OR 'parallel group*':ti,ab,kw OR 'factorial trial':ti,ab,kw OR ((phase NEAR/5 (study OR trial)):ti,ab,kw) OR ((case* NEAR/6 (matched OR control*)):ti,ab,kw) OR ((match* NEAR/6 (pair OR pairs OR cohort* OR control* OR group* OR healthy OR age OR sex OR gender OR patient* OR subject* OR participant*)):ti,ab,kw) OR ((propensity NEAR/6 (scor* OR match*)):ti,ab,kw) OR versus:ti OR vs:ti OR compar*:ti OR ((compar* NEAR/1 study):ti,ab,kw) OR (('major clinical study'/de OR 'clinical study'/de OR 'cohort analysis'/de OR 'observational study'/de OR 'cross-sectional study'/de OR 'multicenter study'/de OR 'correlational study'/de OR 'follow up'/de OR cohort*:ti,ab,kw OR 'follow up':ti,ab,kw OR followup:ti,ab,kw OR longitudinal*:ti,ab,kw OR prospective*:ti,ab,kw OR retrospective*:ti,ab,kw OR observational*:ti,ab,kw OR 'cross sectional*':ti,ab,kw OR cross?ectional*:ti,ab,kw OR multicent*:ti,ab,kw OR 'multi-cent*':ti,ab,kw OR consecutive*:ti,ab,kw) AND (group:ti,ab,kw OR groups:ti,ab,kw OR subgroup*:ti,ab,kw OR versus:ti,ab,kw OR vs:ti,ab,kw OR compar*:ti,ab,kw OR 'odds ratio*':ab OR 'relative odds':ab OR 'risk ratio*':ab OR 'relative risk*':ab OR 'rate ratio':ab OR aor:ab OR arr:ab OR rrr:ab OR ((('or' OR 'rr') NEAR/6 ci):ab))) |
14966536 |
|
#9 |
#4 AND #5 – SR’s |
531 |
|
#10 |
#4 AND #6 NOT #9 – RCT’s |
741 |
|
#11 |
#4 AND (#7 OR #8) NOT (#9 OR #10) – Observationele studies |
1901 |
|
#12 |
#9 OR #10 OR #11 |
3173 |
Ovid/Medline
|
# |
Searches |
Results |
|
1 |
exp Infertility, Male/ or ((exp Infertility/ or Fertility/) and exp Male/) or ((male* or men or man) adj3 (infertil* or hypofertil* or fertil* or subfertil* or infecund* or childless* or reproduct*)).ti,ab,kf. or ((spermatogen* or spermiation or spermiogen*) adj3 failure).ti,ab,kf. or (spermatozoon adj3 (abnormalit* or anomal*)).ti,ab,kf. or ((semen or sperm) adj3 (qualit* or quantit* or parameter*)).ti,ab,kf. or asperm*.ti,ab,kf. or asthenazoosperm*.ti,ab,kf. or asthenosperm*.ti,ab,kf. or asthenozoosperm*.ti,ab,kf. or azoosperm*.ti,ab,kf. or cryptosperm*.ti,ab,kf. or cryptozoosperm*.ti,ab,kf. or hyposperm*.ti,ab,kf. or oligoasthenosperm*.ti,ab,kf. or oligosperm*.ti,ab,kf. or oligozoosperm*.ti,ab,kf. or retrograde ejaculat*.ti,ab,kf. or sco syndrome*.ti,ab,kf. or sertoli cell only syndrome*.ti,ab,kf. or teratozoosperm*.ti,ab,kf. or thermosensitive genic male steril*.ti,ab,kf. or globozoosperm*.ti,ab,kf. or macrozoosperm*.ti,ab,kf. |
99984 |
|
2 |
exp Dietary Supplements/ or exp Iron/ or exp Minerals/ or exp Nutritional Support/ or exp Vitamins/ or exp Zinc/ or exp Selenium/ or exp Glutathione/ or exp Folic Acid/ or exp Ubiquinone/ or exp Carnitine/ or exp Carotenoids/ or exp Lycopene/ or exp beta Carotene/ or exp Ascorbic Acid/ or exp Acetylcysteine/ or exp alpha-Tocopherol/ or exp Fish Oils/ or exp Fatty Acids, Omega-3/ or exp Fatty Acids/ or exp Plant Oils/ or exp Xanthophylls/ or exp Thioctic Acid/ or exp Antioxidants/ or exp Vitamin B Complex/ or exp Arginine/ or exp Flavonoids/ or exp Riboflavin/ or exp Castor Oil/ or exp Olive Oil/ or exp Safflower Oil/ or exp Fatty Acids, Essential/ or exp Arachidonic Acid/ or exp Linoleic Acid/ or exp Fatty Acids, Unsaturated/ or exp alpha-Linolenic Acid/ or exp gamma-Linolenic Acid/ or exp Fatty Acids, Omega-6/ or exp Cod Liver Oil/ or exp Inositol/ or exp Docosahexaenoic Acids/ or exp Magnesium/ or exp Melatonin/ or (((nutritional or iron) adj3 (therap* or treatment* or support*)) or ((plant* or seed* or vegetable* or dietary or edible or ricinus or cast?r or ricin* or olive* or safflower or fish or tuna) adj3 (oil* or fat)) or supplement* or vitamin* or zinc or zincum or selenicum or selenium or glutathine or glutathiol or glutathion or glutathione or glutathioral or glutatiol or tathion or tationil or tiobione or acfol or acifolic or filicine or folacid* or folacin or folart or folate or folavit or foldine or foliamin or folicet or folicid or folina or folinsyre or folitab or folivit or folsan or folsav or folverlan or folvite or ingafol or lafol or lexpec or megafol or neocepri or rubiefol or sodium folate or speciafoldine or vifolin or ubiquinone or coenzyme q* or abedine or biocarn or biomux or cardiogen or carnicor or carnil or carnitene* or carnitin or carnitine* or carnitor or carnovis or cartin or entomin or lefcar or monocamin or nefrocarnit or novain or vitacarn or carotenoid* or carotinoid* or astaxanthin* or astaxanthine* or ovoester or lycopene* or menevit or multivitamin* or b-tene or beta carotene or beta carotin* or betacarotene* or betatene* or carotaben or lurotin or solatene or solvin or acidylina or adenex or agrumina or allercorb or allescorb or arcavit c or arcavite c or arkovital c or ascelat or ascofar or ascomed or asconvita or ascor or ascorbate or ascorbate sodium or ascorbic acid* or ascorbicap or ascorbicin or ascorbico or ascorbin or ascorbina or ascorbinic acid* or ascorbit or ascorbite or ascorbitol or ascorbivit or ascorbivite or ascorbone or ascorbutina or ascorbyl or ascorbyn or ascorcee or ascorgil or ascorin or ascormin or ascorteal or ascorval or ascorvel or ascorvit or ascorvite or ascorvitina or askorbin or austrovit c or austrovite c or bentavit c or bentavite c or cantan or cantaxin or cebetate or cebicure or cebiolon or cebion or cebione or cecap or cecon or cecone or cecorbin or cecorbine or cecorbyl or cecorbyle or cecrisina or cedon or cedone or cedoxon or cedoxone or ceevifil or cegiolan or celaskon or celaskone or celin or celine or cenetone or cenol or cenolate or cequinyl or cereon or cergona or cescorbat or cetamican or cetamid or cetamine or cetebe or ceterapion or ceterapione or cetrinets or cevalin or cevaline or cevatine or cevex or cevibram or cevigal or cevigen or cevigol or cevikap or cevilat or cevimin or cevimine or cevisol or cevit or cevita or cevitamin* or cevitan or cevite or cevitex or cevitil or cevitol or cewin or chewcee or ci drol or ciamin or ciergin or cifilina or cipca or cisir or citamino or cith or citoascorbina or citoxyl or citran or citravite or citritabs or citrovitamin* or civigor or civitin or civitine or concemin or cortalex or dagrascorbin or dagravit c or dancimin c or davitamon c or dayvital or delo c or difvitamin* c or dropice or dumovit c or dumovite c or esuron or esurvit or esurvite or flavettes or hicee or hybrin or ikacee or irocevit or irocevite or jarexin or jarexine or lacivit or lacivite or laroscorbine or lemascorb or levo ascorbate or levo ascorbic acid* or limcee or limo ce or liqui cee or myascorbin or natrascorb or novo ascorbic or nybadol or pascorbin or pharmascorbine or potassium ascorbate or proscorbin or proscorbine or redoxon or redoxon forte or ribena or scorbacid* or scorbettes or scorbex or scorbitol or scorbumine or secorbate or sevalin or sodascorbate or sodium ascorbate or sweetcee or testascorbic or upsavit or vicef or vicelat or vicetrin or viciman or vicin or vicitina or vicon or viforcit or viforcite or viscorin or viscorine or vitac or vitace or vitacee or vitaci or vitacimin or vitacimine or vitacin or vitacine or vitaplex c or vitapric or vitapur c or vitasan c or vitascorbin or vitascorbine or vitascorbol or vitelix c or vitocee or vorange or wandervit c or wandervite c or witamina c or xitix or xyloascorbic acid* or acerac or acetadote or acetain or acetyl cysteine or acetylcystein or acetylcysteine or acypront or airbron or alveolex or bromuc or bronchocil or brunac or cetilan or cetylev or drenaflen or ecomucyl or euronac or exomuc or fabrol or fluimicil or fluimucil or fluimukan or flumil or flumil infantil or fluprowit or flutafin or genac or granon or hidonac or ilube or inspir or lappe or libramucil or lysomucil or lysox or menaxol or mercapturic acid* or mucocil or mucodrill or mucofillin or mucolator or mucomiste or mucomyst or mucomystendo or mucopect or mucoserin or mucosil or mucosof or mucosolvin or mucosten or mucoza or mukolit or muteran or parvolex or pulmosal or reolin or respaire or rinofluimucil or sigamucil or solmucol or spatam or sputoprompt or stecin or tirocular or tixair or zifluvis or ethylcysteine or alpha tocopherol* or alpha tocopheryl acetate or alpha tocopheryl acetic acid* or aquasol e or austrovit e or covitol or dagravit e or dalfatol or davitamon e or dermorelle or detulin or dumovit e or e ferol or e perle or e perte or e recordati or e toplex or e vicotrat or e vimin or e vita or e viterbin or ecoferol or efer or eferol or enoulan forte or ephynal or eplonat or eprolin or erevit or esol or esorb or eterapion or eviabit or evigen or eviol or evion or evit or evitol or godabion e or gonavit or hanobak or ido e or juvela or juvele or livingpherol or metaperex or mulsal e or natopherol or optovit e or phytoferol or pletocol or socopherol or spondyvit or toco 500 or tocoferolo bioglan or tocomine or tocopherex or tocopherol acetate or tocopherol gumpro or tocopheryl acetate or tocophrine or tocovigor or tocovital or toferol or topherol or vibolex e or vidom e or viea or vietal or viprimol or viteolin or viteoline or wandervit e or ameu or efamed or epax or feniko or himega or k 85 or lachs 550 or lipitac or maxepa or olemar or omegaven or optimepa or pikasol or promega or super epa or superepa or omega* or fat* acid* or arginin* or arginyl residue or argivene or bioarginina or r gene 10 or flavonoid* or beflavin* or beflavine* or flavaxin or hyrye or lactoflavin* or ovoflavin or pabriflan or riboflavin* or ribovel or leucocianidol or leucocyanidin* or leucocyanidol or pycnogenol or pygnoforton or pyknogenol or lutein* or xanthophyl* or xantofyl* or xantophyl* or berlithione or biletan or heparlipon or lipoate or lipoic acid* or liponic acid* or neurothioct or protogen or thioctacid* or thioctan or thioctic acid* or thiogamma or thiotocid or tioctan or tioctic acid* or tioctidasi or antioxidant* or antioxidat* or antoxidant* or radical scavenger* or neoloid or lyprinol or arachidonate* or arachidonate sodium or arachidonic acid* or sodium arachidonate* or linoic acid* or linolate* or linoleate* or linoleic acid* or linolic acid* or sodium linoleate* or ufa or unsaturated fat* or unsaturated lipid* or linolenate* or linolenic acid* or efagel or polyunsaturat* fat* or cod* liver oil* or eicosapen or epaisdin or epanova or omega 3 or omega forte or sakana or betitol or cyclohexanehexol or cyclohexitol or dambose or hexol or inosil or inosimesol or inosit* or meat sugar or mesitol or meso inosit* or mesoinosil or mesoinosit* or mesol or mesovil or muscle sugar or myoinosil or myoinosit* or nucite or phaseomannite or phaseomannitol or scyllite or tonozil or dhasco or docosahexaenoate or docosahexaenoic acid* or docosahexenoic acid* or magnesium or romag or adaflex or aritonin or ceyestaeusom or ceyesto or circadin or civasta or eusom or lestinora or mallozen or mecastrin or melabiorytm or melatal or melatan or melatol or melatonin* or melatonite or mellaras or mellozzan or melovine or mucomel or noxarem or orlogin or sental or slenyto or sloremina or syncrodin or waferest or nutr?ceutical*).ti,ab,kf. |
2990044 |
|
3 |
1 and 2 |
14158 |
|
4 |
limit 3 to yr="2010 -Current" |
9392 |
|
5 |
4 not (comment/ or editorial/ or letter/) not ((exp animals/ or exp models, animal/) not humans/) |
5180 |
|
6 |
meta-analysis/ or meta-analysis as topic/ or (metaanaly* or meta-analy* or metanaly*).ti,ab,kf. or systematic review/ or cochrane.jw. or (prisma or prospero).ti,ab,kf. or ((systemati* or scoping or umbrella or "structured literature") adj3 (review* or overview*)).ti,ab,kf. or (systemic* adj1 review*).ti,ab,kf. or ((systemati* or literature or database* or data-base*) adj10 search*).ti,ab,kf. or ((structured or comprehensive* or systemic*) adj3 search*).ti,ab,kf. or ((literature adj3 review*) and (search* or database* or data-base*)).ti,ab,kf. or (("data extraction" or "data source*") and "study selection").ti,ab,kf. or ("search strategy" and "selection criteria").ti,ab,kf. or ("data source*" and "data synthesis").ti,ab,kf. or (medline or pubmed or embase or cochrane).ab. or ((critical or rapid) adj2 (review* or overview* or synthes*)).ti. or (((critical* or rapid*) adj3 (review* or overview* or synthes*)) and (search* or database* or data-base*)).ab. or (metasynthes* or meta-synthes*).ti,ab,kf. |
737147 |
|
7 |
exp clinical trial/ or randomized controlled trial/ or exp clinical trials as topic/ or randomized controlled trials as topic/ or Random Allocation/ or Double-Blind Method/ or Single-Blind Method/ or (clinical trial, phase i or clinical trial, phase ii or clinical trial, phase iii or clinical trial, phase iv or controlled clinical trial or randomized controlled trial or multicenter study or clinical trial).pt. or random*.ti,ab. or (clinic* adj trial*).tw. or ((singl* or doubl* or treb* or tripl*) adj (blind$3 or mask$3)).tw. or Placebos/ or placebo*.tw. |
2709295 |
|
8 |
Epidemiologic studies/ or case control studies/ or exp cohort studies/ or Controlled Before-After Studies/ or Case control.tw. or cohort.tw. or Cohort analy$.tw. or (Follow up adj (study or studies)).tw. or (observational adj (study or studies)).tw. or Longitudinal.tw. or Retrospective*.tw. or prospective*.tw. or consecutive*.tw. or Cross sectional.tw. or Cross-sectional studies/ or historically controlled study/ or interrupted time series analysis/ [Onder exp cohort studies vallen ook longitudinale, prospectieve en retrospectieve studies] |
4692361 |
|
9 |
Case-control Studies/ or clinical trial, phase ii/ or clinical trial, phase iii/ or clinical trial, phase iv/ or comparative study/ or control groups/ or controlled before-after studies/ or controlled clinical trial/ or double-blind method/ or historically controlled study/ or matched-pair analysis/ or single-blind method/ or (((control or controlled) adj6 (study or studies or trial)) or (compar* adj (study or studies)) or ((control or controlled) adj1 active) or "open label*" or ((double or two or three or multi or trial) adj (arm or arms)) or (allocat* adj10 (arm or arms)) or placebo* or "sham-control*" or ((single or double or triple or assessor) adj1 (blind* or masked)) or nonrandom* or "non-random*" or "quasi-experiment*" or "parallel group*" or "factorial trial" or "pretest posttest" or (phase adj5 (study or trial)) or (case* adj6 (matched or control*)) or (match* adj6 (pair or pairs or cohort* or control* or group* or healthy or age or sex or gender or patient* or subject* or participant*)) or (propensity adj6 (scor* or match*))).ti,ab,kf. or (confounding adj6 adjust*).ti,ab. or (versus or vs or compar*).ti. or ((exp cohort studies/ or epidemiologic studies/ or multicenter study/ or observational study/ or seroepidemiologic studies/ or (cohort* or 'follow up' or followup or longitudinal* or prospective* or retrospective* or observational* or multicent* or 'multi-cent*' or consecutive*).ti,ab,kf.) and ((group or groups or subgroup* or versus or vs or compar*).ti,ab,kf. or ('odds ratio*' or 'relative odds' or 'risk ratio*' or 'relative risk*' or aor or arr or rrr).ab. or (("OR" or "RR") adj6 CI).ab.)) |
5659051 |
|
10 |
5 and 6 – SR’s |
352 |
|
11 |
(5 and 7) not 10 – RCT’s |
749 |
|
12 |
(5 and (8 or 9)) not (10 or 11) – Observationele studies |
1062 |
|
13 |
10 or 11 or 12 |
2163 |
Zoekstrategie - 27 juli 2024
Embase.com
|
No. |
Query |
Results |
|
#1 |
'male infertility'/exp OR 'male fertility'/exp OR (('subfertility'/de OR 'childlessness'/exp) AND 'male'/exp) OR (((male* OR men OR man) NEAR/3 (infertil* OR hypofertil* OR 'fertil*' OR subfertil* OR infecund* OR childless* OR reproduct*)):ti,ab,kw) OR (((spermatogen* OR spermiation OR spermiogen*) NEAR/3 failure):ti,ab,kw) OR ((spermatozoon NEAR/3 (abnormalit* OR anomal*)):ti,ab,kw) OR (((semen OR sperm) NEAR/3 (qualit* OR quantit* OR parameter*)):ti,ab,kw) OR asperm*:ti,ab,kw OR asthenazoosperm*:ti,ab,kw OR asthenosperm*:ti,ab,kw OR asthenozoosperm*:ti,ab,kw OR azoosperm*:ti,ab,kw OR cryptosperm*:ti,ab,kw OR cryptozoosperm*:ti,ab,kw OR hyposperm*:ti,ab,kw OR oligoasthenosperm*:ti,ab,kw OR oligosperm*:ti,ab,kw OR oligozoosperm*:ti,ab,kw OR 'retrograde ejaculat*':ti,ab,kw OR 'sco syndrome*':ti,ab,kw OR 'sertoli cell only syndrome*':ti,ab,kw OR teratozoosperm*:ti,ab,kw OR 'thermosensitive genic male steril*':ti,ab,kw OR globozoosperm*:ti,ab,kw OR macrozoosperm*:ti,ab,kw |
112802 |
|
#2 |
'supplementation'/exp OR 'diet supplementation'/exp OR 'iron therapy'/exp OR 'mineral supplementation'/exp OR 'nutritional support'/exp OR 'vitamin supplementation'/exp OR 'zinc'/exp OR 'selenium'/exp OR 'glutathione'/exp OR 'folic acid'/exp OR 'ubiquinone'/exp OR 'carnitine'/exp OR 'carotenoid'/exp OR 'astaxanthin'/exp OR 'lycopene'/exp OR 'menevit'/exp OR 'multivitamin'/exp OR 'beta carotene'/exp OR 'ascorbic acid'/exp OR 'acetylcysteine'/exp OR 'ethylcysteine'/exp OR 'alpha tocopherol'/exp OR 'fish oil'/exp OR 'omega amino acid'/exp OR 'fatty acid'/exp OR 'vegetable oil'/exp OR 'pycnogenol'/exp OR 'xanthophyll'/exp OR 'thioctic acid'/exp OR 'antioxidant'/exp OR 'vitamin'/exp OR 'vitamin b group'/exp OR 'arginine'/exp OR 'flavonoid'/exp OR 'riboflavin'/exp OR 'edible oil'/exp OR 'castor oil'/exp OR 'lyprinol'/exp OR 'olive oil'/exp OR 'safflower oil'/exp OR 'essential fatty acid'/exp OR 'arachidonic acid'/exp OR 'linoleic acid'/exp OR 'unsaturated fatty acid'/exp OR 'linolenic acid'/exp OR 'gamma linolenic acid'/exp OR 'omega 6 fatty acid'/exp OR 'polyunsaturated fatty acid'/exp OR 'cod liver oil'/exp OR 'omega 3 fatty acid'/exp OR 'inositol'/exp OR 'docosahexaenoic acid'/exp OR 'magnesium'/exp OR 'melatonin'/exp OR 'nutraceutical'/exp OR (((nutritional OR iron) NEAR/3 (therap* OR treatment* OR support*)):ti,ab,kw) OR (((plant* OR seed* OR vegetable* OR dietary OR edible OR ricinus OR cast?r OR ricin* OR olive* OR safflower OR fish OR tuna) NEAR/3 (oil* OR fat)):ti,ab,kw) OR 'supplement*':ti,ab,kw OR vitamin*:ti,ab,kw OR 'zinc':ti,ab,kw OR 'zincum':ti,ab,kw OR 'selenicum':ti,ab,kw OR 'selenium':ti,ab,kw OR 'glutathine':ti,ab,kw OR 'glutathiol':ti,ab,kw OR 'glutathion':ti,ab,kw OR 'glutathione':ti,ab,kw OR 'glutathioral':ti,ab,kw OR 'glutatiol':ti,ab,kw OR 'tathion':ti,ab,kw OR 'tationil':ti,ab,kw OR 'tiobione':ti,ab,kw OR 'acfol':ti,ab,kw OR 'acifolic':ti,ab,kw OR 'filicine':ti,ab,kw OR 'folacid*':ti,ab,kw OR 'folacin':ti,ab,kw OR 'folart':ti,ab,kw OR 'folate':ti,ab,kw OR 'folavit':ti,ab,kw OR 'foldine':ti,ab,kw OR 'foliamin':ti,ab,kw OR 'folicet':ti,ab,kw OR 'folicid':ti,ab,kw OR 'folina':ti,ab,kw OR 'folinsyre':ti,ab,kw OR 'folitab':ti,ab,kw OR 'folivit':ti,ab,kw OR 'folsan':ti,ab,kw OR 'folsav':ti,ab,kw OR 'folverlan':ti,ab,kw OR 'folvite':ti,ab,kw OR 'ingafol':ti,ab,kw OR 'lafol':ti,ab,kw OR 'lexpec':ti,ab,kw OR 'megafol':ti,ab,kw OR 'neocepri':ti,ab,kw OR 'rubiefol':ti,ab,kw OR 'sodium folate':ti,ab,kw OR 'speciafoldine':ti,ab,kw OR 'vifolin':ti,ab,kw OR 'ubiquinone':ti,ab,kw OR 'coenzyme q*':ti,ab,kw OR 'abedine':ti,ab,kw OR 'biocarn':ti,ab,kw OR 'biomux':ti,ab,kw OR 'cardiogen':ti,ab,kw OR 'carnicor':ti,ab,kw OR 'carnil':ti,ab,kw OR 'carnitene*':ti,ab,kw OR 'carnitin':ti,ab,kw OR 'carnitine*':ti,ab,kw OR 'carnitor':ti,ab,kw OR 'carnovis':ti,ab,kw OR 'cartin':ti,ab,kw OR 'entomin':ti,ab,kw OR 'lefcar':ti,ab,kw OR 'monocamin':ti,ab,kw OR 'nefrocarnit':ti,ab,kw OR 'novain':ti,ab,kw OR 'vitacarn':ti,ab,kw OR 'carotenoid*':ti,ab,kw OR 'carotinoid*':ti,ab,kw OR 'astaxanthin*':ti,ab,kw OR 'astaxanthine*':ti,ab,kw OR 'ovoester':ti,ab,kw OR 'lycopene*':ti,ab,kw OR 'menevit':ti,ab,kw OR 'multivitamin*':ti,ab,kw OR 'b-tene':ti,ab,kw OR 'beta carotene':ti,ab,kw OR 'beta carotin*':ti,ab,kw OR 'betacarotene*':ti,ab,kw OR 'betatene*':ti,ab,kw OR 'carotaben':ti,ab,kw OR 'lurotin':ti,ab,kw OR 'solatene':ti,ab,kw OR 'solvin':ti,ab,kw OR 'acidylina':ti,ab,kw OR 'adenex':ti,ab,kw OR 'agrumina':ti,ab,kw OR 'allercorb':ti,ab,kw OR 'allescorb':ti,ab,kw OR 'arcavit c':ti,ab,kw OR 'arcavite c':ti,ab,kw OR 'arkovital c':ti,ab,kw OR 'ascelat':ti,ab,kw OR 'ascofar':ti,ab,kw OR 'ascomed':ti,ab,kw OR 'asconvita':ti,ab,kw OR 'ascor':ti,ab,kw OR 'ascorbate':ti,ab,kw OR 'ascorbate sodium':ti,ab,kw OR 'ascorbic acid*':ti,ab,kw OR 'ascorbicap':ti,ab,kw OR 'ascorbicin':ti,ab,kw OR 'ascorbico':ti,ab,kw OR 'ascorbin':ti,ab,kw OR 'ascorbina':ti,ab,kw OR 'ascorbinic acid*':ti,ab,kw OR 'ascorbit':ti,ab,kw OR 'ascorbite':ti,ab,kw OR 'ascorbitol':ti,ab,kw OR 'ascorbivit':ti,ab,kw OR 'ascorbivite':ti,ab,kw OR 'ascorbone':ti,ab,kw OR 'ascorbutina':ti,ab,kw OR 'ascorbyl':ti,ab,kw OR 'ascorbyn':ti,ab,kw OR 'ascorcee':ti,ab,kw OR 'ascorgil':ti,ab,kw OR 'ascorin':ti,ab,kw OR 'ascormin':ti,ab,kw OR 'ascorteal':ti,ab,kw OR 'ascorval':ti,ab,kw OR 'ascorvel':ti,ab,kw OR 'ascorvit':ti,ab,kw OR 'ascorvite':ti,ab,kw OR 'ascorvitina':ti,ab,kw OR 'askorbin':ti,ab,kw OR 'austrovit c':ti,ab,kw OR 'austrovite c':ti,ab,kw OR 'bentavit c':ti,ab,kw OR 'bentavite c':ti,ab,kw OR 'cantan':ti,ab,kw OR 'cantaxin':ti,ab,kw OR 'cebetate':ti,ab,kw OR 'cebicure':ti,ab,kw OR 'cebiolon':ti,ab,kw OR 'cebion':ti,ab,kw OR 'cebione':ti,ab,kw OR 'cecap':ti,ab,kw OR 'cecon':ti,ab,kw OR 'cecone':ti,ab,kw OR 'cecorbin':ti,ab,kw OR 'cecorbine':ti,ab,kw OR 'cecorbyl':ti,ab,kw OR 'cecorbyle':ti,ab,kw OR 'cecrisina':ti,ab,kw OR 'cedon':ti,ab,kw OR 'cedone':ti,ab,kw OR 'cedoxon':ti,ab,kw OR 'cedoxone':ti,ab,kw OR 'ceevifil':ti,ab,kw OR 'cegiolan':ti,ab,kw OR 'celaskon':ti,ab,kw OR 'celaskone':ti,ab,kw OR 'celin':ti,ab,kw OR 'celine':ti,ab,kw OR 'cenetone':ti,ab,kw OR 'cenol':ti,ab,kw OR 'cenolate':ti,ab,kw OR 'cequinyl':ti,ab,kw OR 'cereon':ti,ab,kw OR 'cergona':ti,ab,kw OR 'cescorbat':ti,ab,kw OR 'cetamican':ti,ab,kw OR 'cetamid':ti,ab,kw OR 'cetamine':ti,ab,kw OR 'cetebe':ti,ab,kw OR 'ceterapion':ti,ab,kw OR 'ceterapione':ti,ab,kw OR 'cetrinets':ti,ab,kw OR 'cevalin':ti,ab,kw OR 'cevaline':ti,ab,kw OR 'cevatine':ti,ab,kw OR 'cevex':ti,ab,kw OR 'cevibram':ti,ab,kw OR 'cevigal':ti,ab,kw OR 'cevigen':ti,ab,kw OR 'cevigol':ti,ab,kw OR 'cevikap':ti,ab,kw OR 'cevilat':ti,ab,kw OR 'cevimin':ti,ab,kw OR 'cevimine':ti,ab,kw OR 'cevisol':ti,ab,kw OR 'cevit':ti,ab,kw OR 'cevita':ti,ab,kw OR 'cevitamin*':ti,ab,kw OR 'cevitan':ti,ab,kw OR 'cevite':ti,ab,kw OR 'cevitex':ti,ab,kw OR 'cevitil':ti,ab,kw OR 'cevitol':ti,ab,kw OR 'cewin':ti,ab,kw OR 'chewcee':ti,ab,kw OR 'ci drol':ti,ab,kw OR 'ciamin':ti,ab,kw OR 'ciergin':ti,ab,kw OR 'cifilina':ti,ab,kw OR 'cipca':ti,ab,kw OR 'cisir':ti,ab,kw OR 'citamino':ti,ab,kw OR 'cith':ti,ab,kw OR 'citoascorbina':ti,ab,kw OR 'citoxyl':ti,ab,kw OR 'citran':ti,ab,kw OR 'citravite':ti,ab,kw OR 'citritabs':ti,ab,kw OR 'citrovitamin*':ti,ab,kw OR 'civigor':ti,ab,kw OR 'civitin':ti,ab,kw OR 'civitine':ti,ab,kw OR 'concemin':ti,ab,kw OR 'cortalex':ti,ab,kw OR 'dagrascorbin':ti,ab,kw OR 'dagravit c':ti,ab,kw OR 'dancimin c':ti,ab,kw OR 'davitamon c':ti,ab,kw OR 'dayvital':ti,ab,kw OR 'delo c':ti,ab,kw OR 'difvitamin* c':ti,ab,kw OR 'dropice':ti,ab,kw OR 'dumovit c':ti,ab,kw OR 'dumovite c':ti,ab,kw OR 'esuron':ti,ab,kw OR 'esurvit':ti,ab,kw OR 'esurvite':ti,ab,kw OR 'flavettes':ti,ab,kw OR 'hicee':ti,ab,kw OR 'hybrin':ti,ab,kw OR 'ikacee':ti,ab,kw OR 'irocevit':ti,ab,kw OR 'irocevite':ti,ab,kw OR 'jarexin':ti,ab,kw OR 'jarexine':ti,ab,kw OR 'lacivit':ti,ab,kw OR 'lacivite':ti,ab,kw OR 'laroscorbine':ti,ab,kw OR 'lemascorb':ti,ab,kw OR 'levo ascorbate':ti,ab,kw OR 'levo ascorbic acid*':ti,ab,kw OR 'limcee':ti,ab,kw OR 'limo ce':ti,ab,kw OR 'liqui cee':ti,ab,kw OR 'myascorbin':ti,ab,kw OR 'natrascorb':ti,ab,kw OR 'novo ascorbic':ti,ab,kw OR 'nybadol':ti,ab,kw OR 'pascorbin':ti,ab,kw OR 'pharmascorbine':ti,ab,kw OR 'potassium ascorbate':ti,ab,kw OR 'proscorbin':ti,ab,kw OR 'proscorbine':ti,ab,kw OR 'redoxon':ti,ab,kw OR 'redoxon forte':ti,ab,kw OR 'ribena':ti,ab,kw OR 'scorbacid*':ti,ab,kw OR 'scorbettes':ti,ab,kw OR 'scorbex':ti,ab,kw OR 'scorbitol':ti,ab,kw OR 'scorbumine':ti,ab,kw OR 'secorbate':ti,ab,kw OR 'sevalin':ti,ab,kw OR 'sodascorbate':ti,ab,kw OR 'sodium ascorbate':ti,ab,kw OR 'sweetcee':ti,ab,kw OR 'testascorbic':ti,ab,kw OR 'upsavit':ti,ab,kw OR 'vicef':ti,ab,kw OR 'vicelat':ti,ab,kw OR 'vicetrin':ti,ab,kw OR 'viciman':ti,ab,kw OR 'vicin':ti,ab,kw OR 'vicitina':ti,ab,kw OR 'vicon':ti,ab,kw OR 'viforcit':ti,ab,kw OR 'viforcite':ti,ab,kw OR 'viscorin':ti,ab,kw OR 'viscorine':ti,ab,kw OR 'vitac':ti,ab,kw OR 'vitace':ti,ab,kw OR 'vitacee':ti,ab,kw OR 'vitaci':ti,ab,kw OR 'vitacimin':ti,ab,kw OR 'vitacimine':ti,ab,kw OR 'vitacin':ti,ab,kw OR 'vitacine':ti,ab,kw OR 'vitaplex c':ti,ab,kw OR 'vitapric':ti,ab,kw OR 'vitapur c':ti,ab,kw OR 'vitasan c':ti,ab,kw OR 'vitascorbin':ti,ab,kw OR 'vitascorbine':ti,ab,kw OR 'vitascorbol':ti,ab,kw OR 'vitelix c':ti,ab,kw OR 'vitocee':ti,ab,kw OR 'vorange':ti,ab,kw OR 'wandervit c':ti,ab,kw OR 'wandervite c':ti,ab,kw OR 'witamina c':ti,ab,kw OR 'xitix':ti,ab,kw OR 'xyloascorbic acid*':ti,ab,kw OR 'acerac':ti,ab,kw OR 'acetadote':ti,ab,kw OR 'acetain':ti,ab,kw OR 'acetyl cysteine':ti,ab,kw OR 'acetylcystein':ti,ab,kw OR 'acetylcysteine':ti,ab,kw OR 'acypront':ti,ab,kw OR 'airbron':ti,ab,kw OR 'alveolex':ti,ab,kw OR 'bromuc':ti,ab,kw OR 'bronchocil':ti,ab,kw OR 'brunac':ti,ab,kw OR 'cetilan':ti,ab,kw OR 'cetylev':ti,ab,kw OR 'drenaflen':ti,ab,kw OR 'ecomucyl':ti,ab,kw OR 'euronac':ti,ab,kw OR 'exomuc':ti,ab,kw OR 'fabrol':ti,ab,kw OR 'fluimicil':ti,ab,kw OR 'fluimucil':ti,ab,kw OR 'fluimukan':ti,ab,kw OR 'flumil':ti,ab,kw OR 'flumil infantil':ti,ab,kw OR 'fluprowit':ti,ab,kw OR 'flutafin':ti,ab,kw OR 'genac':ti,ab,kw OR 'granon':ti,ab,kw OR 'hidonac':ti,ab,kw OR 'ilube':ti,ab,kw OR 'inspir':ti,ab,kw OR 'lappe':ti,ab,kw OR 'libramucil':ti,ab,kw OR 'lysomucil':ti,ab,kw OR 'lysox':ti,ab,kw OR 'menaxol':ti,ab,kw OR 'mercapturic acid*':ti,ab,kw OR 'mucocil':ti,ab,kw OR 'mucodrill':ti,ab,kw OR 'mucofillin':ti,ab,kw OR 'mucolator':ti,ab,kw OR 'mucomiste':ti,ab,kw OR 'mucomyst':ti,ab,kw OR 'mucomystendo':ti,ab,kw OR 'mucopect':ti,ab,kw OR 'mucoserin':ti,ab,kw OR 'mucosil':ti,ab,kw OR 'mucosof':ti,ab,kw OR 'mucosolvin':ti,ab,kw OR 'mucosten':ti,ab,kw OR 'mucoza':ti,ab,kw OR 'mukolit':ti,ab,kw OR 'muteran':ti,ab,kw OR 'parvolex':ti,ab,kw OR 'pulmosal':ti,ab,kw OR 'reolin':ti,ab,kw OR 'respaire':ti,ab,kw OR 'rinofluimucil':ti,ab,kw OR 'sigamucil':ti,ab,kw OR 'solmucol':ti,ab,kw OR 'spatam':ti,ab,kw OR 'sputoprompt':ti,ab,kw OR 'stecin':ti,ab,kw OR 'tirocular':ti,ab,kw OR 'tixair':ti,ab,kw OR 'zifluvis':ti,ab,kw OR 'ethylcysteine':ti,ab,kw OR 'alpha tocopherol*':ti,ab,kw OR 'alpha tocopheryl acetate':ti,ab,kw OR 'alpha tocopheryl acetic acid*':ti,ab,kw OR 'aquasol e':ti,ab,kw OR 'austrovit e':ti,ab,kw OR 'covitol':ti,ab,kw OR 'dagravit e':ti,ab,kw OR 'dalfatol':ti,ab,kw OR 'davitamon e':ti,ab,kw OR 'dermorelle':ti,ab,kw OR 'detulin':ti,ab,kw OR 'dumovit e':ti,ab,kw OR 'e ferol':ti,ab,kw OR 'e perle':ti,ab,kw OR 'e perte':ti,ab,kw OR 'e recordati':ti,ab,kw OR 'e toplex':ti,ab,kw OR 'e vicotrat':ti,ab,kw OR 'e vimin':ti,ab,kw OR 'e vita':ti,ab,kw OR 'e viterbin':ti,ab,kw OR 'ecoferol':ti,ab,kw OR 'efer':ti,ab,kw OR 'eferol':ti,ab,kw OR 'enoulan forte':ti,ab,kw OR 'ephynal':ti,ab,kw OR 'eplonat':ti,ab,kw OR 'eprolin':ti,ab,kw OR 'erevit':ti,ab,kw OR 'esol':ti,ab,kw OR 'esorb':ti,ab,kw OR 'eterapion':ti,ab,kw OR 'eviabit':ti,ab,kw OR 'evigen':ti,ab,kw OR 'eviol':ti,ab,kw OR 'evion':ti,ab,kw OR 'evit':ti,ab,kw OR 'evitol':ti,ab,kw OR 'godabion e':ti,ab,kw OR 'gonavit':ti,ab,kw OR 'hanobak':ti,ab,kw OR 'ido e':ti,ab,kw OR 'juvela':ti,ab,kw OR 'juvele':ti,ab,kw OR 'livingpherol':ti,ab,kw OR 'metaperex':ti,ab,kw OR 'mulsal e':ti,ab,kw OR 'natopherol':ti,ab,kw OR 'optovit e':ti,ab,kw OR 'phytoferol':ti,ab,kw OR 'pletocol':ti,ab,kw OR 'socopherol':ti,ab,kw OR 'spondyvit':ti,ab,kw OR 'toco 500':ti,ab,kw OR 'tocoferolo bioglan':ti,ab,kw OR 'tocomine':ti,ab,kw OR 'tocopherex':ti,ab,kw OR 'tocopherol acetate':ti,ab,kw OR 'tocopherol gumpro':ti,ab,kw OR 'tocopheryl acetate':ti,ab,kw OR 'tocophrine':ti,ab,kw OR 'tocovigor':ti,ab,kw OR 'tocovital':ti,ab,kw OR 'toferol':ti,ab,kw OR 'topherol':ti,ab,kw OR 'vibolex e':ti,ab,kw OR 'vidom e':ti,ab,kw OR 'viea':ti,ab,kw OR 'vietal':ti,ab,kw OR 'viprimol':ti,ab,kw OR 'viteolin':ti,ab,kw OR 'viteoline':ti,ab,kw OR 'wandervit e':ti,ab,kw OR 'ameu':ti,ab,kw OR 'efamed':ti,ab,kw OR 'epax':ti,ab,kw OR 'feniko':ti,ab,kw OR 'himega':ti,ab,kw OR 'k 85':ti,ab,kw OR 'lachs 550':ti,ab,kw OR 'lipitac':ti,ab,kw OR 'maxepa':ti,ab,kw OR 'olemar':ti,ab,kw OR 'omegaven':ti,ab,kw OR 'optimepa':ti,ab,kw OR 'pikasol':ti,ab,kw OR 'promega':ti,ab,kw OR 'super epa':ti,ab,kw OR 'superepa':ti,ab,kw OR 'omega*':ti,ab,kw OR 'fat* acid*':ti,ab,kw OR 'arginin*':ti,ab,kw OR 'arginyl residue':ti,ab,kw OR 'argivene':ti,ab,kw OR 'bioarginina':ti,ab,kw OR 'r gene 10':ti,ab,kw OR 'flavonoid*':ti,ab,kw OR 'beflavin*':ti,ab,kw OR 'beflavine*':ti,ab,kw OR 'flavaxin':ti,ab,kw OR 'hyrye':ti,ab,kw OR 'lactoflavin*':ti,ab,kw OR 'ovoflavin':ti,ab,kw OR 'pabriflan':ti,ab,kw OR 'riboflavin*':ti,ab,kw OR 'ribovel':ti,ab,kw OR 'leucocianidol':ti,ab,kw OR 'leucocyanidin*':ti,ab,kw OR 'leucocyanidol':ti,ab,kw OR 'pycnogenol':ti,ab,kw OR 'pygnoforton':ti,ab,kw OR 'pyknogenol':ti,ab,kw OR 'lutein*':ti,ab,kw OR 'xanthophyl*':ti,ab,kw OR 'xantofyl*':ti,ab,kw OR 'xantophyl*':ti,ab,kw OR 'berlithione':ti,ab,kw OR 'biletan':ti,ab,kw OR 'heparlipon':ti,ab,kw OR 'lipoate':ti,ab,kw OR 'lipoic acid*':ti,ab,kw OR 'liponic acid*':ti,ab,kw OR 'neurothioct':ti,ab,kw OR 'protogen':ti,ab,kw OR 'thioctacid*':ti,ab,kw OR 'thioctan':ti,ab,kw OR 'thioctic acid*':ti,ab,kw OR 'thiogamma':ti,ab,kw OR 'thiotocid':ti,ab,kw OR 'tioctan':ti,ab,kw OR 'tioctic acid*':ti,ab,kw OR 'tioctidasi':ti,ab,kw OR 'antioxidant*':ti,ab,kw OR 'antioxidat*':ti,ab,kw OR 'antoxidant*':ti,ab,kw OR 'radical scavenger*':ti,ab,kw OR 'neoloid':ti,ab,kw OR 'lyprinol':ti,ab,kw OR 'arachidonate*':ti,ab,kw OR 'arachidonate sodium':ti,ab,kw OR 'arachidonic acid*':ti,ab,kw OR 'sodium arachidonate*':ti,ab,kw OR 'linoic acid*':ti,ab,kw OR 'linolate*':ti,ab,kw OR 'linoleate*':ti,ab,kw OR 'linoleic acid*':ti,ab,kw OR 'linolic acid*':ti,ab,kw OR 'sodium linoleate*':ti,ab,kw OR 'ufa':ti,ab,kw OR 'unsaturated fat*':ti,ab,kw OR 'unsaturated lipid*':ti,ab,kw OR 'linolenate*':ti,ab,kw OR 'linolenic acid*':ti,ab,kw OR 'efagel':ti,ab,kw OR 'polyunsaturat* fat*':ti,ab,kw OR 'cod* liver oil*':ti,ab,kw OR 'eicosapen':ti,ab,kw OR 'epaisdin':ti,ab,kw OR 'epanova':ti,ab,kw OR 'omega 3':ti,ab,kw OR 'omega forte':ti,ab,kw OR 'sakana':ti,ab,kw OR 'betitol':ti,ab,kw OR 'cyclohexanehexol':ti,ab,kw OR 'cyclohexitol':ti,ab,kw OR 'dambose':ti,ab,kw OR 'hexol':ti,ab,kw OR 'inosil':ti,ab,kw OR 'inosimesol':ti,ab,kw OR 'inosit*':ti,ab,kw OR 'meat sugar':ti,ab,kw OR 'mesitol':ti,ab,kw OR 'meso inosit*':ti,ab,kw OR 'mesoinosil':ti,ab,kw OR 'mesoinosit*':ti,ab,kw OR 'mesol':ti,ab,kw OR 'mesovil':ti,ab,kw OR 'muscle sugar':ti,ab,kw OR 'myoinosil':ti,ab,kw OR 'myoinosit*':ti,ab,kw OR 'nucite':ti,ab,kw OR 'phaseomannite':ti,ab,kw OR 'phaseomannitol':ti,ab,kw OR 'scyllite':ti,ab,kw OR 'tonozil':ti,ab,kw OR 'dhasco':ti,ab,kw OR 'docosahexaenoate':ti,ab,kw OR 'docosahexaenoic acid*':ti,ab,kw OR 'docosahexenoic acid*':ti,ab,kw OR 'magnesium':ti,ab,kw OR 'romag':ti,ab,kw OR 'adaflex':ti,ab,kw OR 'aritonin':ti,ab,kw OR 'ceyestaeusom':ti,ab,kw OR 'ceyesto':ti,ab,kw OR 'circadin':ti,ab,kw OR 'civasta':ti,ab,kw OR 'eusom':ti,ab,kw OR 'lestinora':ti,ab,kw OR 'mallozen':ti,ab,kw OR 'mecastrin':ti,ab,kw OR 'melabiorytm':ti,ab,kw OR 'melatal':ti,ab,kw OR 'melatan':ti,ab,kw OR 'melatol':ti,ab,kw OR 'melatonin*':ti,ab,kw OR 'melatonite':ti,ab,kw OR 'mellaras':ti,ab,kw OR 'mellozzan':ti,ab,kw OR 'melovine':ti,ab,kw OR 'mucomel':ti,ab,kw OR 'noxarem':ti,ab,kw OR 'orlogin':ti,ab,kw OR 'sental':ti,ab,kw OR 'slenyto':ti,ab,kw OR 'sloremina':ti,ab,kw OR 'syncrodin':ti,ab,kw OR 'waferest':ti,ab,kw OR 'nutr?ceutical*':ti,ab,kw |
3851745 |
|
#3 |
#1 AND #2 |
19260 |
|
#4 |
#3 AND [2021-2024]/py NOT ('conference abstract'/it OR 'editorial'/it OR 'letter'/it OR 'note'/it) NOT (('animal'/exp OR 'animal experiment'/exp OR 'animal model'/exp OR 'nonhuman'/exp) NOT 'human'/exp) |
1895 |
|
#5 |
'meta analysis'/exp OR 'meta analysis (topic)'/exp OR metaanaly*:ti,ab OR 'meta analy*':ti,ab OR metanaly*:ti,ab OR 'systematic review'/de OR 'cochrane database of systematic reviews'/jt OR prisma:ti,ab OR prospero:ti,ab OR (((systemati* OR scoping OR umbrella OR 'structured literature') NEAR/3 (review* OR overview*)):ti,ab) OR ((systemic* NEAR/1 review*):ti,ab) OR (((systemati* OR literature OR database* OR 'data base*') NEAR/10 search*):ti,ab) OR (((structured OR comprehensive* OR systemic*) NEAR/3 search*):ti,ab) OR (((literature NEAR/3 review*):ti,ab) AND (search*:ti,ab OR database*:ti,ab OR 'data base*':ti,ab)) OR (('data extraction':ti,ab OR 'data source*':ti,ab) AND 'study selection':ti,ab) OR ('search strategy':ti,ab AND 'selection criteria':ti,ab) OR ('data source*':ti,ab AND 'data synthesis':ti,ab) OR medline:ab OR pubmed:ab OR embase:ab OR cochrane:ab OR (((critical OR rapid) NEAR/2 (review* OR overview* OR synthes*)):ti) OR ((((critical* OR rapid*) NEAR/3 (review* OR overview* OR synthes*)):ab) AND (search*:ab OR database*:ab OR 'data base*':ab)) OR metasynthes*:ti,ab OR 'meta synthes*':ti,ab |
1048530 |
|
#6 |
'clinical trial'/exp OR 'randomization'/exp OR 'single blind procedure'/exp OR 'double blind procedure'/exp OR 'crossover procedure'/exp OR 'placebo'/exp OR 'prospective study'/exp OR rct:ab,ti OR random*:ab,ti OR 'single blind':ab,ti OR 'randomised controlled trial':ab,ti OR 'randomized controlled trial'/exp OR placebo*:ab,ti |
4076308 |
|
#7 |
'major clinical study'/de OR 'clinical study'/de OR 'case control study'/de OR 'family study'/de OR 'longitudinal study'/de OR 'retrospective study'/de OR 'prospective study'/de OR 'comparative study'/de OR 'cohort analysis'/de OR ((cohort NEAR/1 (study OR studies)):ab,ti) OR (('case control' NEAR/1 (study OR studies)):ab,ti) OR (('follow up' NEAR/1 (study OR studies)):ab,ti) OR (observational NEAR/1 (study OR studies)) OR ((epidemiologic NEAR/1 (study OR studies)):ab,ti) OR (('cross sectional' NEAR/1 (study OR studies)):ab,ti) |
8334285 |
|
#8 |
'case control study'/de OR 'comparative study'/exp OR 'control group'/de OR 'controlled study'/de OR 'controlled clinical trial'/de OR 'crossover procedure'/de OR 'double blind procedure'/de OR 'phase 2 clinical trial'/de OR 'phase 3 clinical trial'/de OR 'phase 4 clinical trial'/de OR 'pretest posttest design'/de OR 'pretest posttest control group design'/de OR 'quasi experimental study'/de OR 'single blind procedure'/de OR 'triple blind procedure'/de OR (((control OR controlled) NEAR/6 trial):ti,ab,kw) OR (((control OR controlled) NEAR/6 (study OR studies)):ti,ab,kw) OR (((control OR controlled) NEAR/1 active):ti,ab,kw) OR 'open label*':ti,ab,kw OR (((double OR two OR three OR multi OR trial) NEAR/1 (arm OR arms)):ti,ab,kw) OR ((allocat* NEAR/10 (arm OR arms)):ti,ab,kw) OR placebo*:ti,ab,kw OR 'sham-control*':ti,ab,kw OR (((single OR double OR triple OR assessor) NEAR/1 (blind* OR masked)):ti,ab,kw) OR nonrandom*:ti,ab,kw OR 'non-random*':ti,ab,kw OR 'quasi-experiment*':ti,ab,kw OR crossover:ti,ab,kw OR 'cross over':ti,ab,kw OR 'parallel group*':ti,ab,kw OR 'factorial trial':ti,ab,kw OR ((phase NEAR/5 (study OR trial)):ti,ab,kw) OR ((case* NEAR/6 (matched OR control*)):ti,ab,kw) OR ((match* NEAR/6 (pair OR pairs OR cohort* OR control* OR group* OR healthy OR age OR sex OR gender OR patient* OR subject* OR participant*)):ti,ab,kw) OR ((propensity NEAR/6 (scor* OR match*)):ti,ab,kw) OR versus:ti OR vs:ti OR compar*:ti OR ((compar* NEAR/1 study):ti,ab,kw) OR (('major clinical study'/de OR 'clinical study'/de OR 'cohort analysis'/de OR 'observational study'/de OR 'cross-sectional study'/de OR 'multicenter study'/de OR 'correlational study'/de OR 'follow up'/de OR cohort*:ti,ab,kw OR 'follow up':ti,ab,kw OR followup:ti,ab,kw OR longitudinal*:ti,ab,kw OR prospective*:ti,ab,kw OR retrospective*:ti,ab,kw OR observational*:ti,ab,kw OR 'cross sectional*':ti,ab,kw OR cross?ectional*:ti,ab,kw OR multicent*:ti,ab,kw OR 'multi-cent*':ti,ab,kw OR consecutive*:ti,ab,kw) AND (group:ti,ab,kw OR groups:ti,ab,kw OR subgroup*:ti,ab,kw OR versus:ti,ab,kw OR vs:ti,ab,kw OR compar*:ti,ab,kw OR 'odds ratio*':ab OR 'relative odds':ab OR 'risk ratio*':ab OR 'relative risk*':ab OR 'rate ratio':ab OR aor:ab OR arr:ab OR rrr:ab OR ((('or' OR 'rr') NEAR/6 ci):ab))) |
14879461 |
|
#9 |
#4 AND #5 – SR’s |
256 |
|
#10 |
#4 AND #6 NOT #9 – RCT’s |
269 |
|
#11 |
#4 AND (#7 OR #8) NOT (#9 OR #10) – Observationele studies |
716 |
|
#12 |
#9 OR #10 OR #11 |
1241 |
Ovid/Medline
|
# |
Searches |
Results |
|
1 |
exp Infertility, Male/ or ((exp Infertility/ or Fertility/) and exp Male/) or ((male* or men or man) adj3 (infertil* or hypofertil* or fertil* or subfertil* or infecund* or childless* or reproduct*)).ti,ab,kf. or ((spermatogen* or spermiation or spermiogen*) adj3 failure).ti,ab,kf. or (spermatozoon adj3 (abnormalit* or anomal*)).ti,ab,kf. or ((semen or sperm) adj3 (qualit* or quantit* or parameter*)).ti,ab,kf. or asperm*.ti,ab,kf. or asthenazoosperm*.ti,ab,kf. or asthenosperm*.ti,ab,kf. or asthenozoosperm*.ti,ab,kf. or azoosperm*.ti,ab,kf. or cryptosperm*.ti,ab,kf. or cryptozoosperm*.ti,ab,kf. or hyposperm*.ti,ab,kf. or oligoasthenosperm*.ti,ab,kf. or oligosperm*.ti,ab,kf. or oligozoosperm*.ti,ab,kf. or retrograde ejaculat*.ti,ab,kf. or sco syndrome*.ti,ab,kf. or sertoli cell only syndrome*.ti,ab,kf. or teratozoosperm*.ti,ab,kf. or thermosensitive genic male steril*.ti,ab,kf. or globozoosperm*.ti,ab,kf. or macrozoosperm*.ti,ab,kf. |
101325 |
|
2 |
exp Dietary Supplements/ or exp Iron/ or exp Minerals/ or exp Nutritional Support/ or exp Vitamins/ or exp Zinc/ or exp Selenium/ or exp Glutathione/ or exp Folic Acid/ or exp Ubiquinone/ or exp Carnitine/ or exp Carotenoids/ or exp Lycopene/ or exp beta Carotene/ or exp Ascorbic Acid/ or exp Acetylcysteine/ or exp alpha-Tocopherol/ or exp Fish Oils/ or exp Fatty Acids, Omega-3/ or exp Fatty Acids/ or exp Plant Oils/ or exp Xanthophylls/ or exp Thioctic Acid/ or exp Antioxidants/ or exp Vitamin B Complex/ or exp Arginine/ or exp Flavonoids/ or exp Riboflavin/ or exp Castor Oil/ or exp Olive Oil/ or exp Safflower Oil/ or exp Fatty Acids, Essential/ or exp Arachidonic Acid/ or exp Linoleic Acid/ or exp Fatty Acids, Unsaturated/ or exp alpha-Linolenic Acid/ or exp gamma-Linolenic Acid/ or exp Fatty Acids, Omega-6/ or exp Cod Liver Oil/ or exp Inositol/ or exp Docosahexaenoic Acids/ or exp Magnesium/ or exp Melatonin/ or (((nutritional or iron) adj3 (therap* or treatment* or support*)) or ((plant* or seed* or vegetable* or dietary or edible or ricinus or cast?r or ricin* or olive* or safflower or fish or tuna) adj3 (oil* or fat)) or supplement* or vitamin* or zinc or zincum or selenicum or selenium or glutathine or glutathiol or glutathion or glutathione or glutathioral or glutatiol or tathion or tationil or tiobione or acfol or acifolic or filicine or folacid* or folacin or folart or folate or folavit or foldine or foliamin or folicet or folicid or folina or folinsyre or folitab or folivit or folsan or folsav or folverlan or folvite or ingafol or lafol or lexpec or megafol or neocepri or rubiefol or sodium folate or speciafoldine or vifolin or ubiquinone or coenzyme q* or abedine or biocarn or biomux or cardiogen or carnicor or carnil or carnitene* or carnitin or carnitine* or carnitor or carnovis or cartin or entomin or lefcar or monocamin or nefrocarnit or novain or vitacarn or carotenoid* or carotinoid* or astaxanthin* or astaxanthine* or ovoester or lycopene* or menevit or multivitamin* or b-tene or beta carotene or beta carotin* or betacarotene* or betatene* or carotaben or lurotin or solatene or solvin or acidylina or adenex or agrumina or allercorb or allescorb or arcavit c or arcavite c or arkovital c or ascelat or ascofar or ascomed or asconvita or ascor or ascorbate or ascorbate sodium or ascorbic acid* or ascorbicap or ascorbicin or ascorbico or ascorbin or ascorbina or ascorbinic acid* or ascorbit or ascorbite or ascorbitol or ascorbivit or ascorbivite or ascorbone or ascorbutina or ascorbyl or ascorbyn or ascorcee or ascorgil or ascorin or ascormin or ascorteal or ascorval or ascorvel or ascorvit or ascorvite or ascorvitina or askorbin or austrovit c or austrovite c or bentavit c or bentavite c or cantan or cantaxin or cebetate or cebicure or cebiolon or cebion or cebione or cecap or cecon or cecone or cecorbin or cecorbine or cecorbyl or cecorbyle or cecrisina or cedon or cedone or cedoxon or cedoxone or ceevifil or cegiolan or celaskon or celaskone or celin or celine or cenetone or cenol or cenolate or cequinyl or cereon or cergona or cescorbat or cetamican or cetamid or cetamine or cetebe or ceterapion or ceterapione or cetrinets or cevalin or cevaline or cevatine or cevex or cevibram or cevigal or cevigen or cevigol or cevikap or cevilat or cevimin or cevimine or cevisol or cevit or cevita or cevitamin* or cevitan or cevite or cevitex or cevitil or cevitol or cewin or chewcee or ci drol or ciamin or ciergin or cifilina or cipca or cisir or citamino or cith or citoascorbina or citoxyl or citran or citravite or citritabs or citrovitamin* or civigor or civitin or civitine or concemin or cortalex or dagrascorbin or dagravit c or dancimin c or davitamon c or dayvital or delo c or difvitamin* c or dropice or dumovit c or dumovite c or esuron or esurvit or esurvite or flavettes or hicee or hybrin or ikacee or irocevit or irocevite or jarexin or jarexine or lacivit or lacivite or laroscorbine or lemascorb or levo ascorbate or levo ascorbic acid* or limcee or limo ce or liqui cee or myascorbin or natrascorb or novo ascorbic or nybadol or pascorbin or pharmascorbine or potassium ascorbate or proscorbin or proscorbine or redoxon or redoxon forte or ribena or scorbacid* or scorbettes or scorbex or scorbitol or scorbumine or secorbate or sevalin or sodascorbate or sodium ascorbate or sweetcee or testascorbic or upsavit or vicef or vicelat or vicetrin or viciman or vicin or vicitina or vicon or viforcit or viforcite or viscorin or viscorine or vitac or vitace or vitacee or vitaci or vitacimin or vitacimine or vitacin or vitacine or vitaplex c or vitapric or vitapur c or vitasan c or vitascorbin or vitascorbine or vitascorbol or vitelix c or vitocee or vorange or wandervit c or wandervite c or witamina c or xitix or xyloascorbic acid* or acerac or acetadote or acetain or acetyl cysteine or acetylcystein or acetylcysteine or acypront or airbron or alveolex or bromuc or bronchocil or brunac or cetilan or cetylev or drenaflen or ecomucyl or euronac or exomuc or fabrol or fluimicil or fluimucil or fluimukan or flumil or flumil infantil or fluprowit or flutafin or genac or granon or hidonac or ilube or inspir or lappe or libramucil or lysomucil or lysox or menaxol or mercapturic acid* or mucocil or mucodrill or mucofillin or mucolator or mucomiste or mucomyst or mucomystendo or mucopect or mucoserin or mucosil or mucosof or mucosolvin or mucosten or mucoza or mukolit or muteran or parvolex or pulmosal or reolin or respaire or rinofluimucil or sigamucil or solmucol or spatam or sputoprompt or stecin or tirocular or tixair or zifluvis or ethylcysteine or alpha tocopherol* or alpha tocopheryl acetate or alpha tocopheryl acetic acid* or aquasol e or austrovit e or covitol or dagravit e or dalfatol or davitamon e or dermorelle or detulin or dumovit e or e ferol or e perle or e perte or e recordati or e toplex or e vicotrat or e vimin or e vita or e viterbin or ecoferol or efer or eferol or enoulan forte or ephynal or eplonat or eprolin or erevit or esol or esorb or eterapion or eviabit or evigen or eviol or evion or evit or evitol or godabion e or gonavit or hanobak or ido e or juvela or juvele or livingpherol or metaperex or mulsal e or natopherol or optovit e or phytoferol or pletocol or socopherol or spondyvit or toco 500 or tocoferolo bioglan or tocomine or tocopherex or tocopherol acetate or tocopherol gumpro or tocopheryl acetate or tocophrine or tocovigor or tocovital or toferol or topherol or vibolex e or vidom e or viea or vietal or viprimol or viteolin or viteoline or wandervit e or ameu or efamed or epax or feniko or himega or k 85 or lachs 550 or lipitac or maxepa or olemar or omegaven or optimepa or pikasol or promega or super epa or superepa or omega* or fat* acid* or arginin* or arginyl residue or argivene or bioarginina or r gene 10 or flavonoid* or beflavin* or beflavine* or flavaxin or hyrye or lactoflavin* or ovoflavin or pabriflan or riboflavin* or ribovel or leucocianidol or leucocyanidin* or leucocyanidol or pycnogenol or pygnoforton or pyknogenol or lutein* or xanthophyl* or xantofyl* or xantophyl* or berlithione or biletan or heparlipon or lipoate or lipoic acid* or liponic acid* or neurothioct or protogen or thioctacid* or thioctan or thioctic acid* or thiogamma or thiotocid or tioctan or tioctic acid* or tioctidasi or antioxidant* or antioxidat* or antoxidant* or radical scavenger* or neoloid or lyprinol or arachidonate* or arachidonate sodium or arachidonic acid* or sodium arachidonate* or linoic acid* or linolate* or linoleate* or linoleic acid* or linolic acid* or sodium linoleate* or ufa or unsaturated fat* or unsaturated lipid* or linolenate* or linolenic acid* or efagel or polyunsaturat* fat* or cod* liver oil* or eicosapen or epaisdin or epanova or omega 3 or omega forte or sakana or betitol or cyclohexanehexol or cyclohexitol or dambose or hexol or inosil or inosimesol or inosit* or meat sugar or mesitol or meso inosit* or mesoinosil or mesoinosit* or mesol or mesovil or muscle sugar or myoinosil or myoinosit* or nucite or phaseomannite or phaseomannitol or scyllite or tonozil or dhasco or docosahexaenoate or docosahexaenoic acid* or docosahexenoic acid* or magnesium or romag or adaflex or aritonin or ceyestaeusom or ceyesto or circadin or civasta or eusom or lestinora or mallozen or mecastrin or melabiorytm or melatal or melatan or melatol or melatonin* or melatonite or mellaras or mellozzan or melovine or mucomel or noxarem or orlogin or sental or slenyto or sloremina or syncrodin or waferest or nutr?ceutical*).ti,ab,kf. |
3033172 |
|
3 |
1 and 2 |
14471 |
|
4 |
limit 3 to yr="2021 -Current" |
3678 |
|
5 |
4 not (comment/ or editorial/ or letter/) not ((exp animals/ or exp models, animal/) not humans/) |
2242 |
|
6 |
meta-analysis/ or meta-analysis as topic/ or (metaanaly* or meta-analy* or metanaly*).ti,ab,kf. or systematic review/ or cochrane.jw. or (prisma or prospero).ti,ab,kf. or ((systemati* or scoping or umbrella or "structured literature") adj3 (review* or overview*)).ti,ab,kf. or (systemic* adj1 review*).ti,ab,kf. or ((systemati* or literature or database* or data-base*) adj10 search*).ti,ab,kf. or ((structured or comprehensive* or systemic*) adj3 search*).ti,ab,kf. or ((literature adj3 review*) and (search* or database* or data-base*)).ti,ab,kf. or (("data extraction" or "data source*") and "study selection").ti,ab,kf. or ("search strategy" and "selection criteria").ti,ab,kf. or ("data source*" and "data synthesis").ti,ab,kf. or (medline or pubmed or embase or cochrane).ab. or ((critical or rapid) adj2 (review* or overview* or synthes*)).ti. or (((critical* or rapid*) adj3 (review* or overview* or synthes*)) and (search* or database* or data-base*)).ab. or (metasynthes* or meta-synthes*).ti,ab,kf. |
763261 |
|
7 |
exp clinical trial/ or randomized controlled trial/ or exp clinical trials as topic/ or randomized controlled trials as topic/ or Random Allocation/ or Double-Blind Method/ or Single-Blind Method/ or (clinical trial, phase i or clinical trial, phase ii or clinical trial, phase iii or clinical trial, phase iv or controlled clinical trial or randomized controlled trial or multicenter study or clinical trial).pt. or random*.ti,ab. or (clinic* adj trial*).tw. or ((singl* or doubl* or treb* or tripl*) adj (blind$3 or mask$3)).tw. or Placebos/ or placebo*.tw. |
2756182 |
|
8 |
Epidemiologic studies/ or case control studies/ or exp cohort studies/ or Controlled Before-After Studies/ or Case control.tw. or cohort.tw. or Cohort analy$.tw. or (Follow up adj (study or studies)).tw. or (observational adj (study or studies)).tw. or Longitudinal.tw. or Retrospective*.tw. or prospective*.tw. or consecutive*.tw. or Cross sectional.tw. or Cross-sectional studies/ or historically controlled study/ or interrupted time series analysis/ [Onder exp cohort studies vallen ook longitudinale, prospectieve en retrospectieve studies] |
4786178 |
|
9 |
Case-control Studies/ or clinical trial, phase ii/ or clinical trial, phase iii/ or clinical trial, phase iv/ or comparative study/ or control groups/ or controlled before-after studies/ or controlled clinical trial/ or double-blind method/ or historically controlled study/ or matched-pair analysis/ or single-blind method/ or (((control or controlled) adj6 (study or studies or trial)) or (compar* adj (study or studies)) or ((control or controlled) adj1 active) or "open label*" or ((double or two or three or multi or trial) adj (arm or arms)) or (allocat* adj10 (arm or arms)) or placebo* or "sham-control*" or ((single or double or triple or assessor) adj1 (blind* or masked)) or nonrandom* or "non-random*" or "quasi-experiment*" or "parallel group*" or "factorial trial" or "pretest posttest" or (phase adj5 (study or trial)) or (case* adj6 (matched or control*)) or (match* adj6 (pair or pairs or cohort* or control* or group* or healthy or age or sex or gender or patient* or subject* or participant*)) or (propensity adj6 (scor* or match*))).ti,ab,kf. or (confounding adj6 adjust*).ti,ab. or (versus or vs or compar*).ti. or ((exp cohort studies/ or epidemiologic studies/ or multicenter study/ or observational study/ or seroepidemiologic studies/ or (cohort* or 'follow up' or followup or longitudinal* or prospective* or retrospective* or observational* or multicent* or 'multi-cent*' or consecutive*).ti,ab,kf.) and ((group or groups or subgroup* or versus or vs or compar*).ti,ab,kf. or ('odds ratio*' or 'relative odds' or 'risk ratio*' or 'relative risk*' or aor or arr or rrr).ab. or (("OR" or "RR") adj6 CI).ab.)) |
5746168 |
|
10 |
5 and 6 – SR’s |
176 |
|
11 |
(5 and 7) not 10 – RCT’s |
308 |
|
12 |
(5 and (8 or 9)) not (10 or 11) – Observationele studies |
367 |
|
13 |
10 or 11 or 12 |
851 |