Biomarkers bij MCI
Uitgangsvraag
Wat is de waarde van de toepassing van MRI, PET en liquoronderzoek bij het voorspellen van progressie van MCI naar dementie?
De uitgangsvraag omvat de volgende deelvragen:
- Wat is de waarde van de toepassing van MRI bij het voorspellen van progressie van MCI naar dementie?
- Wat is de waarde van de toepassing van liquoronderzoek bij het voorspellen van progressie van MCI naar dementie?
- Wat is de waarde van de toepassing van PET bij het voorspellen van progressie van MCI naar dementie?
Aanbeveling
1. MRI
Verricht niet standaard een MRI scan bij patiënten met MCI om de progressie naar dementie te kunnen voorspellen.
2. CSF
Verricht niet standaard liquoronderzoek bij patiënten met MCI om de progressie naar dementie te kunnen voorspellen.
Overweeg liquoronderzoek met bepaling van p-tau/Aß of t-tau/Aß ratio als er een sterke wens is om Alzheimerpathologie als oorzaak van de de cognitieve symptomen uit te sluiten en dan met name bij jonge mensen. Hierbij dient het risico op een fout-positieve uitslag zorgvuldig gewogen en besproken te worden met de patiënt. Neem bij twijfel over de interpretatie contact op met een expert.
3. PET
- FDG: Verricht geen FDG PET scan bij patiënten met MCI om de progressie naar dementie te kunnen voorspellen.
- Amyloid: Verricht niet standaard een amyloid PET scan bij patiënten met MCI om de progressie naar dementie te kunnen voorspellen.
Overweeg een amyloid PET als er een sterke wens is om Alzheimerpathologie als oorzaak van de de cognitieve symptomen uit te sluiten en dan met name bij jonge mensen. Hierbij dient het risico op een fout-positieve uitslag zorgvuldig gewogen en besproken te worden met de patiënt. Neem bij twijfel over de interpretatie contact op met een expert.
Overwegingen
Gezien de onderzochte testen en de overwegend geheugenpolikliniek populaties, zijn de aanbevelingen primair van toepassing op de diagnostiek in de tweede en derde lijn. Toch zijn de aanbevelingen van belang voor de eerstelijn, zodat patiënten ook goed geinformeerd kunnen worden over wat aanvullend onderzoek in het ziekenhuis ze wel en niet kan brengen. Dit kan helpen om samen met de patiënt een passende beslissing te nemen over het doel en nut van een verwijzing.
Balans tussen gewenste en ongewenste effecten
Op basis van meerdere systematische reviews blijkt dat er onvoldoende bewijs is dat MRI, liquor onderzoek of PET-scan van waarde zijn om te voorspellen welke mensen met MCI in de jaren erna dementie ontwikkelen. De bewijskracht van de uitgevoerde studies is over het algemeen laag tot zeer laag, vanwege belangrijke methodologsiche tekortkomingen op diverse vlakken wat leidt tot diverse bronnen van bias. Onderzoek dat is opgezet vanuit de klinische vraag bij een representatieve populatie in de eerstelijn of op de geheugenpolikliniek ontbreekt. Hierdoor is onbekend in hoeverre de drie index testen van toegevoegde waarde zouden kunnen zijn bij de voorspelling welke mensen met MCI dementie gaan ontwikkelen, als de klinische gegevens mee worden gewogen.
Het is opvallend dat bij alle drie de indextesten steeds met relatief korte follow-up van maximaal enkele jaren, gemiddeld 1/3 van de patiënten met MCI dementie ontwikkelt. Dit suggereert dat het betrekkelijk geselecteerde populaties met een relatief hoog risico betreft, waarbij deze het meest vergelijkbaar lijken met de populatie in de geheugenkliniek. Het is onzeker in hoeverre deze getallen van toepassing zijn op de patiënten met MCI die bij de huisarts komen.
Er zijn een paar belangrijke kanttekeningen te maken.
In de eerste plaats is het Alzheimer veld in beweging, en veranderen criteria voor de ziekte van Alzheimer. Er zijn verschillende expertise commissies welke verschillende concurrerende criteria hebben opgesteld, waarbij er vooralsnog geen helderheid is over de toepassing en meerwaarde voor de diagnosestelling. Dit werd recent uitgebreid in een overzicht samengevat (van der Molen, 2025). Dit betreft voornamelijk criteria met een meer centrale rol voor biologische markers waaronder amyloid-beta gemeten in liquor, bloed of met PET onderzoek. Ook hier blijft een belangrijke beperking dat het onduidelijk is wat de waarde in de patiëntenzorg is, omdat voor geen van de gebruikte biomarkers - gemeten in liquor, bloed of met PET – overtuigend is aangetoond dat ze betrouwbaar voorspellen welke MCI-patiënt dementie ontwikkelt en wanneer.
In de tweede plaats blijkt uit het huidige onderzoek dat de specificiteit van beschikbare markers steeds laag is, waardoor er veel fout positieve uitslagen komen. Dit is problematisch, omdat ten onrechte de diagnose ziekte van Alzheimer stellen bij iemand die geen dementie ontwikkelt, schadelijk kan zijn. Er zijn klinische situaties denkbaar waarbij het belang van een negatieve test zwaar kan wegen, omdat dan een andere differentiaal diagnose voor de MCI moet worden uitgewerkt, zoals bijvoorbeeld een depressie die gepaard kan gaan met lichte cognitieve stoornissen. Dit vraagt zorgvuldig en uitgebreid overleg met de patiënt en uitleg wat de betekenis van een negatieve en een positieve test is. Dit zijn geen eenvoudige gesprekken. Het is belangrijk dat in deze gesprekken aandacht is voor de prognose. Er zijn studies die getallen rapporteren over conversie van MCI naar dementie bij een positieve amyloid test (Huszár, 2024; Vos, 2015). Echter zijn deze studies niet opgezet vanuit een klinisch perspectief, waardoor er geen duidelijkheid is over de toegevoegde waarde van deze onderzoeken bovenop klinische gegevens.
In de derde plaats hebben we op basis van de literatuur geen onderscheid kunnen maken naar leeftijd. Toch zijn er aanwijzingen dat de specificiteit van biomarker testen op jonge leeftijd hoger is, en er dus minder fout-positieve uitslagen zijn, aangezien de prevalentie van amyloid pathologie in de populatie op jongere leeftijd lager is (Jansen, 2015). Daarmee kan de overweging om een of meerdere van de index testen in te zetten bij relatief jonge mensen anders zijn. Ook kan het voor jonge mensen belangrijker zijn om Alzheimer pathologie uit te sluiten, waarmee progressie naar dementie in de komende jaren onwaarschijnlijk wordt.
In de vierde plaats wordt soms een MRI scan met als doel uitsluiting van (neurochirurgisch) behandelbare aandoeningen overwogen, indien daar aanleiding voor is vanwege een atypisch beloop of focale afwijkingen bij neurologisch onderzoek. In de module “Structurele beeldvorming dementie” staat al beschreven voor de dementie populatie wat de waarde is van CT en MRI voor de (differentiaal) diagnostiek, waaronder een behandelbare aandoening in de differentiaal diagnose (NVKG, 2014).
Tot slot zijn de gebruikte systematische reviews al wat ouder, waardoor de meest recente literatuur niet is meegenomen. Bij bestudering van een selectie van meer recente artikelen, blijkt dat de belangrijkste methodologische beperkingen en bronnen van bias min of meer onveranderd zijn, en dat onderzoek dat is opgezet vanuit de diagnostische of prognostische vraag in de spreekkamer waarin goed geëvalueerd wordt of er patiënt-relevante consequenties zijn aan het doen van een van de index-testen, ontbreekt. De meer recente artikelen maken daarnaast vaker gebruik van predictiemodellen met meerdere variabelen, waarbij geen gebruik wordt gemaakt van diagnostische uitkomstmaten. Daarmee sluit de meer recente literatuur, alhoewel waardevol, niet goed aan bij klinische vraag van de PICO in deze richtlijn. Er zijn onderzoeken die cognitieve achteruitgang proberen te voorspellen op basis van combinaties van biomarkers en klinische kenmerken (bijvoorbeeld Van der Veere, 2024). Deze modellen zijn vooralsnog onvoldoende gevalideerd voor klinische toepassing.
Kwaliteit van bewijs
MRI
De overall kwaliteit van bewijs is zeer laag (zie SoF tabel 1). Dit betekent dat het cluster zeer onzeker is over het gevonden geschatte effect van de cruciale uitkomstmaten.
CSF
De overall kwaliteit van bewijs is zeer laag (zie SoF tabel 2). Dit betekent dat het cluster zeer onzeker is over het gevonden geschatte effect van de cruciale uitkomstmaten.
PET
De overall kwaliteit van bewijs is zeer laag (zie SoF tabel 3). Dit betekent dat het cluster zeer onzeker is over het gevonden geschatte effect van de cruciale uitkomstmaten.
Waarden en voorkeuren van patiënten (en eventueel hun naasten/verzorgers)
Effecten op patiënt relevante uitkomstmaten zijn niet onderzocht. Enkele onderzoeken gebruiken diagnostische zekerheid van de arts als uitkomstmaat, maar dit is geen uitkomstmaat die informatie geeft over of de patiënt baat heeft bij de test.
Er zijn situaties voorstelbaar waarin de hoge negatief voorspellende waarde relevant is, om de ziekte van Alzheimer uit te sluiten. Dit kan bijvoorbeeld relatief jonge mensen betreffen, mensen bij wie de ziekte van Alzheimer bij meerdere familieleden voorkomt of als er een differentiaal diagnose is met een depressie als oorzaak van de lichte cognitieve stoornissen. Dit belang moet altijd afgewogen worden tegen de kans op een fout positieve uitslag en de consequenties die deze kan hebben voor een patiënt.
In het kader van de huidige richtlijn module heeft geen raadpleging van de doelgroep (mensen met MCI en hun naasten) plaatsgevonden.
Kostenaspecten
De kosten verschillen per indextest. Veel mensen met lichte cognitieve stoornissen blijven voor diagnostiek in de eerstelijn, zeker mensen ouder dan 65 jaar. MRI, liquor onderzoek en PET onderzoek zou voor deze mensen betekenen dat er ook een consult bij een medisch specialist bij komt, omdat de onderzoeken niet vanuit de eerstelijn worden aangevraagd (een enkele uitzondering voor MRI daargelaten) en daarmee dus een toename van de kosten. Bij mensen met MCI die met een zorgvraag in de tweedelijns- of derdelijnszorg komen wordt regelmatig structurele beeldvorming (MRI of CT) verricht om andere mogelijk behandelbare oorzaken uit te sluiten, hoewel dat volgens de vigerende richtlijnen niet standaard nodig is. Liquoronderzoek of PET scan in de tweedelijn of derdelijn zijn niet standaard en leiden tot een toename van de kosten. Vanwege de afwezigheid van bewijs dat de indextesten toegevoegde waarde hebben om vast te stellen welke patiënt met MCI de komende jaren dementie ontwikkelt, leveren alle drie de testen een toename van kosten op zonder dat hier een duidelijke winst tegenover staat. Er is beschreven dat het verrichten van een amyloid PET bij een geheugenkliniek populatie in Nederland geassocieerd is met latere opname in een verpleeghuis en lagere zorgkosten (van Maurik, 2023). In een internationale studie is dit verband echter niet zo duidelijk aangetoond, maar was de follow-up periode veel korter (maar 12 maanden). Gedegen kosten-effectiviteits analyses ontbreken (Rabinovici, 2023).
Gezondheidsgelijkheid
Er zijn geen aanwijzingen dat het toevoegen van een van de drie indextesten leidt tot een toename van gezondheidsongelijkheid in Nederland. Diagnostiek naar dementie valt binnen de zorgverzekering, zowel bij de huisarts als in het ziekenhuis, inclusief MRI, liquor onderzoek en FDG PET. Amyloid PET onderzoek is niet volledig verzekerde zorg, maar valt onder het ziekenhuis budget. De beschikbaarheid kan dan ook variëren per locatie. Wel kan in theorie verwijzing naar een specialist om een van de drie testen te laten doen een barrière vormen voor individuele patiënten, omdat deze zorg onder het eigen risico valt, terwijl diagnostiek in de eerstelijn niet onder het eigen risico valt.
Aanvaardbaarheid
Ethische aanvaardbaarheid
Het doen van een MRI-scan van de hersenen lijkt aanvaardbaar voor de patiënten en wordt over het algemeen als weinig belastend ervaren.
Het doen van een lumbaalpunctie lijkt aanvaardbaar. De risico’s van een lumbaalpunctie zijn laag. Wel levert het onderzoek ongemak op voor de betreffende patiënt. Bij gebrek aan duidelijk toegevoegde waarde voor patiënten is het onderzoek vanwege de (lage) risico’s en het ongemak mogelijk niet aanvaardbaar voor een deel van de patiënten.
Het doen van een PET-scan lijkt aanvaardbaar. Het onderzoek is weinig belastend, maar er wordt wel een lage dosis radio-actief materiaal toegediend. Bij gebrek aan duidelijk toegvoegde waarde is het onderzoek voor de meeste patiënten waarschijnlijk niet aanvaardbaar.
Duurzaamheid
Het doen van geen aanvullend onderzoek is het meest duurzaam. Bij afwezige duidelijke toegevoegde waarde van een index-test, is het niet duurzaam om deze test uit te voeren. Een goede onderbouwing waarom het bij een individuele patiënt toch meerwaarde heeft, is nodig.
Haalbaarheid
Het maken van een MRI-scan bij patiënten met MCI is haalbaar bij patiënten die een geheugenpolikliniek bezoeken. Het maken van een MRI hersenen bij patiënten bij wie de diagnostiek in de eerstelijn plaatsvindt is waarschijnlijk niet haalbaar, omdat 1) dit leidt tot een toename van de belasting van de MRI capaciteit; 2) dit leidt tot aanzienlijk hogere kosten in het zorgsysteem en 3) de interpretatie altijd plaats dient te vinden in de klinische context, door een clinicus die de uitslag van de MRI-scan zelf kan interpreteren en kan relateren aan de klinische verschijnselen – wat in de eerstelijn meestal niet mogelijk is door gebrek aan expertise.
Het doen van liquor onderzoek bij alle mensen met MCI is niet haalbaar vanwege capaciteitsproblemen. Het grootste deel van de patiënten met MCI wordt in de eerstelijn of bij de geriater gezien. Beiden doen over het algemeen zelf geen lumbaalpuncties. Op dit moment wordt liquor onderzoek bij minder dan 5-10% van alle mensen met MCI verricht, voornamelijk mensen < 65 jaar.
Het maken van een PET-scan van de hersenen bij alle mensen met MCI is niet haalbaar vanwege capaciteitsproblemen en kosten. Bij patiënten met MCI in de geheugenkliniek kan een PET scan in specifieke gevallen verricht worden (bijvoorbeeld wanneer liquoronderzoek gewenst is, maar niet mogelijk door gebruik van antistollingsmedicatie). Hiervoor dient de juiste expertise in het betreffende centrum aanwezig te zijn.
Rationale van de aanbeveling: weging van argumenten voor en tegen de interventies
Voor MRI is een sterke aanbeveling tegen standaard toepassen opgesteld, omdat er niet voldoende bewijs is voor de meerwaarde bij het voorspellen van progressie van MCI naar dementie. Daarbij is meegewogen dat het uitvoeren van MRI leidt tot een extra handeling met bijbehorende kosten en belasting van de MRI capaciteit.
Voor CSF is een sterke aanbeveling tegen standaard toepassen opgesteld, omdat niet voldoende bewijs is voor de meerwaarde bij het voorspellen van progressie van MCI naar dementie. Daarbij is meegewogen dat het uitvoeren van CSF leidt tot een extra handeling met bijbehorende kosten en een handeling is waarbij patiënten ongemak kunnen ervaren.
Voor PET is een sterke aanbeveling tegen standaard toepassen opgesteld, omdat niet voldoende bewijs is voor de meerwaarde bij het voorspellen van progressie van MCI naar dementie. Daarbij is meegewogen dat het uitvoeren van PET leidt tot een extra handeling met bijbehorende kosten en het toedienen van radio-actief materiaal voor patiënten mogelijk niet aanvaardbaar is gezien het gebrek aan onderbouwing voor de diagnostische meerwaarde.
Eindoordeel: Conditionele aanbeveling.
Onderbouwing
Achtergrond
Patiënts with mild cognitive impairment (MCI) are a heterogeneous group, both in terms of etiology and prognosis. Sometimes an early stage of a neurodegenerative or other brain disease will eventually lead to dementia, but some patiënts with MCI never develop dementia. Better prediction of whether and when patiënts with MCI develop dementia is relevant. Several techniques may possibly contribute to a better prediction of which patiënts with MCI will and will not develop dementia:
- magnetic resonance imaging (MRI), including volumetry of gray and white matter and of specific brain structures;
- nuclear imaging of the brain, such as positron emission tomography (PET) of amyloid plaques and glucose metabolism;
- liquor cerebrospinalis (hereafter: CSF).
Conclusies / Summary of Findings
Summary of Findings
Summary of Findings table index-1: Magnetic Resonance Imaging (MRI) markers for predicting progression from MCI to Alzheimer dementia
Population: Patiënts with Mild Cognitive Impairment (MCI)
Intervention: MRI
Comparator: No marker
Reference: Pathological anatomical (PA) diagnosis or the clinical consensus diagnosis of dementia or diagnostic criteria based on DSM IV or NINCDS-ADRDA with an average minimum follow-up duration of 2 years
|
Outcome
|
Study results and measurements |
Effect estimates* |
Certainty of the Evidence (Quality of evidence) |
Conclusions |
|
Total hippocampus |
||||
|
Specificity (critical)
|
The systematic review and meta-analysis of Lombardi (2020) included 22 studies with 2209 participants to assess the accuracy of total hippocampus volume measured by MRI to detect conversion from MCI to ADD compared to other outcomes. Of the 2209 participants, 687 were diagnosed with ADD. |
0,71 (95% CI 0,65 to 0,77) |
Low Due to serious risk of bias, due to serious inconsistency1,3 |
Total hippocampus volume measured by MRI may not be of added value in accurately diagnosing progression to ADD in patiënts with MCI.
(Lombardi, 2020) |
|
Sensitivity (important) |
0,73 (95% CI 0,64 to 0,80) |
Low Due to serious risk of bias, due to serious inconsistency1,3 |
||
|
Positive likelihood ratio (important) |
2,53 (95% CI 2,09 to 3,06) |
Low Due to serious risk of bias, due to serious inconsistency1,3 |
||
|
Negative likelihood ratio (important)
|
0,38 (95% CI 0,29 to 0,50). |
Low Due to serious risk of bias, due to serious inconsistency1,3 |
||
|
Positive predictive value (important) |
-
|
- |
No GRADE (no evidence was found) |
No evidence was found regarding the positive predictive value of total hippocampus volume measured by MRI to diagnose progression to ADD in patiënts with MCI. |
|
Negative predictive value (important)
|
-
|
- |
No GRADE (no evidence was found) |
No evidence was found regarding the negative predictive value of MRI total hippocampus volume measured by MRI to diagnose progression to ADD in patiënts with MCI. |
|
Medial temporal lobe volume |
||||
|
Specificity (critical)
|
The systematic review and meta-analysis of Lombardi (2020) included seven studies with 1077 participants to assess the accuracy of medial temporal lobe volume measured by MRI to detect conversion from MCI to ADD compared to other outcomes. Of the 1077 participants, 330 were diagnosed with ADD.
|
0,65 (95% CI 0,51 to 0,76) |
Low Due to serious risk of bias, due to serious imprecision1,2 |
Medial temporal lobe volume measured by MRI may not be of added value in accurately diagnosing progression to ADD in patiënts with MCI.
(Lombardi, 2020) |
|
Sensitivity (important) |
0,64 (95% CI 0,53 to 0,73) |
Low Due to serious risk of bias, due to serious imprecision1,2 |
||
|
Positive likelihood ratio (important) |
1,81 (95% CI 1,41 to 2,32) |
Low Due to serious risk of bias, due to serious imprecision1,2 |
||
|
Negative likelihood ratio (important)
|
0,56 (95% CI 0,46 to 0,67) |
Low Due to serious risk of bias, due to serious imprecision1,2 |
||
|
Positive predictive value (important) |
-
|
- |
No GRADE (no evidence was found) |
No evidence was found regarding the positive predictive value of Medial temporal lobe volume measured by MRI to diagnose progression to ADD in patiënts with MCI. |
|
Negative predictive value (important)
|
-
|
- |
No GRADE (no evidence was found) |
No evidence was found regarding the negative predictive value of Medial temporal lobe volume measured by MRI to diagnose progression to ADD in patiënts with MCI. |
|
Total entorhinal cortex volume |
||||
|
Specificity (critical)
|
The systematic review and meta-analysis of Lombardi (2020) included four studies with 529 participants to assess the accuracy of total entorhinal cortex volume measured by MRI to detect conversion from MCI to ADD compared to other outcomes. Of the 529 participants, 229 were diagnosed with ADD. |
Meta-analyses not conducted due to sparse and heterogeneous data. Range: 0,60 to 1,00, |
Very low Due to serious risk of bias, due to serious inconsistency1-3 |
The evidence is very uncertain about whether total entorhinal cortex volume measured by MRI is accurate in diagnosing progression to ADD in patiënts with MCI.
(Lombardi, 2020) |
|
Sensitivity (important) |
Meta-analyses not conducted due to sparse and heterogeneous data. Range: 0,50 to 0,88. |
Very low Due to serious risk of bias, due to serious inconsistency1-3 |
||
|
Positive likelihood ratio (important) |
-
|
- |
No GRADE (no evidence was found) |
No evidence was found regarding the positive likelihood ratio of total entorhinal cortex volume measured by MRI to diagnose progression to ADD in patiënts with MCI. |
|
Negative likelihood ratio (important)
|
-
|
- |
No GRADE (no evidence was found) |
No evidence was found regarding the negative likelihood ratio of total entorhinal cortex volume measured by MRI to diagnose progression to ADD in patiënts with MCI. |
|
Positive predictive value (important) |
-
|
- |
No GRADE (no evidence was found) |
No evidence was found regarding the positive predictive value of total entorhinal cortex volume measured by MRI to diagnose progression to ADD in patiënts with MCI. |
|
Negative predictive value (important)
|
-
|
- |
No GRADE (no evidence was found) |
No evidence was found regarding the negative predictive value of total entorhinal cortex volume measured by MRI to diagnose progression to ADD in patiënts with MCI. |
|
Lateral ventricles volume |
||||
|
Specificity (critical)
|
The systematic review and meta-analysis of Lombardi (2020) included five studies with 1077 participants to assess the accuracy of MRI t-tau to detect conversion from MCI to ADD compared to other outcomes. Of the 1077 participants, 371 were diagnosed with ADD. |
0,64 (95% CI 0,59 to 0,70) |
Low Due to serious risk of bias, due to serious imprecision1,2 |
Lateral ventricles volume measured by MRI may not be of added value in accurately diagnosing progression to ADD in patiënts with MCI.
(Lombardi, 2020) |
|
Sensitivity (important) |
0,57 (95% CI 0,49 to 0,65) |
Low Due to serious risk of bias, due to serious imprecision1,2 |
||
|
Positive likelihood ratio (important) |
1,61 (95% CI 1,39 to 1,87) |
Low Due to serious risk of bias, due to serious imprecision1,2 |
||
|
Negative likelihood ratio (important)
|
0,66 (95% CI 0,57 to 0,78) |
Low Due to serious risk of bias, due to serious imprecision1,2 |
||
|
Positive predictive value (important) |
-
|
- |
No GRADE (no evidence was found) |
No evidence was found regarding the positive predictive value of lateral ventricles volume measured by MRI to diagnose progression to ADD in patiënts with MCI. |
|
Negative predictive value (important)
|
-
|
- |
No GRADE (no evidence was found) |
No evidence was found regarding the negative predictive value of lateral ventricles volume measured by MRI to diagnose progression to ADD in patiënts with MCI. |
|
Whole brain volume |
||||
|
Specificity (critical)
|
The systematic review and meta-analysis of Lombardi (2020) included four studies with 424 participants to assess the accuracy of whole brain volume measured by MRI to detect conversion from MCI to ADD compared to other outcomes. Of the 424 participants, 220 were diagnosed with ADD. |
Meta-analyses not conducted due to sparse and heterogeneous data. Range: 0,41 to 1,00 |
Very low Due to very serious risk of bias, due to serious inconsistency 1,3 |
The evidence is very uncertain about whether whole brain volume measured by MRI is accurate in diagnosing progression to ADD in patiënts with MCI.
(Lombardi, 2020) |
|
Sensitivity (important) |
Meta-analyses not conducted due to sparse and heterogeneous data. Range: 0,33 to 0,92 |
Very low Due to very serious risk of bias, due to serious inconsistency 1,3 |
||
|
Positive predictive value (important) |
-
|
- |
No GRADE (no evidence was found) |
No evidence was found regarding the positive predictive value of whole brain volume measured by MRI to diagnose progression to ADD in patiënts with MCI. |
|
Negative predictive value (important)
|
-
|
- |
No GRADE (no evidence was found) |
No evidence was found regarding the negative predictive value of whole brain volume measured by MRI to diagnose progression to ADD in patiënts with MCI. |
|
Positive likelihood ratio (important) |
-
|
- |
No GRADE (no evidence was found) |
No evidence was found regarding the positive likelihood ratio of whole brain volume measured by MRI to diagnose progression to ADD in patiënts with MCI. |
|
Negative likelihood ratio (important)
|
-
|
- |
No GRADE (no evidence was found) |
No evidence was found regarding the negative likelihood ratio of whole brain volume measured by MRI to diagnose progression to ADD in patiënts with MCI. |
* Summary estimates are based on the summary operating point from a hierarchical summary ROC curve (HSROC) model. CI = confidence interval.
1. Risk of bias: serious. Due to lack of blinding, participant selection (registry data) or lack of reporting on the index test (unclear pre-specified definition for a ‘positive’ result).
2. Imprecision: serious. Wide 95% confidence intervals.
3. Inconsistency: serious. Due to sparse and inconsistent data.
Summary of Findings table index-2: Cerebrospinal fluid (CSF) markers for predicting progression from MCI to Alzheimer dementia
Population: Patiënts with Mild Cognitive Impairment (MCI)
Intervention: CSF markers (amyloidβ1-42, total-tau (t-tau), phosporylated-tau181 (p-tau), ratio t-tau/aβ, ratio p-tau/aβ, neurofilament light)
Comparator: No CSF marker
Reference: Pathological anatomical (PA) diagnosis or the clinical consensus diagnosis of dementia or diagnostic criteria based on DSM IV or NINCDS-ADRDA with an average minimum follow-up duration of 2 years.
|
Outcome |
Study results and measurements |
Effect estimates* |
Certainty of the Evidence (Quality of evidence) |
Conclusions |
|
Amyloidβ42 |
||||
|
Specificity (critical)
|
The systematic review and meta-analysis of Ritchie (2014) included fourteen studies with 1349 participants to assess the accuracy of CSF amyloidß42 to detect conversion from MCI to ADD compared to other outcomes. Of the 1349 participants, 436 were diagnosed with ADD. |
The sensitivities were between 0,36 and 1,00 while the specificities were between 0,29 and 0,91. No summary measures were calculated due to variation in thresholds and measures. To illustrate potential strengths and weaknesses of CSF amyloidβ42, sensitivity of 0,81 (95% CI 0,72 to 0,87) was estimated from an HSROC curve at the included study median specificity of 0,64. This equated to a positive likelihood ratio of 2,22 (95% CI 2,00 to 2,47), and a negative likelihood ratio of 0,31 (95% CI 0,21 to 0,48). |
Very low Due to very serious risk of bias, due to serious inconsistency1,2 |
The evidence is very uncertain about whether CSF amyloidβ42 is accurate in diagnosing progression to ADD in patiënts with MCI. (Ritchie, 2014) |
|
Sensitivity (important) |
Very low Due to very serious risk of bias, due to serious inconsistency1,2 |
|||
|
Positive likelihood ratio (important) |
Very low Due to very serious risk of bias, due to serious inconsistency1,2 |
|||
|
Negative likelihood ratio (important)
|
Very low Due to very serious risk of bias, due to serious inconsistency1,2 |
|||
|
Positive predictive value (important) |
-
|
- |
No GRADE (no evidence was found) |
No evidence was found regarding the positive predictive value of CSF amyloidβ42 to diagnose progression to ADD in patiënts with MCI. |
|
Negative predictive value (important)
|
-
|
- |
No GRADE (no evidence was found) |
No evidence was found regarding the negative predictive value of CSF amyloidβ42 to diagnose progression to ADD in patiënts with MCI. |
|
Total-tau (t-tau) |
||||
|
Specificity (critical)
|
The systematic review and meta-analysis of Ritchie (2017) included seven studies with 709 participants to assess the accuracy of CSF t-tau to detect conversion from MCI to ADD compared to other outcomes. Of the 709 participants, 291 were diagnosed with ADD. |
The sensitivities were between 0,51 and 0,95 while the specificities were between 0,48 and 0,88. No summary measures were calculated due to variation in thresholds and measures. To illustrate potential strengths and weaknesses of CSF t-tau, sensitivity of 0,77 (95% CI 0,67 to 0,85) was estimated from an HSROC curve at the included study median specificity of 0,72. This equated to a positive likelihood ratio of 2,72 (95% CI 2,43 to 3,04), and a negative likelihood ratio of 0,32 (95% CI 0,22 to 0,47). |
Very low Due to very serious risk of bias, due to serious inconsistency1,2 |
The evidence is very uncertain about whether CSF t-tau is accurate in diagnosing progression to ADD in patiënts with MCI. (Ritchie, 2017) |
|
Sensitivity (important) |
Very low Due to very serious risk of bias, due to serious inconsistency1,2 |
|||
|
Positive likelihood ratio (important) |
Very low Due to very serious risk of bias, due to serious inconsistency1,2 |
|||
|
Negative likelihood ratio (important)
|
Very low Due to very serious risk of bias, due to serious inconsistency1,2 |
|||
|
Positive predictive value (important) |
-
|
- |
No GRADE (no evidence was found) |
No evidence was found regarding the positive predictive value of CSF t-tau to diagnose progression to ADD in patiënts with MCI. |
|
Negative predictive value (important)
|
-
|
- |
No GRADE (no evidence was found) |
No evidence was found regarding the negative predictive value of CSF t-tau to diagnose progression to ADD in patiënts with MCI. |
|
Phosphorylated-tau (p-tau) |
||||
|
Specificity (critical)
|
The systematic review and meta-analysis of Ritchie (2017) included six studies with 492 participants to assess the accuracy of CSF p-tau to detect conversion from MCI to ADD compared to other outcomes. Of the 492 participants, 164 were diagnosed with ADD.
|
The sensitivities were between 0,40 and 1,00 while the specificities were between 0,22 and 0,86. No summary measures were calculated due to variation in thresholds and measures. To illustrate potential strengths and weaknesses of CSF t-tau, sensitivity of 0,81 (95% CI 0,64 to 0,92) was estimated from an HSROC curve at the included study median specificity of 0,48. This equated to a positive likelihood ratio of 1,55 (95% CI 1,31 to 1,84), and a negative likelihood ratio of 0,39 (95% CI 0,19 to 0,82). |
Very low Due to very serious risk of bias, due to serious inconsistency1,2 |
The evidence is very uncertain about whether CSF p-tau is accurate in diagnosing progression to ADD in patiënts with MCI. (Ritchie, 2017) |
|
Sensitivity (important) |
Very low Due to very serious risk of bias, due to serious inconsistency1,2 |
|||
|
Positive likelihood ratio (important) |
Very low Due to very serious risk of bias, due to serious inconsistency1,2 |
|||
|
Negative likelihood ratio (important)
|
Very low Due to very serious risk of bias, due to serious inconsistency1,2 |
|||
|
Positive predictive value (important) |
-
|
- |
No GRADE (no evidence was found) |
No evidence was found regarding the positive predictive value of CSF p-tau to diagnose progression to ADD in patiënts with MCI. |
|
Negative predictive value (important)
|
-
|
- |
No GRADE (no evidence was found) |
No evidence was found regarding the negative predictive value of CSF p-tau to diagnose progression to ADD in patiënts with MCI. |
|
t-tau/Aß ratio |
||||
|
Specificity (critical)
|
The systematic review and meta-analysis of Ritchie (2017) included two studies with 251 participants to assess the accuracy of CSF t-tau/Aß to detect conversion from MCI to ADD compared to other outcomes. Of the 251 participants, 102 were diagnosed with ADD. |
One study (n=37) reported a sensitivity of 0,91 and a specificity of 0,50. The other study (n=214) reported a sensitivity of 0,96 and a specificity of 0,51.
|
Very low Due to very serious risk of bias, due to serious imprecision1,3 |
The evidence is very uncertain about whether CSF t-tau/Aß ratio is accurate in diagnosing progression to ADD in patiënts with MCI. (Ritchie, 2017) |
|
Sensitivity (important) |
Very low Due to very serious risk of bias, due to serious imprecision1, |
The evidence is very uncertain about whether CSF t-tau/Aß ratio is accurate in diagnosing progression to ADD in patiënts with MCI. (Ritchie, 2017) |
||
|
Positive likelihood ratio (important) |
- |
- |
No GRADE (no evidence was found) |
No evidence was found regarding the positive likelihood ratio of CSF t-tau/Aß ratio to diagnose progression to ADD in patiënts with MCI. |
|
Negative likelihood ratio (important)
|
- |
- |
No GRADE (no evidence was found) |
No evidence was found regarding the negative likelihood ratio of CSF t-tau/Aß ratio to diagnose progression to ADD in patiënts with MCI. |
|
Positive predictive value (important) |
- |
- |
No GRADE (no evidence was found) |
No evidence was found regarding the positive predictive value of CSF t-tau/Aß ratio to diagnose progression to ADD in patiënts with MCI. |
|
Negative predictive value (important)
|
- |
- |
No GRADE (no evidence was found) |
No evidence was found regarding the negative predictive value of CSF t-tau/Aß ratio to diagnose progression to ADD in patiënts with MCI. |
|
p-tau/Aß ratio |
||||
|
Specificity (critical)
|
The systematic review and meta-analysis of Ritchie (2017) included six studies with 492 participants to assess the accuracy of CSF p-tau to detect conversion from MCI to ADD compared to other outcomes. Of the 492 participants, 164 were diagnosed with ADD. |
Specificity ranged from 0,33 to 0,95 across studies. Sensitivity ranged from 0,80 to 0,96 across studies. No meta-analysis based on the HSROC curve was performed as the studies were few and small. |
Very low Due to very serious risk of bias, due to serious imprecision1,3 |
The evidence is very uncertain about whether CSF p-tau/Aß ratio is accurate in diagnosing progression to ADD in patiënts with MCI. (Ritchie, 2017) |
|
Sensitivity (important) |
Very low Due to very serious risk of bias, due to serious imprecision1,3 |
|||
|
Positive likelihood ratio (important) |
- |
- |
No GRADE (no evidence was found) |
No evidence was found regarding the positive likelihood ratio of CSF p-tau/Aß ratio to diagnose progression to ADD in patiënts with MCI. |
|
Negative likelihood ratio (important)
|
- |
- |
No GRADE (no evidence was found) |
No evidence was found regarding the negative likelihood ratio of CSF p-tau/Aß ratio to diagnose progression to ADD in patiënts with MCI. |
|
Positive predictive value (important) |
- |
- |
No GRADE (no evidence was found) |
No evidence was found regarding the positive predictive value of CSF p-tau/Aß ratio to diagnose progression to ADD in patiënts with MCI. |
|
Negative predictive value (important)
|
- |
- |
No GRADE (no evidence was found) |
No evidence was found regarding the negative predictive value of CSF p-tau/Aß ratio to diagnose progression to ADD in patiënts with MCI. |
|
Neurofilament light |
||||
|
Specificity (critical) |
- |
- |
No GRADE (no evidence was found) |
No evidence was found regarding accuracy measures (specificity, sensitivity, positive likelidhood ratio, negative likelidhood ratio, positive predictive value, negative predictive value) of CSF neurofilament light to diagnose progression to ADD in patiënts with MCI. |
|
Sensitivity (important) |
- |
- |
||
|
Positive predictive value (important) |
- |
- |
||
|
Negative predictive value (important) |
- |
- |
||
|
Positive likelihood ratio (important) |
- |
- |
||
|
Negative likelihood ratio (important) |
- |
- |
||
* The studies used various thresholds for calculating specificity and sensitivity, therefore summary estimates were not computed but derived from the HSROC model at the median value of specificity computed from the included studies. CI = confidence interval.
1. Risk of bias: very serious. Due to lack of reporting on enrolment or lacking consecutive or random enrolment of patiënts; no pre-set cut-off specified for the index test and not all patiënts were accounted for in the analysis or the time interval between the index test and reference standard was not appropriate (duration of follow-up was shorter than one year).
2. Inconsistency: serious. Due to sparse and inconsistent data with conflicting results.
3. Imprecision: very serious. Due to the optimal information size was not achieved.
Summary of Findings table index-3: Positron Emitted Tomography (PET) markers for predicting progression from MCI to Alzheimer dementia
Population: Patiënts with Mild Cognitive Impairment (MCI)
Intervention: Positron Emitted Tomography (PET) markers
Comparator: No marker
Reference: Pathological anatomical (PA) diagnosis or the clinical consensus diagnosis of dementia or diagnostic criteria based on DSM IV or NINCDS-ADRDA with an average minimum follow-up duration of 2 years.
|
Outcome
|
Study results and measurements |
Effect estimates* |
Certainty of the Evidence (Quality of evidence) |
Conclusions |
|
11C-PIB amyloid PET |
||||
|
Specificity (critical)
|
The systematic review and meta-analysis of Zhang (2014) included nine studies with 274 participants to assess the accuracy of 11C-PIB-PET to detect conversion from MCI to ADD compared. Of the 274 participants, 112 were diagnosed with Alzheimer’s dementia.
|
The sensitivities were between 0,83 and 1,00 while the specificities were between 0,46 and 0,88. No summary measures were calculated due to variation in thresholds and measures. To illustrate potential strengths and weaknesses of 11C-PIB-PET, sensitivity of 0,96 (95% confidence interval (CI) 0,87 to 0,99) was estimated from an HSROC curve at the included study median specificity of 0,58. This equated to a positive likelihood ratio of 2,29 (95% CI 2,17 to 2,41), and a negative likelihood ratio of 0,07 (95% CI 0,02 to 0,24) |
Very low Due to very serious risk of bias, due to serious inconsistency1,2 |
The evidence is very uncertain about whether 11C-PIB-PET is accurate in diagnosing progression to ADD in patiënts with MCI.
(Zhang, 2014) |
|
Sensitivity (important) |
Very low Due to very serious risk of bias, due to serious inconsistency1,2 |
|||
|
Positive likelihood ratio (important) |
Very low Due to very serious risk of bias, due to serious inconsistency1,2 |
|||
|
Negative likelihood ratio (important)
|
Very low Due to very serious risk of bias, due to serious inconsistency1,2 |
|||
|
Positive predictive value (important) |
-
|
- |
No GRADE (no evidence was found) |
No evidence was found regarding the positive predictive value of 11C-PIB-PET to diagnose progression to ADD in patiënts with MCI. |
|
Negative predictive value (important)
|
-
|
- |
No GRADE (no evidence was found) |
No evidence was found regarding the negative predictive value of 11C-PIB-PET to diagnose progression to ADD in patiënts with MCI. |
|
18F-florbetapir amyloid PET |
||||
|
Specificity (critical)
|
The systematic review of Cotta Ramusino (2024) included 8 studies with 1806 participants to assess the accuracy of 8F-florbetapir PET to detect conversion from MCI to ADD compared to other outcomes.
The systematic review and meta-analysis of Martinez (2017) included two studies with 448 participants to assess the accuracy of 18F-florbetapir PET to detect conversion from MCI to ADD compared. |
The sensitivities were between 0,64 and 0,94 while the specificities were between 0,48 and 0,93. No summary measures were calculated due to variation in thresholds and measures. |
Low Due to serious risk of bias, due to serious inconsistency1,2 |
18F-florbetapir PET may be accurate in diagnosing progression to ADD in patiënts with MCI.
(Martinez, 2017; Cotta ramusino, 2024) |
|
Sensitivity (important) |
Low Due to serious risk of bias, due to serious inconsistency1,2 |
|||
|
Positive likelihood ratio (important) |
-
|
- |
No GRADE (no evidence was found) |
No evidence was found regarding the positive likelihood ratio of 18F-florbetapir PET to diagnose progression to ADD in patiënts with MCI. |
|
Negative likelihood ratio (important)
|
-
|
- |
No GRADE (no evidence was found) |
No evidence was found regarding the negative likelihood ratio of 18F-florbetapir PET to diagnose progression to ADD in patiënts with MCI. |
|
Positive predictive value (important) |
-
|
- |
No GRADE (no evidence was found) |
No evidence was found regarding the positive predictive value of 18F-florbetapir PET to diagnose progression to ADD in patiënts with MCI. |
|
Negative predictive value (important)
|
-
|
- |
No GRADE (no evidence was found) |
No evidence was found regarding the negative predictive value of 18F-florbetapir PET to diagnose progression to ADD in patiënts with MCI. |
|
18F-FDG PET |
||||
|
Specificity (critical)
|
The systematic review and meta-analysis of Smailagic (2015) included 14 studies with 421 participants to assess the accuracy of 18F-FDG PET to detect conversion from MCI to ADD compared to other outcomes.
The systematic review of Cotta Ramusino (2024) included 25 studies with 6803 participants to assess the accuracy of 18F-FDG PET to detect conversion from MCI to ADD compared to other outcomes.
|
The sensitivities were between 0,25 and 1,00 while the specificities were between 0,15 and 1,00. No summary measures were calculated due to variation in thresholds and measures.
To illustrate potential strengths and weaknesses of 18F-FDG PET, Smailagic (2015) reported a sensitivity of 0,76 (95% CI 0,54 to 0,90) as estimated from an HSROC curve at the included study median specificity of 0,82. This equated to a positive likelihood ratio of 4,03 (95% CI 2,97 to 5,47), and a negative likelihood ratio of 0,34 (95% CI 0,15 to 0,75) |
Very low Due to very serious risk of bias, due to serious inconsistency1,2 |
18F-FDG uptake by brain tissue as measured by PET may be accurate in diagnosing progression to ADD in patiënts with MCI.
(Smailagic, 2015; Cotta ramusino, 2024)
|
|
Sensitivity (important) |
Very low Due to very serious risk of bias, due to serious inconsistency1,2 |
|||
|
Positive likelihood ratio (important) |
Very low Due to very serious risk of bias, due to serious inconsistency1,2 |
|||
|
Negative likelihood ratio (important)
|
Very low Due to very serious risk of bias, due to serious inconsistency1,2 |
|||
|
Positive predictive value (important) |
-
|
- |
No GRADE (no evidence was found) |
No evidence was found regarding the positive predictive value of 18F-FDG PETmeasured by MRI to diagnose progression to ADD in patiënts with MCI. |
|
Negative predictive value (important)
|
-
|
- |
No GRADE (no evidence was found) |
No evidence was found regarding the negative predictive value of 18F-FDG PETmeasured by MRI to diagnose progression to ADD in patiënts with MCI. |
* Summary estimates of sensitivity and specificity were not computed because the studies used various thresholds for calculating specificity and sensitivity.
1. Risk of bias: very serious. Due to lack of blinding or lack of reporting on the index test (for Zhang 2014 and Smailagic 2015 also unclear pre-specified definition for a ‘positive’ result) or reference test as well as the variation of methods used.
2. Inconsistency: serious. Due to sparse and inconsistent data with conflicting results.
Samenvatting literatuur
Description of studies
A total of seven studies were included in the analysis of the literature. One study for index-1 MRI (Lombardi, 2020), two studies for index-2 CSF markers (Ritchie, 2014; Ritchie, 2017) and four studies for index-3 PET (Cotta Ramusino, 2024; Martinez, 2017; Smailagic, 2015 and Zhang, 2014). Important study characteristics and results are summarized in table 2. The assessment of the risk of bias is summarized in the risk of bias tables (under the tab ‘Evidence tabellen’).
Index 1 Magnetic Resonance Imaging (MRI)
Lombardi (2020) performed a systematic review and meta-analysis to determine the diagnostic accuracy of MRI for detecting mild cognitive impairment (MCI) patiënts who convert to Alzheimer's disease dementia (ADD) over time. A systematic literature search was performed up until 29 January 2019. Studies were included if they had 1) prospective cohorts with a clinical follow-up diagnosis of Alzheimer’s disease dementia as a reference standard (delayed verification), 2) baseline MRI documented at or around the time the MCI diagnosis was made, 3) sufficient data to construct two by two tables expressing MRI results by disease status and 4) used either quantitative volumetric measurements or qualitative visual assessment of MRI to detect atrophy in specific brain regions or the whole brain. Case series, case-control and retrospective studies were excluded. 33 studies with 3935 participants evaluated the use of MRI to detect conversion to ADD. Of the 3935 participants, 1341 (34%) developed Alzheimer’s dementia. In the absence of specified thresholds, the authors used a hierarchical summary ROC curve (HSROC) model to estimate pooled accuracy measures as well as to investigate relative diagnostic odds ratios in subgroup analyses (assuming parallel ROC curves in logits). Studies yielded heterogeneous estimates and no clear threshold effects were apparent both graphically and statistically in analyses with more data. The metadas user-written command in Statistical Analysis System (SAS) (version 9.4. SAS Institute Inc., Cary, NC, USA) statistical package was used for the analyses.
Index 2 Cerebrospinal fluid (CSF)
Ritchie (2014) performed a systematic review and meta-analysis to determine the diagnostic accuracy of plasma and CSF Aß levels for detecting mild cognitive impairment (MCI) patiënts who would convert to Alzheimer's disease dementia (ADD) or other forms of dementia over time. A systematic literature search was performed up until 3 December 2012. Studies were included if they had 1) prospectively well-defined cohorts, 2) any accepted definition of MCI but no dementia, 3) baseline CSF or plasma Aß levels, or both, documented at or around the time the MCI diagnosis was made, 4) reference standard for Alzheimer’s dementia diagnosis based on the National Institute for National Institute of Neurological and Communicative Diseases and Stroke/ Alzheimer's Disease and Related Disorders Association (NINCDS- ADRDA) or the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition (DSM-IV) criteria, 5) sufficient data to construct two by two tables expressing biomarker results by disease status. Studies were excluded if they included patiënts with possible other causes for dementia: psychiatric, neurological, metabolic, immunological, hormonal or cerebrovascular disorders, genetic cause or early onset ADD (if studies included patiënts below the age of 50 years, they were excluded). Although Ritchie 2014 was not limited to studies reporting amyloidß42, no studies were identified that reported on diagnostic accuracy for ADD for CSF amyloidß40 or CSF amyloidß42/amyloidß40 ratio. Fourteen studies with 1349 participants evaluated the use of CSF amyloidß42 to detect conversion to ADD. Of the 1349 participants, 436 developed Alzheimer’s dementia. Positive cases were determined based on the amyloidß42 thresholds employed in the respective primary studies. Because of variation in assay thresholds, the authors did not estimate summary sensitivity and specificity. Instead, the authors derived estimates of sensitivity at fixed values of specificity from the model fitted to produce a summary receiver operating characteristic (ROC) curve. The authors used a hierarchical summary receiver operating characteristic (HSROC) model that accounted for between study variability through the inclusion of random effects and derived sensitivities with 95% confidence intervals (CI) at median, lower and upper quartile values of the specificities from the included studies.
Ritchie (2017) performed a systematic review and meta-analysis to determine the diagnostic accuracy of 1) CSF t-tau, 2) CSF p-tau, 3) the CSF t-tau/Aβ ratio and 4) the CSF p-tau/Aβ ratio index tests for detecting MCI patiënts who would convert to ADD or other forms of dementia over time. A systematic literature search was performed in January 2013. Studies were included if they had 1) prospectively well-defined cohorts, 2) any accepted definition of MCI but no dementia, 3) CSF t-tau or p-tau and CSF tau (t-tau or p-tau)/Aβ ratio documented at or around the time the MCI diagnosis was made, 4) reference standard for Alzheimer’s dementia diagnosis based on the NINCDS- ADRDA or DSM-IV criteria, 5) sufficient data to construct two by two tables expressing biomarker results by disease status. Fifteen studies were included with 1282 participants with MCI at baseline. 1172 participants had analysable data, 430 participants developed Alzheimer’s dementia and 130 participants other forms of dementia. Follow-up ranged from less than one year to over four years, but in the majority of studies was in the range of one to three years. Diagnostic accuracy was evaluated for CSF t-tau in seven studies (291 cases and 418 non-cases), CSF p-tau in six studies (164 cases and 328 non-cases), CSF p-tau/Aβ ratio in five studies (140 cases and 293 non-cases) and CSF t-tau/Aβ ratio in only two studies. Because of variation in assay thresholds, the authors did not estimate summary sensitivity and specificity. Instead, the authors derived estimates of sensitivity at fixed values of specificity from the model fitted to produce a summary receiver operating characteristic (ROC) curve. The authors used a hierarchical summary receiver operating characteristic (HSROC) model that accounted for between study variability through the inclusion of random effects and derived sensitivities with 95% confidence intervals (CI) at median, lower and upper quartile values of the specificities from the included studies.
Index 3 Positron Emitted Tomography (PET)
Cotta Ramusino (2024) performed a systematic review to determine the diagnostic accuracy of molecular imaging markers for detecting mild cognitive impairment (MCI) patiënts who convert to Alzheimer's disease dementia (ADD) over time. The intention was to update existing reviews up until 2017. A systematic literature search was performed from 1 January 2017 up until 28 February 2022. Studies were included if they had 1) prospective cohorts with a clinical follow-up diagnosis of Alzheimer’s disease dementia as a reference standard (delayed verification), 2) sample size of at least 50 patiënts with MCI, 3) follow-up of at least 3 years, 4) reported on critical outcome measures, e.g., sensitivity, specificity, accuracy, area under the receiver, or operating characteristic curve (ROC or AUC) and 5) used molecular brain imaging techniques (amyloid-, tau-, [18F] FDG-PETs, DaT-SPECT, and cardiac [123I]-MIBG scintigraphy). No studies on tau-PET were eligible for inclusion. Eight studies with 1806 participants evaluated the use of 18F PET with florbetapir (n=7) or flutametamol (n=1) to detect conversion to ADD. Of the 1806 participants, 549 (30%) developed Alzheimer’s dementia. 25 studies with 6803 participants evaluated the use of 18F-FDG PET to detect conversion to ADD. Of the 6803 participants, 2572 (38%) developed Alzheimer’s dementia.
Martinez (2017) performed a systematic review and meta-analysis to determine the diagnostic accuracy of 18F PET with florbetapir for detecting mild cognitive impairment (MCI) patiënts who convert to Alzheimer's disease dementia (ADD) over time. A systematic literature search was performed up until May 2017. Studies were included if they had 1) prospective cohorts with a clinical follow-up diagnosis of Alzheimer’s disease dementia as a reference standard (delayed verification), 2) baseline PET documented at or around the time the PET diagnosis was made, 3) sufficient data to construct two by two tables expressing PET results by disease status and 4) used the 18F-florbetapir PET scan. A case control study with a delayed verification design was included, this occurred in the context of a cohort study so is a diagnostic nested case-control study (Doraiswamy, 2014). Two studies with 448 participants evaluated the use of 18F-florbetapir PET to detect conversion to ADD (Doraiswamy 2014, Schreiber 2015). Of the 448 participants, 69 (15,4%) developed Alzheimer’s dementia.
Smailagic (2015) performed a systematic review and meta-analysis to determine the diagnostic accuracy of 18F-FDG uptake by brain tissue as measured by PET for detecting mild cognitive impairment (MCI) patiënts who convert to Alzheimer's disease dementia (ADD) over time. A systematic literature search was performed up until January 2013. Studies were included if they had 1) prospective cohorts with a clinical follow-up diagnosis of Alzheimer’s disease dementia as a reference standard (delayed verification), 2) baseline PET documented at or around the time the PET diagnosis was made, 3) sufficient data to construct two by two tables expressing PET results by disease status and 4) used PET to detect 18F-FDFG uptake in the brain. Case control studies with a delayed verification design were included, these occurred in the context of a cohort study so are diagnostic nested case-control studies. 14 studies with 421 participants evaluated the use of 18F-FDG PET to detect conversion to ADD. Of the 421 participants, 150 (36%) developed Alzheimer’s dementia. Different brain regions were examined, all studies included the temporo-parietal lobes, 12 also included the posterior cingulate and part of the frontal lobes. Because of between study variation in thresholds and measures of 18F-FDG uptake, the authors did not estimate summary sensitivity and specificity. Instead, the authors derived estimates of sensitivity at fixed values of specificity from the model fitted to produce a summary receiver operating characteristic (ROC) curve. The authors used a hierarchical summary receiver operating characteristic (HSROC) model that accounted for between study variability through the inclusion of random effects and derived sensitivities with 95% confidence intervals (CI) at median values of the specificities from the included studies.
Zhang (2014) performed a systematic review and meta-analysis to determine the diagnostic accuracy of PET with the 11C-labelled Pittsburgh Compound-B (11C-PIB) ligand for detecting mild cognitive impairment (MCI) patiënts who convert to Alzheimer's disease dementia (ADD) over time. A systematic literature search was performed up until 12 January 2013. Studies were included if they had 1) prospective cohorts with a clinical follow-up diagnosis of Alzheimer’s disease dementia as a reference standard (delayed verification with a minimum of 1 year between time of MCI and time of dementia diagnosis), 2) baseline PET documented at or around the time the MCI diagnosis was made, 3) sufficient data to construct two by two tables expressing PET results by disease status and 4) used PET with the 11C-labelled Pittsburgh Compound-B (11C-PIB) ligand. Case control studies with a delayed verification design were included, these occurred in the context of a cohort study so are diagnostic nested case-control studies. Nine studies with 274 participants evaluated the use of PET to detect conversion to ADD. Of the 274 participants, 112 (41%) developed Alzheimer’s dementia. Because of between study variation in thresholds and measures of 11C-PIB amyloid retention, the authors did not estimate summary sensitivity and specificity. Instead, the authors derived estimates of sensitivity at fixed values of specificity from the model fitted to produce a summary receiver operating characteristic (ROC) curve. The authors used a hierarchical summary receiver operating characteristic (HSROC) model that accounted for between study variability through the inclusion of random effects and derived sensitivities with 95% confidence intervals (CI) at median, lower and upper quartile values of the specificities from the included studies.
Table 2. Characteristics of included systematic reviews
* For further details, see risk of bias table in the appendix
Results
Index 1 Magnetic Resonance Imaging (MRI)
1.1 Total hippocampal volume
1.1.1 Specificity and sensitivity
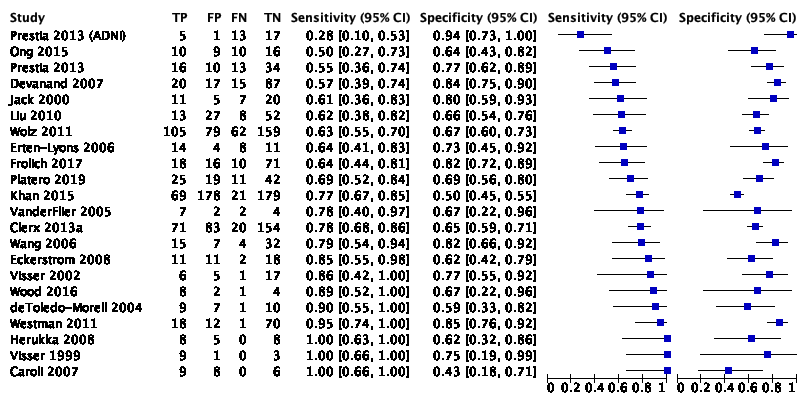
The systematic review and meta-analysis of Lombardi (2020) included 22 studies with 2209 participants to assess the accuracy of total hippocampus volume measured by MRI to detect conversion from MCI to ADD compared to other outcomes. Of the 2209 participants, 687 were diagnosed with ADD. Individual study estimates of sensitivity were between 28% and 100% while the specificities were between 43% and 94%. Results are shown in Figure 1.
1.1.2 Positive predictive value
Lombardi (2020) did not report on the outcome positive predictive value.
1.1.3 Negative predictive value
Lombardi (2020) did not report on the outcome negative predictive value.
1.1.4 Positive likelihood ratio
Lombardi (2020) reported a positive likelihood ratio of 2.53 (95% CI 2.09 to 3.06).
1.1.5 Negative likelihood ratio
Lombardi (2020) reported a negative likelihood ratio of 0.38 (95% CI 0.29 to 0.50).
Figure 1. Forest plot of total hippocampal volume measured by structural MRI for early diagnosis of dementia due to Alzheimer's disease in people with mild cognitive impairment (Lombardi 2020)
Plot shows study-specific estimates of sensitivity and specificity (squares) with 95% confidence interval (black line) and study. Studies are ordered according to the estimates of sensitivity. TP: true positive; FP: false positive; FN: false negative; TN: true negative
1.2 Atrophy medial temporal lobe
1.2.1 Specificity and sensitivity
The systematic review and meta-analysis of Lombardi (2020) included seven studies with 1077 participants to assess the accuracy of medial temporal lobe volume measured by MRI to detect conversion from MCI to ADD compared to other outcomes. Of the 1077 participants, 330 were diagnosed with ADD. Individual study estimates of sensitivity were between 40% and 86% while the specificities were between 44% and 85%. Results are shown in Figure 2.
1.2.2 Positive predictive value
Lombardi (2020) did not report on the outcome positive predictive value.
1.2.3 Negative predictive value
Lombardi (2020) did not report on the outcome negative predictive value.
1.2.4 Positive likelihood ratio
Lombardi (2020) reported a positive likelihood ratio of 1.81 (95% CI 1.41 to 2.32).
1.2.5 Negative likelihood ratio
Lombardi (2020) reported a negative likelihood ratio of 0.56 (95% CI 0.46 to 0.67).
Figure 2. Forest plot of medial temporal lobe volume measured by structural MRI for early diagnosis of dementia due to Alzheimer's disease in people with mild cognitive impairment (Lombardi 2020)
Plot shows study-specific estimates of sensitivity and specificity (squares) with 95% confidence interval (black line) and study. TP: true positive; FP: false positive; FN: false negative; TN: true negative
1.3 Lateral ventricles volume
1.3.1 Specificity and sensitivity
The systematic review and meta-analysis of Lombardi (2020) included five studies with 1077 participants to assess the accuracy of MRI t-tau to detect conversion from MCI to ADD compared to other outcomes. Of the 1077 participants, 371 were diagnosed with ADD. Individual study estimates of sensitivity were between 51% and 75% while the specificities were between 47% and 73%. Results are shown in Figure 3.
1.3.2 Positive predictive value
Lombardi (2020) did not report on the outcome positive predictive value.
1.3.3 Negative predictive value
Lombardi (2020) did not report on the outcome negative predictive value.
1.3.4 Positive likelihood ratio
Lombardi (2020) reported a positive likelihood ratio of 1.61 (95% CI 1.39 to 1.87).
1.3.5 Negative likelihood ratio
Lombardi (2020) reported a negative likelihood ratio of 0.66 (95% CI 0.57 to 0.78).
Figure 3. Forest plot of lateral ventricles volume measured by structural MRI for early diagnosis of dementia due to Alzheimer's disease in people with mild cognitive impairment (Lombardi 2020)
Plot shows study-specific estimates of sensitivity and specificity (squares) with 95% confidence interval (black line) and study. TP: true positive; FP: false positive; FN: false negative; TN: true negative
1.4 Total entrorhinal cortex volume
1.4.1 Specificity and sensitivity
The systematic review and meta-analysis of Lombardi (2020) included four studies with 529 participants to assess the accuracy of total entorhinal cortex volume measured by MRI to detect conversion from MCI to ADD compared to other outcomes. Of the 529 participants, 229 were diagnosed with ADD. Individual study estimates of sensitivity were between 50% and 88% while the specificities were between 60% and 100%. Results are shown in Figure 4.
1.4.2 Positive predictive value
Lombardi (2020) did not report on the outcome positive predictive value.
1.4.3 Negative predictive value
Lombardi (2020) did not report on the outcome negative predictive value.
1.4.4 Positive likelihood ratio
Lombardi (2020) did not report a positive likelihood ratio. Meta-analyses were not conducted due to sparse and heterogeneous data
1.4.5 Negative likelihood ratio
Lombardi (2020) did not report a negative likelihood ratio. Meta-analyses were not conducted due to sparse and heterogeneous data.
Figure 4. Forest plot of total entrorhinal cortex volume measured by structural MRI for early diagnosis of dementia due to Alzheimer's disease in people with mild cognitive impairment (Lombardi 2020)
Plot shows study-specific estimates of sensitivity and specificity (squares) with 95% confidence interval (black line) and study. TP: true positive; FP: false positive; FN: false negative; TN: true negative
1.5 Whole brain volume
1.5.1 Specificity and sensitivity
The systematic review and meta-analysis of Lombardi (2020) included four studies with 424 participants to assess the accuracy of whole brain volume measured by MRI to detect conversion from MCI to ADD compared to other outcomes. Of the 424 participants, 220 were diagnosed with ADD. Individual study estimates of sensitivity were between 33% and 92% while the specificities were between 41% and 100%. Results are shown in Figure 5.
1.5.2 Positive predictive value
Lombardi (2020) did not report on the outcome positive predictive value.
1.5.3 Negative predictive value
Lombardi (2020) did not report on the outcome negative predictive value.
1.5.4 Positive likelihood ratio
Lombardi (2020) did not report a positive likelihood ratio. Meta-analyses were not conducted due to sparse and heterogeneous data
1.5.5 Negative likelihood ratio
Lombardi (2020) did not report a negative likelihood ratio. Meta-analyses were not conducted due to sparse and heterogeneous data.
Figure 5. Forest plot of whole brain volume measured by structural MRI for early diagnosis of dementia due to Alzheimer's disease in people with mild cognitive impairment (Lombardi 2020)
Plot shows study-specific estimates of sensitivity and specificity (squares) with 95% confidence interval (black line) and study. TP: true positive; FP: false positive; FN: false negative; TN: true negative
Index 2 Cerebrospinal fluid (CSF)
2.1 Amyloidβ42
2.1.1 Specificity and sensitivity
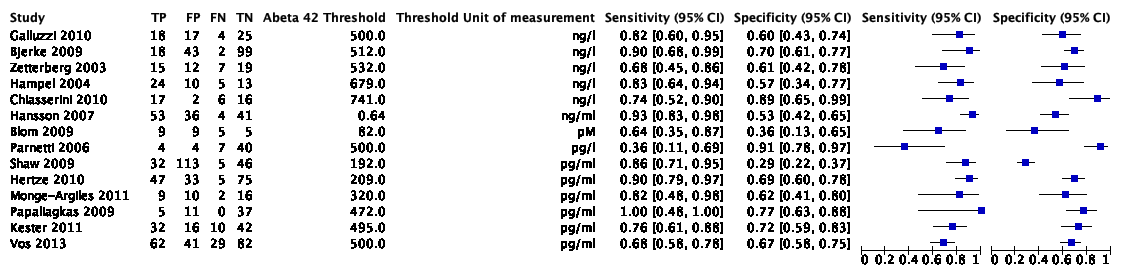
The systematic review and meta-analysis of Ritchie (2014) included fourteen studies with 1349 participants to assess the accuracy of CSF amyloidß42 to detect conversion from MCI to ADD compared to other outcomes. Of the 1349 participants, 436 were diagnosed with ADD. Individual study estimates of sensitivity were between 36% and 100% while the specificities were between 29% and 91%. Results are shown in Figure 6.
2.1.2 Positive predictive value
Ritchie (2014) did not report on the outcome positive predictive value.
2.1.3 Negative predictive value
Ritchie (2014) did not report on the outcome negative predictive value.
Figure 6. Forest plot of CSF amyloidß42 for early diagnosis of dementia due to Alzheimer's disease in people with mild cognitive impairment (Ritchie 2014)
Plot shows study-specific estimates of sensitivity and specificity (squares) with 95% confidence interval (black line) and study. TP: true positive; FP: false positive; FN: false negative; TN: true negative
2.2. Total-tau (CSF t-tau)
2.2.1 Specificity and sensitivity
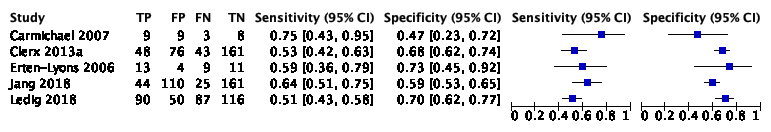
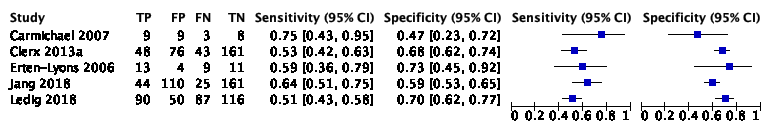
The systematic review and meta-analysis of Ritchie (2017) included seven studies with 709 participants to assess the accuracy of CSF t-tau to detect conversion from MCI to ADD compared to other outcomes. Of the 709 participants, 291 were diagnosed with ADD. Individual study estimates of sensitivity were between 51% and 95% while the specificities were between 48% and 88%. Results are shown in Figure 7.
2.2.2 Positive predictive value
Ritchie (2017) did not report on the outcome positive predictive value.
2.2.3 Negative predictive value
Ritchie (2017) did not report on the outcome negative predictive value.
Figure 7. Forest plot of CSF t-tau for early diagnosis of dementia due to Alzheimer's disease in people with mild cognitive impairment (Ritchie 2017)
Plot shows study-specific estimates of sensitivity and specificity (squares) with 95% confidence interval (black line) and study. TP: true positive; FP: false positive; FN: false negative; TN: true negative; CI = confidence interval
2.3. Phosphorylated tau (CSF p-tau)
2.3.1 Specificity and sensitivity
The systematic review and meta-analysis of Ritchie (2017) included six studies with 492 participants to assess the accuracy of CSF p-tau to detect conversion from MCI to ADD compared to other outcomes. Of the 492 participants, 164 were diagnosed with ADD. Individual study estimates of sensitivity were between 40% and 100% while the specificities were between 22% and 86%. Results are shown in Figure 8.
2.3.2 Positive predictive value
Ritchie (2017) did not report on the outcome positive predictive value.
2.3.3 Negative predictive value
Ritchie (2017) did not report on the outcome negative predictive value.
Figure 8. Forest plot of CSF p-tau for early diagnosis of dementia due to Alzheimer's disease in people with mild cognitive impairment (Ritchie, 2017)
Plot shows study-specific estimates of sensitivity and specificity (squares) with 95% confidence interval (black line) and study. TP: true positive; FP: false positive; FN: false negative; TN: true negative; CI = confidence interval
2.4. CSF t-tau/Aß ratio
The systematic review and meta-analysis of Ritchie (2017) included two studies with 251 participants to assess the accuracy of CSF t-tau/Aß to detect conversion from MCI to ADD compared to other outcomes. Of the 251 participants, 102 were diagnosed with ADD. One study (n=37) reported a sensitivity of 91% and a specificity of 50% based on TP = 10, FP = 13, FN = 1, TN = 13. The other study (n=214) reported a sensitivity of 96% and a specificity of 51% based on TP = 87, FP = 60, FN = 4, TN = 63.
2.5. CSF p-tau/Aß ratio
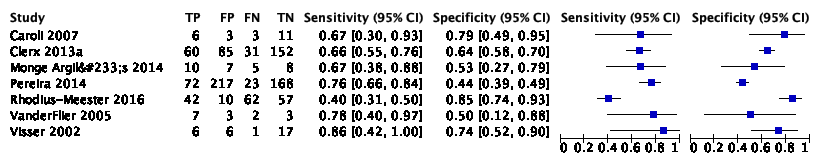
2.5.1 Specificity and sensitivity
The systematic review and meta-analysis of Ritchie (2017) included five studies with 433 participants to assess the accuracy of CSF p-tau/Aß to detect conversion from MCI to ADD compared to other outcomes. Of the 433 participants, 140 were diagnosed with ADD. Individual study estimates of sensitivity were between 80% and 96% while the specificities were between 33% and 95%. Results are shown in Figure 9.
2.5.2 Positive predictive value
Ritchie (2017) did not report on the outcome positive predictive value.
2.5.3 Negative predictive value
Ritchie (2017) did not report on the outcome negative predictive value.
Figure 9. Forest plot of CSF p-tau/ Aß ratio for early diagnosis of dementia due to Alzheimer's disease in people with mild cognitive impairment (Ritchie 2017)
Plot shows study-specific estimates of sensitivity and specificity (squares) with 95% confidence interval (black line) and study. TP: true positive; FP: false positive; FN: false negative; TN: true negative; CI = confidence interval
2.6. Neurofilament light
None of the included studies (Ritchie 2014, Ritchie 2017) reported on the diagnostic accuracy (specificity, sensitivity, positive predictive value or negative predictive value) of neurofilament light.
Index 3 Positron Emitted Tomography (PET)
3.1 Amyloid 11C-PIB-PET
3.1.1 Specificity and sensitivity
The systematic review and meta-analysis of Zhang (2014) included nine studies with 274 participants to assess the accuracy of 11C-PIB-PET to detect conversion from MCI to ADD compared. Of the 274 participants, 112 were diagnosed with Alzheimer’s dementia. Individual study estimates of sensitivity were between 83% and 100% while the specificities were between 46% and 88%. Results are shown in Figure 10.
3.1.2 Positive predictive value
Zhang (2014) did not report on the outcome positive predictive value.
3.1.3 Negative predictive value
Zhang (2014) did not report on the outcome negative predictive value.
3.1.4 Positive likelihood ratio
Zhang (2014) did not report on the outcome positive likelihood ratio.
3.1.5 Negative likelihood ratio
Zhang (2014) did not report on the outcome negative likelihood ratio.
Figure 10. Forest plot of 11C-PIB-PET for early diagnosis of dementia due to Alzheimer's disease in people with mild cognitive impairment (Zhang 2014)
Plot shows study-specific estimates of sensitivity and specificity (squares) with 95% confidence interval (black line) and study. TP: true positive; FP: false positive; FN: false negative; TN: true negative; CI = confidence interval
3.2 Amyloid 18F-florbetapir
3.2.1 Specificity and sensitivity
The systematic review and meta-analysis of Martinez (2017) included two studies with 448 participants to assess the accuracy of 18F-florbetapir PET to detect conversion from MCI to ADD compared. Of the 448 participants, 69 were diagnosed with Alzheimer’s dementia. One study (n = 401) examined follow-up between one to less than two years and reported a sensitivity of 89% (95% CI 78 to 95) and a specificity of 58% (95% CI 53 to 64) by visual assessment, and a sensitivity of 87% (95% CI 76 to 94) and a specificity of 51% (95% CI 45 to 56) by quantitative assessment by the standardised uptake value ratio (SUVR). The other study (n=47) examined follow-up between two years to less than four years and reported a sensitivity of 67% (95% CI 30 to 93) and a specificity of 71% (95% CI 54 to 85) by visual assessment.
The systematic review of Cotta Ramusino (2024) included 8 studies with 1806 participants to assess the accuracy of 8F-florbetapir PET to detect conversion from MCI to ADD compared to other outcomes. Of the 1806 participants, 549 (30%) developed Alzheimer’s dementia. Individual study estimates of sensitivity were between 0,64 and 0,94 while the specificities were between 0,48 and 0,93.
Results are shown in table 4.
3.2.2 Positive predictive value
Martinez (2017) and Cotta Ramusino (2024) did not report on the outcome positive predictive value.
3.2.3 Negative predictive value
Martinez (2017) and Cotta Ramusino (2024) did not report on the outcome negative predictive value.
3.2.4 Positive likelihood ratio
Martinez (2017) and Cotta Ramusino (2024) did not report on the outcome positive likelihood ratio.
3.2.5 Negative likelihood ratio
Martinez (2017) and Cotta Ramusino (2024) did not report on the outcome negative likelihood ratio.
Table 4. Sensitivity and specificity of 18F-florbetapir PET for early diagnosis of dementia due to Alzheimer's disease in people with mild cognitive impairment (Martinez 2017, Cotta Ramusino 2024)
|
Review |
Study |
N |
Sensitivity (95% confidence interval)* |
Specificity (95% confidence interval)* |
|
Cotta Ramusino, 2024 |
Wolk, 2018 |
224 |
0,64 (0,53-0,75) |
0,69 (0,60 - 0,76) |
|
|
Choi, 2018 |
171 |
0,86 |
0,75 |
|
|
Bouallègue, 2018 |
289 |
0,76 |
0,71 |
|
|
Blazhenets, 2020 |
319 |
0,94 (0,80-0,99) |
0,48 (0,39-0,57) |
|
|
Bouallegue, 2017 |
209 |
pontine SUVr 0,83, cerebellar SUVr 0,84, composite SUVr 0.88 |
pontine SUVr 0,82, cerebellar SUVr 0,81, composite SUVr 0,81 |
|
|
Beyer, 2020 |
396 |
visual read 0,79; CBL reference 0,79; BST reference 0,79; WM reference: 0,79 |
visual read 0,74; CBL reference 0,72; BST reference: 0,74; WM reference: 0,77 |
|
|
Gupta, 2020 |
61 |
ROI: 1,00, VOI: 0,90 |
ROI: 0,86, VOI: 0,93 |
|
|
Popescu, 2020 |
206 |
0,67 |
0,67 |
|
Martinez, 2017 |
Schreiber, 2015 |
401 |
Visual: 0,89 (0,78-0,95) Quantitative: 0,87 (0,76-0,94) |
Visual: 0,58 (0,53-0,64) Quantitative: 0,51 (0,45-0,56) |
|
|
Doraiswamy, 2014 |
47 |
0,67 (0,30-0,93) |
0,71 (0,54-0,85) |
* Some studies used multiple methods to assess the PET scan and thus establish the diagnosis. When applicable this is indicated by giving the name of the method used
3.3 Glucose metabolism ([18F]FDG-PET)
3.3.1 Specificity and sensitivity
The systematic review and meta-analysis of Smailagic (2015) included 14 studies with 421 participants to assess the accuracy of [18F]FDG-PET to detect conversion from MCI to ADD compared to other outcomes. Of the 421 participants, 150 were diagnosed with ADD. Individual study estimates of sensitivity were between 0,25 and 1,00 while the specificities were between 0,15 and 1,00.
The systematic review of Cotta Ramusino (2024) included 25 studies with 6803 participants to assess the accuracy of 18F-FDG PET to detect conversion from MCI to ADD compared to other outcomes. Of the 6803 participants, 2572 (38%) developed Alzheimer’s dementia. Individual study estimates of sensitivity were between 0,25 and 1,00 while the specificities were between 0,15 and 1,00.
Results are shown in table 5.
3.3.2 Positive predictive value
Neither Smailagic (2015) or Cotta Ramusino (2024) reported on the outcome positive predictive value.
3.3.3 Negative predictive value
Neither Smailagic (2015) or Cotta Ramusino (2024) reported on the outcome negative predictive value.
Table 5. Sensitivity and specificity of [18F]FDG-PET for early diagnosis of dementia due to Alzheimer's disease in people with mild cognitive impairment (Smailagic 2015, Cotta Ramusino 2024)
|
Review |
Study |
N |
Sensitivity (95% confidence interval)* |
Specificity (95% confidence interval)* |
|
Cotta Ramusino, 2024 |
Liu, 2017 |
234 |
0,57 |
0,82 |
|
|
Pagani, 2017 |
122 |
0,83 (0,77-0,89) |
0,85 (0,72-0,98) |
|
|
Meles, 2017 |
122 |
0,84 |
0,67 |
|
|
Pagani, 2017 |
122 |
0,87 (0,81-0,94) |
0,93 (0,83-1,00) |
|
|
Katako, 2018 |
366 |
SVM-ISDA 0,87; SVM SMO 0,63 |
SVM-ISDA 0,60; SVM SMO 0,90 |
|
|
Iaccarino, 2019 |
259 |
0,77 |
0,65 |
|
|
Teng, 2020 |
79 |
0,88 (0,72-0,97) |
0,87 (0,74-0,95) |
|
|
Huang, 2019 |
767 |
0,68 |
0,84 |
|
|
Yee, 2020 |
637 |
0,74(0,70-0,78) |
0,75 (0,72-0,78) |
|
|
Tripathi, 2019 |
87 |
0,87 (0,66-0,97) |
0,94 (0,85–0,98) |
|
|
Blum, 2019 |
220 |
0,83(0,77-0,89) |
0,88 (0,77-0,94) |
|
|
Lu, 2018 |
521 |
0,81 (0,73-0,88) |
0,83 (0,79-0,86) |
|
|
Choi, 2018 |
171 |
0,71 |
0,79 |
|
|
Bouallègue, 2018 |
289 |
0,71 |
0,66 |
|
|
Santangelo, 2020 |
77 |
0,43(0,26-0,61) |
0,93(0,81-0,99) |
|
|
Yoshitaka, 2018 |
114 |
PALZ 0,70; 3D-SSP 0,94 |
PALZ 0,90; 3D-SSP 0,53 |
|
|
Pan, 2021 |
526 |
Voxel-wise: 0,55; ROI-wise: 0,56; 2D CNN: 0,52; 3D CNN: 0,59; MiSePyNet: 0,72 |
Voxel-wise 0,84; ROI-wise: 0,84; 2D CNN: 0,85; 3D CNN: 0,88; MiSePyNet: 0,88 |
|
|
Wang, 2020 |
420 |
SVM-ROI uptake:0,82 ± 0,02 SVM-MCI pattern:0,84 ± 0,05 SVM-Connectome:0,88 ± 0,03 LR model-ROI uptake:0,81 ± 0,06 LR model-MCI pattern:0,82 ± 0,02 LR model-Connectome:0,81 ± 0,03 Random forest-ROI uptake:0,81 ± 0,04 Random forest-MCI pattern:0,85 ± 0,03 Random forest-Connectome:0,88 ± 0,03 |
SVM-ROI uptake:0,67 ± 0,03 SVM-MCI pattern:0,67 ± 0,06 SVM-Connectome:0,81 ± 0,04 LR model-ROI uptake:0,61 ± 0,05 LR model-MCI pattern:0,67 ± 0,06 LR model-Connectome:0,84 ± 0,07 Random forest-ROI uptake:0,59 ± 0,06 Random forest-MCI pattern:0,61 ± 0,09 Random forest-Connectome:0,63 ± 0,07 |
|
|
Zhou, 2021 |
355 |
AlexNet=0,73 ± 0,03 ZF-Net=0,71 ± 0,03 InceptionV3=0,69 ± 0,06 ResNet18=0,69 ± 0,04 Xception=0,71 ± 0,04 SUVR method= 0,68 ± 0,00 |
AlexNet=0,75 ± 0,04 ZF-Net=0,77 ± 0,04 InceptionV3=0,77 ± 0,07 ResNet18=0,67 ± 0,03 Xception=0,69 ± 0,05 SUVR method= 0,68 ± 2,41 |
|
|
Tondo, 2021 |
142 |
0,90 (0,79-0,96) |
0,89 (0,80-0,95) |
|
|
Gupta, 2020 |
61 |
ROI: 0,87, VOI: 0,86 |
ROI: 0,87, VOI: 0,80 |
|
Smailagic, 2015 |
Anchisi 2005 |
48 |
0,93 [0,66, 1,00] |
0,82 [0,65, 0,93] |
|
|
Arnaiz 2001 |
20 |
0,67 [0,30, 0,93] |
0,82 [0,48, 0,98] |
|
|
Berent 1999 |
20 |
0,70 [0,35, 0,93] |
0,70 [0,35, 0,93] |
|
|
Chetelat 2003 |
17 |
1,00 [0,59, 1,00] |
1,00 [0,69, 1,00] |
|
|
Clerici 2009 |
26 |
0,92 [0,64, 1,00] |
0,15 [0,02, 0,45] |
|
|
Drzezga 2005 |
30 |
0,92 [0,62, 1,00] |
0,89 [0,65, 0,99] |
|
|
Fellgiebel 2007 |
16 |
1,00 [0,40, 1,00] |
0,75 [0,43, 0,95] |
|
|
Galluzzi 2010 |
38 |
0,79 [0,49, 0,95] |
0,29 [0,13, 0,51] |
|
|
Herholz 2011 |
94 |
0,57 [0,37, 0,75] |
0,67 [0,54, 0,78] |
|
|
Mosconi 2004 |
37 |
0,38 [0,09, 0,76] |
0,97 [0,82, 1,00] |
|
|
Nobili 2008 |
33 |
0,82 [0,48, 0,98] |
0,91 [0,71, 0,99] |
|
|
Ossenkoppele 2012a |
12 |
0,75 [0,19, 0,99] |
0,88 [0,47, 1,00] |
|
|
Ossenkoppele 2012b |
12 |
0,83 [0,36, 1,00] |
1,00 [0,54, 1,00] |
|
|
Pardo 2010 |
18 |
0,25 [0,03, 0,65] |
0,60 [0,26, 0,88] |
* Some studies used multiple methods to assess the PET scan and thus establish the diagnosis. When applicable this is indicated by giving the name of the method used
Zoeken en selecteren
The ideal body of evidence would include studies that directly compare the test strategies under consideration (i.e., randomized trials) and the resulting interventions and consequences for patiënts (i.e., patiënt-important outcomes). Such studies would, by design, address all of the issues in the analytical framework and allow guideline panelists to apply the familiar GRADE approach for interventions’ (Schünemann, 2019). If this direct evidence does not exist, a search question on test accuracy outcomes is formulated. Connecting test accuracy to downstream consequences is required for decision-making.
A systematic review of the literature was performed to answer the following question(s):
- What is the value of MRI in predicting progression from MCI to dementia?
- What is the value of CSF testing in predicting progression from MCI to dementia?
- What is the value of PET in predicting progression from MCI to dementia?
Table PICRO
| Patients | Patients with mild cognitive impairment |
|
Index test-1 Index test-2
Index test-3 |
Magnetic Resonance Imaging (MRI) Cerebrospinal fluid (CSF) biomarkers (amyloidβ42, total-tau, phosporylated tau, ratio ptau/aβ, neurofilament light) Positron Emitted Tomography (PET) biomarkers (amyloid, glucose metabolism imaging) |
| Comparator test | No use of the biomarker |
| Reference test | Pathological anatomical (PA) confirmed clinical diagnosis or the clinical consensus diagnosis of dementia or diagnostic criteria based on DSM IV or NINCDS-ADRDA with an average minimum follow-up duration of 2 years |
| Outcomes | Diagnostic accuracy measures for the progression to Alzheimer dementia (sensitivity, specificity, positive predictive value, negative predictive value, positive likelihood ratio (LR+), negative likelihood ratio (LR-)) |
| Other selection criteria | Study design: Systematic reviews of studies with a longitudinal design. Timing/setting: Biomarker testing at the time of MCI diagnosis and outcome upon follow-up with the index test with an average minimum follow-up duration of 2 years |
Relevant outcome measures
The guideline panel considered specificity as critical outcome measure for decision making; and sensitivity, positive predictive value, negative predictive value as important outcome measures for decision making. From a clinical perspective high specificity is more important than high sensitivity. In other words, avoiding false positive diagnoses is more important than avoiding false negative diagnoses, due to the absence of available disease-modifying treatments (see table 1).
Table 1. Consequences of diagnostic test characteristics
|
Outcome |
Consequences |
Relevance |
|
True positives (TP) |
Monitoring for the development of dementia can be justified. Prognosis of potential cognitive decline in upcoming years. |
Important |
|
True negatives (TN) |
No further action is justified. Progression to dementia in the coming years is unlikely. |
Critical |
|
False positives (FP) |
Monitoring for development of dementia is not warranted. May lead to wrong expectations about potential cognitive decline in the upcoming years. |
Critical |
|
False negatives (FN) |
Possibly further monitoring was justified. May lead to unjustified relief of good prognosis. |
Important |
A priori, the guideline panel did not define the outcome measures listed above but used the definitions used in the studies.
The guideline panel deliberately did not define minimal clinically (patiënt) important differences. Rather, evaluated progression to a clinical dementia syndrome. From a clinical perspective, progression to dementia is most clear and unambiguous. Using a measure of cognitive decline may still result in uncertainty, because the magnitude of relevant cognitive decline is uncertain and may be different for each person, and cognitive test results tend to fluctuate over time in persons with MCI, which may lead to improvement after an initial deterioration.
Search and select (Methods)
The databases Medline (via OVID) and Embase (via Embase.com) were searched with relevant search terms (biomarkers (MRI/PET/CSF) for mild cognitive impairment) from 2010 until 20 August 2024. The detailed search strategy is listed under the tab ‘Literature search strategy’. The systematic literature search resulted in 595 hits. Studies were selected based on the following criteria:
- Systematic reviews (searched in at least two databases, detailed search strategy with search date, in- and exclusion criteria, exclusion table, risk of bias assessment and results of individual studies available).
- Full-text English language publication and
- Studies according to the PICROTS.
Because of the large body of literature and the knowledge on beforehand that several high-quality systematic reviews exist, the guideline committee decided to initially search for systematic reviews only, with the option to extend the search to individual studies if the results from the systematic reviews would be deemed insufficient.
Initially, 22 studies were selected based on title and abstract screening. After reading the full text, 15 studies were excluded (see the exclusion table under the tab ‘Evidence tabellen’) and seven studies were included.
Referenties
- Cotta Ramusino M, Massa F, Festari C, Gandolfo F, Nicolosi V, Orini S, Nobili F, Frisoni GB, Morbelli S, Garibotto V; European Inter-Societal Consensus on the Biomarker-Based Diagnosis of Dementia. Diagnostic performance of molecular imaging methods in predicting the progression from mild cognitive impairment to dementia: an updated systematic review. Eur J Nucl Med Mol Imaging. 2024 Jun;51(7):1876-1890. doi: 10.1007/s00259-024-06631-y. Epub 2024 Feb 15. PMID: 38355740.
- Groot C, Smith R, Collij LE, Mastenbroek SE, Stomrud E, Binette AP, Leuzy A, Palmqvist S, Mattsson-Carlgren N, Strandberg O, Cho H, Lyoo CH, Frisoni GB, Peretti DE, Garibotto V, La Joie R, Soleimani-Meigooni DN, Rabinovici G, Ossenkoppele R, Hansson O. Tau Positron Emission Tomography for Predicting Dementia in Individuals With Mild Cognitive Impairment. JAMA Neurol. 2024 Aug 1;81(8):845-856. doi: 10.1001/jamaneurol.2024.1612. PMID: 38857029; PMCID: PMC11165418.
- Huszár Z, Engh MA, Pavlekovics M, Sato T, Steenkamp Y, Hanseeuw B, Terebessy T, Molnár Z, Hegyi P, Csukly G. Risk of conversion to mild cognitive impairment or dementia among subjects with amyloid and tau pathology: a systematic review and meta-analysis. Alzheimers Res Ther. 2024 Apr 12;16(1):81. doi: 10.1186/s13195-024-01455-2. PMID: 38610055; PMCID: PMC11015617.
- Jansen WJ, Ossenkoppele R, Knol DL, Tijms BM, Scheltens P, Verhey FR, Visser PJ; Amyloid Biomarker Study Group; Aalten P, Aarsland D, Alcolea D, Alexander M, Almdahl IS, Arnold SE, Baldeiras I, Barthel H, van Berckel BN, Bibeau K, Blennow K, Brooks DJ, van Buchem MA, Camus V, Cavedo E, Chen K, Chetelat G, Cohen AD, Drzezga A, Engelborghs S, Fagan AM, Fladby T, Fleisher AS, van der Flier WM, Ford L, Förster S, Fortea J, Foskett N, Frederiksen KS, Freund-Levi Y, Frisoni GB, Froelich L, Gabryelewicz T, Gill KD, Gkatzima O, Gómez-Tortosa E, Gordon MF, Grimmer T, Hampel H, Hausner L, Hellwig S, Herukka SK, Hildebrandt H, Ishihara L, Ivanoiu A, Jagust WJ, Johannsen P, Kandimalla R, Kapaki E, Klimkowicz-Mrowiec A, Klunk WE, Köhler S, Koglin N, Kornhuber J, Kramberger MG, Van Laere K, Landau SM, Lee DY, de Leon M, Lisetti V, Lleó A, Madsen K, Maier W, Marcusson J, Mattsson N, de Mendonça A, Meulenbroek O, Meyer PT, Mintun MA, Mok V, Molinuevo JL, Møllergård HM, Morris JC, Mroczko B, Van der Mussele S, Na DL, Newberg A, Nordberg A, Nordlund A, Novak GP, Paraskevas GP, Parnetti L, Perera G, Peters O, Popp J, Prabhakar S, Rabinovici GD, Ramakers IH, Rami L, Resende de Oliveira C, Rinne JO, Rodrigue KM, Rodríguez-Rodríguez E, Roe CM, Rot U, Rowe CC, Rüther E, Sabri O, Sanchez-Juan P, Santana I, Sarazin M, Schröder J, Schütte C, Seo SW, Soetewey F, Soininen H, Spiru L, Struyfs H, Teunissen CE, Tsolaki M, Vandenberghe R, Verbeek MM, Villemagne VL, Vos SJ, van Waalwijk van Doorn LJ, Waldemar G, Wallin A, Wallin ÅK, Wiltfang J, Wolk DA, Zboch M, Zetterberg H. Prevalence of cerebral amyloid pathology in persons without dementia: a meta-analysis. JAMA. 2015 May 19;313(19):1924-38. doi: 10.1001/jama.2015.4668. PMID: 25988462; PMCID: PMC4486209.
- Janelidze S, Bali D, Ashton NJ, Barthélemy NR, Vanbrabant J, Stoops E, Vanmechelen E, He Y, Dolado AO, Triana-Baltzer G, Pontecorvo MJ, Zetterberg H, Kolb H, Vandijck M, Blennow K, Bateman RJ, Hansson O. Head-to-head comparison of 10 plasma phospho-tau assays in prodromal Alzheimer's disease. Brain. 2023 Apr 19;146(4):1592-1601. doi: 10.1093/brain/awac333. PMID: 36087307; PMCID: PMC10115176.
- Lombardi G, Crescioli G, Cavedo E, Lucenteforte E, Casazza G, Bellatorre AG, Lista C, Costantino G, Frisoni G, Virgili G, Filippini G. Structural magnetic resonance imaging for the early diagnosis of dementia due to Alzheimer's disease in people with mild cognitive impairment. Cochrane Database Syst Rev. 2020 Mar 2;3(3):CD009628. doi: 10.1002/14651858.CD009628.pub2. PMID: 32119112; PMCID: PMC7059964.
- Martínez G, Vernooij RW, Fuentes Padilla P, Zamora J, Bonfill Cosp X, Flicker L. 18F PET with florbetapir for the early diagnosis of Alzheimer's disease dementia and other dementias in people with mild cognitive impairment (MCI). Cochrane Database Syst Rev. 2017 Nov 22;11(11):CD012216. doi: 10.1002/14651858.CD012216.pub2. PMID: 29164603; PMCID: PMC6486090.
- van Maurik IS, Broulikova HM, Mank A, Bakker ED, de Wilde A, Bouwman FH, Stephens AW, van Berckel BNM, Scheltens P, van der Flier WM. A more precise diagnosis by means of amyloid PET contributes to delayed institutionalization, lower mortality, and reduced care costs in a tertiary memory clinic setting. Alzheimers Dement. 2023 May;19(5):2006-2013. doi: 10.1002/alz.12846. Epub 2022 Nov 23. PMID: 36419238.
- van der Molen LH, Boenink M, van lente H, Richard E. Changing definitions of disease: Transformations in the diagnostic criteria for Alzheimer’s Disease. Alz & Dem 2025. In print.
- Ossenkoppele R, Rabinovici GD, Smith R, Cho H, Schöll M, Strandberg O, Palmqvist S, Mattsson N, Janelidze S, Santillo A, Ohlsson T, Jögi J, Tsai R, La Joie R, Kramer J, Boxer AL, Gorno-Tempini ML, Miller BL, Choi JY, Ryu YH, Lyoo CH, Hansson O. Discriminative Accuracy of [18F]flortaucipir Positron Emission Tomography for Alzheimer Disease vs Other Neurodegenerative Disorders. JAMA. 2018 Sep 18;320(11):1151-1162. doi: 10.1001/jama.2018.12917. PMID: 30326496; PMCID: PMC6233630.
- Pichet Binette A, Palmqvist S, Bali D, Farrar G, Buckley CJ, Wolk DA, Zetterberg H, Blennow K, Janelidze S, Hansson O. Combining plasma phospho-tau and accessible measures to evaluate progression to Alzheimer's dementia in mild cognitive impairment patients. Alzheimers Res Ther. 2022 Mar 29;14(1):46. doi: 10.1186/s13195-022-00990-0. PMID: 35351181; PMCID: PMC8966264.
- Rabinovici GD, Carrillo MC, Apgar C, Gareen IF, Gutman R, Hanna L, Hillner BE, March A, Romanoff J, Siegel BA, Smith K, Song Y, Weber C, Whitmer RA, Gatsonis C. Amyloid Positron Emission Tomography and Subsequent Health Care Use Among Medicare Beneficiaries With Mild Cognitive Impairment or Dementia. JAMA Neurol. 2023 Nov 1;80(11):1166-1173. doi: 10.1001/jamaneurol.2023.3490. PMID: 37812437; PMCID: PMC10562987.
- Ritchie C, Smailagic N, Noel-Storr AH, Takwoingi Y, Flicker L, Mason SE, McShane R. Plasma and cerebrospinal fluid amyloid beta for the diagnosis of Alzheimer's disease dementia and other dementias in people with mild cognitive impairment (MCI). Cochrane Database Syst Rev. 2014 Jun 10;2014(6):CD008782. doi: 10.1002/14651858.CD008782.pub4. PMID: 24913723; PMCID: PMC6465069.
- Ritchie C, Smailagic N, Noel-Storr AH, Ukoumunne O, Ladds EC, Martin S. CSF tau and the CSF tau/ABeta ratio for the diagnosis of Alzheimer's disease dementia and other dementias in people with mild cognitive impairment (MCI). Cochrane Database Syst Rev. 2017 Mar 22;3(3):CD010803. doi: 10.1002/14651858.CD010803.pub2. PMID: 28328043; PMCID: PMC6464349.
- Schünemann HJ, Mustafa RA, Brozek J, Santesso N, Bossuyt PM, Steingart KR, Leeflang M, Lange S, Trenti T, Langendam M, Scholten R, Hooft L, Murad MH, Jaeschke R, Rutjes A, Singh J, Helfand M, Glasziou P, Arevalo-Rodriguez I, Akl EA, Deeks JJ, Guyatt GH; GRADE Working Group. GRADE guidelines: 22. The GRADE approach for tests and strategies-from test accuracy to patiënt-important outcomes and recommendations. J Clin Epidemiol. 2019 Jul;111:69-82. doi: 10.1016/j.jclinepi.2019.02.003. Epub 2019 Feb 7. PMID: 30738926.
- Smailagic N, Vacante M, Hyde C, Martin S, Ukoumunne O, Sachpekidis C. ¹⁸F-FDG PET for the early diagnosis of Alzheimer's disease dementia and other dementias in people with mild cognitive impairment (MCI). Cochrane Database Syst Rev. 2015 Jan 28;1(1):CD010632. doi: 10.1002/14651858.CD010632.pub2. PMID: 25629415; PMCID: PMC7081123.
- van der Veere PJ, Hoogland J, Visser LNC, Van Harten AC, Rhodius-Meester HF, Sikkes SAM, Venkatraghavan V, Barkhof F, Teunissen CE, van de Giessen E; Alzheimer's Disease Neuroimaging Initiative (ADNI),; Berkhof J, Van Der Flier WM. Predicting Cognitive Decline in Amyloid-Positive Patients With Mild Cognitive Impairment or Mild Dementia. Neurology. 2024 Aug 13;103(3):e209605. doi: 10.1212/WNL.0000000000209605. Epub 2024 Jul 10. PMID: 38986053; PMCID: PMC11238942.
- Vos SJ, Verhey F, Frölich L, Kornhuber J, Wiltfang J, Maier W, Peters O, Rüther E, Nobili F, Morbelli S, Frisoni GB, Drzezga A, Didic M, van Berckel BN, Simmons A, Soininen H, Kłoszewska I, Mecocci P, Tsolaki M, Vellas B, Lovestone S, Muscio C, Herukka SK, Salmon E, Bastin C, Wallin A, Nordlund A, de Mendonça A, Silva D, Santana I, Lemos R, Engelborghs S, Van der Mussele S; Alzheimer’s Disease Neuroimaging Initiative; Freund-Levi Y, Wallin ÅK, Hampel H, van der Flier W, Scheltens P, Visser PJ. Prevalence and prognosis of Alzheimer's disease at the mild cognitive impairment stage. Brain. 2015 May;138(Pt 5):1327-38. doi: 10.1093/brain/awv029. Epub 2015 Feb 17. PMID: 25693589; PMCID: PMC5013930.
- Zhang S, Smailagic N, Hyde C, Noel-Storr AH, Takwoingi Y, McShane R, Feng J. (11)C-PIB-PET for the early diagnosis of Alzheimer's disease dementia and other dementias in people with mild cognitive impairment (MCI). Cochrane Database Syst Rev. 2014 Jul 23;2014(7):CD010386. doi: 10.1002/14651858.CD010386.pub2. PMID: 25052054; PMCID: PMC6464750.
Evidence tabellen
Risk of Bias tables
Table of quality assessment for systematic reviews of diagnostic studies
|
Study
|
Appropriate and clearly focused question?1
Yes/no/unclear |
Comprehensive and systematic literature search?2
Yes/no/unclear |
Description of included and excluded studies?3
Yes/no/unclear |
Description of relevant characteristics of included studies?4
Yes/no/unclear |
Assessment of scientific quality of included studies?5
Yes/no/unclear |
Enough similarities between studies to make combining them reasonable?6 Yes/no/unclear |
Potential risk of publication bias taken into account?7
Yes/no/unclear |
Potential conflicts of interest reported?8
Yes/no/unclear |
|
Cotta ramusino, 2024 |
Yes |
No, only one database searched |
Yes |
Yes, partly lacking patiënt characteristics |
Yes |
Unclear |
No |
Unclear |
|
Lombardi, 2020 |
Yes |
Yes |
Yes |
Yes |
Yes |
No, authors do not combine results. |
No |
No |
|
Martinez, 2017 |
Yes |
Yes |
Yes |
Yes |
Yes |
No, authors do not combine results. |
No |
Yes |
|
Ritchie, 2014 |
Yes |
Yes |
Yes |
Yes |
Yes |
No, authors do not combine results. |
No |
No |
|
Ritchie, 2017 |
Yes |
Yes |
Yes |
Yes |
Yes |
No, authors do not combine results. |
No |
No |
|
Smailagic, 2015 |
Yes |
Yes |
Yes |
Yes |
Yes |
No, authors do not combine results. |
No |
No |
|
Zhang, 2014 |
Yes |
Yes |
Yes |
Yes |
Yes |
No, authors do not combine results. |
No |
No |
1. Research question (PICO) and inclusion criteria should be appropriate (in relation to the research question to be answered in the clinical guideline) and predefined
2. Search period and strategy should be described; at least Medline searched
3. Potentially relevant studies that are excluded at final selection (after reading the full text) should be referenced with reasons
4. Characteristics of individual studies relevant to the research question (PICO) should be reported
5. Quality of individual studies should be assessed using a quality scoring tool or checklist (preferably QUADAS-2; COSMIN checklist for measuring instruments) and taken into account in the evidence synthesis
6. Clinical and statistical heterogeneity should be assessed; clinical: enough similarities in patiënt characteristics, diagnostic tests (strategy) to allow pooling? For pooled data: at least 5 studies available for pooling; assessment of statistical heterogeneity and, more importantly (see Note), assessment of the reasons for heterogeneity (if present)? Note: sensitivity and specificity depend on the situation in which the test is being used and the thresholds that have been set, and sensitivity and specificity are correlated; therefore, the use of heterogeneity statistics (p-values; I2) is problematic, and rather than testing whether heterogeneity is present, heterogeneity should be assessed by eye-balling (degree of overlap of confidence intervals in Forest plot), and the reasons for heterogeneity should be examined.
7. There is no clear evidence for publication bias in diagnostic studies, and an ongoing discussion on which statistical method should be used. Tests to identify publication bias are likely to give false-positive results, among available tests, Deeks’ test is most valid. Irrespective of the use of statistical methods, you may score “Yes” if the authors discuss the potential risk of publication bias.
Table of excluded studies
|
Reference |
Reason for exclusion |
|
Arbizu J, Festari C, Altomare D, Walker Z, Bouwman F, Rivolta J, Orini S, Barthel H, Agosta F, Drzezga A, Nestor P, Boccardi M, Frisoni GB, Nobili F; EANM-EAN Task Force for the Prescription of FDG-PET for Dementing Neurodegenerative Disorders. Clinical utility of FDG-PET for the clinical diagnosis in MCI. Eur J Nucl Med Mol Imaging. 2018 Jul;45(9):1497-1508. doi: 10.1007/s00259-018-4039-7. Epub 2018 Apr 27. PMID: 29704037. |
Wrong design |
|
Blennow K, Shaw LM, Stomrud E, Mattsson N, Toledo JB, Buck K, Wahl S, Eichenlaub U, Lifke V, Simon M, Trojanowski JQ, Hansson O. Predicting clinical decline and conversion to Alzheimer's disease or dementia using novel Elecsys Aβ(1-42), pTau and tTau CSF immunoassays. Sci Rep. 2019 Dec 13;9(1):19024. doi: 10.1038/s41598-019-54204-z. PMID: 31836810; PMCID: PMC6911086. |
This individual study was submitted post hoc and was therefore not assessed within the systematic review–based evidence selection used for this module. Upon screening, the study outcomes are not relevant to the PICO, limiting applicability to this guideline. |
|
Ferreira D, Perestelo-Pérez L, Westman E, Wahlund LO, Sarría A, Serrano-Aguilar P. Meta-Review of CSF Core Biomarkers in Alzheimer's Disease: The State-of-the-Art after the New Revised Diagnostic Criteria. Front Aging Neurosci. 2014 Mar 24;6:47. doi: 10.3389/fnagi.2014.00047. PMID: 24715863; PMCID: PMC3970033. |
Wrong design (meta-review) |
|
Ferreira D, Rivero-Santana A, Perestelo-Pérez L, Westman E, Wahlund LO, Sarría A, Serrano-Aguilar P. Improving CSF Biomarkers' Performance for Predicting Progression from Mild Cognitive Impairment to Alzheimer's Disease by Considering Different Confounding Factors: A Meta-Analysis. Front Aging Neurosci. 2014 Oct 16;6:287. doi: 10.3389/fnagi.2014.00287. PMID: 25360114; PMCID: PMC4199277. |
Wrong reference (not defined) |
|
Herukka SK, Simonsen AH, Andreasen N, Baldeiras I, Bjerke M, Blennow K, Engelborghs S, Frisoni GB, Gabryelewicz T, Galluzzi S, Handels R, Kramberger MG, Kulczyńska A, Molinuevo JL, Mroczko B, Nordberg A, Oliveira CR, Otto M, Rinne JO, Rot U, Saka E, Soininen H, Struyfs H, Suardi S, Visser PJ, Winblad B, Zetterberg H, Waldemar G. Recommendations for cerebrospinal fluid Alzheimer's disease biomarkers in the diagnostic evaluation of mild cognitive impairment. Alzheimers Dement. 2017 Mar;13(3):285-295. doi: 10.1016/j.jalz.2016.09.009. Epub 2016 Oct 27. PMID: 28341066. |
Wrong method (only one database searched) |
|
Huszár Z, Engh MA, Pavlekovics M, Sato T, Steenkamp Y, Hanseeuw B, Terebessy T, Molnár Z, Hegyi P, Csukly G. Risk of conversion to mild cognitive impairment or dementia among subjects with amyloid and tau pathology: a systematic review and meta-analysis. Alzheimers Res Ther. 2024 Apr 12;16(1):81. doi: 10.1186/s13195-024-01455-2. PMID: 38610055; PMCID: PMC11015617. |
Wrong outcome measures (prognostic), wrong index (combination CSF and PET), wrong reference (neurocognitive tests) |
|
Ma Y, Brettschneider J, Collingwood JF. A Systematic Review and Meta-Analysis of Cerebrospinal Fluid Amyloid and Tau Levels Identifies Mild Cognitive Impairment Patiënts Progressing to Alzheimer's Disease. Biomedicines. 2022 Jul 15;10(7):1713. doi: 10.3390/biomedicines10071713. PMID: 35885018; PMCID: PMC9313367. |
Wrong outcome measures (prognostic association measure) |
|
Ma Y, Zhang S, Li J, Zheng DM, Guo Y, Feng J, Ren WD. Predictive accuracy of amyloid imaging for progression from mild cognitive impairment to Alzheimer disease with different lengths of follow-up: a meta-analysis. [Corrected]. Medicine (Baltimore). 2014 Dec;93(27):e150. doi: 10.1097/MD.0000000000000150. Erratum in: Medicine (Baltimore). 2015 Jan;94(2):1. Erratum in: Medicine (Baltimore). 2015 Jan;94(2):1. doi: 10.1097/01.md.0000460809.30837.cf. PMID: 25501055; PMCID: PMC4602781. |
Same studies as Zhang 2014, less documentation methodology. |
|
Monge-Argilés JA, Sánchez-Payá J, Muñoz-Ruiz C, Pampliega-Pérez A, Montoya-Gutiérrez J, Leiva-Santana C. Biomarcadores en el liquido cefalorraquideo de pacientes con deterioro cognitivo leve: metaanalisis de su capacidad predictiva para el diagnostico de la enfermedad de Alzheimer [Biomarkers in the cerebrospinal fluid of patiënts with mild cognitive impairment: a meta-analysis of their predictive capacity for the diagnosis of Alzheimer's disease]. Rev Neurol. 2010 Feb 16-28;50(4):193-200. Spanish. PMID: 20198590. |
Wrong language (Spanish) |
|
Noel-Storr AH, Flicker L, Ritchie CW, Nguyen GH, Gupta T, Wood P, Walton J, Desai M, Solomon DF, Molena E, Worrall R, Hayen A, Choudhary P, Ladds E, Lanctôt KL, Verhey FR, McCleery JM, Mead GE, Clare L, Fioravanti M, Hyde C, Marcus S, McShane R. Systematic review of the body of evidence for the use of biomarkers in the diagnosis of dementia. Alzheimers Dement. 2013 May;9(3):e96-e105. doi: 10.1016/j.jalz.2012.01.014. Epub 2012 Oct 27. PMID: 23110863. |
Wrong publication type, describes how studies report without presenting the diagnostic outcomes reported in studies. |
|
Olsson B, Lautner R, Andreasson U, Öhrfelt A, Portelius E, Bjerke M, Hölttä M, Rosén C, Olsson C, Strobel G, Wu E, Dakin K, Petzold M, Blennow K, Zetterberg H. CSF and blood biomarkers for the diagnosis of Alzheimer's disease: a systematic review and meta-analysis. Lancet Neurol. 2016 Jun;15(7):673-684. doi: 10.1016/S1474-4422(16)00070-3. Epub 2016 Apr 8. PMID: 27068280. |
Wrong outcome (fold change) |
|
Ruan D, Sun L. Amyloid-β PET in Alzheimer's disease: A systematic review and Bayesian meta-analysis. Brain Behav. 2023 Jan;13(1):e2850. doi: 10.1002/brb3.2850. Epub 2022 Dec 27. PMID: 36573329; PMCID: PMC9847612. |
Wrong method (only one database searched) |
|
Ruan Q, D'Onofrio G, Sancarlo D, Bao Z, Greco A, Yu Z. Potential neuroimaging biomarkers of pathologic brain changes in Mild Cognitive Impairment and Alzheimer's disease: a systematic review. BMC Geriatr. 2016 May 16;16:104. doi: 10.1186/s12877-016-0281-7. PMID: 27184250; PMCID: PMC4869390. |
Wrong design (narrative review) |
|
Sanchez-Catasus CA, Stormezand GN, van Laar PJ, De Deyn PP, Sanchez MA, Dierckx RA. FDG-PET for Prediction of AD Dementia in Mild Cognitive Impairment. A Review of the State of the Art with Particular Emphasis on the Comparison with Other Neuroimaging Modalities (MRI and Perfusion SPECT). Curr Alzheimer Res. 2017;14(2):127-142. doi: 10.2174/1567205013666160629081956. PMID: 27357645. |
Wrong method (only one database searched) |
|
Zhang S, Han D, Tan X, Feng J, Guo Y, Ding Y. Diagnostic accuracy of 18 F-FDG and 11 C-PIB-PET for prediction of short-term conversion to Alzheimer's disease in subjects with mild cognitive impairment. Int J Clin Pract. 2012 Feb;66(2):185-98. doi: 10.1111/j.1742-1241.2011.02845.x. PMID: 22257044. |
Wrong reference (short follow-up) |
|
Zhu L, Zhao W, Chen J, Li G, Qu J. Systematic review and meta-analysis of diagnostic test accuracy (DTA) studies: the role of cerebral perfusion imaging in prognosis evaluation of mild cognitive impairment. Ann Palliat Med. 2022 Feb;11(2):673-683. doi: 10.21037/apm-21-3916. PMID: 35249345. |
Wrong reporting (lacking relevant PICROTS information due to unclear reference) |
Verantwoording
Beoordelingsdatum en geldigheid
Publicatiedatum : 07-01-2026
Beoordeeld op geldigheid : 07-01-2026
Algemene gegevens
De verantwoording zal op de Richtlijnendatabase (Richtlijnendatabase.nl) bij elke geprioriteerde module die is ontwikkeld binnen dit cluster worden geplaatst. De betrokken expertiseleden, de kwalitatieve raming van mogelijke financiële gevolgen in het kader van de Wet kwaliteit, klachten en geschillen zorg (Wkkgz) en de autoriserende partijen kunnen variëren per module.
De ontwikkeling van deze richtlijnmodule werd ondersteund door het Kennisinstituut van de Federatie Medisch Specialisten (www.demedischspecialist.nl/kennisinstituut) en werd gefinancierd uit de Stichting Kwaliteitsgelden Medisch Specialisten (SKMS). De financier heeft geen enkele invloed gehad op de inhoud van de richtlijnmodule.
Samenstelling werkgroep
Voor het ontwikkelen van de richtlijnmodule is in 2021 een multidisciplinair cluster ingesteld. Het cluster Cognitieve stoornissen en dementie bestaat uit meerdere richtlijnen (zie hier de actuele clusterindeling). De stuurgroep bewaakt het proces van modulair onderhoud binnen het cluster. De expertisegroepsleden brengen hun expertise in, indien nodig. De volgende personen uit het cluster zijn betrokken geweest bij de herziening van deze module:
Clusterstuurgroepleden
- Dhr. prof. dr. M.G.M. (Marcel) Olde Rikkert (voorzitter), klinisch geriater; NVKG
- Dhr. prof. dr. A.R. (Tony) Absalom, anesthesioloog; NVA
- Dhr. dr. J.H.J.M. (Jeroen) de Bresser, radioloog; NVvR
- Mevr. dr. I.K. (Indrag) Lampe, psychiater; NVvP
- Mevr. R. (Renske) Leistra, patiëntvertegenwoordiger; Alzheimer Nederland
- Mevr. prof. dr. B.C. (Barbara) van Munster, internist; NIV
- Dhr. prof. dr. E. (Edo) Richard, neuroloog; NVN
- Mevr. prof. dr. Ir. C. (Charlotte) Teunissen, klinisch chemicus; NVKC
- Dhr. dr. R.A.W. (Ronald) Verhagen, orthopedisch chirurg; NOV
Betrokken clusterexpertisegroepleden
- Dhr. dr. A.P.A. (Auke) Appelman, radioloog; NVvR
- Mevr. dr. R.L. (Rozemarijn) van Bruchem-van Visser, internist ouderengeneeskunde; NIV
- Dhr. drs. B.P.H. (Bas) ter Brugge, specialist ouderengeneeskunde; Verenso
- Dhr. dr. J.A.H.R. (Jurgen) Claassen, klinisch geriater; NVKG
- Dhr. dr. P.L.J. (Paul) Dautzenberg, klinisch geriater; NVKG
- Mevr. dr. M.E.A. (Marlise) van Eersel, internist; NIV
- Mevr. drs. C.M. (Christa) de Geus, neurogeneticus; NVKG
- Mevr. dr. E.M. (Elsmarieke van de Giessen), radioloog; NVNG
- Mevr. drs. M. (Marjolein) Groeneveld, verpleegkundig Consulent Geriatrie, klinisch epidemioloog; V&VN
- Mevr. drs. H. (Hiltsje) Heemskerk, klinisch geriater; NVKG
- Dhr. dr. R.B. (Rients) Huitema, klinisch neuropsycholoog; NIP
- Dhr. drs. A. (Ali) Lahdidioui, internist; NIV
- Dhr. dr. J. (Jules) Lavalaye, nucleair geneeskundige; NVNG
- Mevr. drs. L. (Lieke) Mitrov, ziekenhuisapotheker; NVZA
- Mevr. dr. M. (Marieke) Perry, huisarts/onderzoeker; NHG
- Dhr. dr. G. (Gerwin) Roks, neuroloog; NVN
- Mevr. dr. T.R. (Rikje) Ruiter, internist; NIV
- Mevr. A.J.B.P. (Astrid) Schoonbrood, ergotherapeut; EN
- Mevr. dr. N. (Niki) Schoonenboom, neuroloog; NVN
- Dhr. dr. H. (Harro) Seelaar, neuroloog; NVN
- Dhr. dr. K.S. (Koen) Simons, intensivist-internist; NVIC
- Mevr. drs. M.M.E. (Marlies) Sleegers-Kerkenaar, klinisch geriater; NVKG
- Mevr. drs. VCJ (Vera) van Stek-Smits, neuropsycholoog-gezondheidszorgpsycholoog; NIP
- Mevr. dr. E.M. (Eveline) Veltman, psychiater; NVvP
- Dhr. drs. D. (Dave) Verkaik, AIOS geriatrie; NVKG
- Mevr. prof. dr. M. (Meike) Vernooij, radioloog; NVvR
- Dhr. dr. E.G.B. (Jort) Vijverberg, neuroloog; NVN
- Mevr. dr. M.A. (Marjolein) Wijngaarden, internist; NIV
Met ondersteuning van
- Mevr. dr. C.T.J. (Charlotte) Michels, senior adviseur, Kennisinstituut van de Federatie Medisch Specialisten
- Mevr. dr. L.C. (Lotte) Houtepen, adviseur, Kennisinstituut van de Federatie Medisch Specialisten
- Mevr. drs. L.C. (Laura) van Wijngaarden, junior adviseur, Kennisinstituut van de Federatie Medisch Specialisten
Belangenverklaringen
Een overzicht van de belangen van de clusterleden en het oordeel over het omgaan met eventuele belangen vindt u in onderstaande tabel. De ondertekende belangenverklaringen zijn op te vragen bij het secretariaat van het Kennisinstituut van de Federatie Medisch Specialisten via secretariaat@kennisinstituut.nl.
Clusterstuurgroepleden
|
Clusterlid |
Functie |
Nevenfuncties |
Gemelde belangen |
Ondernomen actie |
|
Olde Rikkert* |
Hoogleraar Geriatrie, Radboudumc, Nijmegen |
Hoofdredacteur Nederlands Tijdschrift voor Geneeskunde |
Geen; uitsluitend ZonMw gefinancierd onderzoek dat overheidsbelang centraal stelt. Sinds 2017 geen farma-onderzoek meer. |
Geen restrictie |
|
Absalom |
Hoogleraar Anesthesiologie, UMCG, Groningen |
Consultancy werkzaamheden (betaald, alle betalingen aan UMCG) 6. Consultancy werk voor Becton Dickinson (Eysins, Switzerland) en Terumo (Tokyo, Japan) – technische advies over spuitpompen. Niet gerelateerd aan dementie/ MCI/ delier. |
Extern gefinancierd onderzoeken, maar financier heeft geen belangen bij de richtlijn. * Rigel Pharmaceuticals (San Francisco, USA) (PAST) * The Medicines Company (Parsippany, NJ, USA)(PAST) |
Geen restricties, omdat adviseurswerk niet gerelateerd is aan de afbakening van het cluster |
|
De Bresser |
- Neuroradioloog 1.0fte, LUMC, Leiden |
Geen |
Mijn onderzoek wordt mede gesponsord door Alzheimer Nederland. Deze financier heeft geen belang bij bepaalde uitkomsten van de richtlijn. |
Geen restrictie |
|
Lampe |
Psychiater, OLVG Ziekenhuis, Amsterdam |
Geen |
Geen |
Geen restrictie |
|
Leistra |
Belangenbehartiger, Alzheimer Nederland |
Casemanager Dementie bij de King Arthur Groep |
Geen |
Geen restrictie |
|
Richard |
Hoogleraar neurologie: * afdeling neurologie Radboudumc (0.8fte) * afdeling Public and Occupational Health Amsterdam UMC (0.2 fte) |
- Neuroloog-onderzoeker AmsterdamUMC, locatie AMC, gastvrijheidsaanstelling. - Hoofdredacteur Leerboek Neurologie (Bohn Staffleu van Loghem) |
Geen, uitsluitend onderzoek financiering van non-profit instellingen (e.g. ZonMw, Europese Commissie). |
Geen restrictie |
|
Teunissen |
Hoofd Neurochemisch laboratorium, Afdeling Klinische Chemie, AmsterdamUMC, lokatie VUmc, Amsterdam |
*Adviseur voor educatief blad: Mednet Neurologie (betaald). * Alle betalingen zijn aan het AmsterdamUMC. |
*Wetenschappelijke samenwerking met ADxNeurosciences, Olink, Quanterix, Roche in kader van o.a. Marie Curie subsidie. Het doel van het Marie Curie project is om een nieuwe generatie van onderzoekers in het biomarker veld op te leiden, tot experts in alle aspecten van biomarker onderzoek. Aan het einde van de ontwikkeling van biomarkers zullen deze via bedrijven op de markt moeten komen, en omdat niet alle wetenschappers uiteindelijk in de academische wereld blijven, is het belangrijk dat jonge onderzoekers ervaring opdoen met de manier van onderzoek doen in het bedrijfsleven. In marie curie projecten gebeurt dat door een deel van het onderzoek bij een bedrijf uit te voeren. *Het Neurochemisch laboratorium doet contractresearch voor Acumen, ADx Neurosciences, AC-Immune, Alamar, Aribio, Axon Neurosciences, Beckman-Coulter, BioConnect, Bioorchestra, Brainstorm Therapeutics, Celgene, Cognition Therapeutics, EIP Pharma, Eisai, Eli Lilly, Fujirebio, Instant Nano Biosensors, Novo Nordisk, Olink, PeopleBio, Quanterix, Roche, Toyama, Vivoryon. Dit zijn meestal biomarker-analyses voor hun trials, bij inclusies en als uitkomstmaten. *Grants: Research of CET is supported by the European Commission (Marie Curie International Training Network, grant agreement No 860197 (MIRIADE) and TAME, Innovative Medicines Initiatives 3TR (Horizon 2020, grant no 831434) EPND ( IMI 2 Joint Undertaking (JU), grant No. 101034344) and JPND (bPRIDE, CCAD), European Partnership on Metrology, co-financed from the European Union’s Horizon Europe Research and Innovation Programme and by the Participating States ((22HLT07 NEuroBioStand), CANTATE project funded by the Alzheimer Drug Discovery Foundation, Alzheimer Association, Michael J Fox Foundation, Health Holland, the Dutch Research Council (ZonMW), Alzheimer Drug Discovery Foundation, The Selfridges Group Foundation, Alzheimer Netherlands. CT is recipient of ABOARD, which is a public-private partnership receiving funding from ZonMW (#73305095007) and Health~Holland, Topsector Life Sciences & Health (PPP-allowance; #LSHM20106). CT is recipient of TAP-dementia, a ZonMw funded project (#10510032120003) in the context of the Dutch National Dementia Strategy. |
Geen restrictie |
|
Van Munster |
* Hoogleraar Interne Geneeskunde, Ouderengeneeskunde/Geriatrie, UMCG, Groningen. *Plaatsvervangend opleider Geriatrie, UMCG, Groningen. |
- 2020 – heden Voorzitter Alzheimer Centrum Groningen (alle functies zijn onbetaald) |
*2022 ZONMw: Young Onset Dementia- INCLUDED: Advance care planning. 2022 ZEGG/ZONMw: "The impact of a comprehensive geriatric assessment including advance care planning in acutely hospitalized frail patients with cognitive disorders: the GOAL study" |
Geen restrictie |
|
Verhagen |
Orthopedisch chirurg/opleider in Tergooi MC |
Geen |
Geen |
Geen restrictie |
Clusterexpertisegroepleden
Richtlijn Dementie: Module ‘Antipsychotica bij dementie’
|
Clusterlid |
Functie |
Nevenfuncties |
Gemelde belangen |
Ondernomen actie |
|
Mitrov |
Ziekenhuisapotheker, Flevoziekenhuis, Almere |
Special Interest Group Farmacotherapie bij ouderen (NVKG/NVZA), onbetaald |
Geen |
Geen restrictie |
|
Verkaik |
Aios geriatrie, betaalde functie. In opleiding vanuit Gelre ziekenhuizen Apeldoorn en Zutphen. |
Deelname pvc commissie als aios lid voor kwaliteits visitaties (onbetaald) |
Geen |
Geen restrictie |
Richtlijn Dementie: Module ‘Antidepressiva bij dementie’
|
Clusterlid |
Functie |
Nevenfuncties |
Gemelde belangen |
Ondernomen actie |
|
Heemskerk |
Klinisch geriater, Arkin Ouderen (GGZ, Amsterdam) |
NVKG, commissie ouderenpsychiatrie |
Geen |
Geen restrictie |
|
Veltman |
Psychiater, UMCU |
Geen |
Geen |
Geen restrictie |
Richtlijn MCI: Module 'Biomarkers’
|
Clusterlid |
Functie |
Nevenfuncties |
Gemelde belangen |
Ondernomen actie |
|
Van de Giessen |
Radioloog (aandachtsgebeid nucleaire geneeskunde), Amsterdam UMC |
- NVNG, commissie onderwijs en themagroep neuro |
Beoordelingen PET scans voor Ixico (betaald aan instituut) Contract research voor Roche (betaald aan instituut) Onderzoeksfinanciering van non-profit instelingen (o.a. ZonMw, Alzheimer Nederland, Hersenstichting, Health~Holland, KWF) |
Geen restrictie |
|
Richard |
Hoogleraar neurologie: * afdeling neurologie Radboudumc (0.8fte) * afdeling Public and Occupational Health Amsterdam UMC (0.2 fte) |
- Neuroloog-onderzoeker AmsterdamUMC, locatie AMC, gastvrijheidsaanstelling. - Hoofdredacteur Leerboek Neurologie (Bohn Staffleu van Loghem) |
Geen, uitsluitend onderzoek financiering van non-profit instellingen (e.g. ZonMw, Europese Commissie). |
Geen restrictie |
Richtlijn MCI: Module ‘Leefstijladviezen’
|
Clusterlid
|
Functie |
Nevenfuncties |
Gemelde belangen |
Ondernomen actie |
|
Groeneveld |
Verpleegkundig consulent geriatrie (16 uur) Verpleegkundig onderzoeker Waardengedreven zorg (16 uur) |
Geen
|
Extern gefinancieerd onderzoek: * Catharina onderzoeksfonds: Prestatie en acceptatie van een predictiemodel voor vallen in het ziekenhuis (rol als projectleider) *KIPZ subsidie t/m 31 december: Prestatie en acceptatie van een predictiemodel voor vallen in het ziekenhuis (ja) |
Geen restrictie |
|
Ruiter |
*Internist ouderen geneeskunde en klinische farmacologie Maasstad Ziekenhuis 0,8fte *Post-doctoraal onderzoeker & Epidemioloog B Erasmus MC dept. Epidemiologie 0,1 fte * Lid van het College ter beoordeling van geneesmiddelen |
*Lid raad van toezicht Stichting Landelijk Wonen Klein Houtdijk (Onbetaald). *Secretaris Forum Visitatorum NIV (Onbetaald). *Redactielid Tijdschrift Geriatrie en Gerontologie (Onbetaald). * Voorzitter commissie beroepsbelangen kerngroep ouderengeneeskunde NIV. |
Geen |
Geen restrictie |
|
Claassen |
Universitair hoofddocent en klinisch geriater (1.0 fte), Radboudumc, Nijmegen |
Geen |
*Fase 3 onderzoek Novo Nordisk (EVOKE, NCT04777396. Wereldwijde geneesmiddelenstudie (semaglutide) over Alzheimer. Rol als lokale PI. Middel komt niet aan bod in huidige vijf modules. *MOCIA-project, gefinancierd door ZonMW (www.mocia.nl). *ABOARD onderzoek (gefinancierd door ZonMW en Health Holland, projectnummer: 73305095007) |
Geen restrictie |
Richtlijn MCI: Module ‘Follow-up’
|
Clusterlid |
Functie |
Nevenfuncties |
Gemelde belangen |
Ondernomen actie |
|
Claassen |
Universitair hoofddocent en klinisch geriater (1.0 fte), Radboudumc, Nijmegen |
Geen |
*Fase 3 onderzoek Novo Nordisk (EVOKE, NCT04777396. Wereldwijde geneesmiddelenstudie (semaglutide) over Alzheimer. Rol als lokale PI. Middel komt niet aan bod in huidige vijf modules. *MOCIA-project, gefinancierd door ZonMW (www.mocia.nl). *ABOARD onderzoek (gefinancierd door ZonMW en Health Holland, projectnummer: 73305095007) |
Geen restrictie |
|
Perry |
* Huisarts, Huisartsenpraktijk Velp, 0.5 fte * Senior-onderzoeker afdelingen eerstelijnsgeneeskunde en Geriatrie van het Radboudumc en bij het Radboud Alzheimer Centrum, 0.5 fte |
Auteur hoofdstuk Vergeetachtigheid in Álledaagse klachten 2020 (onkostenvergoeding) Commissielid werkgroep multidisciplinaire richtlijn Dementie 2018 - 2020 (onkostenvergoeding) Commissielid werkgroep heziening NHG standaard Dementie 2017 - 2020 (onkostenvergoeding) Commissielid werkgroep Addendum MCI bij multidisciplinaire richtlijn Dementie 20 1 6-20 1 B (onkostenvergoeding) Auteur online nascholing dementie Accredidact huisartsen 2016 en doktersassistenten 2017 (betaald) Auteur twee boekhoofdstukken dementie (palliatieve zorg en diagnostische verrichtingen) 2017 en 2018 in opdracht van het NHG (onkostenvergoeding) Auteur hoofdtsuk Vergeetachtigheid (Alledaagse klachten in de huisartsgeneeskunde) 2020 Onkostenvergoeding Expert bij www.dementie.nl tot heden (vrijwillig) Columnist Alz (donateursblaadje Alzheimer Nederland) tot 2016 (vrijwillig) |
Projectleider DementieNet (financiering door Giekes-Strijbis fonds, Alzheimer Nederland en ZonMw) Andere ZonMw/Memorabel projecten: - Decidem (anticiperende besluitvorming met mensen met dementie door huisartsen), medeprojectleider - Crisisreductie in de dementiezorg, medeaanvrager - SHiMMy en SHARED (relatie sociale gezondheid en ontstaan en progressie van dementie), medeaanvrager - S-Decided (gezamelijke besluitvorming bij diagnostiek bij geheugenklachten), medeaanvrager
Diverse projecten omtrent transmurale/interprofessionele aanpak van advance care planning van Zorginstituut Nederland, Stoffels-Hornstra fonds, ZonMw EPOS: Extramurale praktijkontwikkeling specialist ouderengeneeskunde UNICITY: dementie op jonge leeftijd, oa signalering door huisartsen en onderscheid met depressie, burn-out
*DementieNet aanpak, generieke benadering voor lokale eerstelijns netwerkzorg *Expert bij www.dementie.nl, website van Alzheimer Nederland (vrijwillig) *Columnist Alz (donateursblaadje Alzheimer Nederland) tot 2016 (vrijwillig) *Diverse malen gastspreker bij verschillende Alzheimer Cafés (vrijwillig) |
Geen restrictie |
|
Roks |
Neuroloog ETZ Tilburg |
METC Brabant, lid en vice voorzitter (onkostenvergoeding) |
Geen |
Geen restrictie |
Algemene herziening: Nieuwe concept indeling (richtlijn Dementie en richtlijn MCI)
|
Clusterlid |
Functie |
Nevenfuncties |
Gemelde belangen |
Ondernomen actie |
|
Lampe |
Psychiater, OLVG ziekenhuis |
Geen |
Geen |
Geen restrictie |
|
Olde Rikkert* |
Hoogleraar Geriatrie, Radboudumc, Nijmegen |
Hoofdredacteur Nederlands Tijdschrift voor Geneeskunde |
Geen; uitsluitend ZonMw gefinancierd onderzoek dat overheidsbelang centraal stelt. Sinds 2017 geen farma-onderzoek meer. |
Geen restrictie |
|
Perry |
* Huisarts, Huisartsenpraktijk Velp, 0.5 fte * Senior-onderzoeker afdelingen eerstelijnsgeneeskunde en Geriatrie van het Radboudumc en bij het Radboud Alzheimer Centrum, 0.5 fte |
Auteur hoofdstuk Vergeetachtigheid in Álledaagse klachten 2020 (onkostenvergoeding) Commissielid werkgroep multidisciplinaire richtlijn Dementie 2018 - 2020 (onkostenvergoeding) Commissielid werkgroep heziening NHG standaard Dementie 2017 - 2020 (onkostenvergoeding) Commissielid werkgroep Addendum MCI bij multidisciplinaire richtlijn Dementie 20 1 6-20 1 B (onkostenvergoeding) Auteur online nascholing dementie Accredidact huisartsen 2016 en doktersassistenten 2017 (betaald) Auteur twee boekhoofdstukken dementie (palliatieve zorg en diagnostische verrichtingen) 2017 en 2018 in opdracht van het NHG (onkostenvergoeding) Auteur hoofdtsuk Vergeetachtigheid (Alledaagse klachten in de huisartsgeneeskunde) 2020 Onkostenvergoeding Expert bij www.dementie.nl tot heden (vrijwillig) Columnist Alz (donateursblaadje Alzheimer Nederland) tot 2016 (vrijwillig) |
Projectleider DementieNet (financiering door Giekes-Strijbis fonds, Alzheimer Nederland en ZonMw) Andere ZonMw/Memorabel projecten: - Decidem (anticiperende besluitvorming met mensen met dementie door huisartsen), medeprojectleider - Crisisreductie in de dementiezorg, medeaanvrager - SHiMMy en SHARED (relatie sociale gezondheid en ontstaan en progressie van dementie), medeaanvrager - S-Decided (gezamelijke besluitvorming bij diagnostiek bij geheugenklachten), medeaanvrager
Diverse projecten omtrent transmurale/interprofessionele aanpak van advance care planning van Zorginstituut Nederland, Stoffels-Hornstra fonds, ZonMw EPOS: Extramurale praktijkontwikkeling specialist ouderengeneeskunde UNICITY: dementie op jonge leeftijd, oa signalering door huisartsen en onderscheid met depressie, burn-out
*DementieNet aanpak, generieke benadering voor lokale eerstelijns netwerkzorg *Expert bij www.dementie.nl, website van Alzheimer Nederland (vrijwillig) *Columnist Alz (donateursblaadje Alzheimer Nederland) tot 2016 (vrijwillig) *Diverse malen gastspreker bij verschillende Alzheimer Cafés (vrijwillig) |
Geen restrictie |
|
Richard |
Hoogleraar neurologie: * afdeling neurologie Radboudumc (0.8fte) * afdeling Public and Occupational Health Amsterdam UMC (0.2 fte) |
- Neuroloog-onderzoeker AmsterdamUMC, locatie AMC, gastvrijheidsaanstelling. - Hoofdredacteur Leerboek Neurologie (Bohn Staffleu van Loghem) |
Geen, uitsluitend onderzoek financiering van non-profit instellingen (e.g. ZonMw, Europese Commissie). |
Geen restrictie |
Inbreng patiëntenperspectief
Kwalitatieve raming van mogelijke financiële gevolgen in het kader van de Wkkgz
Bij de richtlijnmodule voerden de clusterleden conform de Wet kwaliteit, klachten en geschillen zorg (Wkkgz) een kwalitatieve raming uit om te beoordelen of de aanbevelingen mogelijk leiden tot substantiële financiële gevolgen. Bij het uitvoeren van deze beoordeling is de richtlijnmodule op verschillende domeinen getoetst (zie het stroomschema bij Werkwijze).
Uit de kwalitatieve raming blijkt dat er waarschijnlijk geen substantiële financiële gevolgen zijn, zie onderstaande tabel.
| Module |
Uitkomst raming |
Toelichting |
|
Biomarkers bij MCI |
Geen substantiële financiële gevolgen |
Hoewel uit de toetsing volgt dat de aanbeveling(en) breed toepasbaar zijn (>40.000 patiënten), volgt uit de toetsing dat het overgrote deel van de zorgverleners al aan de norm voldoet, het geen nieuwe manier van zorgverlening betreft, het geen toename in het aantal voltijdsequivalenten of wijziging in het opleidingsniveau van zorgverleners betreft. Er worden daarom geen financiële gevolgen verwacht. |
Werkwijze
Voor meer details over de gebruikte richtlijnmethodologie verwijzen wij u naar de Werkwijze. Relevante informatie voor de ontwikkeling/herziening van deze richtlijnmodule is hieronder weergegeven.
Zoekverantwoording
Algemene informatie
|
Cluster/richtlijn: Cognitieve stoornissen en Dementie |
|
|
Uitgangsvraag/modules: UV3 Wat is de waarde van de toepassing van MRI, PET en liquoronderzoek bij het voorspellen van progressie van MCI naar dementie? |
|
|
Database(s): Embase.com, Ovid/Medline |
Datum: 20 augustus 2024 |
|
Periode: vanaf 2010 |
Talen: geen restrictie |
|
Literatuurspecialist: Alies Oost |
Rayyan: |
|
BMI-zoekblokken: voor verschillende opdrachten wordt (deels) gebruik gemaakt van de zoekblokken van BMI-Online https://blocks.bmi-online.nl/ Deduplication: voor het ontdubbelen is gebruik gemaakt van http://dedupendnote.nl:9777/ |
|
Zoekopbrengst
|
|
EMBASE |
OVID/MEDLINE |
Ontdubbeld |
|
SR |
470 |
368 |
595* |
|
RCT |
1221 |
1373 |
2101 |
|
Observationele studies |
5920 |
4508 |
7186 |
|
Totaal |
7611 |
6249 |
9882 |
*in Rayyan
Zoekstrategie
Embase.com
|
No. |
Query |
Results |
|
#1 |
'mild cognitive impairment'/exp OR 'mild cognitive impair*':ti,ab,kw OR mci:ti,ab,kw OR (('cognitive impair*' NEAR/3 ('no dement*' OR 'without dement*' OR 'not dement*')):ti,ab,kw) OR cind:ti,ab,kw |
72118 |
|
#2 |
'nuclear magnetic resonance imaging'/exp OR 'mri scanner'/exp OR ('magnetic resonance':ab,ti AND (image:ab,ti OR images:ab,ti OR imaging:ab,ti)) OR mri:ab,ti OR mris:ab,ti OR nmr:ab,ti OR mra:ab,ti OR mras:ab,ti OR zeugmatograph*:ab,ti OR 'mr tomography':ab,ti OR 'mr tomographies':ab,ti OR 'mr tomographic':ab,ti OR 'mr imag*':ti,ab,kw OR 'proton spin':ab,ti OR ((magneti*:ab,ti OR 'chemical shift':ab,ti) AND imaging:ab,ti) OR fmri:ab,ti OR fmris:ab,ti OR rsfmri:ti,ab,kw OR 'computer assisted emission tomography'/exp OR spect:ti,ab,kw OR petscan*:ti,ab,kw OR pet:ti,ab,kw OR (((emission OR positron) NEAR/3 tomograph*):ti,ab,kw) OR radionuclid*:ti,ab,kw OR (('glucose metabolism' NEAR/3 imag*):ti,ab,kw) OR 'cerebrospinal fluid'/exp OR 'cerebrospinal fluid level'/exp OR 'cerebrospinal fluid examination'/exp OR csf:ti,ab,kw OR liquor*:ti,ab,kw OR (((cerebrospinal* OR cerebral) NEAR/3 (fluid* OR biomarker*)):ti,ab,kw) OR 'amyloid beta protein'/exp OR 'amyloid beta protein[1-42]'/exp OR 'amyloid beta protein[1-40]'/exp OR (((amyloid OR a) NEAR/3 (beta* OR β*)):ti,ab,kw) OR ((amyloid NEAR/3 (plaque* OR imag*)):ti,ab,kw) OR 'abeta*':ti,ab,kw OR aβ*:ti,ab,kw OR amyloidβ*:ti,ab,kw OR amyloidbeta:ti,ab,kw OR abp:ti,ab,kw OR 'tau protein'/exp OR 'p tau protein'/exp OR 't tau protein'/exp OR 'total tau protein'/exp OR 'phosphorylated tau protein'/exp OR 'phosphorylated tau 181'/exp OR 'phosphorylated tau 181 protein'/exp OR 'phosphorylated tau'/exp OR tau:ti,ab,kw OR ttau:ti,ab,kw OR ptau*:ti,ab,kw OR tau181:ti,ab,kw OR 'neurofilament l'/exp OR 'neurofilament light chain'/exp OR 'neurofilament light chain protein'/exp OR 'neurofilament light protein'/exp OR 'neurofilament light'/exp OR 'neurofilament l*':ti,ab,kw OR nfl:ti,ab,kw OR 'biological marker'/exp OR biomarker*:ti,ab,kw OR (((biological OR diagnostic OR imaging) NEAR/3 marker*):ti,ab,kw) OR 'hippocampal atrophy'/exp OR 'hippocampal volume'/exp OR 'medial temporal atrophy'/exp OR 'brain atrophy'/exp OR 'cortical thickness (brain)'/exp OR 'white matter'/exp OR 'gray matter'/exp OR ((hippocamp* NEAR/3 (atroph* OR volume)):ti,ab,kw) OR 'medial temporal atrophy':ti,ab,kw OR ((brain NEAR/3 (atroph* OR shrink* OR abiotroph*)):ti,ab,kw) OR (((cortex OR cortical) NEAR/3 (thickness OR thinning)):ti,ab,kw) OR 'white matter':ti,ab,kw OR 'gray matter':ti,ab,kw OR hyperintensit*:ti,ab,kw OR wmh:ti,ab,kw |
3403012 |
|
#3 |
'area under the curve'/exp OR 'brier score'/exp OR 'computer prediction'/exp OR 'c statistic'/exp OR 'c statistics'/exp OR 'integrated discrimination improvement'/exp OR 'net reclassification improvement'/exp OR 'net reclassification index'/exp OR 'prediction'/exp OR 'predictive model'/exp OR 'predictive modeling'/exp OR 'predictive validity'/exp OR 'predictive value'/exp OR 'regression analysis'/exp OR 'statistical model'/exp OR 'area under the curve':ti,ab,kw OR 'brier score*':ti,ab,kw OR 'c statistic*' OR 'computer prediction':ti,ab,kw OR 'decision curve anal*':ti,ab,kw OR (('net reclassification' NEAR/2 (improvement OR index)):ti,ab,kw) OR (((predict* OR statistical*) NEAR/3 (model* OR validity OR value)):ti,ab,kw) OR 'proportional hazards model*':ti,ab,kw OR 'r square*':ti,ab,kw OR regression:ti,ab,kw OR predict*:ti OR multivariate:ti,ab,kw OR multivariab*:ti,ab,kw |
2968462 |
|
#4 |
'sensitivity and specificity'/de OR sensitivity:ab,ti OR specificity:ab,ti OR predict*:ab,ti OR 'roc curve':ab,ti OR 'receiver operator':ab,ti OR 'receiver operators':ab,ti OR likelihood:ab,ti OR 'diagnostic error'/exp OR 'diagnostic accuracy'/exp OR 'diagnostic test accuracy study'/exp OR 'inter observer':ab,ti OR 'intra observer':ab,ti OR interobserver:ab,ti OR intraobserver:ab,ti OR validity:ab,ti OR kappa:ab,ti OR reliability:ab,ti OR reproducibility:ab,ti OR ((test NEAR/2 're-test'):ab,ti) OR ((test NEAR/2 'retest'):ab,ti) OR 'reproducibility'/exp OR accuracy:ab,ti OR 'differential diagnosis'/exp OR 'validation study'/de OR 'measurement precision'/exp OR 'diagnostic value'/exp OR 'reliability'/exp OR 'predictive value'/exp OR ppv:ti,ab,kw OR npv:ti,ab,kw OR (((false OR true) NEAR/3 (negative OR positive)):ti,ab) |
6541201 |
|
#5 |
#1 AND #2 AND (#3 OR #4) NOT ('conference abstract'/it OR 'editorial'/it OR 'letter'/it OR 'note'/it) NOT (('animal'/exp OR 'animal experiment'/exp OR 'animal model'/exp OR 'nonhuman'/exp) NOT 'human'/exp) NOT (('adolescent'/exp OR 'child'/exp OR adolescent*:ti,ab,kw OR child*:ti,ab,kw OR schoolchild*:ti,ab,kw OR infant*:ti,ab,kw OR girl*:ti,ab,kw OR boy*:ti,ab,kw OR teen:ti,ab,kw OR teens:ti,ab,kw OR teenager*:ti,ab,kw OR youth*:ti,ab,kw OR pediatr*:ti,ab,kw OR paediatr*:ti,ab,kw OR puber*:ti,ab,kw) NOT ('adult'/exp OR 'aged'/exp OR 'middle aged'/exp OR adult*:ti,ab,kw OR man:ti,ab,kw OR men:ti,ab,kw OR woman:ti,ab,kw OR women:ti,ab,kw)) |
10308 |
|
#6 |
#5 AND [2010-2024]/py |
9043 |
|
#7 |
'meta analysis'/exp OR 'meta analysis (topic)'/exp OR metaanaly*:ti,ab OR 'meta analy*':ti,ab OR metanaly*:ti,ab OR 'systematic review'/de OR 'cochrane database of systematic reviews'/jt OR prisma:ti,ab OR prospero:ti,ab OR (((systemati* OR scoping OR umbrella OR 'structured literature') NEAR/3 (review* OR overview*)):ti,ab) OR ((systemic* NEAR/1 review*):ti,ab) OR (((systemati* OR literature OR database* OR 'data base*') NEAR/10 search*):ti,ab) OR (((structured OR comprehensive* OR systemic*) NEAR/3 search*):ti,ab) OR (((literature NEAR/3 review*):ti,ab) AND (search*:ti,ab OR database*:ti,ab OR 'data base*':ti,ab)) OR (('data extraction':ti,ab OR 'data source*':ti,ab) AND 'study selection':ti,ab) OR ('search strategy':ti,ab AND 'selection criteria':ti,ab) OR ('data source*':ti,ab AND 'data synthesis':ti,ab) OR medline:ab OR pubmed:ab OR embase:ab OR cochrane:ab OR (((critical OR rapid) NEAR/2 (review* OR overview* OR synthes*)):ti) OR ((((critical* OR rapid*) NEAR/3 (review* OR overview* OR synthes*)):ab) AND (search*:ab OR database*:ab OR 'data base*':ab)) OR metasynthes*:ti,ab OR 'meta synthes*':ti,ab |
733409 |
|
#8 |
'clinical trial'/exp OR 'randomization'/exp OR 'single blind procedure'/exp OR 'double blind procedure'/exp OR 'crossover procedure'/exp OR 'placebo'/exp OR 'prospective study'/exp OR rct:ab,ti OR random*:ab,ti OR 'single blind':ab,ti OR 'randomised controlled trial':ab,ti OR 'randomized controlled trial'/exp OR placebo*:ab,ti |
3302394 |
|
#9 |
'major clinical study'/de OR 'clinical study'/de OR 'case control study'/de OR 'family study'/de OR 'longitudinal study'/de OR 'retrospective study'/de OR 'prospective study'/de OR 'comparative study'/de OR 'cohort analysis'/de OR ((cohort NEAR/1 (study OR studies)):ab,ti) OR (('case control' NEAR/1 (study OR studies)):ab,ti) OR (('follow up' NEAR/1 (study OR studies)):ab,ti) OR (observational NEAR/1 (study OR studies)) OR ((epidemiologic NEAR/1 (study OR studies)):ab,ti) OR (('cross sectional' NEAR/1 (study OR studies)):ab,ti) |
6767914 |
|
#10 |
'case control study'/de OR 'comparative study'/exp OR 'control group'/de OR 'controlled study'/de OR 'controlled clinical trial'/de OR 'crossover procedure'/de OR 'double blind procedure'/de OR 'phase 2 clinical trial'/de OR 'phase 3 clinical trial'/de OR 'phase 4 clinical trial'/de OR 'pretest posttest design'/de OR 'pretest posttest control group design'/de OR 'quasi experimental study'/de OR 'single blind procedure'/de OR 'triple blind procedure'/de OR (((control OR controlled) NEAR/6 trial):ti,ab,kw) OR (((control OR controlled) NEAR/6 (study OR studies)):ti,ab,kw) OR (((control OR controlled) NEAR/1 active):ti,ab,kw) OR 'open label*':ti,ab,kw OR (((double OR two OR three OR multi OR trial) NEAR/1 (arm OR arms)):ti,ab,kw) OR ((allocat* NEAR/10 (arm OR arms)):ti,ab,kw) OR placebo*:ti,ab,kw OR 'sham-control*':ti,ab,kw OR (((single OR double OR triple OR assessor) NEAR/1 (blind* OR masked)):ti,ab,kw) OR nonrandom*:ti,ab,kw OR 'non-random*':ti,ab,kw OR 'quasi-experiment*':ti,ab,kw OR crossover:ti,ab,kw OR 'cross over':ti,ab,kw OR 'parallel group*':ti,ab,kw OR 'factorial trial':ti,ab,kw OR ((phase NEAR/5 (study OR trial)):ti,ab,kw) OR ((case* NEAR/6 (matched OR control*)):ti,ab,kw) OR ((match* NEAR/6 (pair OR pairs OR cohort* OR control* OR group* OR healthy OR age OR sex OR gender OR patiënt* OR subject* OR participant*)):ti,ab,kw) OR ((propensity NEAR/6 (scor* OR match*)):ti,ab,kw) OR versus:ti OR vs:ti OR compar*:ti OR ((compar* NEAR/1 study):ti,ab,kw) OR (('major clinical study'/de OR 'clinical study'/de OR 'cohort analysis'/de OR 'observational study'/de OR 'cross-sectional study'/de OR 'multicenter study'/de OR 'correlational study'/de OR 'follow up'/de OR cohort*:ti,ab,kw OR 'follow up':ti,ab,kw OR followup:ti,ab,kw OR longitudinal*:ti,ab,kw OR prospective*:ti,ab,kw OR retrospective*:ti,ab,kw OR observational*:ti,ab,kw OR 'cross sectional*':ti,ab,kw OR cross?ectional*:ti,ab,kw OR multicent*:ti,ab,kw OR 'multi-cent*':ti,ab,kw OR consecutive*:ti,ab,kw) AND (group:ti,ab,kw OR groups:ti,ab,kw OR subgroup*:ti,ab,kw OR versus:ti,ab,kw OR vs:ti,ab,kw OR compar*:ti,ab,kw OR 'odds ratio*':ab OR 'relative odds':ab OR 'risk ratio*':ab OR 'relative risk*':ab OR 'rate ratio':ab OR aor:ab OR arr:ab OR rrr:ab OR ((('or' OR 'rr') NEAR/6 ci):ab))) |
15326988 |
|
#11 |
#6 AND #7 - SR |
470 |
|
#12 |
#6 AND #8 NOT #11 - RCT |
1221 |
|
#13 |
#6 AND (#9 OR #10) NOT (#11 OR #12) - observationeel |
5920 |
|
#14 |
#11 OR #12 OR #13 |
7611 |
Ovid/Medline
|
# |
Searches |
Results |
|
1 |
Cognitive Dysfunction/ or 'mild cognitive impair*'.ti,ab,kf. or mci.ti,ab,kf. or ('cognitive impair*' adj3 ('no dement*' or 'without dement*' or 'not dement*')).ti,ab,kf. or cind.ti,ab,kf. |
65090 |
|
2 |
exp magnetic resonance imaging/ or ("magnetic resonance" and (image or images or imaging)).ti,ab,kf. or mri.ti,ab,kf. or mris.ti,ab,kf. or nmr.ti,ab,kf. or mra.ti,ab,kf. or mras.ti,ab,kf. or zeugmatograph*.ti,ab,kf. or "mr tomography".ti,ab,kf. or "mr tomographies".ti,ab,kf. or "mr tomographic".ti,ab,kf. or 'mr imag*'.ti,ab,kf. or "proton spin".ti,ab,kf. or ((magneti* or "chemical shift") and imaging).ti,ab,kf. or fmri.ti,ab,kf. or fmris.ti,ab,kf. or rsfmri.ti,ab,kf. or exp Tomography, Emission-Computed/ or spect.ti,ab,kf. or petscan*.ti,ab,kf. or pet.ti,ab,kf. or ((emission or positron) adj3 tomograph*).ti,ab,kf. or radionuclid*.ti,ab,kf. or ('glucose metabolism' adj3 imag*).ti,ab,kf. or exp Cerebrospinal Fluid/ or csf.ti,ab,kf. or liquor*.ti,ab,kf. or ((cerebrospinal* or cerebral) adj3 (fluid* or biomarker*)).ti,ab,kf. or exp Amyloid beta-Peptides/ or ((amyloid or a) adj3 beta*).ti,ab,kf. or (amyloid adj3 (plaque* or imag*)).ti,ab,kf. or 'abeta*'.ti,ab,kf. or amyloidbeta.ti,ab,kf. or abp.ti,ab,kf. or exp tau Proteins/ or tau.ti,ab,kf. or ttau.ti,ab,kf. or ptau*.ti,ab,kf. or tau181.ti,ab,kf. or exp Neurofilament Proteins/ or 'neurofilament l*'.ti,ab,kf. or nfl.ti,ab,kf. or Biomarkers/ or biomarker*.ti,ab,kf. or ((biological or diagnostic or imaging) adj3 marker*).ti,ab,kf. or (exp Brain/ and exp Atrophy/) or exp Gray Matter/ or exp White Matter/ or (hippocamp* adj3 (atroph* or volume)).ti,ab,kf. or 'medial temporal atrophy'.ti,ab,kf. or (brain adj3 (atroph* or shrink* or abiotroph*)).ti,ab,kf. or ((cortex or cortical) adj3 (thickness or thinning)).ti,ab,kf. or 'white matter'.ti,ab,kf. or 'gray matter'.ti,ab,kf. or hyperintensit*.ti,ab,kf. or wmh.ti,ab,kf. |
2354373 |
|
3 |
exp "Sensitivity and Specificity"/ or (sensitivity or specificity).ti,ab. or (predict* or ROC-curve or receiver-operator*).ti,ab. or (likelihood or LR*).ti,ab. or exp Diagnostic Errors/ or (inter-observer or intra-observer or interobserver or intraobserver or validity or kappa or reliability).ti,ab. or reproducibility.ti,ab. or (test adj2 (re-test or retest)).ti,ab. or "Reproducibility of Results"/ or accuracy.ti,ab. or Diagnosis, Differential/ or Validation Study/ or ((false or true) adj3 (negative or positive)).ti,ab. |
5170952 |
|
4 |
Area Under Curve/ or exp Forecasting/ or "Predictive Value of Tests"/ or exp Multivariate Analysis/ or exp Regression Analysis/ or exp Models, Statistical/ or area under the curve.ti,ab,kf. or brier score*.ti,ab,kf. or c statistic*.ti,ab,kf. or computer prediction.ti,ab,kf. or decision curve anal*.ti,ab,kf. or (net reclassification adj2 (improvement or index)).ti,ab,kf. or ((predict* or statistical*) adj3 (model* or validity or value)).ti,ab,kf. or proportional hazards model*.ti,ab,kf. or r square*.ti,ab,kf. or regression.ti,ab,kf. or predict*.ti. or multivaria*.ti,ab,kf. |
2623690 |
|
5 |
(1 and 2 and (3 or 4)) not (comment/ or editorial/ or letter/) not ((exp animals/ or exp models, animal/) not humans/) not ((Adolescent/ or Child/ or Infant/ or adolescen*.ti,ab,kf. or child*.ti,ab,kf. or schoolchild*.ti,ab,kf. or infant*.ti,ab,kf. or girl*.ti,ab,kf. or boy*.ti,ab,kf. or teen.ti,ab,kf. or teens.ti,ab,kf. or teenager*.ti,ab,kf. or youth*.ti,ab,kf. or pediatr*.ti,ab,kf. or paediatr*.ti,ab,kf. or puber*.ti,ab,kf.) not (Adult/ or adult*.ti,ab,kf. or man.ti,ab,kf. or men.ti,ab,kf. or woman.ti,ab,kf. or women.ti,ab,kf.)) |
10633 |
|
6 |
limit 5 to yr="2010 -Current" |
9479 |
|
7 |
meta-analysis/ or meta-analysis as topic/ or (metaanaly* or meta-analy* or metanaly*).ti,ab,kf. or systematic review/ or cochrane.jw. or (prisma or prospero).ti,ab,kf. or ((systemati* or scoping or umbrella or "structured literature") adj3 (review* or overview*)).ti,ab,kf. or (systemic* adj1 review*).ti,ab,kf. or ((systemati* or literature or database* or data-base*) adj10 search*).ti,ab,kf. or ((structured or comprehensive* or systemic*) adj3 search*).ti,ab,kf. or ((literature adj3 review*) and (search* or database* or data-base*)).ti,ab,kf. or (("data extraction" or "data source*") and "study selection").ti,ab,kf. or ("search strategy" and "selection criteria").ti,ab,kf. or ("data source*" and "data synthesis").ti,ab,kf. or (medline or pubmed or embase or cochrane).ab. or ((critical or rapid) adj2 (review* or overview* or synthes*)).ti. or (((critical* or rapid*) adj3 (review* or overview* or synthes*)) and (search* or database* or data-base*)).ab. or (metasynthes* or meta-synthes*).ti,ab,kf. |
768680 |
|
8 |
exp clinical trial/ or randomized controlled trial/ or exp clinical trials as topic/ or randomized controlled trials as topic/ or Random Allocation/ or Double-Blind Method/ or Single-Blind Method/ or (clinical trial, phase i or clinical trial, phase ii or clinical trial, phase iii or clinical trial, phase iv or controlled clinical trial or randomized controlled trial or multicenter study or clinical trial).pt. or random*.ti,ab. or (clinic* adj trial*).tw. or ((singl* or doubl* or treb* or tripl*) adj (blind$3 or mask$3)).tw. or Placebos/ or placebo*.tw. |
2766390 |
|
9 |
Epidemiologic studies/ or case control studies/ or exp cohort studies/ or Controlled Before-After Studies/ or Case control.tw. or cohort.tw. or Cohort analy$.tw. or (Follow up adj (study or studies)).tw. or (observational adj (study or studies)).tw. or Longitudinal.tw. or Retrospective*.tw. or prospective*.tw. or consecutive*.tw. or Cross sectional.tw. or Cross-sectional studies/ or historically controlled study/ or interrupted time series analysis/ [Onder exp cohort studies vallen ook longitudinale, prospectieve en retrospectieve studies] |
4805971 |
|
10 |
Case-control Studies/ or clinical trial, phase ii/ or clinical trial, phase iii/ or clinical trial, phase iv/ or comparative study/ or control groups/ or controlled before-after studies/ or controlled clinical trial/ or double-blind method/ or historically controlled study/ or matched-pair analysis/ or single-blind method/ or (((control or controlled) adj6 (study or studies or trial)) or (compar* adj (study or studies)) or ((control or controlled) adj1 active) or "open label*" or ((double or two or three or multi or trial) adj (arm or arms)) or (allocat* adj10 (arm or arms)) or placebo* or "sham-control*" or ((single or double or triple or assessor) adj1 (blind* or masked)) or nonrandom* or "non-random*" or "quasi-experiment*" or "parallel group*" or "factorial trial" or "pretest posttest" or (phase adj5 (study or trial)) or (case* adj6 (matched or control*)) or (match* adj6 (pair or pairs or cohort* or control* or group* or healthy or age or sex or gender or patiënt* or subject* or participant*)) or (propensity adj6 (scor* or match*))).ti,ab,kf. or (confounding adj6 adjust*).ti,ab. or (versus or vs or compar*).ti. or ((exp cohort studies/ or epidemiologic studies/ or multicenter study/ or observational study/ or seroepidemiologic studies/ or (cohort* or 'follow up' or followup or longitudinal* or prospective* or retrospective* or observational* or multicent* or 'multi-cent*' or consecutive*).ti,ab,kf.) and ((group or groups or subgroup* or versus or vs or compar*).ti,ab,kf. or ('odds ratio*' or 'relative odds' or 'risk ratio*' or 'relative risk*' or aor or arr or rrr).ab. or (("OR" or "RR") adj6 CI).ab.)) |
5764659 |
|
11 |
6 and 7 - SR |
368 |
|
12 |
(6 and 8) not 11 - RCT |
1373 |
|
13 |
(6 and (9 or 10)) not (11 or 12) - observationeel |
4508 |
|
14 |
11 or 12 or 13 |
6249 |