Infragenuaal: Gecoate ballonnen en stents
Uitgangsvraag
Wat is de optimale behandeling van het infragenuale traject?
De uitgangsvraag bevat de volgende deelvragen:
- Wat is het optimale type ballon (gecoat/ongecoat) bij endovasculaire behandeling van kritieke ischemie onder de knie?
- Wat is het optimale type stent (gecoat/ongecoat) bij endovasculaire behandeling van kritieke ischemie onder de knie?
Aanbeveling
Kies bij endovasculaire behandeling van patiënten met kritieke ischemie en crurale stenosen of occlusies bij voorkeur primair voor PTA met niet-gecoate ballonnen.
Plaats bij voorkeur een gecoate stent bij onvoldoende resultaat (persisterende stenose of flow-limiterende disssectie) na PTA met een niet-gecoate ballon.
Overweeg bij primaire behandeling van focale (minder dan drie centimeter) proximale crurale stenosen of occlusies plaatsing van een gecoate stent.
Overwegingen
Voor- en nadelen van de interventie en de kwaliteit van het bewijs
Er is literatuuronderzoek gedaan naar de voor- en nadelen van de behandeling van crurale arteriële stenosen en occlusies middels ballonnen en stents gecoat met een geneesmiddel vergeleken met niet-gecoate ballonnen of stents bij patiënten met kritieke ischemie. Als cruciale uitkomstmaten werden majeure amputatie en klinisch gedreven revascularisatie en mortaliteit gedefinieerd. Patency/restenose, wondgenezing en kwaliteit van leven werden beschouwd als belangrijke uitkomstmaten voor de besluitvorming.
Er werden tien trials gevonden. Op basis van deze trials werd na een jaar een betere patency en dientengevolge ook een reductie in klinisch gedreven revascularisatie ten opzichte van niet-gecoate stents/ballonnen gezien. Ook resulteert een behandeling middels gecoate ballonnen en stents na een jaar waarschijnlijk in een betere wondgenezing. Echter, er werd een duidelijke trend gezien richting meer majeure amputaties bij de behandeling middels gecoate ballonnen ten opzichte van niet-gecoate ballonnen, terwijl gecoate stents het aantal majeure amputaties juist leken te verminderen ten opzichte van niet-gecoate stents. Een verschil in mortaliteit tussen de groepen leek er niet te zijn. Kwaliteit van leven werd niet beschreven. Hier ligt een kennislacune.
Op basis van de literatuur lijkt behandeling met gecoate ballonnen en stents effectiever op het gebied van patency, reïnterventie en wondgenezing dan behandeling met niet-gecoate ballonnen en stents bij patiënten met kritieke ischemie en crurale arteriële stenosen en occlusies. Gecoate ballonnen lijken echter het risico op majeure amputatie te vergroten. Ondanks de verschillen in coating en anatomie, is er in de literatuuranalyse geen onderscheid gemaakt in subanalyses tussen evrolimus gecoate stents en paclitaxel gecoate stents. De effecten van beide stents waren vergelijkbaar en niet tegenstrijdig, waardoor poolen van de resultaten geen vertekend effect heeft gegeven.
Waarden en voorkeuren van patiënten (en evt. hun verzorgers)
Voor de patiënt met CLTI is het belangrijk om de wond(en) aan de voet te laten genezen en een (majeure) amputatie te voorkomen. Uit de literatuur blijkt dat dit doel het beste behaald kan worden door behandeling met een niet-gecoate ballon. In geval van onvoldoende resultaat kan een korte, gecoate stent geplaatst worden. Bij korte letsels kan primair gekozen worden voor een gecoate stent.
Kosten (middelenbeslag)
Gecoate ballonnen en stents zijn duur (400-1000 euro). Echter, het gebruik van deze producten bij CLTI-patiënten heeft als doel om een amputatie te voorkomen. De kosten voor de maatschappij na een majeure amputatie zijn aanzienlijk en derhalve lijkt het gebruik van deze dure producten goed te verdedigen.
Aanvaardbaarheid, haalbaarheid en implementatie
Atherosclerotische letsels in de crurale arteriën zijn vaak lang (meer dan tien centimeter). Er zijn talloze ballonnen beschikbaar in alle denkbare lengtes en diameters om deze letsels te behandelen. Echter, dit geldt niet voor stents. De stents, die gebruikt worden bij de behandeling van crurale afwijkingen, komen vanuit de cardiologie. Ondanks dat de diameters van de crurale arteriën en de coronairen in grote lijnen overeenkomen, zijn de lengtes van de afwijkingen in de coronairen over het algemeen veel korter dan in de cruraal arteriën. De beschikbare gecoate stents hebben dan ook een maximale lengte van circa 3 centimeter. Derhalve wordt het gebruik van gecoate stents bij de behandeling van crurale afwijkingen gereserveerd voor de kortere letsels en als bail-out bij onvoldoende resultaat na PTA. Daarbij is ook de locatie van de afwijkingen van belang bij de keuze voor een gecoate stent. Proximaal in het onderbeen zijn de crurale arteriën redelijk gefixeerd, maar richting de enkel worden deze arteriën steeds mobieler. Distaal in het onderbeen wordt derhalve het gebruik van stents door de werkgroep afgeraden.
Rationale van de aanbeveling: weging van argumenten voor en tegen de interventies
Op basis van de literatuur lijkt behandeling met gecoate ballonnen en stents effectiever op het gebied van patency, reïnterventie en wondgenezing dan behandeling met niet-gecoate ballonnen en stents bij patiënten met kritieke ischemie en crurale arteriële stenosen en occlusies. Gecoate ballonnen lijken echter het risico op majeure amputatie te vergroten. Voor een patiënt is het zeer belangrijk amputatie te voorkomen. Bij behandeling van patiënten met kritieke ischemie en arteriële stenosen en occlusies onder de knie gaat de voorkeur daarom uit naar niet-gecoate ballonnen. Mocht dit onvoldoende resultaat geven dan wordt bail-out stenting geadviseerd middels een gecoate stent.
Onderbouwing
Endovascular treatment of patients with peripheral arterial disease has increased dramatically in recent decades due to improved techniques and increased expertise. The major advantage of endovascular treatment is its minimally invasive nature, however, the disadvantage is its limited patency compared to surgical revascularisation. To improve the patency of endovascular treatment, several technical developments have become available, such as the use of stents and coating balloons and stents with antiproliferative drugs. Balloons and stents are coated with, for example, Paclitaxel or Sirolimus to reduce the risk of intima hyperplasia and restenosis. Particularly in the femoro-popliteal, but also in the infragenicular pathway, there is a lot of experience with coated endovascular balloons and stents.
Treatment of stenotic and occluding vasculature is indicated in patients presenting with critical ischaemia (Chronic Limb Threatening Ischemia; CLTI). Patients with intermittent claudication (CI) who do not respond adequately to treatment by supervised gait training are also eligible for endovascular revascularisation.
Treatment of patients with CI is aimed at improving walking distance. Patients with CLTI often have one or more wounds on the foot and the aim of treatment is therefore to heal these wounds. Treatment of the infragenicular pathway is basically reserved for patients with CLTI.
Critical outcomes
|
Low GRADE |
Treatment with drug-coated balloon angioplasty may increase major amputation at 12 months when compared with uncoated balloon angioplasty in CLTI patients with below-the-knee arterial stenoses and occlusions.
Ipema, 2020; Jia, 2020; DEBATE-BTK; ACOART-BTK; IN.PACT; SINGA-PACLI. |
|
Low GRADE |
Treatment with drug-eluting stents may reduce major amputation at 12 months when compared with uncoated stents in CLTI patients with below-the-knee arterial stenoses and occlusions.
Source: DESTINY; PADI. |
|
Low GRADE |
Treatment with drug-coated balloons may reduce clinically driven target lesion revascularization at 12 months when compared with uncoated balloons in CLTI patients with below-the-knee arterial stenoses and occlusions.
Source: Ipema, 2020; Jia, 2020; DEBATE-BTK; ACOART-BTK; IN.PACT; SINGA-PACLI. |
|
Low GRADE |
Treatment with drug-eluting stents may have little to no effect on clinically driven target lesion revascularization at 12 months when compared with uncoated stents in CLTI patients with below-the-knee arterial stenoses and occlusions.
Source: DESTINY; PADI. |
|
Moderate GRADE |
Treatment with drug-coated balloons or stents likely has little to no effect on mortality at 12 months when compared with uncoated balloons or stents in CLTI patients with below-the-knee arterial stenoses and occlusions.
Source: Ipema, 2020; Jia, 2020; DEBATE-BTK; ACOART-BTK; IN.PACT; SINGA-PACLI; DESTINY; PADI. |
Important outcomes
|
Moderate GRADE |
Treatment with drug-coated balloons or stents likely reduces binary restenosis at 12 months when compared with uncoated balloons or stents in CLTI patients with below-the-knee arterial stenoses and occlusions.
Source: Ipema, 2020; Jia, 2020; DEBATE-BTK; ACOART-BTK; IN.PACT; SINGA-PACLI; DESTINY; PADI. |
|
Low GRADE |
Treatment with drug-coated balloons may improve wound healing at 12 months when compared with uncoated balloons in CLTI patients with below-the-knee arterial stenoses and occlusions.
Source: Jia, 2020; ACOART-BTK; SINGA-PACLI. |
|
No GRADE |
No evidence was found about the effect of treatment with drug-eluting stents on wound healing when compared with uncoated stents in CLTI patients with below-the-knee arterial stenoses and occlusions. |
|
No GRADE |
No evidence was found about the effect of treatment with drug-coated balloons or stents on quality of life when compared with uncoated balloons or stents in CLTI patients with below-the-knee arterial stenoses and occlusions. |
Description of studies
Table 1 outlines the main study characteristics of the included publications.
Ipema (2020) performed a systematic review and meta-analysis of drug-coated balloon angioplasty versus standard percutaneous transluminal angioplasty in below the knee peripheral arterial disease and included studies published between January 2008 and November 2018. Ten studies representing 1593 patients met the inclusion criteria. The current analysis only included the four randomized controlled trials (Haddad, 2017; Liistro, 2013; Zeller, 2014; Zeller, 2015).
Jia (2020) reported results of the AcoArt II–BTK study, a prospective, multicenter,
randomized controlled trial that sought to assess the safety and efficacy of Litos or Tulip DCBs versus uncoated balloons for patients with stenotic or occlusive infrapopliteal lesions. The trial enrolled 120 patients who were randomly assigned to angioplasty with either a DCB (n=61; mean age 70.7±7.4 years; 36 men) or a conventional balloon catheter (n=59; mean age 70.8±9.0 years; 36 men). Most patients had diabetes and hypertension, and nearly all patients (119/120) suffered from CLTI (Rutherford category 4–6). The primary efficacy outcome was primary patency at 6 months, defined as the absence of target lesion occlusion on angiography or the need for clinically-driven target lesion revascularization (CD-TLR) and no major amputation of the target limb. The follow-up window at 6 months ranged from 5 to 9 months.
In the ACOART-BTK randomized controlled single-center study, Liistro (2020) compared the efficacy and safety of the Litos drug-coated balloon (N=52) versus plain old balloon angioplasty (N=53) in patients with critical limb ischemia undergoing below-the-knee intervention. Inclusion criteria were critical limb ischemia (Rutherford class ≥4) and significant stenosis or occlusion >40 mm of at least 1 vessel below the knee with distal runoff successfully treated with angioplasty. Six-month angiographic late lumen loss was the primary endpoint. Occlusive restenosis at 6 months and clinically driven target lesion revascularization at 12 months were secondary endpoints.
Liistro (2022) assessed the safety and effectiveness of the IN.PACT 014 paclitaxel-coated balloon versus conventional percutaneous transluminal angioplasty for infrapopliteal chronic total occlusions in patients with chronic limb-threatening ischaemia. In the IN.PACT BTK prospective, multicentre, randomised pilot study, fifty participants (Rutherford clinical category 4-5) were randomised 1:1 to DCB (N=23) or PTA (N=27). The primary effectiveness endpoint was late lumen loss at 9 months post procedure. Safety outcomes up to 9 months included all-cause mortality, major target limb amputation, and clinically driven target lesion revascularisation.
The single-center, randomized DEBATE-BTK trial enrolled 132 diabetic patients with critical limb ischemia. Subjects were randomized 1:1 to IN.PACT paclitaxel-coated or plain old balloon angioplasty. Major clinical endpoints were, freedom from all–cause death, freedom from clinically driven target lesion revascularization and the occurrence of major amputation at 12 months. Liistro (2013) reported twelve-month results and Liistro (2022) results up to 5 years.
In the SINGA-PACLI trial, a prospective, randomized, two-center, double-blind superiority study, Patel (2021) included participants with critical limb ischemia with rest pain or tissue loss with atherosclerotic disease in the native below-the-knee arteries. Patient were randomly assigned to paclitaxel-coated balloon angioplasty or conventional angioplasty, after stratification for diabetes and renal failure between November 2013 and October 2017. The primary efficacy end point was angiographic primary patency at 6 months analyzed on an intention-to-treat basis. Secondary end points through 12 months were composed of major adverse events including death and major amputations, wound healing, limb salvage, clinically driven target-lesion revascularization, and amputation-free survival.
Bosiers (2012) reported results of the DESTINY trial, a randomized comparison of everolimus-eluting versus bare-metal stents in patients with critical limb ischemia and infrapopliteal arterial occlusive disease. 74 patients were treated with Xience V and 66 patients were treated with Vision. Follow-up patient assessments were conducted at 1, 6,
and 12 months and included a history and physical examination, assessment of the clinical category of CLI according to the Rutherford categorization, and a color-flow Doppler
ultrasound investigation.
The PADI trial compared paclitaxel-eluting stents with bare-metal stents for infrapopliteal lesions in adults with critical limb ischemia (Rutherford category ≥4). Spreen (2016) and Spreen (2017) reported the 12-month outcomes and results up to 5 years, respectively. Primary end point was 6-month primary binary patency of treated lesions, defined as ≤50% stenosis on computed tomographic angiography. Loss of >50% of luminal diameter on CT, treatment in interim by means of infrapopliteal bypass or endovascular reintervention, major amputation, and death related to CLI were considered as treatment failure. An ordinal score was used to grade the severity of treatment failure from vessel restenosis, through vessel occlusion to treatment in interim, major amputation, or CLI-related death. Additional secondary end points were ischemic categorization of the treated leg by means of Rutherford classification, minor and major amputation (at or below versus above ankle level, respectively) of the trial leg, and periprocedural (within 30 days) complications, serious adverse events, and death.
Table 3. Study characteristics.
|
Study |
Disease severity |
Comparison |
N (I/C) |
Follow-up |
|
Bosiers, 2012 DESTINY trial |
Rutherford category 4 or 5 |
Xience-V everolimus-eluting stent vs. bare-metal stent |
74/66 |
12 months |
|
Haddad, 2017 |
Rutherford 4 to 6 |
Luminor 14 paclitaxel-coated vs. uncoated balloon |
48/48 |
12 months |
|
Jia, 2021 |
Rutherford category 4 to 6 |
Litos or Tulip paclitaxel-coated vs. uncoated balloon |
61/59 |
12 months |
|
Liistro, 2020 ACOART-BTK trial |
Rutherford category 4 or higher |
Litos paclitaxel-coated vs. uncoated balloon |
52/53 |
12 months |
|
Liistro, 2013 Liistro, 2022 DEBATE-BTK |
Rutherford category 4 or higher |
IN.PACT paclitaxel-coated vs. uncoated balloon |
65/67 |
12 months, 5 years |
|
Liistro, 2022 IN.PACT trial |
Rutherford category 4 or 5 |
IN.PACT paclitaxel-coated vs. uncoated balloon |
23/27 |
9 months |
|
Patel, 2021 SINGA-PACLI trial |
Rutherford category 4, 5, or 6 |
Passeo-18 Lux paclitaxel-coated vs. uncoated balloon |
70/68 |
12 months |
|
Spreen, 2016 Spreen, 2017 PADI trial |
Rutherford category 4 or higher |
paclitaxel-eluting stent vs. bare-metal stent |
73/64 |
12 months, 5 years |
|
Zeller, 2014 IN.PACT DEEP |
CLI |
IN.PACT Ampherion paclitaxel-coated vs. uncoated balloon |
239/119 |
12 months |
|
Zeller, 2015 BIOLUX-PII |
CLI |
Passeo-18 Lux paclitaxel-coated vs. uncoated balloon |
36/36 |
12 months |
Results
Major amputation (critical outcome)
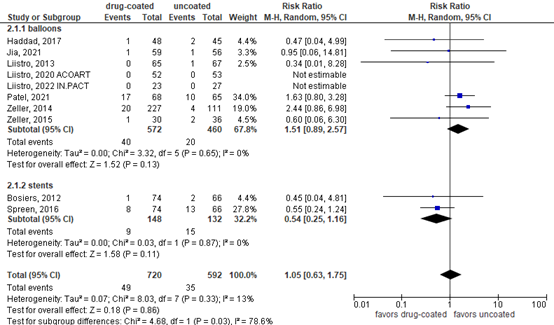
Major amputation was reported in eight publications comparing balloons, and two comparing stents (figure 1). For balloons, the pooled RR was 1.51 (95% CI 0.89 to 2.57, N=1032) in favor of uncoated balloons. The difference was considered clinically relevant. At longer follow-up, Liistro (2022) reported major amputations at 5 years in 1/65 (2%) patients with drug-coated balloons versus 2/67 (3%) patients with uncoated balloons.
In contrast, for stents, the pooled RR was 0.54 (95% CI 0.63 to 1.75, N=280) in favor of drug-eluting stents. The difference was considered clinically relevant. At longer follow-up, Spreen (2017) reported major amputations at 2 years in 9/68 (13.2%) patients with drug-coated balloons versus 15/60 (24.8%) patients with uncoated balloons. At 5 years, major amputations were performed in 11/57 (19.3%) versus 17/50 (34%) patients.
Figure 1. Major amputation at 12 months
Random effects model; df: degrees of freedom; I2: statistical heterogeneity; CI: confidence interval; Z: p-value of pooled effect.
Clinically driven target lesion revascularization (critical outcome)
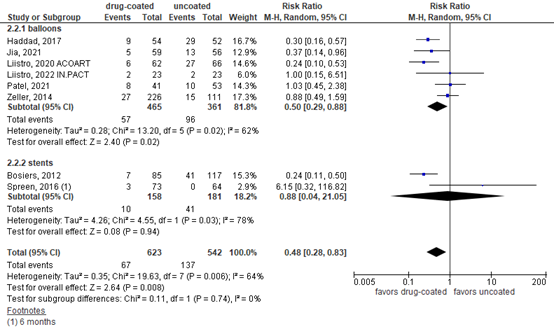
Six publications reported CD-TLR at 12 months for drug-coated versus uncoated balloons and two studies reported CD-TLR for drug-eluting versus bare metal stents at 6 to 12 months (figure 2). For balloons, the pooled RR was 0.50 in favor of drug-coated balloon angioplasty, (95% CI 0.29 to 0.88, N=826). For stents, the results were inconsistent. Bosiers (2012) found a RR of 0.24 (95% CI 0.11 to 0.50) in favor of drug-eluting stents, whereas Spreen (2016) reported a RR of 6.15 (95% CI 0.32 to 116.8) in favor of bare metal stents.
Figure 2. Clinically driven target lesion revascularization at 12 months
Random effects model; df: degrees of freedom; I2: statistical heterogeneity; CI: confidence interval; Z: p-value of pooled effect.
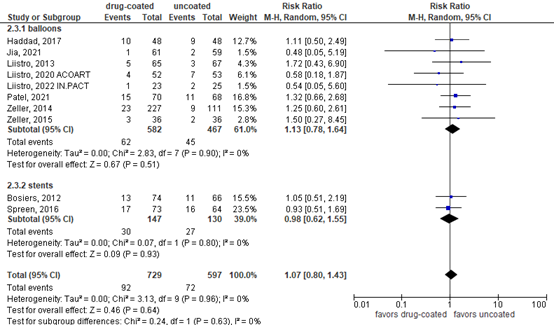
Mortality (critical outcome)
Eight publications reported mortality at 12 months balloon drug-coated and uncoated balloon angioplasty. Two studies reported mortality comparing stents (figure 3). The pooled RR for balloons was 1.13 (95% CI 0.78 to 1.64; N=1049) in favor of uncoated balloons. At longer follow-up, Liistro (2022) reported a 5-year mortality of 24/65 (36.9%) with drug-coated balloons versus 32/67 (46.3%) in uncoated balloons.
For stents, the pooled RR was 0.98 (95% CI 0.62 to 1.55, N=277) in favor of uncoated stents. Taken together, the RR was 1.07 (95% CI 0.80 to 1.43; N=1326) in favor of drug-eluting stents. The differences were not considered clinically relevant.
Figure 3. Mortality at 12 months
Random effects model; df: degrees of freedom; I2: statistical heterogeneity; CI: confidence interval; Z: p-value of pooled effect.
At longer follow-up, Spreen (2017) reported a 2-year mortality of 17/73 (23.3%) with drug-eluting stents versus 19/64 (29.7%) in bare metal stents.
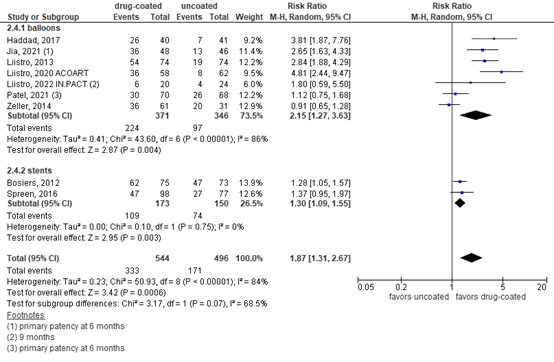
Patency
Seven publications reported patency of freedom from binary restenosis at 6 to 12 months in drug-coated versus balloon angioplasty, and three in drug-eluting versus bare metal stents. For balloons, the pooled RR was 2.15 (95% CI 1.27 to 3.63, N=717) in favor of drug-coated balloon angioplasty, as indicated in figure 1. For stents, the RR was 1.30 (95% CI 1.09 to 1.55, N=323) in favor of drug-coated stents. Taken together, the RR was 1.87 in favor of drug-coated devices, with a 95% CI from 1.31 to 2.67 (N=1040). The differences were considered clinically relevant.
Figure 4. Patency or freedom of binary restenosis at 12 months
Random effects model; df: degrees of freedom; I2: statistical heterogeneity; CI: confidence interval; Z: p-value of pooled effect.
In addition, Spreen (2017) reported freedom from binary restenosis at 2 years of 15/48 (31.3%) with drug-eluting stents versus 11/45 (24.4%) with bare metal stents, and 5/43 (11.6%) versus 3/35 (8.6%), respectively, at 5 years.
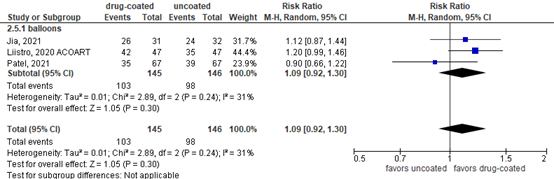
Wound healing
Three publications reported wound healing at 12 months comparing balloon drug-coated and uncoated balloon angioplasty (figure 5). The included studies comparing stents did not report wound healing. The pooled RR for balloons was 1.09 (95% CI 0.92 to 1.30; N=291) in favor of drug-coated balloons.
The difference was not considered clinically relevant.
Figure 5. Wound healing at 12 months
Random effects model; df: degrees of freedom; I2: statistical heterogeneity; CI: confidence interval; Z: p-value of pooled effect.
Quality of life
Quality of life was not reported in the included studies
Level of evidence of the literature
The level of evidence for all outcomes was based on randomized controlled trials and therefore started high.
The level of evidence regarding the outcome measure major amputation was downgraded by two levels to LOW due to conflicting results (inconsistency, -1) and due to the limited number of events (imprecision, -1).
The level of evidence regarding the outcome measure clinically driven target lesion revascularization was downgraded by one level to LOW due to conflicting results (inconsistency, -1) and due to the limited number of events (imprecision, -1).
The level of evidence regarding the outcome measure mortality was downgraded by one level to MODERATE, due to conflicting results (inconsistency, -1).
The level of evidence regarding the outcome measure patency was downgraded by one level to MODERATE, due to conflicting results (inconsistency, -1).
The level of evidence regarding the outcome measure wound healing was downgraded by two levels to LOW, due to conflicting results (inconsistency, -1), and because the confidence interval crossed one limit of clinical decision making (imprecision, -1).
A systematic review of the literature was performed to answer the following question:
What are the benefits and harms of drug-coated balloons or stents compared with uncoated balloons or stents in patients with chronic limb threatening ischemia below the knee?
Table 1. PICO 1
|
Patients |
patients with chronic limb threatening ischemia below the knee |
|
Intervention |
drug-coated balloon |
|
Control |
uncoated balloon |
|
Outcomes |
major amputation, mortality, clinically driven re-intervention, patency/restenosis, quality of life, wound healing. |
|
Other selection criteria |
Study design: systematic reviews and randomized controlled trials |
Table 2. PICO 2
|
Patients |
patients with chronic limb threatening ischemia below the knee |
|
Intervention |
drug-coated stent |
|
Control |
uncoated stent |
|
Outcomes |
major amputation, mortality, clinically driven re-intervention, patency/restenosis, quality of life, wound healing. |
|
Other selection criteria |
Study design: systematic reviews and randomized controlled trials |
Relevant outcome measures
The guideline development group considered major amputation, clinically driven re-intervention, and mortality as critical outcome measures for decision making; and patency, quality of life and wound healing as important outcome measures for decision making.
The working group did not define the outcome measures listed above a priori, but used the definitions used in the studies.
The working group defined 25% as a minimal clinically (patient) important difference for dichotomous outcomes (relative risk ≤0.80 or ≥0.25).
Search and select (Methods)
On the 10th of August, relevant search terms were used to search for systematic reviews, RCT and observational studies about the optimal treatment for patients with infragenicularl/infrapopliteal arterial occlusive disease in the databases Embase.com and Ovid/Medline. The search resulted in 475 unique hits. Studies were selected based on the following criteria: (1) randomized controlled trials in (2) patients with critical limb ischemia below the knee, in which (3) drug-coated balloons or stents were compared with uncoated balloons or stents. Twenty-six publications were initially selected based on title and abstract screening. After reading the full text, sixteen publications were excluded (see the table with reasons for exclusion under the tab Methods), and ten publications were included.
Results
One systematic review and nine additional randomized trials were included in the analysis of the literature. Important study characteristics and results are summarized in the evidence tables. The assessment of the risk of bias is summarized in the risk of bias tables.
- Bosiers M, Scheinert D, Peeters P, Torsello G, Zeller T, Deloose K, Schmidt A, Tessarek J, Vinck E, Schwartz LB. Randomized comparison of everolimus-eluting versus bare-metal stents in patients with critical limb ischemia and infrapopliteal arterial occlusive disease. J Vasc Surg. 2012 Feb;55(2):390-8. doi: 10.1016/j.jvs.2011.07.099. Epub 2011 Dec 14. PMID: 22169682.
- Ipema J, Huizing E, Schreve MA, de Vries JPM, Ünlü Ç. Editor's Choice - Drug Coated Balloon Angioplasty vs. Standard Percutaneous Transluminal Angioplasty in Below the Knee Peripheral Arterial Disease: A Systematic Review and Meta-Analysis. Eur J Vasc Endovasc Surg. 2020 Feb;59(2):265-275. doi: 10.1016/j.ejvs.2019.10.002. Epub 2019 Dec 27. PMID: 31889657.
- Jia X, Zhuang B, Wang F, Gu Y, Zhang J, Lu X, Dai X, Liu Z, Bi W, Liu C, Wang S, Liistro F, Guo W. Drug-Coated Balloon Angioplasty Compared With Uncoated Balloons in the Treatment of Infrapopliteal Artery Lesions (AcoArt II-BTK). J Endovasc Ther. 2021 Apr;28(2):215-221. doi: 10.1177/1526602820969681. Epub 2020 Oct 29. PMID: 33118432.
- Liistro F, Angioli P, Ventoruzzo G, Ducci K, Reccia MR, Ricci L, Falsini G, Scatena A, Pieroni M, Bolognese L. Randomized Controlled Trial of Acotec Drug-Eluting Balloon Versus Plain Balloon for Below-the-Knee Angioplasty. JACC Cardiovasc Interv. 2020 Oct 12;13(19):2277-2286. doi: 10.1016/j.jcin.2020.06.045. Epub 2020 Sep 16. PMID: 32950416.
- Liistro F, Porto I, Angioli P, Grotti S, Ricci L, Ducci K, Falsini G, Ventoruzzo G, Turini F, Bellandi G, Bolognese L. Drug-eluting balloon in peripheral intervention for below the knee angioplasty evaluation (DEBATE-BTK): a randomized trial in diabetic patients with critical limb ischemia. Circulation. 2013 Aug 6;128(6):615-21. doi: 10.1161/CIRCULATIONAHA.113.001811. PMID: 23797811.
- Liistro F, Weinberg I, Almonacid Popma A, Shishehbor MH, Deckers S, Micari A. Paclitaxel-coated balloons versus percutaneous transluminal angioplasty for infrapopliteal chronic total occlusions: the IN.PACT BTK randomised trial. EuroIntervention. 2022 Apr 1;17(17):e1445-e1454. doi: 10.4244/EIJ-D-21-00444. PMID: 34602386; PMCID: PMC9896391.
- Patel A, Irani FG, Pua U, Tay KH, Chong TT, Leong S, Chan ES, Tan GWL, Burgmans MC, Zhuang KD, Quek LHH, Kwan J, Damodharan K, Gogna A, Tan BP, Too CW, Chan SXJM, Chng SP, Yuan W, Tan BS. Randomized Controlled Trial Comparing Drug-coated Balloon Angioplasty versus Conventional Balloon Angioplasty for Treating Below-the-Knee Arteries in Critical Limb Ischemia: The SINGA-PACLI Trial. Radiology. 2021 Sep;300(3):715-724. doi: 10.1148/radiol.2021204294. Epub 2021 Jul 6. PMID: 34227886.
- Spreen MI, Martens JM, Hansen BE, Knippenberg B, Verhey E, van Dijk LC, de Vries JP, Vos JA, de Borst GJ, Vonken EJ, Wever JJ, Statius van Eps RG, Mali WP, van Overhagen H. Percutaneous Transluminal Angioplasty and Drug-Eluting Stents for Infrapopliteal Lesions in Critical Limb Ischemia (PADI) Trial. Circ Cardiovasc Interv. 2016 Feb;9(2):e002376. doi: 10.1161/CIRCINTERVENTIONS.114.002376. PMID: 26861113; PMCID: PMC4753788.
- Spreen MI, Martens JM, Knippenberg B, van Dijk LC, de Vries JPM, Vos JA, de Borst GJ, Vonken EPA, Bijlstra OD, Wever JJ, Statius van Eps RG, Mali WPTM, van Overhagen H. Long-Term Follow-up of the PADI Trial: Percutaneous Transluminal Angioplasty Versus Drug-Eluting Stents for Infrapopliteal Lesions in Critical Limb Ischemia. J Am Heart Assoc. 2017 Apr 14;6(4):e004877. doi: 10.1161/JAHA.116.004877. PMID: 28411244; PMCID: PMC5533004.
|
Study reference |
Study characteristics |
Patient characteristics |
Intervention (I) |
Comparison / control (C)
|
Follow-up |
Outcome measures and effect size |
Comments |
|
Ipema, 2020
PS., study characteristics and results are extracted from the SR (unless stated otherwise) |
SR and meta-analysis of RCTs, comparative studies and case series. Only RCTs are included in current analysis.
Literature search up to November 2018
A: Liistro, 2013 B: Zeller, 2014 C: Zeller, 2015 D: Haddad, 2017
Study design: RCT
Setting and Country: The Netherlands
Source of funding and conflicts of interest: CONFLICT OF INTEREST None. FUNDING None
|
Inclusion criteria SR: Articles were eligible if they included DCB angioplasty or compared DCB angioplasty with standard PTA of infrapopliteal arteries in patients with peripheral arterial disease, were published in English, included human subjects, and had a full text available.
Exclusion criteria SR: case reports, articles with fewer than 50 infrapopliteal angioplasties, the use of other types of balloons or stents, cutting balloon, cryoplasty, or laser technique, reviews, commentaries, letters to the editor, or conference abstracts.
4 RCTs included
Important patient characteristics at baseline: N, mean age A: 132, 74/75y B: 358, 73/71y C: 72, 73/70y D: 93, NR
Sex (% male) A: 80.3 B: 74.3 C: 79.1 D: NR
Diabetes mellitus A: 100% B: 73.5% C: 66.7% D: 95.7%
Range of Rutherford category A: 4-6 B: 3-6 C: 2-5 D: 4-6
A: Liistro B: Zeller C: Zeller D: Haddad
Groups were comparable at baseline |
Describe intervention:
A: DCB B: DCB C: DCB D: DCB
|
Describe control:
A: PTA B: PTA C: PTA D: PTA
|
End-point of follow-up: For review: 12 months
For how many participants were no complete outcome data available? Numbers specified under outcomes
|
Restenosis >50% recurrent stenosis on duplex ultrasound or angiography, or a peak systolic velocity rate ≥2.5 on duplex ultrasound, at 12 months A: I: 20/74; C: 55/74 B: I: 25/61; C: 11/31 C: - D: I: 14/40; C: 34/41
Target lesion revascularization A clinically driven repeat percutaneous intervention of the target lesion or bypass surgery of the target vessel, at 12 months A: - B: I: 27/226; C: 15/111 C: I: 12/40; C: 15/49 D: I: 9/54; C: 29/52
Amputation Limb salvage (avoidance of major amputation) at 12 months A: I: 65/65; C: 66/67 B: I: 207/227; C: 107/111 C: I: 29/30; C: 34/36 D: I: 47/48; C: 43/45
Mortality Overall survival at 12 months A: I: 60/65; C: 64/67 B: I: 204/227; C: 102/111 C: I: 33/36; C: 34/36 D: I: 38/48; C: 39/48
Quality of life Not reported
Wound healing Not reported
|
Risk of bias (high, some concerns or low): Tool used by authors: Cochrane tool for assessing risk of bias
A: unclear for blinding outcome assessment, high for blinding participants and personnel B: unclear for incomplete outcome data and other bias, high for selective reporting and blinding participants and personnel C: unclear for blinding participants and personnel and for blinding outcome assessment D: unclear for random sequence generation, allocation concealment, blinding participants and personnel and for blinding outcome assessment
Author’s conclusion In patients with peripheral arterial disease who underwent infrapopliteal angioplasty, no significant differences in limb salvage, survival, restenosis, TLR, and AFS rates were found when DCB angioplasty was compared with standard PTA.
|
Risk of bias tabel
|
Study reference
(first author, publication year) |
Was the allocation sequence adequately generated?
Definitely yes Probably yes Probably no Definitely no |
Was the allocation adequately concealed?
Definitely yes Probably yes Probably no Definitely no |
Blinding: Was knowledge of the allocated interventions adequately prevented?
Were patients blinded?
Were healthcare providers blinded?
Were data collectors blinded?
Were outcome assessors blinded?
Were data analysts blinded?
Definitely yes Probably yes Probably no Definitely no |
Was loss to follow-up (missing outcome data) infrequent?
Definitely yes Probably yes Probably no Definitely no |
Are reports of the study free of selective outcome reporting?
Definitely yes Probably yes Probably no Definitely no |
Was the study apparently free of other problems that could put it at a risk of bias?
Definitely yes Probably yes Probably no Definitely no |
Overall risk of bias If applicable/necessary, per outcome measure
LOW Some concerns HIGH
|
|
Fanelli, 2014 |
Definitely yes;
Reason: Patients were randomized (1:1) without stratification using computer-generated assignments when they entered the angiographic suite. |
Probably yes;
Reason: not specified |
Probably no;
Reason: health care providers were not blinded, but patients and outcome assessors were blinded. |
Definitely yes;
Reason: No loss to follow-up reported. |
Probably yes;
Reason: All relevant outcomes were reported |
Probably yes;
Reason: No other problems noted |
Some concerns for CD-TLR
|
|
Jia, 2020 |
Definitely yes;
Reason: All patients were randomly assigned to the DCB or control group (1:1 ratio) by a central randomization computer system. |
Definitely yes;
Reason: Opaque, sealed envelopes were used and opened by an independent researcher. |
Definitely no;
Reason: No specific measures were taken to blind the person who performed the repeat angiography or who made the decision about CD-TLR, whereas quantitative evaluation of the angiograms was performed by a core laboratory blinded to the type of treatment provided. |
Definitely yes;
Reason: Loss to follow-up was infrequent in intervention and control group. |
Probably yes;
Reason: All relevant outcomes were reported |
Probably yes;
Reason: No other problems noted |
Some concerns for CD-TLR
|
|
Liistro, 2020 |
Definitely yes;
Reason: Target lesions were randomly assigned to 1 of the 2 study arms after optimal angioplasty. Randomization was performed in 2 parallel blocks of 10 with the use of computer-generated random digits. |
Probably yes;
Reason: Prior to randomization, single or sequential balloon dilatations were performed to reach an optimal angiographic result. In case of randomization to POBA, the procedure ended with final angiography. |
Definitely no;
Reason: patients were blinded, but health care providers were not. Outcomes of interest are not likely affected by lack of blinding, except CD-TLR |
Definitely yes;
Reason: Loss to follow-up was infrequent in intervention and control group. |
Probably yes;
Reason: All relevant outcomes were reported |
Probably yes;
Reason: No other problems noted |
Some concerns for CD-TLR
|
|
Liistro, 2022 IN.PACT BTK |
Probably yes;
Reason: mentioned but not specified. |
Probably yes;
Reason: mentioned but not specified. |
Definitely no;
Due to the nature of the procedure, it was not possible to blind the operator or study site staff. In addition, participants were not blinded due to inherent differences in procedure between the DCB and PTA groups |
Definitely yes;
Reason: Loss to follow-up was infrequent in intervention and control group. |
Probably yes;
Reason: All relevant outcomes were reported |
Probably yes;
Reason: No other problems noted |
Some concerns for CD-TLR
|
|
Liistro, 2022 DEBATE-BTK |
Definitely yes;
Lesions were randomly assigned to 1 of the 2 study arms after successful passage of the guidewire. Randomization was performed in blocks of 10 with the use of computer-generated random digits. |
Definitely yes;
Group assignments were placed in sealed envelopes. |
Definitely no;
The study was not blinded. |
No information |
Probably yes;
Reason: All relevant outcomes were reported |
Probably yes;
Reason: No other problems noted |
Some concerns for CD-TLR
|
|
Patel, 2021 SINGA-PACLI |
Definitely yes;
The randomization sequence was computer generated by our academic collaborator (Singapore Clinical Research Institute), with permuted block lengths of four and six, and stratification by diabetes mellitus, end-stage renal failure status, and study site. |
Definitely yes;
To conceal treatment allocation, we used tamper-proof serially numbered opaque envelopes accessible only to the operator performing the procedure. |
Definitely no;
Participants and study personnel who performed the follow-up imaging (duplex US and angiography) assessments were blinded to the treatment assignment. Clinicians deciding revascularization were not blinded. |
Probably no;
For later time points, loss to follow-up was considerable and different between intervention and control group (20 vs 10%) |
Probably yes;
Reason: All relevant outcomes were reported |
Probably yes;
Reason: No other problems noted |
Some concerns for CD-TLR and outcomes at 12 months |
|
Bosiers, 2020 DESTINY |
Definitely yes;
Block randomization and stratification per center were conducted using block sizes of eight, which resulted in slightly unequal study populations. |
Definitely yes;
After successful wire traversal, patients were randomized 1:1 using preprovided sealed envelopes. |
Definitely no;
Patients were blinded to their treatment assignment, and study site personnel were counseled on nondisclosure. The physician performing the procedure was not blinded to the assigned treatment. |
Probably no;
Loss to follow-up up to one year was limited for clinical outcomes, but high for outcomes based on imaging. |
Probably yes;
Reason: All relevant outcomes were reported |
Probably yes;
Reason: No other problems noted |
Some concerns for CD-TLR and patency at 12 months |
|
Falkowski, 2008 |
Definitely yes;
Patients were randomly divided into two groups using statistica StatSoft |
No information |
Definitely no;
Open-label study |
Probably yes;
Loss to follow-up up was infrequent |
Probably yes;
Reason: All relevant outcomes were reported |
Probably yes;
Reason: No other problems noted |
Some concerns for CD-TLR
|
|
Rastan, 2011 Rastan, 2021 |
Definitely yes;
We allocated patients to the two treatment groups using a computer-generated random sequence, set in blocks for each participating centre. Patients were randomly assigned to the groups in a 1:1 ratio. |
Probably yes;
Not specified, but likely. |
Definitely yes;
Physicians and patients were unaware of the treatment-group assignment. All clinical endpoints were adjudicated by an independent clinical-events committee. All physicians including the medical staffs, the research coordinators, and the clinical-events committee were blinded for the treatment-group assignments. |
Probably no;
Loss to follow-up up was limited for clinical outcomes, but high for outcomes based on imaging. |
Probably yes;
Reason: All relevant outcomes were reported |
Probably yes;
Reason: No other problems noted |
Some concerns for patency
|
|
Spreen, 2016 Spreen, 2017 PADI |
Definitely yes;
Computer-generated random sequence on a 1:1 basis. andomization was per limb and stratified in blocks per center. |
Definitely yes;
The attending radiological technician opened the sealed, opaque envelope. The block size (n=4) was known only to the statistician. |
Definitely no;
Patients, operators, and investigators were not blinded |
Probably yes;
Reason: Loss to follow-up up was infrequent |
Probably yes;
Reason: All relevant outcomes were reported |
Probably yes;
Reason: No other problems noted |
Some concerns for CD-TLR
|
Exclusie tabel
|
Reference |
Reason for exclusion |
|
Falkowski A, Poncyljusz W, Wilk G, Szczerbo-Trojanowska M. The evaluation of primary stenting of sirolimus-eluting versus bare-metal stents in the treatment of atherosclerotic lesions of crural arteries. Eur Radiol. 2009 Apr;19(4):966-74. doi: 10.1007/s00330-008-1225-1. Epub 2008 Nov 26. PMID: 19034460. |
Wrong population: majority patients with claudication |
|
Haddad SE, Shishani JM, Qtaish I, Rawashdeh MA, Qtaishat BS. One Year Primary Patency of Infrapopliteal Angioplasty Using Drug- Eluting Balloons: Single Center Experience at King Hussein Medical Center. J Clin Imaging Sci. 2017 Aug 3;7:31. doi: 10.4103/jcis.JCIS_34_17. PMID: 28852581; PMCID: PMC5559924. |
Data extracted from systematic review |
|
Rastan A, Brechtel K, Krankenberg H, Zahorsky R, Tepe G, Noory E, Schwarzwälder U, Macharzina R, Schwarz T, Bürgelin K, Sixt S, Tübler T, Neumann FJ, Zeller T. Sirolimus-eluting stents for treatment of infrapopliteal arteries reduce clinical event rate compared to bare-metal stents: long-term results from a randomized trial. J Am Coll Cardiol. 2012 Aug 14;60(7):587-91. doi: 10.1016/j.jacc.2012.04.035. PMID: 22878166. |
Wrong population: majority patients with claudication |
|
Rastan A, Tepe G, Krankenberg H, Zahorsky R, Beschorner U, Noory E, Sixt S, Schwarz T, Brechtel K, Böhme C, Neumann FJ, Zeller T. Sirolimus-eluting stents vs. bare-metal stents for treatment of focal lesions in infrapopliteal arteries: a double-blind, multi-centre, randomized clinical trial. Eur Heart J. 2011 Sep;32(18):2274-81. doi: 10.1093/eurheartj/ehr144. Epub 2011 May 26. PMID: 21622669. |
Wrong population: majority patients with claudication |
|
Bosiers MJ, Deloose K, Peeters P, Torsello G, Zeller T, Scheinert D, Schmidt A, Maene L, Keirse K, Varcoe R, Bosiers M. Outcome of a drug-eluting stent in longer below-the-knee lesions in patients with critical limb ischemia. J Cardiovasc Surg (Torino). 2017 Feb;58(1):49-54. doi: 10.23736/S0021-9509.16.09546-X. Epub 2016 Jul 26. PMID: 27455888. |
Wrong study design |
|
Fanelli F, Cannavale A, Corona M, Lucatelli P, Wlderk A, Salvatori FM. The "DEBELLUM"--lower limb multilevel treatment with drug eluting balloon--randomized trial: 1-year results. J Cardiovasc Surg (Torino). 2014 Apr;55(2):207-16. PMID: 24670828. |
Wrong population: femoro-popliteal region |
|
Katsanos K, Spiliopoulos S, Diamantopoulos A, Siablis D, Karnabatidis D, Scheinert D. Wound Healing Outcomes and Health-Related Quality-of-Life Changes in the ACHILLES Trial: 1-Year Results From a Prospective Randomized Controlled Trial of Infrapopliteal Balloon Angioplasty Versus Sirolimus-Eluting Stenting in Patients With Ischemic Peripheral Arterial Disease. JACC Cardiovasc Interv. 2016 Feb 8;9(3):259-267. doi: 10.1016/j.jcin.2015.10.038. Epub 2016 Jan 6. PMID: 26777329. |
Wrong comparison |
|
Konijn LCD, Wakkie T, Spreen MI, de Jong PA, van Dijk LC, Wever JJ, Veger HTC, Statius van Eps RG, Mali WPTM, van Overhagen H. 10-Year Paclitaxel Dose-Related Outcomes of Drug-Eluting Stents Treated Below the Knee in Patients with Chronic Limb-Threatening Ischemia (The PADI Trial). Cardiovasc Intervent Radiol. 2020 Dec;43(12):1881-1888. doi: 10.1007/s00270-020-02602-6. Epub 2020 Jul 28. PMID: 32725411; PMCID: PMC7649154. |
Wrong study design |
|
Scheinert D, Katsanos K, Zeller T, Koppensteiner R, Commeau P, Bosiers M, Krankenberg H, Baumgartner I, Siablis D, Lammer J, Van Ransbeeck M, Qureshi AC, Stoll HP; ACHILLES Investigators. A prospective randomized multicenter comparison of balloon angioplasty and infrapopliteal stenting with the sirolimus-eluting stent in patients with ischemic peripheral arterial disease: 1-year results from the ACHILLES trial. J Am Coll Cardiol. 2012 Dec 4;60(22):2290-5. doi: 10.1016/j.jacc.2012.08.989. PMID: 23194941. |
Wrong comparison |
|
Sivapragasam N, Matchar DB, Zhuang KD, Patel A, Pua U, Win HH, Chandramohan S, Venkatanarasimha N, Chua JME, Tan GWL, Irani FG, Leong S, Tay KH, Chong TT, Tan BS. Cost-Effectiveness of Drug-Coated Balloon Angioplasty Versus Conventional Balloon Angioplasty for Treating Below-the-Knee Arteries in Chronic Limb-Threatening Ischemia: The SINGA-PACLI Trial. Cardiovasc Intervent Radiol. 2022 Nov;45(11):1663-1669. doi: 10.1007/s00270-022-03073-7. Epub 2022 Mar 2. PMID: 35237860. |
Wrong study design |
|
Zeller T, Micari A, Scheinert D, Baumgartner I, Bosiers M, Vermassen FEG, Banyai M, Shishehbor MH, Wang H, Brodmann M; IN.PACT DEEP Trial Investigators. The IN.PACT DEEP Clinical Drug-Coated Balloon Trial: 5-Year Outcomes. JACC Cardiovasc Interv. 2020 Feb 24;13(4):431-443. doi: 10.1016/j.jcin.2019.10.059. PMID: 32081236. |
Wrong study design |
|
Zeller T, Beschorner U, Pilger E, Bosiers M, Deloose K, Peeters P, Scheinert D, Schulte KL, Rastan A, Brodmann M. Paclitaxel-Coated Balloon in Infrapopliteal Arteries: 12-Month Results From the BIOLUX P-II Randomized Trial (BIOTRONIK'S-First in Man study of the Passeo-18 LUX drug releasing PTA Balloon Catheter vs. the uncoated Passeo-18 PTA balloon catheter in subjects requiring revascularization of infrapopliteal arteries). JACC Cardiovasc Interv. 2015 Oct;8(12):1614-22. doi: 10.1016/j.jcin.2015.07.011. PMID: 26493253. |
Data extracted from systematic review |
|
Zeller T, Baumgartner I, Scheinert D, Brodmann M, Bosiers M, Micari A, Peeters P, Vermassen F, Landini M, Snead DB, Kent KC, Rocha-Singh KJ; IN.PACT DEEP Trial Investigators. Drug-eluting balloon versus standard balloon angioplasty for infrapopliteal arterial revascularization in critical limb ischemia: 12-month results from the IN.PACT DEEP randomized trial. J Am Coll Cardiol. 2014 Oct 14;64(15):1568-76. doi: 10.1016/j.jacc.2014.06.1198. PMID: 25301459. |
Data extracted from systematic review |
|
Cassese S, Ndrepepa G, Liistro F, Fanelli F, Kufner S, Ott I, Laugwitz KL, Schunkert H, Kastrati A, Fusaro M. Drug-Coated Balloons for Revascularization of Infrapopliteal Arteries: A Meta-Analysis of Randomized Trials. JACC Cardiovasc Interv. 2016 May 23;9(10):1072-80. doi: 10.1016/j.jcin.2016.02.011. Epub 2016 Apr 27. PMID: 27131439. |
More recent systematic review used |
|
Chen X, Li J, Zheng C, He Y, Jia J, Wang X, Li D, Shang T, Li M. Drug-delivering endovascular treatment versus angioplasty in artery occlusion diseases: a systematic review and meta-analysis. Curr Med Res Opin. 2018 Jan;34(1):95-105. doi: 10.1080/03007995.2017.1372114. Epub 2017 Nov 6. PMID: 28837370. |
More recent systematic review used |
|
Matsuoka EK, Hasebe T, Ishii R, Miyazaki N, Soejima K, Iwasaki K. Comparative performance analysis of interventional devices for the treatment of ischemic disease in below-the-knee lesions: a systematic review and meta-analysis. Cardiovasc Interv Ther. 2022 Jan;37(1):145-157. doi: 10.1007/s12928-021-00758-7. Epub 2021 Feb 6. PMID: 33547627; PMCID: PMC8789697. |
More complete systematic review used |
Beoordelingsdatum en geldigheid
Publicatiedatum : 02-04-2025
Beoordeeld op geldigheid : 01-04-2025
Algemene gegevens
De ontwikkeling/herziening van deze richtlijnmodule werd ondersteund door het Kennisinstituut van de Federatie Medisch Specialisten (www.demedischspecialist.nl/kennisinstituut) en werd gefinancierd uit de Kwaliteitsgelden Medisch Specialisten (SKMS).
De financier heeft geen enkele invloed gehad op de inhoud van de richtlijnmodule.
Samenstelling werkgroep
Voor het ontwikkelen van de richtlijnmodule is in 2021 een multidisciplinaire werkgroep ingesteld, bestaande uit vertegenwoordigers van alle relevante specialismen (zie hiervoor de Samenstelling van de werkgroep) die betrokken zijn bij de zorg rondom patiënten met (chronisch) perifeer arterieel vaatlijden van de onderste extremiteit(en).
Werkgroep
- Dr. B. (Bram) Fioole (voorzitter), Nederlandse Vereniging voor Heelkunde
- Dr. C. (Çagdas) Ünlü, Nederlandse Vereniging voor Heelkunde
- Drs. J.L. (Jorg) de Bruin, Nederlandse Vereniging voor Heelkunde
- Prof. dr. B.M.E. (Barend) Mees, Nederlandse Vereniging voor Heelkunde
- Drs. D.A.F. (Daniel) van den Heuvel, Nederlandse Vereniging voor Radiologie
- Dr. S.W. (Sanne) de Boer, Nederlandse Vereniging voor Radiologie
- Dr. R. (Rinske) Loeffen), Nederlandse Internisten Vereniging
- Dr. M.E.L. (Marie-Louise) Bartelink, Nederlands Huisartsen Genootschap
- E. (Emilien) Wegerif, Nederlandse Vereniging voor Heelkunde
- J. (Jenny) Zwiggelaar, Koninklijk Nederlands Genootschap voor Fysiotherapie / Vereniging van Oefentherapeuten Cesar en Mensendieck
- G. (Gilaine) Kleian, Harteraad
- P.A.H. (Patricia) van Mierlo – van den Broek, MANP, Verpleegkundigen & Verzorgenden Nederland / VS Vaatchirurgie Netwerk
Met ondersteuning van:
- Dr. W. (Wouter) Harmsen, senior adviseur, Kennisinstituut van de Federatie Medisch Specialisten (tot 2023)
- Dr. R. (Romy) Zwarts - van de Putte, adviseur, Kennisinstituut van de Federatie Medisch Specialisten (tot 2023)
- Dr. M.S. (Matthijs) Ruiter, senior adviseur, Kennisinstituut van de Federatie Medisch Specialisten (vanaf 2023)
- M. (Mitchel) Griekspoor, MSc, adviseur, Kennisinstituut van de Federatie Medisch Specialisten (vanaf 2023)
Belangenverklaringen
De Code ter voorkoming van oneigenlijke beïnvloeding door belangenverstrengeling is gevolgd. Alle werkgroepleden hebben schriftelijk verklaard of zij in de laatste drie jaar directe financiële belangen (betrekking bij een commercieel bedrijf, persoonlijke financiële belangen, onderzoeksfinanciering) of indirecte belangen (persoonlijke relaties, reputatiemanagement) hebben gehad. Gedurende de ontwikkeling of herziening van een module worden wijzigingen in belangen aan de voorzitter doorgegeven. De belangenverklaring wordt opnieuw bevestigd tijdens de commentaarfase.
Een overzicht van de belangen van werkgroepleden en het oordeel over het omgaan met eventuele belangen vindt u in onderstaande tabel. De ondertekende belangenverklaringen zijn op te vragen bij het secretariaat van het Kennisinstituut van de Federatie Medisch Specialisten.
|
Werkgroeplid |
Hoofdfunctie |
Project Tekstveld |
Nevenwerkzaamheden |
Persoonlijke Financiële belangen |
Persoonlijke relaties |
Extern gefinancierd onderzoek |
Intell. Belangen en reputatie |
Overige belangen |
Ondernomen actie |
|
Bram Fioole (vz.) |
Vaatchirurg |
Richtlijn Perifeer arterieel vaatlijden |
Opleider vaatchirurgie en algemene heelkunde Maasstadziekenhuis, onbetaald
Secretaris Stichting DEAll (stichting ter bevordering van wetenschappelijk onderzoek op het gebied van de vaatchirurgie en interventieradiologie), onbetaald.
|
Geen. |
Geen. |
Stichting DEAll/FOREST trial/projectleider-> ja Getinge/DISCOVER trial/projectleider-> ja Cook/Zephyr registry/ projectleider -> ja |
Geen. |
Geen. |
Geen restricties voor deelname aan de richtlijn. |
|
Marie-Louise Bartelink |
* 80% huisartsdocent en -onderzoeker, associate professor * 20% huisartsdocent
|
Richtlijn Perifeer arterieel vaatlijden |
Geen. |
Geen. |
Geen. |
ZonMw - EBM-leren in de huisartspraktijk, kwalittatieve studies - Projectleider |
Geen. |
Geen. |
Geen restricties voor deelname aan de richtlijn. |
|
Patricia van Mierlo – van den Broek |
Verpleegkundig Specialist in het specialisme Algemene Gezondheidszorg werkzaam bij de afdeling Vaatchirurgie.
|
Richtlijn Perifeer arterieel vaatlijden |
Als vrijwilliger werkzaam (onbetaald) als collectant voor een aantal goede doelen (KWF en dierenbescherming) |
Geen. |
Geen. |
Betrokken bij de "FOREST"-trial (randomized comparison of FemORal drug-Eluting balloons and Stents), een gerandomiseerd prospectief multicenter onderzoek waarbij de langere termijn resultaten van drug-eluting ballonnen en stents in het AFS traject onderzocht wordt (NL57055.101.16). Deze trial wordt gesponsord door de stichting DEAII (Dutch Endovascular Alliance).
|
Geen. |
Geen. |
Geen restricties voor deelname aan de richtlijn. |
|
Emilien Wegerif |
Arts onderzoeker |
Richtlijn Perifeer arterieel vaatlijden
|
Eigenaresse Caro-Jo Amsterdam, eenmanszaak betaald Caro-Jo Amsterdam is een Chaar sierraden merk
|
Ik heb geen financiële belangen bij de uitkomsten van de publicaties |
Geen. |
Geen. |
Geen. |
Geen. |
Geen restricties voor deelname aan de richtlijn. |
|
Jenny Zwiggelaar |
*Fysiotherapeut, hart-vaat-long gespecialiseerd
*Projectmedewerker Chronisch ZorgNet
Beide functies in loondienst |
Richtlijn Perifeer arterieel vaatlijden
|
Geen. |
Geen, enkel de bekende werkrelaties. |
Geen. |
Geen. |
Geen persoonlijk belangen of reputatiegewin. Ik treed in deze werkgroep als afgevaardigde van de beroepsgroep KNGF en zal enkel de kennis vanuit ChronischZorgnet (voorheen ClaudicatioNet) meebrengen.
|
Chronisch ZorgNet is op de hoogte en ondersteund de inbreng in deze werkgroep. Vanuit KNGF is de duidelijke opdracht om alle fysiotherapeuten met de specialisatie PAV te vertegenwoordigen. Dit zal naar verwachting ook beter zichtbaar worden bij het invoeren van het aantekenregister Vaat (als onderdeel van deelregistratie hart-vaat-long-fysiotherapeut). Er is een open en prettige communicatie onderling en geen belangenverstrengeling.
|
Geen restricties voor deelname aan de richtlijn. |
|
Rinske Loeffen |
Internist vasculair geneeskundige |
Richtlijn Perifeer arterieel vaatlijden
|
* Redactielid Focus Vasculair (onbetaald) * Werkgroep Tabaksontmoediging NVALT (onbetaald) * Werkgroep SKMS project Stop met Roken Zorg (onbetaald) * Werkgroep Acute boekje vasculaire geneeskunde (onbetaald).
|
Geen. |
Geen. |
Geen. |
Geen. |
Geen. |
Geen restricties voor deelname aan de richtlijn. |
|
Sanne de Boer |
Interventieradioloog |
Richtlijn Perifeer arterieel vaatlijden |
* Verschillende wetenschappelijke en beleidscommissies binnen de NVIR (onbetaald) * Institutioneel contract met Brainlab voor consultant werkzaamheden * Institutioneel contract met MUMC/CTCM * Incidenteel consultant werkzaamheden voor Philips, ook via institutioneel contract.
|
Zie nevenwerkzaamheden. |
Geen. |
|
Geen. |
Geen. |
Geen restricties. Onderwerp van advieswerk valt buiten de afbakening van de richtlijn. |
|
Çagdas Ünlü |
Vaatchirurg |
Richtlijn Perifeer arterieel vaatlijden
|
Projectleider CLEARPATH, studie na antistolling na endovasculaire behandeling ZonMw subsidie |
Geen. |
Geen. |
ZonMw (Clearpath) |
Geen. |
Geen. |
Geen restricties voor deelname aan de richtlijn. |
|
Daniel van den Heuvel |
Interventie Radioloog |
Richtlijn Perifeer arterieel vaatlijden
|
Proctor voor bedrijf LimFlow SA, Parijs. Betaald. Adjudicator voor Bioreact studie, Biotronic. Betaald. |
Zie nevenwerkzaamheden. Verder geen persoonlijke financiële belangen. |
Geen. |
Philips/Illumenate BTK PMS. Effectiviteit en veiligheid van de Stellarex .014 Ballon bij patiënten met kritieke ischemie ->ja. Micromedical/Heal study, PMS. Effectiviteit en veiligheid van de Microstent bij patiënten met kritieke ischemie -> ja. Philips/REPEAT study. Reproduceerbaarheid van 2D perfusie angiografie-> ja. |
Geen. |
Geen. |
Geen restricties voor deelname aan de richtlijn. |
|
Jorg de Bruin |
Vaatchirurg |
Richtlijn Perifeer arterieel vaatlijden
|
EMC; Klinisch en wetenschappelijk werk vaatchirurgie in academisch ziekenhuis |
Geen conflict of specifiek belang. |
Geen. |
Geen. |
Geen. |
Geen. |
Geen restricties voor deelname aan de richtlijn. |
|
Ilse Verstraaten |
Beleidsadviseur Harteraard (betaald) |
Richtlijn Perifeer arterieel vaatlijden
|
Geen. |
Geen. |
Geen. |
Geen. |
Geen. |
Geen. |
Geen restricties voor deelname aan de richtlijn.
|
|
Gilaine Kleian |
Junior projectmedewerker, Harteraad |
Richtlijn Perifeer arterieel vaatlijden
|
Geen. |
Geen. |
Geen. |
Geen. |
Geen. |
Geen. |
Geen restricties voor deelname aan de richtlijn.
|
|
Barend Mees |
Vaatchirurg |
Richtlijn Perifeer arterieel vaatlijden
|
* Secretaris bestuur Nederlandse Vereniging voor Vaatchirurgie * Lid Commissie Richtlijnen NVvH * Council Member European Society for Vascular Surgery * Member ESVS Steering Guideline Committee * Director European Vascular Course. * Bestuur Stichting Drie Lichten * Lid Onderzoekspijler Dutch Cardiovascular Alliance * Lid adviesraad Marfan patienten * Consultancy werkzaamheden voor Philips, Bentley Innomed. |
Geen. |
Geen. |
* Regmed XB: Cardiovascular Moonshot (Moonshot leader) * EU-Horizon 2020: PAPA-ARTIS (local PI) * InScIte: XS-graft (onderzoeksleider) * Philips: SAVER-registry (National PI) * ID3: Supersurg trial (local PI) * Cook: Zephyr registry (local PI) * Philips: FORS registry (local co-PI) * ZonMw: GENPAD study (local PI) |
*Ontwikkelaar van de mazeBox, een training tool voor endovasculaire technieken. |
Geen. |
Geen restricties voor deelname aan richtlijn. Genoemde onderzoek valt buiten de afbakening van de richtlijn. |
Inbreng patiëntenperspectief
Er werd aandacht besteed aan het patiëntenperspectief door het uitnodigen van Harteraad voor de schriftelijke knelpuntenanalyse. Het verslag hiervan is besproken in de werkgroep. De verkregen input is meegenomen bij het opstellen van de uitgangsvragen, de keuze voor de uitkomstmaten en bij het opstellen van de overwegingen. De conceptrichtlijn is tevens voor commentaar voorgelegd aan de deelnemers van de schriftelijke knelpuntenanalyse en de eventueel aangeleverde commentaren zijn bekeken en verwerkt.
Kwalitatieve raming van mogelijke financiële gevolgen in het kader van de Wkkgz
Bij de richtlijnmodule is conform de Wet kwaliteit, klachten en geschillen zorg (Wkkgz) een kwalitatieve raming uitgevoerd om te beoordelen of de aanbevelingen mogelijk leiden tot substantiële financiële gevolgen. Bij het uitvoeren van deze beoordeling is de richtlijnmodule op verschillende domeinen getoetst.
|
Module |
Uitkomst raming |
Toelichting |
|
Module: Infragenuaal: Gecoate ballonnen en stents |
Geen substantiële financiële gevolgen |
Uit de toetsing volgt dat de aanbevelingen mogelijk een kostenbesparend effect hebben door slechts op indicatie de duurdere behandeloptie te overwegen. |
Werkwijze
AGREE
Deze richtlijnmodule is opgesteld conform de eisen vermeld in het rapport Medisch Specialistische Richtlijnen 2.0 van de adviescommissie Richtlijnen van de Raad Kwaliteit. Dit rapport is gebaseerd op het AGREE II instrument (Appraisal of Guidelines for Research & Evaluation II; Brouwers, 2010).
Knelpuntenanalyse en uitgangsvragen
Tijdens de voorbereidende fase inventariseerde de werkgroep de knelpunten in de zorg voor patiënten met (chronisch) perifeer arterieel vaatlijden van de onderste extremiteit(en). Tevens zijn er knelpunten aangedragen via een schriftelijke knelpuntenanalyse. Een verslag hiervan is opgenomen onder aanverwante producten.
Op basis van de uitkomsten van de knelpuntenanalyse zijn door de werkgroep concept-uitgangsvragen opgesteld en definitief vastgesteld.
Uitkomstmaten
Na het opstellen van de zoekvraag behorende bij de uitgangsvraag inventariseerde de werkgroep welke uitkomstmaten voor de patiënt relevant zijn, waarbij zowel naar gewenste als ongewenste effecten werd gekeken. Hierbij werd een maximum van acht uitkomstmaten gehanteerd. De werkgroep waardeerde deze uitkomstmaten volgens hun relatieve belang bij de besluitvorming rondom aanbevelingen, als cruciaal (kritiek voor de besluitvorming), belangrijk (maar niet cruciaal) en onbelangrijk. Tevens definieerde de werkgroep tenminste voor de cruciale uitkomstmaten welke verschillen zij klinisch (patiënt) relevant vonden.
Methode literatuursamenvatting
Een uitgebreide beschrijving van de strategie voor zoeken en selecteren van literatuur is te vinden onder ‘Zoeken en selecteren’ onder Onderbouwing. Indien mogelijk werd de data uit verschillende studies gepoold in een random-effects model. [Review Manager 5.4] werd gebruikt voor de statistische analyses. De beoordeling van de kracht van het wetenschappelijke bewijs wordt hieronder toegelicht.
Beoordelen van de kracht van het wetenschappelijke bewijs
De kracht van het wetenschappelijke bewijs werd bepaald volgens de GRADE-methode. GRADE staat voor ‘Grading Recommendations Assessment, Development and Evaluation’ (zie http://www.gradeworkinggroup.org/). De basisprincipes van de GRADE-methodiek zijn: het benoemen en prioriteren van de klinisch (patiënt) relevante uitkomstmaten, een systematische review per uitkomstmaat, en een beoordeling van de bewijskracht per uitkomstmaat op basis van de acht GRADE-domeinen (domeinen voor downgraden: risk of bias, inconsistentie, indirectheid, imprecisie, en publicatiebias; domeinen voor upgraden: dosis-effect relatie, groot effect, en residuele plausibele confounding).
GRADE onderscheidt vier gradaties voor de kwaliteit van het wetenschappelijk bewijs: hoog, redelijk, laag en zeer laag. Deze gradaties verwijzen naar de mate van zekerheid die er bestaat over de literatuurconclusie, in het bijzonder de mate van zekerheid dat de literatuurconclusie de aanbeveling adequaat ondersteunt (Schünemann, 2013; Hultcrantz, 2017).
|
GRADE |
Definitie |
|
Hoog |
|
|
Redelijk |
|
|
Laag |
|
|
Zeer laag |
|
Bij het beoordelen (graderen) van de kracht van het wetenschappelijk bewijs in richtlijnen volgens de GRADE-methodiek spelen grenzen voor klinische besluitvorming een belangrijke rol (Hultcrantz, 2017). Dit zijn de grenzen die bij overschrijding aanleiding zouden geven tot een aanpassing van de aanbeveling. Om de grenzen voor klinische besluitvorming te bepalen moeten alle relevante uitkomstmaten en overwegingen worden meegewogen. De grenzen voor klinische besluitvorming zijn daarmee niet één op één vergelijkbaar met het minimaal klinisch relevant verschil (Minimal Clinically Important Difference, MCID). Met name in situaties waarin een interventie geen belangrijke nadelen heeft en de kosten relatief laag zijn, kan de grens voor klinische besluitvorming met betrekking tot de effectiviteit van de interventie bij een lagere waarde (dichter bij het nuleffect) liggen dan de MCID (Hultcrantz, 2017).
Overwegingen (van bewijs naar aanbeveling)
Om te komen tot een aanbeveling zijn naast (de kwaliteit van) het wetenschappelijke bewijs ook andere aspecten belangrijk en worden meegewogen, zoals aanvullende argumenten uit bijvoorbeeld de biomechanica of fysiologie, waarden en voorkeuren van patiënten, kosten (middelenbeslag), aanvaardbaarheid, haalbaarheid en implementatie. Deze aspecten zijn systematisch vermeld en beoordeeld (gewogen) onder het kopje ‘Overwegingen’ en kunnen (mede) gebaseerd zijn op expert opinion. Hierbij is gebruik gemaakt van een gestructureerd format gebaseerd op het evidence-to-decision framework van de internationale GRADE Working Group (Alonso-Coello, 2016a; Alonso-Coello 2016b). Dit evidence-to-decision framework is een integraal onderdeel van de GRADE methodiek.
Formuleren van aanbevelingen
De aanbevelingen geven antwoord op de uitgangsvraag en zijn gebaseerd op het beschikbare wetenschappelijke bewijs en de belangrijkste overwegingen, en een weging van de gunstige en ongunstige effecten van de relevante interventies. De kracht van het wetenschappelijk bewijs en het gewicht dat door de werkgroep wordt toegekend aan de overwegingen, bepalen samen de sterkte van de aanbeveling. Conform de GRADE-methodiek sluit een lage bewijskracht van conclusies in de systematische literatuuranalyse een sterke aanbeveling niet a priori uit, en zijn bij een hoge bewijskracht ook zwakke aanbevelingen mogelijk (Agoritsas, 2017; Neumann, 2016). De sterkte van de aanbeveling wordt altijd bepaald door weging van alle relevante argumenten tezamen. De werkgroep heeft bij elke aanbeveling opgenomen hoe zij tot de richting en sterkte van de aanbeveling zijn gekomen.
In de GRADE-methodiek wordt onderscheid gemaakt tussen sterke en zwakke (of conditionele) aanbevelingen. De sterkte van een aanbeveling verwijst naar de mate van zekerheid dat de voordelen van de interventie opwegen tegen de nadelen (of vice versa), gezien over het hele spectrum van patiënten waarvoor de aanbeveling is bedoeld. De sterkte van een aanbeveling heeft duidelijke implicaties voor patiënten, behandelaars en beleidsmakers (zie onderstaande tabel). Een aanbeveling is geen dictaat, zelfs een sterke aanbeveling gebaseerd op bewijs van hoge kwaliteit (GRADE gradering HOOG) zal niet altijd van toepassing zijn, onder alle mogelijke omstandigheden en voor elke individuele patiënt.
|
Implicaties van sterke en zwakke aanbevelingen voor verschillende richtlijngebruikers |
||
|
|
||
|
|
Sterke aanbeveling |
Zwakke (conditionele) aanbeveling |
|
Voor patiënten |
De meeste patiënten zouden de aanbevolen interventie of aanpak kiezen en slechts een klein aantal niet. |
Een aanzienlijk deel van de patiënten zouden de aanbevolen interventie of aanpak kiezen, maar veel patiënten ook niet. |
|
Voor behandelaars |
De meeste patiënten zouden de aanbevolen interventie of aanpak moeten ontvangen. |
Er zijn meerdere geschikte interventies of aanpakken. De patiënt moet worden ondersteund bij de keuze voor de interventie of aanpak die het beste aansluit bij zijn of haar waarden en voorkeuren. |
|
Voor beleidsmakers |
De aanbevolen interventie of aanpak kan worden gezien als standaardbeleid. |
Beleidsbepaling vereist uitvoerige discussie met betrokkenheid van veel stakeholders. Er is een grotere kans op lokale beleidsverschillen. |
Organisatie van zorg
In de knelpuntenanalyse en bij de ontwikkeling van de richtlijnmodule is expliciet aandacht geweest voor de organisatie van zorg: alle aspecten die randvoorwaardelijk zijn voor het verlenen van zorg (zoals coördinatie, communicatie, (financiële) middelen, mankracht en infrastructuur). Randvoorwaarden die relevant zijn voor het beantwoorden van deze specifieke uitgangsvraag zijn genoemd bij de overwegingen. Meer algemene, overkoepelende, of bijkomende aspecten van de organisatie van zorg worden behandeld in module 14 en 15.
Commentaar- en autorisatiefase
De conceptrichtlijnmodule werd aan de betrokken (wetenschappelijke) verenigingen en (patiënt) organisaties voorgelegd ter commentaar. De commentaren werden verzameld en besproken met de werkgroep. Naar aanleiding van de commentaren werd de conceptrichtlijnmodule aangepast en definitief vastgesteld door de werkgroep. De definitieve richtlijnmodule werd aan de deelnemende (wetenschappelijke) verenigingen en (patiënt) organisaties voorgelegd voor autorisatie en door hen geautoriseerd dan wel geaccordeerd.
Literatuur
Agoritsas T, Merglen A, Heen AF, Kristiansen A, Neumann I, Brito JP, Brignardello-Petersen R, Alexander PE, Rind DM, Vandvik PO, Guyatt GH. UpToDate adherence to GRADE criteria for strong recommendations: an analytical survey. BMJ Open. 2017 Nov 16;7(11):e018593. doi: 10.1136/bmjopen-2017-018593. PubMed PMID: 29150475; PubMed Central PMCID: PMC5701989.
Alonso-Coello P, Schünemann HJ, Moberg J, Brignardello-Petersen R, Akl EA, Davoli M, Treweek S, Mustafa RA, Rada G, Rosenbaum S, Morelli A, Guyatt GH, Oxman AD; GRADE Working Group. GRADE Evidence to Decision (EtD) frameworks: a systematic and transparent approach to making well informed healthcare choices. 1: Introduction. BMJ. 2016 Jun 28;353:i2016. doi: 10.1136/bmj.i2016. PubMed PMID: 27353417.
Alonso-Coello P, Oxman AD, Moberg J, Brignardello-Petersen R, Akl EA, Davoli M, Treweek S, Mustafa RA, Vandvik PO, Meerpohl J, Guyatt GH, Schünemann HJ; GRADE Working Group. GRADE Evidence to Decision (EtD) frameworks: a systematic and transparent approach to making well informed healthcare choices. 2: Clinical practice guidelines. BMJ. 2016 Jun 30;353:i2089. doi: 10.1136/bmj.i2089. PubMed PMID: 27365494.
Brouwers MC, Kho ME, Browman GP, Burgers JS, Cluzeau F, Feder G, Fervers B, Graham ID, Grimshaw J, Hanna SE, Littlejohns P, Makarski J, Zitzelsberger L; AGREE Next Steps Consortium. AGREE II: advancing guideline development, reporting and evaluation in health care. CMAJ. 2010 Dec 14;182(18):E839-42. doi: 10.1503/cmaj.090449. Epub 2010 Jul 5. Review. PubMed PMID: 20603348; PubMed Central PMCID: PMC3001530.
Hultcrantz M, Rind D, Akl EA, Treweek S, Mustafa RA, Iorio A, Alper BS, Meerpohl JJ, Murad MH, Ansari MT, Katikireddi SV, Östlund P, Tranæus S, Christensen R, Gartlehner G, Brozek J, Izcovich A, Schünemann H, Guyatt G. The GRADE Working Group clarifies the construct of certainty of evidence. J Clin Epidemiol. 2017 Jul;87:4-13. doi: 10.1016/j.jclinepi.2017.05.006. Epub 2017 May 18. PubMed PMID: 28529184; PubMed Central PMCID: PMC6542664.
Medisch Specialistische Richtlijnen 2.0 (2012). Adviescommissie Richtlijnen van de Raad Kwalitieit. http://richtlijnendatabase.nl/over_deze_site/over_richtlijnontwikkeling.html
Neumann I, Santesso N, Akl EA, Rind DM, Vandvik PO, Alonso-Coello P, Agoritsas T, Mustafa RA, Alexander PE, Schünemann H, Guyatt GH. A guide for health professionals to interpret and use recommendations in guidelines developed with the GRADE approach. J Clin Epidemiol. 2016 Apr;72:45-55. doi: 10.1016/j.jclinepi.2015.11.017. Epub 2016 Jan 6. Review. PubMed PMID: 26772609.
Schünemann H, Brożek J, Guyatt G, et al. GRADE handbook for grading quality of evidence and strength of recommendations. Updated October 2013. The GRADE Working Group, 2013. Available from http://gdt.guidelinedevelopment.org/central_prod/_design/client/handbook/handbook.html.
Zoekverantwoording
Zoekverantwoording
Algemene informatie
|
Cluster/richtlijn: Perifeer arterieel vaatlijden - Infragenuaal: Gecoate ballonnen en stents |
|
|
Uitgangsvraag/modules: Wat is de optimale behandeling van het infragenuale traject? |
|
|
Database(s): Ovid/Medline, Embase.com |
Datum: 10 augustus 2022 |
|
Periode: 2000 - heden |
Talen: Engels, Nederlands |
|
Literatuurspecialist: Miriam van der Maten |
|
|
BMI-zoekblokken: voor verschillende opdrachten wordt (deels) gebruik gemaakt van de zoekblokken van BMI-Online https://blocks.bmi-online.nl/ Bij gebruikmaking van een volledig zoekblok zal naar de betreffende link op de website worden verwezen. |
|
|
Toelichting: Voor deze vraag is gezocht op de elementen:
De opgegeven sleutelartikelen worden gevonden met de search.
|
|
|
Te gebruiken voor richtlijnen tekst: Nederlands In de databases Embase.com en Ovid/Medline is op 10 augustus 2022 met relevante zoektermen gezocht naar systematische reviews, RCT en observationele studies over de optimale behandeling van het infragenuale traject. De literatuurzoekactie leverde 475 unieke treffers op.
Engels On the 10th of August, relevant search terms were used to search for systematic reviews, RCT and observational studies about the optimal treatment for patients with infragenual/infrapopliteal arterial occlusive disease in the databases Embase.com and Ovid/Medline. The search resulted in 475 unique hits. |
|
Zoekopbrengst
|
|
EMBASE |
OVID/MEDLINE |
Ontdubbeld |
|
SRs |
61 |
45 |
64 |
|
RCT |
89 |
101 |
114 |
|
Observationele studies |
151 |
233 |
297 |
|
Totaal |
301 |
379 |
475 |
Zoekstrategie
Embase.com
|
No. |
Query |
Results |
|
#21 |
#18 OR #19 OR #20 |
301 |
|
#20 |
#14 AND #17 NOT (#18 OR #19) = Observationeel |
151 |
|
#19 |
#14 AND #16 NOT #18 = RCT |
89 |
|
#18 |
#14 AND #15 = SR |
61 |
|
#17 |
'comparative study'/exp OR 'control group'/de OR 'controlled study'/de OR 'controlled clinical trial'/de OR 'crossover procedure'/de OR 'double blind procedure'/de OR 'phase 2 clinical trial'/de OR 'phase 3 clinical trial'/de OR 'phase 4 clinical trial'/de OR 'pretest posttest design'/de OR 'pretest posttest control group design'/de OR 'quasi experimental study'/de OR 'single blind procedure'/de OR 'triple blind procedure'/de OR (((control OR controlled) NEAR/6 trial):ti,ab,kw) OR (((control OR controlled) NEAR/6 (study OR studies)):ti,ab,kw) OR (((control OR controlled) NEAR/1 active):ti,ab,kw) OR 'open label*':ti,ab,kw OR (((double OR two OR three OR multi OR trial) NEAR/1 (arm OR arms)):ti,ab,kw) OR ((allocat* NEAR/10 (arm OR arms)):ti,ab,kw) OR placebo*:ti,ab,kw OR 'sham-control*':ti,ab,kw OR (((single OR double OR triple OR assessor) NEAR/1 (blind* OR masked)):ti,ab,kw) OR nonrandom*:ti,ab,kw OR 'non-random*':ti,ab,kw OR 'quasi-experiment*':ti,ab,kw OR crossover:ti,ab,kw OR 'cross over':ti,ab,kw OR 'parallel group*':ti,ab,kw OR 'factorial trial':ti,ab,kw OR ((phase NEAR/5 (study OR trial)):ti,ab,kw) OR ((case* NEAR/6 (matched OR control*)):ti,ab,kw) OR ((match* NEAR/6 (pair OR pairs OR cohort* OR control* OR group* OR healthy OR age OR sex OR gender OR patient* OR subject* OR participant*)):ti,ab,kw) OR ((propensity NEAR/6 (scor* OR match*)):ti,ab,kw) OR versus:ti OR vs:ti OR compar*:ti OR ((compar* NEAR/1 study):ti,ab,kw) OR (('major clinical study'/de OR 'clinical study'/de OR 'cohort analysis'/de OR 'observational study'/de OR 'cross-sectional study'/de OR 'multicenter study'/de OR 'correlational study'/de OR 'follow up'/de OR cohort*:ti,ab,kw OR 'follow up':ti,ab,kw OR followup:ti,ab,kw OR longitudinal*:ti,ab,kw OR prospective*:ti,ab,kw OR retrospective*:ti,ab,kw OR observational*:ti,ab,kw OR 'cross sectional*':ti,ab,kw OR cross?ectional*:ti,ab,kw OR multicent*:ti,ab,kw OR 'multi-cent*':ti,ab,kw OR consecutive*:ti,ab,kw) AND (group:ti,ab,kw OR groups:ti,ab,kw OR subgroup*:ti,ab,kw OR versus:ti,ab,kw OR vs:ti,ab,kw OR compar*:ti,ab,kw OR 'odds ratio*':ab OR 'relative odds':ab OR 'risk ratio*':ab OR 'relative risk*':ab OR 'rate ratio':ab OR aor:ab OR arr:ab OR rrr:ab OR ((('or' OR 'rr') NEAR/6 ci):ab))) OR 'major clinical study'/de OR 'clinical study'/de OR 'case control study'/de OR 'family study'/de OR 'longitudinal study'/de OR 'retrospective study'/de OR 'prospective study'/de OR 'comparative study'/de OR 'cohort analysis'/de OR ((cohort NEAR/1 (study OR studies)):ab,ti) OR (('case control' NEAR/1 (study OR studies)):ab,ti) OR (('follow up' NEAR/1 (study OR studies)):ab,ti) OR (observational NEAR/1 (study OR studies)) OR ((epidemiologic NEAR/1 (study OR studies)):ab,ti) OR (('cross sectional' NEAR/1 (study OR studies)):ab,ti) |
15094884 |
|
#16 |
'randomized controlled trial'/exp OR random*:ti,ab OR (((pragmatic OR practical) NEAR/1 'clinical trial*'):ti,ab) OR ((('non inferiority' OR noninferiority OR superiority OR equivalence) NEAR/3 trial*):ti,ab) OR rct:ti,ab,kw |
1943343 |
|
#15 |
'meta analysis'/exp OR 'meta analysis (topic)'/exp OR metaanaly*:ti,ab OR 'meta analy*':ti,ab OR metanaly*:ti,ab OR 'systematic review'/de OR 'cochrane database of systematic reviews'/jt OR prisma:ti,ab OR prospero:ti,ab OR (((systemati* OR scoping OR umbrella OR 'structured literature') NEAR/3 (review* OR overview*)):ti,ab) OR ((systemic* NEAR/1 review*):ti,ab) OR (((systemati* OR literature OR database* OR 'data base*') NEAR/10 search*):ti,ab) OR (((structured OR comprehensive* OR systemic*) NEAR/3 search*):ti,ab) OR (((literature NEAR/3 review*):ti,ab) AND (search*:ti,ab OR database*:ti,ab OR 'data base*':ti,ab)) OR (('data extraction':ti,ab OR 'data source*':ti,ab) AND 'study selection':ti,ab) OR ('search strategy':ti,ab AND 'selection criteria':ti,ab) OR ('data source*':ti,ab AND 'data synthesis':ti,ab) OR medline:ab OR pubmed:ab OR embase:ab OR cochrane:ab OR (((critical OR rapid) NEAR/2 (review* OR overview* OR synthes*)):ti) OR ((((critical* OR rapid*) NEAR/3 (review* OR overview* OR synthes*)):ab) AND (search*:ab OR database*:ab OR 'data base*':ab)) OR metasynthes*:ti,ab OR 'meta synthes*':ti,ab |
847757 |
|
#14 |
#11 AND #12 AND #13 AND ([english]/lim OR [dutch]/lim) AND [2000-2022]/py NOT ('conference abstract'/it OR 'editorial'/it OR 'letter'/it OR 'note'/it) NOT (('animal experiment'/exp OR 'animal model'/exp OR 'nonhuman'/exp) NOT 'human'/exp) |
394 |
|
#13 |
'drug eluting cardiovascular stent'/exp OR 'drug-eluting balloon catheter'/exp OR 'drug-coated balloon'/exp OR (((eluting OR drug OR coated OR paclitaxel OR sirolimus OR everolimus) NEAR/3 (stent* OR balloon*)):ti,ab,kw) OR 'paclitaxel'/exp OR 'sirolimus'/exp OR 'everolimus'/exp OR 'vessel preparation':ti,ab,kw OR 'lithotripsy'/exp OR 'intravascular lithotripsy'/exp OR 'intravascular lithotripsy':ti,ab,kw OR 'cutting balloon catheter'/exp OR ((balloon* NEAR/3 (cutting OR scoring)):ti,ab,kw) OR 'laser'/exp OR laser*:ti,ab,kw OR 'orbital atherectomy'/exp OR 'orbital atherectomy device'/exp OR 'orbital atherectomy*':ti,ab,kw OR csi:ti,ab,kw |
601485 |
|
#12 |
((below NEAR/3 knee*):ti,ab,kw) OR btk:ti,ab,kw OR infrapopliteal:ti,ab,kw OR infragenual:ti,ab,kw OR genicular:ti,ab,kw |
16771 |
|
#11 |
'peripheral vascular disease'/exp OR 'artery disease'/exp OR 'peripheral occlusive artery disease'/exp OR 'critical limb ischemia'/exp OR 'leg ischemia'/exp OR ((isch?em* NEAR/3 ('lower limb*' OR leg*)):ti,ab,kw) OR 'chronic limb-threatening isch?em*':ti,ab,kw OR clti:ti,ab,kw OR 'critical limb isch?em*':ti,ab,kw OR isch?em*:ti,ab,kw |
221788 |
Ovid/Medline
|
# |
Searches |
Results |
|
23 |
20 or 21 or 22 |
379 |
|
22 |
(16 and 19) not (20 or 21) = Observationeel |
233 |
|
21 |
(15 and 19) not 20 = RCT |
101 |
|
20 |
14 and 19 = SR |
45 |
|
19 |
limit 18 to ((english language or dutch) and yr="2000 -Current") |
481 |
|
18 |
17 not (comment/ or editorial/ or letter/ or ((exp animals/ or exp models, animal/) not humans/)) |
571 |
|
17 |
11 and 12 and 13 |
616 |
|
16 |
Epidemiologic studies/ or case control studies/ or exp cohort studies/ or Controlled Before-After Studies/ or Case control.tw. or cohort.tw. or Cohort analy$.tw. or (Follow up adj (study or studies)).tw. or (observational adj (study or studies)).tw. or Longitudinal.tw. or Retrospective*.tw. or prospective*.tw. or consecutive*.tw. or Cross sectional.tw. or Cross-sectional studies/ or historically controlled study/ or interrupted time series analysis/ or Case-control Studies/ or clinical trial, phase ii/ or clinical trial, phase iii/ or clinical trial, phase iv/ or comparative study/ or control groups/ or controlled before-after studies/ or controlled clinical trial/ or double-blind method/ or historically controlled study/ or matched-pair analysis/ or single-blind method/ or (((control or controlled) adj6 (study or studies or trial)) or (compar* adj (study or studies)) or ((control or controlled) adj1 active) or "open label*" or ((double or two or three or multi or trial) adj (arm or arms)) or (allocat* adj10 (arm or arms)) or placebo* or "sham-control*" or ((single or double or triple or assessor) adj1 (blind* or masked)) or nonrandom* or "non-random*" or "quasi-experiment*" or "parallel group*" or "factorial trial" or "pretest posttest" or (phase adj5 (study or trial)) or (case* adj6 (matched or control*)) or (match* adj6 (pair or pairs or cohort* or control* or group* or healthy or age or sex or gender or patient* or subject* or participant*)) or (propensity adj6 (scor* or match*))).ti,ab,kf. or (confounding adj6 adjust*).ti,ab. or (versus or vs or compar*).ti. or ((exp cohort studies/ or epidemiologic studies/ or multicenter study/ or observational study/ or seroepidemiologic studies/ or (cohort* or 'follow up' or followup or longitudinal* or prospective* or retrospective* or observational* or multicent* or 'multi-cent*' or consecutive*).ti,ab,kf.) and ((group or groups or subgroup* or versus or vs or compar*).ti,ab,kf. or ('odds ratio*' or 'relative odds' or 'risk ratio*' or 'relative risk*' or aor or arr or rrr).ab. or (("OR" or "RR") adj6 CI).ab.)) |
6935662 |
|
15 |
exp randomized controlled trial/ or randomized controlled trials as topic/ or random*.ti,ab. or rct?.ti,ab. or ((pragmatic or practical) adj "clinical trial*").ti,ab,kf. or ((non-inferiority or noninferiority or superiority or equivalence) adj3 trial*).ti,ab,kf. |
1536826 |
|
14 |
meta-analysis/ or meta-analysis as topic/ or (metaanaly* or meta-analy* or metanaly*).ti,ab,kf. or systematic review/ or cochrane.jw. or (prisma or prospero).ti,ab,kf. or ((systemati* or scoping or umbrella or "structured literature") adj3 (review* or overview*)).ti,ab,kf. or (systemic* adj1 review*).ti,ab,kf. or ((systemati* or literature or database* or data-base*) adj10 search*).ti,ab,kf. or ((structured or comprehensive* or systemic*) adj3 search*).ti,ab,kf. or ((literature adj3 review*) and (search* or database* or data-base*)).ti,ab,kf. or (("data extraction" or "data source*") and "study selection").ti,ab,kf. or ("search strategy" and "selection criteria").ti,ab,kf. or ("data source*" and "data synthesis").ti,ab,kf. or (medline or pubmed or embase or cochrane).ab. or ((critical or rapid) adj2 (review* or overview* or synthes*)).ti. or (((critical* or rapid*) adj3 (review* or overview* or synthes*)) and (search* or database* or data-base*)).ab. or (metasynthes* or meta-synthes*).ti,ab,kf. |
611021 |
|
13 |
exp *Angioplasty, Balloon/ or exp Drug-Eluting Stents/ or ((eluting or drug or coated or paclitaxel or sirolimus or everolimus) adj3 (stent* or balloon*)).ti,ab,kf. or exp Paclitaxel/ or exp Sirolimus/ or exp Everolimus/ or 'vessel preparation'.ti,ab,kf. or exp Lithotripsy/ or exp Lithotripsy, Laser/ or exp Lasers/ or 'intravascular lithotripsy'.ti,ab,kf. or laser*.ti,ab,kf. or (balloon* adj3 (cutting or scoring)).ti,ab,kf. or 'orbital atherectomy*'.ti,ab,kf. or CSI.ti,ab,kf. |
427852 |
|
12 |
((below adj3 knee*) or btk or infrapopliteal or infragenual or genicular).ti,ab,kf. |
10209 |
|
11 |
exp Ischemia/ or isch?em*.ti,ab,kf. or clti.ti,ab,kf. or exp Arterial Occlusive Diseases/ or exp Peripheral Vascular Diseases/ or exp Peripheral Vascular Diseases/ |
878584 |