Dose regimen oxytocin augmentation
Uitgangsvraag
Moet oxytocine als bijstimulatie worden toegediend in een hoog- of een laagdosis schema tijdens de ontsluitingsfase van de bevalling bij nulliparae à terme met een niet-vorderende ontsluiting?
Clinical question
Should labour augmentation with oxytocin be started in a high or a low dose regime during the first stage (the dilatation phase) of labour in nulliparous women at term with arrested or protracted labour?
Aanbeveling
Overweeg het gebruik van een laagdosis schema van oxytocine bij nulliparae à terme met een niet-vorderende ontsluiting.
Recommendation
Consider using a low dose oxytocin regimen for labour augmentation in nulliparous women at term with arrested or protracted labour.
Overwegingen
Considerations – evidence to decision
Pros and cons of the intervention and quality of the evidence
Overall, the evidence GRADE is low to moderate, with the majority of recommendations being of low GRADE. There are no clinically important differences in the selected crucial and important outcomes between high and low dose regimens of oxytocin for labour augmentation in nulliparous women at term with arrested or protracted labour. The only exception is a possible reduction in the risk of metabolic acidosis in children exposed to a high dose compared to a low dose of oxytocin. There are no data on the following outcomes: Sarnat score, Thomson score and encephalopathy. This makes it difficult to estimate the importance of the difference in the incidence of metabolic acidosis.
A high dose oxytocin may have no effect on the risk of a caesarean section or instrumental delivery in comparison with a low dose oxytocin (RR 0.81, 95% CI 0.57 to 1.16 and RR 0.97, 95% CI 0.80 to 1.18, respectively). The level of evidence, however, is low to moderate.
Another study, not included in the literature summary because the experimental intervention was pulsatile instead of continuous administration of oxytocin, reported a significant decrease in instrumental deliveries with a pulsatile regimen (Liu, 2018). This study also reported less instrumental deliveries with a high dose compared to a low dose regimen.
In the literature summary of this module, the incidence of hyperstimulation and the need for intra-uterine rescusitation has not been examined, but a low dose regimen may be favourable for this outcome. Tachysystoly occurred more often in the high dose regimen group in the study of Selin (2019), in which tachysystoly was seen in 43.2% of cases in the high dose and 33.5% of cases in the low dose arm (RD 9.6%, 95% CI 4.2 to 15.1%), without differences in the adverse neonatal outcomes. The study of Liu (2018) also reported significant decrease in hyperstimulation with a low dose regimen. Kenyon (2013) reported no evidence of an effect of a high versus low dose regimen on hyperstimulation (RR 1.47, 95%CI 0.73 to 2.94).
No subgroups that could benefit from one regimen compared to the other have been identified.
A major limitation of the performed meta-analyses, is the heterogeneity of regimens used in the individual studies, lowering the strength of the available evidence.
Over het algemeen is de bewijskracht laag tot matig, waarbij het merendeel van de aanbevelingen gebaseerd is op bewijs van lage kwaliteit (GRADE). Er zijn geen klinisch relevante verschillen gevonden in de geselecteerde cruciale en belangrijke uitkomstmaten tussen een hoog- en laagdosis schema van oxytocine bij nulliparae à terme met een niet-vorderende ontsluiting. De enige uitzondering is een mogelijke vermindering van het risico op metabole acidose bij kinderen die zijn blootgesteld aan een hoge dosis oxytocine in vergelijking met een lage dosis. Er zijn geen gegevens beschikbaar over de volgende uitkomsten: Sarnat-score, Thompson-score en neonatale encefalopathie. Dit maakt het moeilijk om het klinische belang van het verschil in de incidentie van metabole acidose in te schatten.
Een hoge dosis oxytocine lijkt geen effect te hebben op het risico op een sectio of een instrumentele bevalling in vergelijking met een lage dosis (RR 0,81, 95% BI 0,57 tot 1,16 en RR 0,97, 95% BI 0,80 tot 1,18, respectievelijk). De bewijskracht is echter laag tot matig.
Een andere studie, die niet is opgenomen in de literatuursamenvatting omdat de experimentele interventie bestond uit pulsatiele in plaats van continue toediening van oxytocine, rapporteerde een significante afname van instrumentele bevallingen bij een pulsatiel regime (Liu, 2018). Deze studie rapporteerde ook minder instrumentele bevallingen bij een hoge dosering in vergelijking met een lage dosering.
In de literatuursamenvatting van deze module is de incidentie van hyperstimulatie en de noodzaak tot intra-uteriene reanimatie niet onderzocht, maar een laag doserings schema zou voor deze uitkomstmaat gunstiger zijn. Tachysystolie kwam vaker voor in de groep met een hoge dosering in de studie van Selin (2019), waarin tachysystolie werd gezien in 43,2% van de gevallen bij een hoge dosis en 33,5% van de gevallen bij een lage dosis (RD 9,6%, 95% BI 4,2 tot 15,1%), zonder verschillen in de nadelige neonatale uitkomstmaten. Ook de studie van Liu (2018) rapporteerde ook een significante afname van hyperstimulatie bij een laagdosis schema. Kenyon (2013) vond geen bewijs voor een effect van een hoge versus lage dosering op hyperstimulatie (RR 1,47, 95% BI 0,73 tot 2,94).
Er zijn geen subgroepen geïdentificeerd die mogelijk baat zouden hebben bij het ene doseringsschema boven het andere.
Een belangrijke beperking van de uitgevoerde meta-analyses is de heterogeniteit van de gebruikte doseringsschema’s in de individuele studies, wat de kracht van het beschikbare bewijs vermindert.
Values and preferences of patients (and their caregivers)
The most important goal of the patient is a spontaneous delivery of a neonate without need for admission at the neonatal care unit and with a pH > 7.05 and hence no expected long-term consequences related to the birth process. There is no evidence in favour of any regimen, except for metabolic acidosis, which was less frequent in the high dose arm. This evidence, however, was reported in only one study (Selin, 2019) and the effect was very uncertain, shown by the wide confidence interval (RR 0.56, 95% CI 0.19 to 1.65).
It is reassuring that there seem to be no differences in childbirth experiences between high and low dose regimens (low grade). However, there is only low grade evidence from one study.
There are patients who prefer to labour as naturally as possible and patients who might have a preference of receiving doses as low as possible, which is to be supported considering the available evidence. It is important for women that their preferences are taken into account and a choice is made based on the principles of shared decision making.
Het belangrijkste doel van de barende is een spontane bevalling van een pasgeborene zonder noodzaak tot opname op de neonatale intensive care en met een pH > 7,05, en daarmee zonder te verwachten langdurige gevolgen gerelateerd aan het geboorteproces. Er is geen bewijs dat één van de doseringsschema’s de voorkeur verdient, met uitzondering van metabole acidose, die minder vaak voorkwam na toediening bij een hoge dosering oxytocine. Dit werd echter slechts in één studie gerapporteerd (Selin, 2019) en het effect was zeer onzeker, zoals blijkt uit het brede betrouwbaarheidsinterval (RR 0,56, 95% BI 0,19 tot 1,65).
Het is geruststellend dat er geen verschillen lijken te zijn in bevallingservaringen tussen hoge en lage doseringen oxytocine (lage GRADE). Deze conclusie is echter gebaseerd op beperkt bewijs (lage GRADE) uit één studie.
Er zijn barenden die de voorkeur geven aan een zo natuurlijk mogelijk verloop van de bevalling, en barenden die liever zo laag mogelijke doseringen oxytocine ontvangen. Deze voorkeuren verdienen ondersteuning, gezien het beschikbare bewijs. Het is belangrijk dat de voorkeuren van vrouwen worden meegenomen in de besluitvorming en dat de keuzes worden gemaakt op basis van de principes van gezamenlijke besluitvorming.
Costs
The possible additional costs of administrating a higher dosage of oxytocin are low since oxytocin is a low-cost medication.
De mogelijke extra kosten van toediening een hogere dosering oxytocine zijn gering, aangezien oxytocine een goedkoop geneesmiddel is.
Acceptability, feasibility and implementation
To our knowledge, no quantitative or qualitative research has been performed to evaluate the acceptability or feasibility of the intervention. We expect that the implementation of any dose regimen would be feasible, since it only entails a possible change in dosage of an indicated intervention. Patients might object to higher doses, as might physicians. Since no differences in the pre-specified outcomes have been found, the working group proposes to opt for a low dose regimen.
Physicians could compare their local protocol to the low and high dose regimens described in Table 1. If their regimen corresponds to a high dose regimen, they could consider to lower the dosage, since there are no differences in the critical and important outcomes considered in this guideline. A low dose is preferred over a high dose since the efficacy is similar and the risk of side effects is lower.
We expect no health inequity, nor do we think there are limiting factors concerning the implementation of our recommendation, except for maybe the low certainty of the evidence. Nevertheless, there are no evidence-based reasons to advise against administrating a high dose regimen. Monitoring of the fetal heartbeat, as well as the frequency of the contractions (to avoid hyperstimulation) is possibly more important than adhering to a certain regimen.
Voor zover bekend is er geen kwantitatief of kwalitatief onderzoek uitgevoerd naar de acceptatie of haalbaarheid van deze interventie. De werkgroep verwacht echter dat implementatie van elk doseringsschema haalbaar is, aangezien het slechts een aanpassing betreft van de dosering van een reeds geïndiceerde interventie. Zowel barenden als zorgverleners zouden mogelijk bezwaar kunnen hebben tegen hogere doseringen. Aangezien er geen verschillen zijn gevonden in de vooraf gespecificeerde uitkomstmaten, adviseert de werkgroep om te kiezen voor een laagdosis schema.
Zorgverleners kunnen hun lokale protocol vergelijken met de lage en hoge doseringsschema’s zoals weergegeven in Tabel 1. Indien het lokale schema overeenkomt met een hoog dosisschema, kan overwogen worden de dosering te verlagen, aangezien er geen verschillen zijn in de kritieke en belangrijke uitkomstmaten zoals beschreven in deze richtlijn. Een laagdosisschema heeft de voorkeur boven een hoog dosis schema, omdat de effectiviteit vergelijkbaar is en het risico op bijwerkingen lager ligt.
De werkgroep verwacht geen gezondheidsongelijkheid of andere belemmerende factoren voor de implementatie van deze aanbeveling, met uitzondering van de lage zekerheid van het bewijs. Er zijn echter geen op bewijs gebaseerde redenen om het gebruik van een hoog dosisschema af te raden. Het monitoren van de foetale hartslag en de frequentie van de contracties (om hyperstimulatie te voorkomen) is mogelijk belangrijker dan het strikt volgen van aan een bepaald dosisschema.
Differences between countries
The decision to use high dose or low dose oxytocin administration, provided that the condition of the fetus is stable, varies between countries, and sometimes even within countries.
De beslissing om oxytocine toe te dienen in een hoog- of laagdosis schema , mits de conditie van de foetus stabiel is, verschilt tussen landen en soms zelfs binnen landen.
Recommendation
Rationale of the recommendation: weighing arguments in favour and against the intervention
Although the high dose regimen may lower the risk of metabolic acidosis, the effect is very uncertain and the evidence is low grade. Since there was no evidence of an effect of a high vs. low dose regimen of oxytocin augmentation on other outcome measures, the working group proposes to use an as low as clinically possible dose regimen.
We suggest that clinicians compare their local protocol to the regimens described in the literature summary of this guideline. If the local regimen corresponds to a low dose regimen, current practice can be continued. If the local regimen corresponds to a high dose regimen, we propose to consider lowering the dosage. Examples of low dose regimens are given in Table 1. Based on the available evidence, continuing current practice, even if a high dose regimen is given, is acceptable. However, we advise not to use higher doses than the doses described as the high dose regimens in Table 1.
Hoewel een hoog dosis schema van oxytocine het risico op metabole acidose mogelijk kan verlagen, is dit effect zeer onzeker en is de bewijskracht laag. Aangezien er geen bewijs is voor een effect van een hoog- versus laagdosis schema op andere uitkomstmaten, adviseert de werkgroep om een zo laag als klinisch verantwoord doseringsschema te gebruiken.
De werkgroep stelt voor dat zorgverleners hun lokale protocol vergelijken met de doseringsschema’s zoals beschreven in de literatuursamenvatting van deze richtlijn. Als het lokale regime overeenkomt met een laagdosis schema, kan de huidige praktijk worden voortgezet. Als het overeenkomt met een hoog dosis schema, wordt geadviseerd de dosering te verlagen. Voorbeelden van laagdosis schema’s worden gegeven in Tabel 1. Op basis van het beschikbare bewijs is het voortzetten van de huidige praktijk, zelfs bij gebruik van een hoog dosis schema, aanvaardbaar. De werkgroep raadt echter af om hogere doseringen te gebruiken dan de welke in Tabel 1 als hoog dosisschema zijn beschreven.
Onderbouwing
Achtergrond
Multiple dosage regimens of oxytocin administration are used for labour augmentation in women at term with arrested or protracted labour. In literature, comparisons of low and high dose regimens are made. Since a clinical dose as low as possible is preferred, we decided to explore if there is a difference in outcomes between low and high dose regimens.
Conclusies / Summary of Findings
Conclusions
|
Very low GRADE |
The evidence is very uncertain about the effect of a high dose regimen versus a low dose regimen oxytocin administration on perinatal death in nulliparous women at term with arrested or protracted labour.
Source: Kenyon (2013), Selin (2019) |
|
Low GRADE |
A high dose oxytocin augmentation may not reduce or increase the risk of an Apgar score <7 at 5 minutes in comparison with a low dose oxytocin augmentation in nulliparous women at term with arrested or protracted labour.
Source: Kenyon (2013), Dy (2018), Selin (2019) |
|
Low GRADE |
A high dose oxytocin augmentation may reduce the risk of umbilical cord (artery) pH <7 in comparison with a low dose oxytocin augmentation in nulliparous women at term with arrested or protracted labour.
Source: Selin (2019) |
|
No GRADE |
The outcome measures Sarnat score, Thompson score and encephalopathy were not reported in the included studies. |
|
Low GRADE |
A high dose oxytocin augmentation may not reduce or increase the risk of a caesarean section in comparison with a low dose oxytocin augmentation in nulliparous women at term with arrested or protracted labour.
Source: Kenyon (2013), Selin (2019), Xenakis (1995) |
|
Moderate GRADE |
A high dose oxytocin augmentation likely does not reduce or increase the risk of an instrumental vaginal delivery in comparison with a low dose oxytocin augmentation in nulliparous women at term with arrested or protracted labour.
Source: Kenyon (2013), Selin (2019), Xenakis (1995) |
|
Low GRADE |
A high dose oxytocin augmentation may reduce the labour duration in comparison with a low dose oxytocin augmentation in nulliparous women at term with arrested or protracted labour.
Source: Kenyon (2013) |
|
Low GRADE |
A high dose oxytocin augmentation may result in little to no difference in the risk of anal sphincter rupture in comparison with a low dose oxytocin augmentation in nulliparous women at term with arrested or protracted labour.
Source: Selin (2019) |
|
No GRADE |
The outcome measure incontinence was not reported. |
|
Moderate GRADE |
A high dose oxytocin augmentation likely does not reduce or increase the risk of haemorrhage in comparison with a low dose oxytocin augmentation in nulliparous women at term with arrested or protracted labour.
Source: Kenyon (2013), Selin (2019) |
|
No GRADE |
The outcome measure infection was not reported. |
|
Low GRADE |
A high dose oxytocin augmentation may not reduce or increase satisfaction/childbirth experience in comparison with a low dose oxytocin augmentation in nulliparous women at term with arrested or protracted labour.
Source: Selin (2021) |
|
No GRADE |
The outcome measures breastfeeding, bonding and later caesarean section at maternal request were not reported. |
Samenvatting literatuur
Description of studies
Kenyon (2013) performed a systematic review and meta-analysis of the literature to compare a high-dose oxytocin regimen versus a low-dose oxytocin regimen for labour augmentation in women with delayed or slow progress of labour. The Cochrane Pregnancy and Childbirth Group’s Trials Register (including trials identified from CENTRAL, MEDLINE, EMBASE, hand searches of 30 journals, major conferences and 44 journals from BioMed Central) was searched up to June 18th, 2013.
The systematic review included published and unpublished RCTs and quasi-randomized trials investigating women with delayed or slow progress of labour who required oxytocin for labour augmentation and comparing a high-dose regimen (i.e. starting dose and increment >4 mU/min) with a low-dose regimen (i.e. starting dose and increment <4 mU/min) with a 15-40 minute increment interval. The review included three randomized controlled trials in nulliparous women (Bidgood, 1987; Jamal, 2004; Kenyon, 2013) and one quasi-randomised controlled trial in nulli- and multiparous women (Xenakis, 1995). In total, 645 women were randomized in these four studies, of which 320 received a high dosage of oxytocin and 324 received a low dosage of oxytocin.
Selin (2019, 2021) performed a RCT in healthy, nulliparous women at term (37+0 to 41+6 wks) with a confirmed delay of labour progress. Women were randomly assigned to the high-dose group (i.e. infusion started with 6.6 mU oxytocin per minute and could be increased every 20 minutes by 6.6 mU to a maximum dose of 59.4 mU/min) or to the low-dose group (i.e. infusion started with 3.3 mU oxytocin per minute and could be increased every 20 minutes by 3.3 mU to a maximum dose of 29.7 mU/min). In the RCT, 1295 women were included in the intention to treat analysis (high-dose group, n=647; low-dose group, n=648) to evaluate the rate of caesarean sections (main outcome measure) and maternal and neonatal outcomes (secondary outcome measures) (Selin, 2019). In the second publication, childbirth experiences and labour pain were evaluated in 1008 women (high-dose group, n=497; low-dose group, n=511) (Selin, 2021).
Dy (2018) performed a double-blinded pilot RCT in nulliparous women at term (37+0 to 41+6 wks) with labour dystocia. Women were randomized to the accelerated oxytocin titration group (high-dose group; infusion of a solution concentration of 40 U oxytocine in 1000 ml started at 4 mU/min and was increased every 30 minutes to a maximum of 40 mU/min) or the gradual oxytocin titration group (low dose; infusion of a solution concentration of 20 U oxytocine in 1000 ml started at 2 mU oxytocine per minute and was increased every 30 minutes to a maximum infusion of 20 mU/min). For this study, 79 women were randomized (high-dose group, n=40; low-dose group, n=39) to evaluate feasibility of a large multicenter trial (primary outcome), the rate of caesarean sections and maternal and foetal indicators of safety (secondary outcomes).
An overview of the different dosage regimens of the included studies is provided in Table 1.
Table 1. Overview of dosage regimens in the included studies.
|
Study |
Intervention (high dose) |
Control (low dose) |
|
Bidgood (1987) (Source: Kenyon, 2013) |
Starting dose of 7 mU oxytocin per min, increased by 7 mU/min every 15 minutes, to a maximum of a frequency of 7 contractions in 15 minutes or by abnormality in the fetal heart rate tracing |
oxytocin started at 2 mU/min, increased by 2 mU/min every 15 minutes, to a maximum of 40 mU/min until a table phase of uterine activity was detected or uterine activity integral exceeded 1500 kPas/15 minutes |
|
Jamal (2004) (Source: Kenyon, 2013) |
Starting dose of 4.5 mU oxytocin per minute, increased by 4.5 mU/min every 30 minutes |
Starting dose of 1.5 mU oxytocin per minute, increased by 1.5 mU/min every 30 minutes |
|
Kenyon (2013) (Source: Kenyon, 2013) |
Starting dose of 4mU oxytocin per minute, increased every 30 minutes to 64 mU/min |
Starting dose of 2mU oxytocin per minute, increased every 30 minutes to 32 mU/min |
|
Xenakis (1995) (Source: Kenyon, 2013) |
Starting dose of 4 mU oxytocin per minute, increased by 4 mU/min every 15 minutes |
Starting dose of 1 mU oxytocin per minute, increased by 1 mU/min every 30 minutes |
|
Selin (2019, 2021) |
Starting dose of 6.6 mU oxytocin per minute, increased by 6.6 mU/min every 20 minutes. |
Starting dose of 3.3 mU oxytocin per minute, increased by 3.3 mU/min every 20 minutes. Maximum dose of 29.7 mU oxytocin per minute |
|
Dy (2018) |
Starting dose of 4 mU oxytocine per minute, increased every 30 minutes to a maximum of 40 mU/min |
Starting dose of 2 mU oxytocine per minute, increased every 30 minutes to a maximum of 20 mU/min |
Results
Crucial outcome measures
1. Perinatal death
Three studies included in the systematic review (Jamal, 2004; Kenyon, 2013; Xenakis, 1995) and one RCT (Selin, 2019) reported on the outcome measure perinatal death.
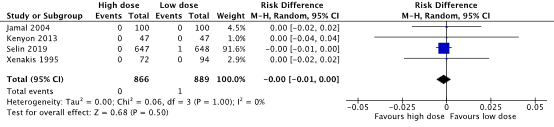
In Selin (2019), one case of perinatal death was reported in the low dose oxytocin group due to severe hypoxic encephalopathy, which was considered not to be related to the study drug (Figure 1). Based on the risk difference (RD -0.00, 95% CI -0.01 to 0.00), there is no clinically relevant difference in perinatal death between the low and high dose oxytocin regimen groups.
Figure 1. Perinatal death, high dose oxytocin versus low dose oxytocin regimens.
Source: Kenyon (2013), Selin (2019). Z: p-value of pooled effect, df: degrees of freedom, I2: statistical heterogeneity, CI: confidence interval.
2. Apgar score <7 at 5 minutes
Three studies included in the systematic review (Bidgood, 1987; Kenyon, 2013; Xenakis, 1995) and two RCTs (Dy, 2018; Selin, 2019) reported on the outcome measure Apgar score.
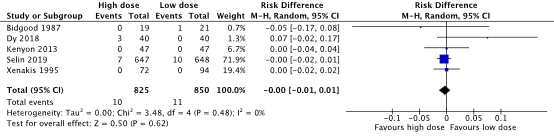
An Apgar score <7 at 5 minutes was found in 10/825 (1.2%) babies in the high dose oxytocin group and in 11/850 (1.3%) babies in the low dose oxytocin group (RD 0.00, 95%CI -0.01 to 0.01) (Figure 2). Based on the risk difference, no clinically important difference in the number of babies having Apgar scores <7 at 5 minutes was found between the high dose and low dose oxytocin groups.
Figure 2. Apgar score <7 at 5 minutes, high dose oxytocin versus low dose oxytocin regimens.
Source: Kenyon (2013), Dy (2018), Selin (2019). Z: p-value of pooled effect, df: degrees of freedom, I2: statistical heterogeneity, CI: confidence interval.
3. Umbilical cord (artery) pH <7
One study (Selin, 2019) reported on the outcome umbilical cord (artery) pH as metabolic acidosis (defined as pH <7.05 and base excess ≥-12 or pH <7.0).
Metabolic acidosis was found in 5/527 (0.9%) babies in the high dose oxytocin group and in 9/527 (1.7%) babies in the low dose oxytocin group (RR 0.56, 95% CI 0.19 to 1.65). There was a clinically important difference in metabolic acidosis in favour of the high dose oxytocin regimen.
4. Sarnat score
Not reported.
5. Thompson score
Not reported.
6. Encephalopathy
Not reported.
Important outcome measures
7. Caesarean section
Three studies included in the systematic review (Bidgood, 1987; Kenyon, 2013; Xenakis, 1995) and two RCTs (Dy, 2018; Selin, 2019) reported on the outcome measure caesarean section.
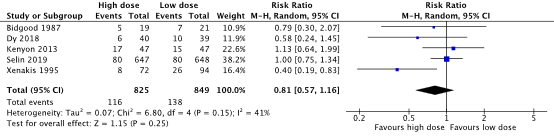
A caesarean section was performed in 116/825 (14.1%) women in the high dose oxytocin group and in 138/849 (16.3%) women in the low dose oxytocin group (RR 0.81, 95% CI 0.57 to 1.16) (Figure 3). No clinically important difference in the number of caesarean sections between the high dose oxytocin and low dose oxytocin groups was found.
Figure 3. Caesarean section, high dose oxytocin versus low dose oxytocin regimens.
Source: Kenyon (2013), Selin (2019). Z: p-value of pooled effect, df: degrees of freedom, I2: statistical heterogeneity, CI: confidence interval.
8. Instrumental vaginal delivery
Three studies included in the systematic review (Bidgood 1987; Kenyon, 2013; Xenakis, 1995) and two RCTs (Dy, 2018; Selin, 2019) reported on the outcome measure instrumental vaginal delivery.
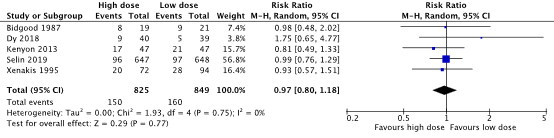
The number of instrumental vaginal deliveries was 150/825 (18.2%) in the high dose oxytocin group and 160/849 (18.8%) in the low dose oxytocin group (RR 0.97, 95% CI 0.80 to 1.18) (Figure 4). There was no clinically relevant difference between the number of instrumental vaginal deliveries between the high dose oxytocin and the low dose oxytocin group in nulliparous women.
Figure 4. Instrumental vaginal delivery, high dose oxytocin versus low dose oxytocin regimens.
Source: Kenyon (2013), Selin (2019). Z: p-value of pooled effect, df: degrees of freedom, I2: statistical heterogeneity, CI: confidence interval.
9. Duration of labour
Duration of labour was reported in five studies but was defined differently in these studies. The working group chose to define duration of labour as the time from oxytocin augmentation to delivery. This was reported by one study included in the systematic review (Bidgood, 1987).
Duration of labour was 7.8 ± 2.7 hours in the high dose oxytocin group, while labour duration was 11.3 ± 6.1 hours in the low dose oxytocin group (mean difference -3.50, 95% CI -6.38 to -0.62 hours). This difference in duration of labour was clinically relevant in favor of the high dose oxytocin group.
10. Anal sphincter rupture
One study (Selin, 2019) reported on anal sphincter rupture, which was defined as anal sphincter injury graded 3 or 4.
Anal sphincter injury (grade 3 or 4) was reported in 26/567 (4.6%) women in the high dose oxytocin group and in 29/568 (5.1%) women in the low dose oxytocin group (RR 0.90, 95% CI 0.54 to 1.51). There was no clinically relevant difference in the number of anal sphincter injuries between the low and the high dose oxytocin groups.
11. Incontinence
Not reported.
12. Haemorrhage >1000 mL
One study included in the systematic review (Kenyon, 2013) and one RCT (Selin, 2019) reported on the outcome measure haemorrhage >1000 mL.
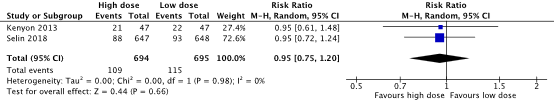
Haemorrhage was reported in 109/694 (15.7%) women in the high dose oxytocin group and in 115/695 (16.5%) women in the low dose oxytocin group (RR 0.95, 95% CI 0.75 to 1.20) (Figure 5). There was no clinically relevant difference in haemorrhage between the high dose and low dose oxytocin groups.
Figure 5. Haemorrhage, high dose oxytocin versus low dose oxytocin regimens.
Source: Kenyon (2013), Selin (2019). Z: p-value of pooled effect, df: degrees of freedom, I2: statistical heterogeneity, CI: confidence interval.
13. Infection
Not reported
14. Satisfaction/childbirth experience
One study evaluated childbirth experiences (Selin, 2021), measured with the Childbirth Experience Questionnaire version 2 in four domains: own capacity, perceived safety, professional support and participation. Most questionnaire items are rated on a 4-point Likert scale, ranging from 1 (totally disagree) to 4 (totally agree). Experience of labour pain, sense of security and control were rated on a 0-100 VAS-scale and VAS-scores were transformed on a 4-point scale (i.e. VAS 0-40=1, VAS 41-60=2, VAS 61-80=3 and VAS 81-100=4). For each domain, a mean score was calculated with higher scores reflecting more positive experiences.
There was no clinically relevant difference in childbirth experience on any of the four domains between the high dose oxytocin group and the low dose oxytocin group (Table 2).
Table 2. Childbirth experiences, high dose oxytocin versus low dose oxytocin regimens.
|
Outcome measure as reported by Selin (2021) |
High dose |
Low dose |
Mean difference (95% CI) |
|
Own capacity |
2.43 (0.55) |
2.46 (0.56) |
-0.03 [-0.10 to 0.04] |
|
Perceived safety |
3.03 (0.72) |
3.06 (0.71) |
-0.03 [-0.12 to 0.06] |
|
Professional support |
3.52 (0.57) |
3.53 (0.52) |
-0.01 [-0.08 to 0.06] |
|
Participation |
3.50 (0.65) |
3.53 (0.6) |
-0.03 [-0.11 to 0.05] |
Source: Selin (2021). CI: confidence interval. Values are Mean (SD)
15. Breastfeeding
Not reported.
16. Bonding
Not reported.
17. Later caesarean section at maternal request
Not reported
Level of evidence of the literature
According to GRADE, systematic reviews of RCTs and RCTs start at a high level of evidence.
Critical outcome measures
The level of evidence regarding the outcome measure perinatal death was downgraded by three levels to a very low GRADE, because of study limitations (-1; risk of bias) and a very small number of events (-2; imprecision).
The level of evidence regarding the outcome measure Apgar score <7 at 5 minutes was downgraded by two levels to a low GRADE, because of study limitations (-1; risk of bias) and conflicting results (-1; inconsistency).
The level of evidence regarding the outcome measure umbilical cord (artery) pH <7
was downgraded by two levels to a low GRADE, because the confidence intervals crossed both margins of clinical relevance (-2, imprecision).
Important outcome measures
The level of evidence regarding the outcome measure caesarean section was downgraded by two levels to a low GRADE, because of conflicting results (-1; inconsistency) and because the 95% confidence interval crossed the lower margin of clinical relevance (-1, imprecision).
The level of evidence regarding the outcome measure instrumental vaginal delivery was downgraded by 1 level to moderate GRADE, because of study limitations (-1; risk of bias).
The level of evidence regarding the outcome measure duration of labour was downgraded by two levels to a low GRADE, because of study limitations (-1; risk of bias) and because of a very small number of included patients (-2; imprecision).
The level of evidence regarding the outcome measure anal sphincter rupture was downgraded by 2 levels to a low GRADE, because the confidence intervals crossed both margins of clinical relevance (-2, imprecision).
The level of evidence regarding the outcome measure haemorrhage was downgraded by one level to a moderate GRADE, because the 95% confidence interval crossed the lower margin of clinical relevance (-1; imprecision).
The level of evidence regarding the outcome measure childbirth experiences was downgraded by two level to a low GRADE, because of a high percentage of non-responders (16%) (-2; risk of bias due to high number of missing results).
Zoeken en selecteren
A systematic review of the literature was performed to answer the following question:
Should oxytocin augmentation be started in a high or a low dose regimen during the first stage (the dilatation phase) of labour in nulliparous women at term with arrested or protracted labour?
The working group did not define which regimen is low or high dose but considered the regimens as defined in the studies themselves as low or high dose.
|
Patients |
Nulliparous women during first stage (the dilatation phase) of spontaneous or induced labour, at term (gestational age ≥37+0) and with a fetus in cephalic presentation, with arrested or protracted labour. |
|
Intervention |
Oxytocin dosage regimen 1, high dose. |
|
Control |
Oxytocin dosage regimen 2, low dose. |
|
Outcomes |
Perinatal death, Apgar score <7 at 5 minutes, umbilical cord (artery) pH <7, Sarnat score, Thompson score, encephalopathy, caesarean section, instrumental vaginal delivery, duration of labour, anal sphincter rupture, incontinence, haemorrhage >1000 ml, infection, satisfaction/childbirth experience, breastfeeding, bonding and later caesarean section at maternal request. |
Relevant outcome measures
The guideline development group considered perinatal death, Apgar score <7 at 5 minutes, umbilical cord (artery) pH <7, Sarnat score, Thompson score and encephalopathy as critical outcome measures for decision making; and caesarean section, instrumental vaginal delivery, duration of labour, anal sphincter rupture, incontinence, haemorrhage >1000 mL, infection, satisfaction/childbirth experience, breastfeeding, bonding and later caesarean section at maternal request as important outcome measures for decision making.
A priori, the working group did not define the outcome measures listed above but used the definitions used in the studies.
The working group defined RR<0.95 or RR>1.05 as a minimal clinically (patient) important difference for the outcome measures perinatal death and encephalopathy. For the other outcomes, RR<0.8 or RR>1.25 was considered as a minimal clinically (patient) important difference. For continuous outcomes a difference of 0.5 Standard Deviation (SD) was considered clinically (patient) relevant.
Search and select (Methods)
The databases Medline (via OVID) and Embase (via Embase.com) were searched with relevant search terms until July 12th, 2022. The detailed search strategy is depicted under the tab Methods. The systematic literature search resulted in 315 hits. Studies were selected based on the following criteria:
- Systematic reviews, randomized controlled trials (RCTs) or comparative observational studies
- Women with arrested or protracted labour during the first stage of spontaneous or induced labour
- Comparison of a high oxytocin dosage (regimen 1) versus a low oxytocin dosage (regimen 2). Augmentation with oxytocin should be administered by continuous administration. Studies using pulsatile administration of oxytocin were excluded.
- Assessing one or more of the predefined outcomes
- Articles published from 1990 to July 7th, 2022
- Publications in English.
Fifteen articles were initially selected based on title and abstract screening. After reading the full text, eleven articles were excluded (see the table with reasons for exclusion), and four articles about three studies were included.
Results
Three studies were included in the analysis of the literature, of which one systematic review and meta-analysis (Kenyon, 2013) and three publications about two randomized controlled trials (Selin, 2019 and 2021; Dy, 2018). Important study characteristics and results are summarized in the evidence tables. The assessment of the risk of bias is summarized in the risk of bias tables.
Referenties
- Dy J, Rainey J, Walker MC, Fraser W, Smith GN, White RR, Waddell P, Janoudi G, Corsi DJ, Wei SQ. Accelerated Titration of Oxytocin in Nulliparous Women with Labour Dystocia: Results of the ACTION Pilot Randomized Controlled Trial. J Obstet Gynaecol Can. 2018 Jun;40(6):690-697.
- Kenyon S, Tokumasu H, Dowswell T, Pledge D, Mori R. High-dose versus low-dose oxytocin for augmentation of delayed labour. Cochrane Database Syst Rev. 2013 Jul 13;(7):CD007201.
- Liu J, Yi Y, Weiwei X. Effects of Increased Frequency, High Dose, and Pulsatile Oxytocin Regimens on Abnormal Labor Delivery. Med Sci Monit. 2018 Apr 7;24:2063-2071.
- Selin L, Wennerholm UB, Jonsson M, Dencker A, Wallin G, Wiberg-Itzel E, Almström E, Petzold M, Berg M. High-dose versus low-dose of oxytocin for labour augmentation: a randomised controlled trial. Women Birth. 2019 Aug;32(4):356-363.
- Selin L, Berg M, Wennerholm UB, Dencker A. Dosage of oxytocin for augmentation of labor and women's childbirth experiences: A randomized controlled trial. Acta Obstet Gynecol Scand. 2021 May;100(5):971-978.
Verantwoording
Beoordelingsdatum en geldigheid
Publicatiedatum : 01-04-2026
Beoordeeld op geldigheid : 01-04-2026
Validity period
The Board of the Dutch Society of Obstetrics and Gynaecology (NVOG) will assess whether these guidelines are still up to date in 2029 at the latest. If necessary, a new working group will be appointed to revise the guideline. The guideline’s validity may lapse earlier if new developments demand revision at an earlier date.
As the holder of this guideline, the NVOG is chiefly responsible for keeping the guideline up to date. Other scientific organizations participating in the guideline or users of the guideline share the responsibility to inform the chiefly responsible party about relevant developments within their fields.
Algemene gegevens
In collaboration with:
- Deutsche Gesellschaft für Gynäkologie und Geburtshilfe
- Vlaamse Vereniging voor Obstetrie en Gynaecologie
- Federação das Sociedades Portuguesas de Obstetricia e Ginecologia
- Svensk Förening För Obstetrik & Gynekologi
- Hellenic Society of Obstetric and Gynecological Emergency
- Schweizerische Gesellschaft für Gynäkologie und Geburtshilfe
- Dansk Selskab for Obstetrik og Gynækologi
- Česká gynekologická a porodnická společnost
- European Midwives Association
Samenstelling werkgroep
Composition guideline development panel
An international panel for the development of the guidelines was formed in 2019. The panel consisted of representatives from all relevant medical disciplines that are involved in medical care for pregnant women.
All panel members have been officially delegated for participation in the guideline development panel by their (scientific) societies. The panel has developed the guidelines in the period from January 2022 until May 2024.
The guideline development panel is responsible for the entire text of this guideline.
All panel members have been officially delegated for participation in the guideline development panel by their scientific societies. The guideline development panel is responsible for the entire text of this guideline.
Members of the EAPM Standing Committee on Guideline Development (SCGD)
- J.J. Duvekot, obstetrician, Consultant Obstetrics and Gynaecology, Erasmus Medical Centre, Rotterdam, the Netherlands (chair)
- D. Ayres de Campos, obstetrician, Consultant Obstetrics and Gynaecology, Faculdade de Medicina, Lisbon, Portugal
- S. Brismar-Wendel, obstetrician, Consultant Obstetrics and Gynaecology, Danderyd Hospital, Stockholm, Sweden
- G. Daskalakis, obstetrician, Consultant Obstetrics and Gynaecology, National & Kapodistrian University, Athens, Creece.
- I. Dehaene, obstetrician, Consultant Obstetrics and Gynaecology, Ghent University Hospital Belgium
- M. Kacerovsky, obstetrician, Consultant Obstetrics and Gynaecology, University Hospital Hradec Kralove, Czech Republic
- S. Kehl, obstetrician, Consultant Obstetrics and Gynaecology, Erlangen University Hospital, Erlangen, Germany
- Julie Glavind, obstetrician, Consultant Obstetrics and Gynaecology, Aarhus University Hospital, Aarhus, Denmark
- A. Hamza, obstetrician, Consultant Obstetrics and Gynaecology, University Medical Center of Saarland, Homburg an der Saar, Germany
- M.A. Ledingham, obstetrician, Consultant Obstetrics and Gynaecology, the Queen Elizabeth Hospital Glasgow, UK
- B. Magowan, obstetrician, Consultant Obstetrics and Gynaecology, and previous Co-Chair UK RCOG Guidelines Committee, NHS Borders, Scotland, UK
Advisor committee (part of the committee since September 2023)
- E. Mestdagh, Director of Research and Development, AP University of Applied Sciences and Arts Antwerp, Belgium, Scientific advisor and guideline developer, KNOV, The Netherlands
- I. Wilsens, senior program coördinator development & innovation, KNOV, the Netherlands
- I. van Ee, patient representative, Dutch Patient Federation, the Netherlands
- P. Stenbäck, Midwife, MCHS, Program Director - Midwifery education. School of Business and Healthcare, Arcada University of Applied Sciences, Helsinki, Treasurer, board member EMA, European Midwifes Association, Belgium
- C. Matteo, European Midwives Association
Methodological support
- J. Tuijtelaars, advisor, Knowledge Institute of the Dutch Association of Medical Specialists
- J.H. van der Lee, senior advisor, Knowledge Institute of the Dutch Association of Medical Specialists
Belangenverklaringen
Declarations of interests
The Code for the prevention of improper influence due to conflicts of interest was followed.
The working group members have provided written statements about (financially supported) relations with commercial companies, organisations or institutions related to the subject matter of the guideline during the past three years. Furthermore, inquiries have been made regarding personal financial interests, interests due to personal relationships, interests related to reputation management, interest related to externally financed research and interests related to knowledge valorisation. The chair of the guideline development panel is informed about changes in interests during the development process. The declarations of interests are reconfirmed during the commentary phase. The declarations of interests can be requested at the administrative office of the Knowledge Institute of the Dutch Association of Medical Specialists and are summarised below.
|
Last name |
Principal position |
Ancillary position(s) |
Declared interests |
Action |
|
Duvekot (chair) |
Consultant Obstetrics and Gynaecology, Erasmus MC, Rotterdam |
Director 'Medisch Advies en Expertise Bureau Duvekot' - making expertises on medical calamities |
None |
None |
|
Dehaene |
Consultant Obstetrics and Gynaecology, Ghent University Hospital |
None |
None |
None |
|
Hamza |
Leading Consultant at the department of obstetrics and prenatal medicine Kantonspital Baden |
At the cantonal hospital I am involved in the clinical work, research and development. |
Inovolvement in research and development to improve maternofenal health.
|
None |
|
Ledingham |
Consultant in Maternal and Fetal Medicine, Queen Elizabeth Hospital, Glasgow |
Guideline developer for sign (scottisch intercollegiate guidelines group) |
None |
None |
|
Magowan |
Consultant Obstetrics and Gynaecology, and Co-Chair UK RCOG Guidelines Committee, NHS Borders, Scotland |
Previous Co-chair RCOG Guidelines committee |
None |
None |
|
Stenbäck |
Midwife, MCHS, Program Director - Midwifery education. School of Business and Healthcare. Arcada University of Applied Sciences, Helsinki. Treasurer, board member EMA, European Midwifes, Association, Belgium. |
Program director of Midwifery education, Arcada University of Applied Sciences, Helsinki. full time employment.
Treasurer of European Midwives Association, is a position of trust. EMA is a non-profit and non-governmental organisation of midwives, representing midwifery organisations and associations from the member states of the European Union (EU), the European Economic Area (EEA) and EU applicant countries and Council of Europe. |
None |
None |
|
Brismar Wendel |
MD, senior consulant, specialist in obstetrics and gynaecology. Department of Women's Health, Danderyd Hospital |
None |
None |
None |
|
Ayres de Campos |
Full Professor of Obstetrics and Gynecology. Medical School, University of Lisbon |
None |
None |
None |
|
Daskalakis |
Professor OB/GYN National and Kapodistrian University of Athens |
General obstetrics and Gynecology duties. |
None |
None |
|
Wilsens |
Senior program coördinator development & innovation, KNOV, the Netherlands |
None |
None |
None |
|
Glavind |
Senior Consultant in obstetrics, Aarhus University Hospital, Denmark |
Member of the Danish National Obstetrics Guideline Steering Comittee: Responsible for coordinating the process in developing, revising and approving Danish national guidelines in obstetrics including the planning of an annual national guideline meeting to discuss new and revised guidlines. This position is not remunerated. |
None |
None |
|
Kehl |
Delegate of the German Society of Perinatal Medicine and German Society of Gynecology and Obstetrics |
None |
None |
None |
|
Mestdagh |
Director of Research and Development - AP University of Applied Sciences and Arts Antwerp - Belgium |
Director of Research and development: |
None |
None |
|
Van Ee |
Dutch Patient federation – adviser patient perspective (fulltime) |
Psoriasis patiens Netherlands - coördinator patient participation and research (unpaid)
|
None |
None |
|
Kacerovsky |
Professor, University Hospital Hradec Kralove, Charles University, Faculty of Medicine in Hradec Kralove, Consultant in Maternal-Fetal Medicine, Hospital Most |
None |
None |
None |
Inbreng patiëntenperspectief
Representation of the patient perspective
Representatives of the Dutch Patient Federation provided review comments. The comments were discussed and where relevant incorporated by the guideline development panel.
This guideline accounts for everyone who gives birth, even though we speak of women and her/she in the guideline.
Implementatie
Implementation
Guideline implementation and practical applicability of the recommendations was taken into consideration during various stages of guideline development. Factors that may promote or hinder implementation of the guideline in daily practice were given specific attention.
The guideline is distributed digitally among all relevant professional groups. The guideline can also be downloaded from the Dutch guideline website: www.richtlijnendatabase.nl.
Werkwijze
Method
AGREE
This guideline has been developed conforming to the requirements of the report of Guidelines for Medical Specialists 3.0 by the advisory committee of the Quality Counsel. This report is based on the AGREE II instrument (Appraisal of Guidelines for Research & Evaluation II) (www.agreetrust.org)(Brouwers, 2010), a broadly accepted instrument in the international community and on the national quality standards for guidelines: “Guidelines for guidelines” (www.zorginstituutnederland.nl).
Identification of subject matter
Since this was a pilot project, the content of the questions and the support base in clinical practice was considered of less importance than the process of international collaboration and learning from each other. The panel made an inventory of clinical questions to be addressed based on their expert opinion and their knowledge of existing guidelines. The ‘Danish national clinical guideline concerning primiparous women with dystocia (lack of progress)’ published in 2015 was also used as a source of clinical questions.
Clinical questions and outcomes
The guideline development panel formulated definitive clinical questions and defined relevant outcome measures (both beneficial land harmful effects). The panel rated the outcome measures as critical, important and not important. Furthermore, where applicable, the panel defined relevant clinical differences.
Strategy for search and selection of literature
For the separate clinical questions, specific search terms were formulated and published scientific articles were searched for in (several) electronic databases. Furthermore, studies were scrutinized by cross-referencing for other included studies. The studies with potentially the highest quality of research were searched for first. The panel members selected literature in pairs (independently of each other) based on title and abstract. A second selection was performed based on full text. The databases, search terms and selection criteria are described in the modules containing the clinical questions.
No definitions were available at the time of performing the search. Therefore, definitions as used in the papers were used.
Quality assessment of individual studies
Individual studies were systematically assessed, based on methodological quality criteria that were determined prior to the search, so that risk of bias could be estimated. This is described in the “risk of bias” tables.
Summary of literature
The relevant research findings of all selected articles are shown in evidence tables. The most important findings in the literature are described in literature summaries. In case there were enough similarities between studies, the study data were pooled.
Grading quality of evidence and strength of recommendations
The strength of the conclusions of the scientific publications was determined using the GRADE-method. GRADE stands for Grading Recommendations Assessment, Development and Evaluation (see http://www.gradeworkinggroup.org/).
GRADE defines four gradations for the quality of scientific evidence: high, moderate, low or very low. These gradations provide information about the amount of certainty about the literature conclusions (http://www.guidelinedevelopment.org/handbook/).
The basic principles of the GRADE method are: formulating and prioritising clinical (patient) relevant outcome measures, a systematic review for each outcome measure, and appraisal of the evidence for each outcome measure based on the eight GRADE domains (domains for downgrading: risk of bias, inconsistency, indirectness, imprecision, and publication bias; domains for upgrading: dose-effect association, large effect, and residual plausible confounding).
GRADE distinguishes four levels for the quality of the scientific evidence: high, moderate, low and very low. These levels refer to the amount of certainty about the conclusion based on the literature, in particular the amount of certainty that the conclusion based on the literature adequately supports the recommendation (Schünemann, 2013; Hultcrantz, 2017).
|
GRADE |
Definition |
|
High |
|
|
Moderate |
|
|
Low |
|
|
Very low |
|
The limits of clinical decision making are very important in grading the evidence in guideline development according to the GRADE methodology (Hultcrantz, 2017). Exceedance of these limits would give rise to adaptation of the recommendation. All relevant outcome measures and considerations need to be taken into account to define the limits of clinical decision making. Therefore, the limits of clinical decision making are not one to one comparable to the minimal clinically relevant difference. In particular for interventions of low costs and without important drawbacks the limit of clinical decision making regarding the effectiveness of the intervention may be lower (i.e. closer to no effect) than the Minimal Clinically Important Difference (MCID) (Hultcrantz, 2017).
Considerations (evidence to decision)
Aspects such as expertise of working group members, patient preferences, costs, availability of facilities, and organisation of healthcare aspects are important to consider when formulating a recommendation. For each clinical question, these aspects are discussed in the paragraph Considerations, using a structured format based on the evidence-to-decision framework of the international GRADE Working Group (Alonso-Coello, 2016a; Alonso-Coello, 2016b). The evidence-to-decision framework is an integral part of the GRADE methodology.
Formulating recommendations
Recommendations provide an answer to the primary question and are based on the best scientific evidence available and the most important considerations. The level of scientific evidence and the importance given to considerations by the working group jointly determine the strength of the recommendation. In accordance with the GRADE method, a low level of evidence for conclusions in the systematic literature review does not rule out a strong recommendation, while a high level of evidence may be accompanied by weak recommendations. The strength of the recommendation is always determined by weighing all relevant arguments.
Knowledge gaps
During the development of this guideline, systematic searches were conducted for research contributing to answering the primary questions. For each primary question, the working group determined whether (additional) scientific research is desirable.
Commentary and authorisation phase
The draft guideline was subjected to commentaries by the scientific societies and patient organisations involved. The draft guideline was also submitted to members of the European Midwives Association (EMA). The comments were collected and discussed with the working group. The feedback was used to improve the guideline; afterwards the working group made the guideline definitive. The final version of the guideline was offered for authorization to the involved scientific societies and patient organisations and was authorized or approved, respectively.
Legal standing of guidelines
Guidelines are not legal prescriptions but contain evidence-based insights and recommendations that care providers should meet in order to provide high quality care. As these recommendations are primarily based on ‘general evidence for optimal care for the average patient’, care providers may deviate from the guideline based on their professional autonomy when they deem it necessary for individual cases. Deviating from the guideline may even be necessary in some situations. If care providers choose to deviate from the guideline, this should be done in consultation with the patient, where relevant. Deviation from the guideline should always be justified and documented.
Literature
Agoritsas T, Merglen A, Heen AF, Kristiansen A, Neumann I, Brito JP, Brignardello-Petersen R, Alexander PE, Rind DM, Vandvik PO, Guyatt GH. UpToDate adherence to GRADE criteria for strong recommendations: an analytical survey. BMJ Open 2017;7:e018593.
Alonso-Coello P, Schünemann HJ, Moberg J, Brignardello-Petersen R, Akl EA, Davoli M, Treweek S, Mustafa RA, Rada G, Rosenbaum S, Morelli A, Guyatt GH, Oxman AD; GRADE Working Group. GRADE Evidence to Decision (EtD) frameworks: a systematic and transparent approach to making well informed healthcare choices. 1: Introduction. BMJ 2016;353:i2016.
Alonso-Coello P, Oxman AD, Moberg J, Brignardello-Petersen R, Akl EA, Davoli M, Treweek S, Mustafa RA, Vandvik PO, Meerpohl J, Guyatt GH, Schünemann HJ; GRADE Working Group. GRADE Evidence to Decision (EtD) frameworks: a systematic and transparent approach to making well informed healthcare choices. 2: Clinical practice guidelines. BMJ. 2016 Jun 30;353:i2089.
Brouwers MC, Kho ME, Browman GP, Burgers JS, Cluzeau F, Feder G, Fervers B, Graham ID, Grimshaw J, Hanna SE, Littlejohns P, Makarski J, Zitzelsberger L; AGREE Next Steps Consortium. AGREE II: advancing guideline development, reporting and evaluation in health care. CMAJ. 2010 Dec 14;182(18):E839-42.
Hultcrantz M, Rind D, Akl EA, Treweek S, Mustafa RA, Iorio A, Alper BS, Meerpohl JJ, Murad MH, Ansari MT, Katikireddi SV, Östlund P, Tranæus S, Christensen R, Gartlehner G, Brozek J, Izcovich A, Schünemann H, Guyatt G. The GRADE Working Group clarifies the construct of certainty of evidence. J Clin Epidemiol. 2017 Jul;87:4-13.
Medisch Specialistische Richtlijnen 2.0 (2012). Adviescommissie Richtlijnen van de Raad Kwalitieit. http://richtlijnendatabase.nl/over_deze_site/over_richtlijnontwikkeling.html
Neumann I, Santesso N, Akl EA, Rind DM, Vandvik PO, Alonso-Coello P, Agoritsas T, Mustafa RA, Alexander PE, Schünemann H, Guyatt GH. A guide for health professionals to interpret and use recommendations in guidelines developed with the GRADE approach. J Clin Epidemiol. 2016 Apr;72:45-55. doi: 10.1016/j.jclinepi.2015.11.017. Epub 2016 Jan 6. Review. PubMed PMID: 26772609.
Schünemann H, Brożek J, Guyatt G, et al. GRADE handbook for grading quality of evidence and strength of recommendations. Updated October 2013. The GRADE Working Group, 2013. Available from http://gdt.guidelinedevelopment.org/central_prod/_design/client/handbook/handbook.html.
Schünemann HJ, Oxman AD, Brozek J, Glasziou P, Jaeschke R, Vist GE, Williams JW Jr, Kunz R, Craig J, Montori VM, Bossuyt P, Guyatt GH; GRADE Working Group. Grading quality of evidence and strength of recommendations for diagnostic tests and strategies. BMJ. 2008 May 17;336(7653):1106-10.
Schünemann, A Holger J [corrected to Schünemann, Holger J]. PubMed PMID: 18483053; PubMed Central PMCID: PMC2386626.
Wessels M, Hielkema L, van der Weijden T. How to identify existing literature on patients' knowledge, views, and values: the development of a validated search filter. J Med Libr Assoc. 2016 Oct;104(4):320-324.