Augmentation with oxytocin
Uitgangsvraag
Moet oxytocine bijstimulatie onmiddellijk of uitgesteld worden toegediend na de diagnose van niet-vorderende ontsluiting, tijdens de eerste fase (de ontsluitingsfase) van een spontane of geïnduceerde bevalling bij nulliparae à terme?
Clinical question
Should oxytocin as augmentation of labour be given immediately or delayed after diagnosing protracted or arrested labour, during the first stage of spontaneous or induced labour in nulliparous women at term?
Aanbeveling
Informeer de barende dat er geen wetenschappelijk bewijs is voor voordelen van vroege (<1 uur) of uitgestelde toediening van oxytocine. De toediening kan mogelijk invloed hebben op de duur van de bevalling. Neem samen een beslissing op basis van klinisch inzicht en de waarden en voorkeuren van de barende.
Recommendation
Inform the patient that there is no scientific evidence about the beneficial effects of immediate (<1 hour) or delayed administration of oxytocin. It might impact the duration of labour. Decide together with the patient based on clinical judgment as well as the patient’s values and preferences.
Overwegingen
Considerations – evidence to decision
Pros and cons of the intervention and quality of the evidence
The literature analysis for this module was based on one Cochrane review (Bugg, 2013) that compared early use of intravenous oxytocin versus delayed use of oxytocin. Even though the evidence as derived from this study was considered low, immediate oxytocin augmentation may slightly reduce labour duration in comparison with delayed oxytocin augmentation in women with protracted or arrested labour. Most women that participated in the studies were nulliparous. There was no clinically relevant or statistically significant difference in satisfaction or childbirth experience between groups with immediate or delayed oxytocin augmentation. The evidence regarding the effect of immediate versus delayed oxytocin augmentation on perinatal death, Apgar score <7 at 5 minutes, caesarean section and instrumental vaginal delivery was very low, and therefore, no conclusions were formulated. No evidence was found for the effect of immediate versus delayed oxytocin on umbilical cord (artery pH), Sarnat score, Thompson score, encephalopathy, anal sphincter rupture, incontinence, breastfeeding, bonding, and later caesarean section at maternal request.
The literature search did not yield any studies that revealed adverse effects of immediate administration of oxytocin augmentation in the first stage of labour of nulliparous women with protracted or arrested labour. No difference was found in reported outcomes, except a shorter duration of labour in those who received immediate administration of oxytocin. This should be considered as a potential pro of an early oxytocin augmentation. No specific cons of immediate administration of oxytocin were revealed in the literature available.
De literatuuranalyse voor deze module is gebaseerd op één Cochrane-review (Bugg, 2013) waarin vroege versus uitgestelde toediening van intraveneuze oxytocine werd vergeleken. Hoewel de bewijskracht van deze studie als laag wordt beoordeeld, kan vroege toediening van oxytocine de duur van de bevalling enigzins verkorten in vergelijking met uitgestelde toediening van oxytocine bij vrouwen met een niet-vorderende ontsluiting. De meeste deelnemers aan de studies waren nulliparae. Er werd geen klinisch relevant of statistisch significant verschil gevonden in tevredenheid of bevallingservaring tussen de groepen met vroege of uitgestelde toediening van oxytocine. Het bewijs over het effect van vroege versus uitgestelde toediening van oxytocine op perinatale sterfte, Apgar-score <7 na 5 minuten, het aantal sectio’s en instrumentele vaginale bevallingen was zeer beperkt, waardoor hierover geen conclusies konden worden geformuleerd. Er werd geen bewijs gevonden voor effecten op arteriële navelstreng-pH, Sarnat-score, Thompson-score, neonatale encefalopathie, anale sfincterrupturen, incontinentie, borstvoeding, hechting en latere sectio op verzoek van de moeder.
Er werden geen studies gevonden die nadelige effecten rapporteerden van vroege toediening van oxytocine tijdens de eerste fase van de bevalling bij nulliparae met een niet-vorderende ontsluiting. Het enige verschil dat werd gevonden,was een kortere duur van de bevalling bij barenden die vroege toediening van oxytocine kregen. Dit kan worden beschouwd als een mogelijk voordeel van vroege toediening van oxytocine. Er werden geen specifieke nadelen van vroege toediening van oxytocine gevonden in de beschikbare literatuur.
Values and preferences of patients (and their caregivers)
Since there are no differences in satisfaction/childbirth experience identified between immediate and delayed oxytocin administration in nulliparous women with protracted or arrested labour in the dilatation phase, it is important that women are informed about both options including pros and cons as part of shared decision making. Women should not be overwhelmed by the immediate administration of oxytocin but informed to make a deliberate decision.
Aangezien er geen verschillen in tevredenheid of bevallingservaring zijn vastgesteld tussen vroege en uitgestelde toediening van oxytocine bij nulliparae met een niet-vorderende ontsluiting, is het belangrijk dat vrouwen over beide opties worden geïnformeerd, inclusief de voor- en nadelen, als onderdeel van gezamenlijke besluitvorming. Vrouwen mogen niet overrompeld worden door een onmiddellijke toediening van oxytocine, maar moeten juist goed geïnformeerd worden zodat zij een weloverwogen keuze kunnen maken.
Sommige vrouwen geven de voorkeur aan een kortere duur van de bevalling. Voor anderen is een zo kort mogelijke bevalling geen prioriteit, zij verkiezen een zo fysiologisch mogelijk verloop van de bevalling en vinden dat het proces de tijd mag nemen die nodig is. Daarom is het van belang dat vrouwen weten dat er geen overtuigend wetenschappelijk bewijs is dat vroege of uitgestelde toediening van oxytocine superieur is. Vrouwen moeten erop kunnen vertrouwen dat zij hun keuze mogen baseren op hun eigen voorkeuren en intuïtie.
Costs
A longer duration of labour can be translated into increased costs associated with a longer stay in the delivery wards. The rates of caesarean section that can have a direct impact on the costs were not different between immediate and delayed oxytocin augmentation. The information about the outcomes associated with a stay at NICU, such as umbilical cord blood pH, Sarnat and Thompson scores, and encephalopathy, that create the direct economic cost were not reported in the literature available, but we do have information about Apgar score <7 at 5 min, which is strongly related to these outcomes as well. Since there is no clinically relevant difference in Apgar score <7 between immediate and delayed oxytocin, we expect no difference in these outcomes and no difference in NICU admission.
Een langere duur van de bevalling kan leiden tot hogere kosten, onder andere door een langere verblijfsduur op de verloskamers. Het percentage sectio’s, wat een directe invloed kan hebben op de zorgkosten, verschilde niet tussen vroege en uitgestelde toediening van oxytocine. Informatie over uitkomstmaten die verband houden met een opname op de NICU, zoals de pH van het navelstrengbloed, Sarnat- en Thompson-scores en neonatale encefalopathie, werden niet gerapporteerd in de beschikbare literatuur. Wel is er informatie beschikbaar over de Apgar-score <7 na 5 minuten, die sterk geassocieerd is met de uitkomstmaten. Aangezien er geen klinisch relevant verschil werd gevonden in Apgar-score <7 tussen vroege en uitgestelde toediening van oxytocine, verwachten we ook geen verschil in deze gerelateerde uitkomstmaten en geen verschil in NICU-opnames.
Acceptability, feasibility, and implementation
Regardless the fact that no differences in satisfaction/childbirth experience were identified between immediate and delayed oxytocin administrations in nulliparous women after diagnosing protracted or arrested labour in the dilatation phase, a shorter duration of labour may be important for some women. For others, childbirth does not have to be as short as possible. They prefer a delivery as physiological as possible and/or or believe that the process of giving birth to a child may take as long as necessary.
There are no obstacles for this intervention that could oppose implementation. After the diagnosis of protracted or arrest of labour is made, in all situations it is possible to start augmentation of labour with oxytocin.
Cephalo-pelvic disproportion and fetal distress, however, should always be excluded before starting augmentation.
Ondanks het feit dat er geen verschillen zijn vastgesteld in tevredenheid of bevallingservaring tussen vroege en uitgestelde toediening van oxytocine bij nulliparae na de diagnose van niet-vorderende ontsluiting, kan een kortere duur van de bevalling voor sommige vrouwen belangrijk zijn. Voor anderen hoeft de bevalling niet zo kort mogelijk te zijn. Zij geven de voorkeur aan een zo fysiologisch mogelijk verloop van de bevalling en vinden dat het proces van het baren van een kind de tijd mag nemen die nodig is.
Er zijn geen belemmeringen voor de implementatie van deze interventie. Nadat de diagnose van niet vorderende ontsluiting is gesteld, is het in alle situaties mogelijk om oxytocine toe te dienen. Wel moet cefalopelvische disproportie en foetale nood altijd worden uitgesloten voordat met augmentatie wordt gestart.
Differences between countries
The decision to start oxytocin augmentation immediately or delayed after diagnosing protracted or arrested labour in the first stage of the labour differs between the countries and/or even within the countries.
De beslissing om oxytocine vroeg of uitgesteld toe te dienen na de diagnose van een niet-vorderende ontsluiting in de eerste fase van de bevalling verschilt tussen landen en soms zelfs binnen landen.
Recommendation
Rationale of the recommendation: weighing arguments in favour and against the intervention
Immediate administration of oxytocin (< 1 hour) after diagnosing protracted or arrested labour is associated with a reduction of duration of labour compared to delayed administration of oxytocin in nulliparous women in the first stage of spontaneous labour. However, quality of the evidence is low. Therefore, the working group gave the recommendation to consider immediate initiation of oxytocin augmentation after diagnosing protracted or arrested labour, based on practical arguments like logistics and a shorter duration of labour.
Discuss with the woman the possibility of immediate initiation of augmentation with oxytocin (<1 hour) after diagnosing protracted or arrested labour in nulliparous women during the first stage of spontaneous labour. Inform women that there is no scientific evidence about immediate or delayed administration of oxytocin being more beneficial and decide together based on the values and preferences of the women.
The evidence for the use of immediate or delayed administration of oxytocin is not available for women with induced labour. However, there is no reason to believe that these women have other pros and cons for immediate or delayed oxytocin administration than women with spontaneous labour. Therefore, the working group recommends to consider giving women with spontaneous labour the same information as women with induced labour.
Vroege toediening van oxytocine (< 1 uur) na het diagnosticeren van niet-vorderende ontsluiting wordt geassocieerd met een kortere duur van de bevalling in vergelijking met uitgestelde toediening van oxytocine bij nulliparae in de eerste fase van een spontane bevalling. De kwaliteit van het bewijs is echter laag. Daarom geeft de werkgroep de aanbeveling om vroege toediening van oxytocine te overwegen op basis van praktische overwegingen, zoals logistiek en een kortere bevallingsduur.
Bespreek met de vrouw de mogelijkheid om vroegtijdig (< 1 uur) te starten met oxytocine-toediening na het diagnosticeren van niet-vorderende ontsluiting bij nulliparae in de eerste fase van een spontane bevalling. Informeer barenden dat er geen wetenschappelijk bewijs is dat vroege of uitgestelde toediening van oxytocine superieur is, en neem samen een beslissing op basis van de waarden en voorkeuren van de barende.
Voor vrouwen bij wie de bevalling is geïnduceerd, is er geen bewijs beschikbaar over het effect van vroege versus uitgestelde toediening van oxytocine. De werkgroep ziet echter geen reden om aan te nemen dat de voor- en nadelen voor deze groep anders zouden zijn dan bij vrouwen met een spontane bevalling. Daarom wordt aanbevolen om vrouwen met een geïnduceerde bevalling dezelfde informatie te geven als vrouwen met een spontane bevalling.
Onderbouwing
Achtergrond
In clinical practice many different schedules to start oxytocin augmentation after the diagnosis of protracted or arrested labour are used. Comparisons of immediate (<1 hour) and delayed (>1 hour) administration of oxytocin augmentation in the first stage of labour have been described in nulliparous women with protracted or arrested labour. There is uncertainty whether early oxytocin augmentation is beneficial and safe for mothers and their newborns, or if this intervention should be delayed.
Conclusies / Summary of Findings
Conclusions
Induced labour
|
No GRADE |
For induced labour, the effect of immediate oxytocin administration versus delayed oxytocin administration for women with protracted or arrested labour was not reported. |
Spontaneous labour - Critical outcome measures
|
Very low GRADE |
The evidence is very uncertain about the effect of immediate oxytocin administration versus delayed oxytocin administration on perinatal death in spontaneously labouring women with protracted or arrested labour during the dilatation phase.
Source: Bugg (2013) |
|
Very low GRADE |
The evidence is very uncertain about the effect of immediate oxytocin administration versus delayed oxytocin administration on Apgar score <7 at 5 minutes in spontaneously labouring women with protracted or arrested labour during the dilatation phase.
Source: Bugg (2013) |
|
No GRADE |
The outcome umbilical cord artery pH, Sarnat score, Thomson score and encephalopathy were not reported. |
Spontaneous labour - Important outcome measures
|
Very low GRADE |
The evidence is very uncertain about the effect of immediate oxytocin administration versus delayed oxytocin administration on caesarean section in spontaneously labouring women with protracted or arrested labour during the dilatation phase.
Source: Bugg (2013) |
|
Very low GRADE |
The evidence is very uncertain about the effect of immediate oxytocin administration versus delayed oxytocin administration on instrumental vaginal delivery in spontaneously labouring women with protracted or arrested labour during the dilatation phase.
Source: Bugg (2013) |
|
Low GRADE |
Immediate oxytocin administration may result in a reduction in duration of labour compared to delayed oxytocin administration in spontaneously labouring women with protracted or arrested labour during the dilatation phase.
Source: Bugg (2013) |
|
No GRADE |
The outcome anal sphincter rupture and incontinence were not reported. |
|
Low GRADE |
Immediate oxytocin administration may result in little to no difference in haemorrhage when compared to delayed oxytocin administration in spontaneously labouring women with protracted or arrested labour during the dilatation phase.
Source: Bugg (2013) |
|
No GRADE |
The outcome infection was not reported. |
|
Low GRADE |
Immediate oxytocin administration may result in little to no difference in satisfaction/childbirth experience compared to delayed oxytocin administration in spontaneously labouring women with protracted or arrested labour during the dilatation phase.
Source: Bugg (2013) |
|
No GRADE |
The outcome breastfeeding, bonding and later caesarean section at maternal request were not reported. |
Samenvatting literatuur
Description of the study
Bugg (2013) performed a systematic review and meta-analysis of the literature to determine if the use of oxytocin for the treatment of slow progress of labour in the first stage of spontaneous labour could have a positive effect on delivery outcomes and maternal and neonatal morbidity and mortality. The first comparison described in Bugg (2013) was intravenous oxytocin versus placebo/no treatment (not included in this literature analysis) and the second comparison was early use of intravenous oxytocin (immediate administration) versus delayed use of oxytocin (i.e. delayed by at least one hour). The Cochrane Pregnancy and Childbirth Group’s Trials Register (including trials identified from CENTRAL, MEDLINE, EMBASE, hand searches of 30 journals, major conferences and 44 journals from BioMed Central) was searched up to May 29th, 2013.
The systematic review included RCTs investigating low-risk pregnant women with slow progress in the first stage of spontaneous labour at term (37-42 weeks) with a singleton foetus presenting by the vertex. Women who had a previous caesarean section or women who were being induced from the outset were excluded. The review included five trials for the comparison immediate versus delayed (from 3 to 8 hours after diagnosis of labor with arrest or protracted labor) use of oxytocin, of which two multicentre (Dencker, 2009 and Hinshaw, 2008) and three single centre trials (Bidgood, 1987; Blanch, 1998 and Hemminki, 1985). In total 1.200 women were included in the active stage of labour, of which 610 received early intravenous oxytocin and 590 received delayed oxytocin.
Results
Crucial outcome measures
1. Perinatal death
Two trials reported on the outcome ‘perinatal death’ which was defined as serious neonatal morbidity or perinatal death, however the only two confirmed cases reported were perinatal deaths (Hemminki, 1995 and Hinshaw, 2008).
One case of perinatal death was reported in each arm of Hinshaw (2008) (RR 0.98, 95%CI 0.06 to 15.57), unrelated to delivery. Hemminki (1985) reported no perinatal deaths (Table 1).
Table 1. Perinatal death, immediate oxytocin augmentation versus delayed oxytocin augmentation.
|
Author, year |
Immediate oxytocin |
Delayed oxytocin |
Risk ratio (95% CI) |
|
Hemminki, 1985 |
0/27 |
0/30 |
Not estimable |
|
Hinshaw, 2008 |
1/208 |
1/204 |
0.98 (0.06 to 15.57) |
|
Total (95%CI) |
1/235 |
1/234 |
0.98 (0.06 to 15.57) |
Source: Bugg (2013). CI:Confindence interval.
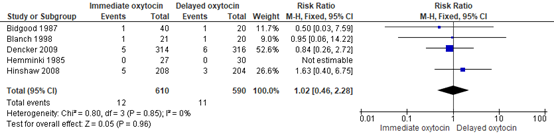
2. Apgar score <7 at 5 minutes
Five trials reported on the outcome Apgar score <7 at 5 minutes (Bidgood, 1987; Blanch, 1998; Dencker, 2009; Hemminki, 1985 and Hinshaw, 2008).
An Apgar score <7 at 5 minutes was found in 12/610 (2%) neonates in the immediate oxytocin group and in 11/590 (1.9%) neonates in the delayed oxytocin group (RR 1.02, 95%CI 0.46 to 2.28) (Figure 1). This difference was considered not clinically relevant.
Figure 1. Apgar score <7 at 5 minutes, immediate oxytocin augmentation versus delayed oxytocin augmentation.
Source: Bugg (2013). Z: p-value of pooled effect, df: degrees of freedom, I2: statistical heterogeneity, CI: confidence interval.
3. Umbilical cord (artery) pH <7.0
An outcome close to this outcome was only reported by Dencker (2009), who reported ‘cord artery metabolic acidosis’, defined as pH <7.0 together with base excess ≤12. Umbilical cord blood was only examined in 466 of the 630 neonates, and the number of missing values per intervention group was not reported. Therefore, the published results are considered unreliable.
4. Sarnat score
Not reported.
5. Thompson score
Not reported.
6. Encephalopathy
Not reported.
Important outcome measures
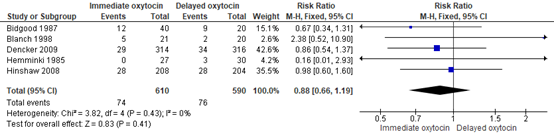
7. Caesarean section
Five trials reported on the outcome caesarean section (Bidgood, 1987; Blanch, 1998; Dencker, 2009; Hemminki, 1985 and Hinshaw, 2008).
Caesarean section was reported in 74/610 (12.1%) of childbirths in the immediate oxytocin group and in 76/590 (12.9%) of childbirths in the delayed oxytocin group (RR 0.88, 95% CI 0.66 to 1.19) (Figure 2). The point estimate, RR 0.88 in favour of immediate oxytocin, did not exceed the predefined threshold of clinically important effect of RR≤0.8.
Figure 2. Caesarean section, immediate oxytocin augmentation versus delayed oxytocin augmentation
Source: Bugg (2013). Z: p-value of pooled effect, df: degrees of freedom, I2: statistical heterogeneity, CI: confidence interval.
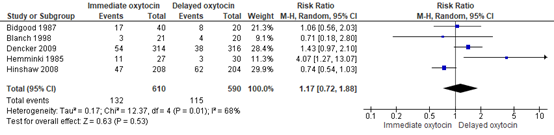
8. Instrumental vaginal delivery
Five trials reported on the outcome instrumental vaginal delivery (Bidgood, 1987; Blanch, 1998; Dencker, 2009; Hemminki, 1985 and Hinshaw, 2008).
Instrumental vaginal delivery was reported in 132/610 (21.6%) of childbirths in the immediate oxytocin group and in 115/590 (19.5%) of childbirths in the delayed oxytocin group (RR 1.17, 95% CI 0.72 to 1.88) (Figure 3). The difference, RR 1.17 in favour of delayed oxytocin, did not exceed the predefined clinical important difference threshold of RR≥1.25, which means that there is no clinically relevant difference.
Figure 3. Instrumental vaginal delivery, immediate oxytocin augmentation versus delayed oxytocin augmentation
Source: Bugg (2013). Z: p-value of pooled effect, df: degrees of freedom, I2: statistical heterogeneity, CI: confidence interval.
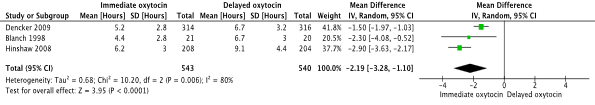
9. Duration of labour
Three studies reported on the outcome duration of labour, described as the time from
randomisation to delivery (Blanch, 1998; Dencker, 2009; Hinshaw, 2008).
The mean difference in duration of labour was 2 hours and 11 minutes (2.19) in favour of the
immediate oxytocin group (95% CI -3.28 to -1.10) (Figure 4). The difference, in favour of immediate oxytocin, was regarded of clinical relevance, exceeding the predefined clinical difference of 0.5 SD.
Figure 4. Duration of labour (in hours), immediate oxytocin augmentation versus delayed oxytocin augmentation
Source: Bugg (2013). Z: p-value of pooled effect, df: degrees of freedom, I2: statistical heterogeneity, CI: confidence interval.
10. Anal sphincter rupture
Not reported.
11. Incontinence
Not reported.
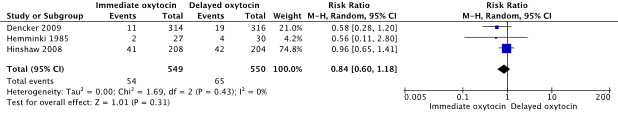
12. Haemorrhage
Three trials reported on the outcome measure haemorrhage. The definition of haemorrhage as described in Bugg (2013) was defined by individual trial authors. Dencker (2009) reported blood loss > 1000 mL, while Hemminki (1985) and Hinshaw (2008) reported blood loss of > 500 mL.
Haemorrhage was reported in 54/549 (9.8%) women after childbirth in the immediate oxytocin group and in 65/550 (11.8%) women after childbirth in the delayed oxytocin group (RR 0.84, 95% CI 0.60 to 1.18) (Figure 5). The difference, RR=0.84 in favour of immediate oxytocin, did not exceed the predefined clinical important difference threshold of RR≤0.8.
Figure 5. Haemorrhage, immediate oxytocin augmentation versus delayed oxytocin augmentation
Source: Bugg (2013). Z: p-value of pooled effect, df: degrees of freedom, I2: statistical heterogeneity, CI: confidence interval.
13. Infection
Not reported.
14. Satisfaction/childbirth experience
The outcome satisfaction/childbirth experience was reported by Bugg (2013) as ‘number of women with negative memories of childbirth’ (based on two questionnaire items of the Childbirth Experience Questionnaire).
One trial reported on the outcome women who were not satisfied (Dencker, 2009). The number of women with a negative childbirth experience was 100/233 (44.8%) in the immediate oxytocin group versus 86/209 (41.1%) in the delayed group (RR 1.04, 95% CI 0.84 to 1.13) (table 2). No clinically important difference in childbirth experiences was found.
15. Breastfeeding
Not reported.
16. Bonding
Not reported.
17. Later caesarean section at maternal request
Not reported.
Level of evidence of the literature
According to GRADE, systematic reviews of RCTs and RCTs start at a high level of evidence.
Critical outcome measures
The level of evidence regarding the outcome measure perinatal death was downgraded by four levels to very low GRADE because of an unclear methodology (-1, risk of bias) and because of a small sample size in the study groups (-3, imprecision).
The level of evidence regarding the outcome measure Apgar score <7 at 5 minutes was downgraded by three levels to very low GRADE because of lack of blinding (-1, risk of bias) and because the 95% confidence intervals crossed both margins of clinical relevance (-2, imprecision).
The outcome measures umbilical cord artery pH, Sarnat score, Thompson score and encephalopathy were not reported and therefore the level of evidence for these outcomes was not assessed.
Important outcome measures
The level of evidence regarding the outcome measure caesarean section was downgraded by three levels to a very low GRADE because of lack of blinding (-1, risk of bias), different directions of the effect (-1; inconsistency) and because the 95% confidence intervals crossed one margin of clinical relevance (-1, imprecision).
The level of evidence regarding the outcome measure instrumental vaginal delivery was downgraded by four levels to a very low GRADE because of a lack of blinding (-1, risk of bias), different directions of the effect (-1, inconsistency) and because the confidence intervals crossed both margins of clinical relevance (-2, imprecision).
The level of evidence regarding the outcome measure duration of labour was downgraded by two levels to low GRADE because of a lack of blinding (-1, risk of bias) and differences in measurement method (-1, indirectness).
The outcome measures anal sphincter rupture and incontinence were not reported and therefore the level of evidence for these outcomes was not assessed.
The level of evidence regarding the outcome measure haemorrhage was downgraded by two levels to low GRADE because of high risk of bias in two of three included studies due to methodological considerations (-1, risk of bias) and because of inconsistency (two studies show a clinically relevant effect, while one study shows no effect of immediate oxytocin) (-1, inconsistency).
The level of evidence regarding the outcome measure satisfaction/childbirth experience was downgraded by two levels to low GRADE because of a lack of blinding (-1, risk of bias) and the number of included participants (442 participants) (-1, imprecision).
The outcome measures infection, breastfeeding, bonding, and later caesarean section at maternal request were not reported and therefore the level of evidence for these outcomes was not assessed.
Zoeken en selecteren
A systematic review of the literature was performed to answer the following question:
What is the effect of immediate oxytocin augmentation compared to delayed oxytocin augmentation (one hour or more) in nulliparous women at term, diagnosed with protracted or arrested labour during the first stage of spontaneous or induced labour?
|
Patients |
Nulliparous women at term (gestational age ≥37+0 weeks) during the first stage of spontaneous or induced labour and a foetus in cephalic presentation, diagnosed with protracted or arrested labour |
|
Intervention |
Immediate (<1 hour) oxytocin augmentation after diagnosing protracted or arrested labour. |
|
Control |
Delayed oxytocin augmentation (>1 hour) after diagnosing protracted or arrested labour. |
|
Outcomes |
Perinatal death, Apgar score <7 at 5 minutes, umbilical cord (artery) pH <7, Sarnat score, Thompson score, encephalopathy, caesarean section, instrumental vaginal delivery, duration of labour, anal sphincter rupture, incontinence, haemorrhage >1000 ml, infection, satisfaction/childbirth experience, breastfeeding, bonding, and later caesarean section at maternal request. |
Relevant outcome measures
The guideline development group considered perinatal death, Apgar score <7 at 5 minutes, umbilical cord (artery) pH <7.0, Sarnat score, Thompson score, encephalopathy as critical outcome measures for decision making; and caesarean section, instrumental vaginal delivery, duration of labour, anal sphincter rupture, incontinence, haemorrhage >1.000 ml, infection, satisfaction/childbirth experience, breastfeeding, bonding and later caesarean section at maternal request as important outcome measures for decision making.
A priori, the working group did not define the outcome measures listed above but used the definitions used in the studies.
The working group defined RR<0.95 or RR>1.05 as a minimal clinically (patient) important difference for the outcome measures perinatal death and encephalopathy. For the other outcomes, RR<0.8 or RR>1.25 was considered as a minimal clinically (patient) important difference. For the continuous outcome ‘duration of labour’, a difference of 0.5 Standard Deviation (SD) was considered clinically (patient) relevant.
Search and select (Methods)
The databases Medline (via OVID) and Embase (via Embase.com) were searched with relevant search terms until July 7th, 2022. The detailed search strategy is depicted under the tab Methods. The systematic literature search resulted in 315 hits. Studies were selected based on the following criteria:
- Systematic reviews, randomized controlled trials (RCTs) or comparative observational studies
- Women with protracted or arrested labour during the first stage of spontaneous or induced labour
- Comparison of immediate oxytocin augmentation versus delayed administration (one hour or more).
- Assessing one or more of the predefined outcomes.
- Articles published from 1990 to July 7th 2022
- Publications in English.
Forty-three studies were initially selected based on title and abstract. After reading the full text, 42 studies were excluded (see the exclusion table with reasons for exclusion under the tab Methods), and one study was included.
Results
One Cochrane review (Bugg, 2013) was identified from the literature analysis. This review included five studies on the comparison of early use of intravenous oxytocin versus delayed use, which is described as comparison 2 in the review. Important study characteristics and results are summarized in the evidence table and the risk of bias is assessed in the quality assessment table.
Referenties
- Bugg GJ, Siddiqui F, Thornton JG. Oxytocin versus no treatment or delayed treatment for slow progress in the first stage of spontaneous labour. Cochrane Database Syst Rev. 2013 Jun 23;(6):CD007123.
Verantwoording
Beoordelingsdatum en geldigheid
Publicatiedatum : 01-04-2026
Beoordeeld op geldigheid : 01-04-2026
Validity period
The Board of the Dutch Society of Obstetrics and Gynaecology (NVOG) will assess whether these guidelines are still up to date in 2029 at the latest. If necessary, a new working group will be appointed to revise the guideline. The guideline’s validity may lapse earlier if new developments demand revision at an earlier date.
As the holder of this guideline, the NVOG is chiefly responsible for keeping the guideline up to date. Other scientific organizations participating in the guideline or users of the guideline share the responsibility to inform the chiefly responsible party about relevant developments within their fields.
Algemene gegevens
In collaboration with:
- Deutsche Gesellschaft für Gynäkologie und Geburtshilfe
- Vlaamse Vereniging voor Obstetrie en Gynaecologie
- Federação das Sociedades Portuguesas de Obstetricia e Ginecologia
- Svensk Förening För Obstetrik & Gynekologi
- Hellenic Society of Obstetric and Gynecological Emergency
- Schweizerische Gesellschaft für Gynäkologie und Geburtshilfe
- Dansk Selskab for Obstetrik og Gynækologi
- Česká gynekologická a porodnická společnost
- European Midwives Association
Samenstelling werkgroep
Composition guideline development panel
An international panel for the development of the guidelines was formed in 2019. The panel consisted of representatives from all relevant medical disciplines that are involved in medical care for pregnant women.
All panel members have been officially delegated for participation in the guideline development panel by their (scientific) societies. The panel has developed the guidelines in the period from January 2022 until May 2024.
The guideline development panel is responsible for the entire text of this guideline.
All panel members have been officially delegated for participation in the guideline development panel by their scientific societies. The guideline development panel is responsible for the entire text of this guideline.
Members of the EAPM Standing Committee on Guideline Development (SCGD)
- J.J. Duvekot, obstetrician, Consultant Obstetrics and Gynaecology, Erasmus Medical Centre, Rotterdam, the Netherlands (chair)
- D. Ayres de Campos, obstetrician, Consultant Obstetrics and Gynaecology, Faculdade de Medicina, Lisbon, Portugal
- S. Brismar-Wendel, obstetrician, Consultant Obstetrics and Gynaecology, Danderyd Hospital, Stockholm, Sweden
- G. Daskalakis, obstetrician, Consultant Obstetrics and Gynaecology, National & Kapodistrian University, Athens, Creece.
- I. Dehaene, obstetrician, Consultant Obstetrics and Gynaecology, Ghent University Hospital Belgium
- M. Kacerovsky, obstetrician, Consultant Obstetrics and Gynaecology, University Hospital Hradec Kralove, Czech Republic
- S. Kehl, obstetrician, Consultant Obstetrics and Gynaecology, Erlangen University Hospital, Erlangen, Germany
- Julie Glavind, obstetrician, Consultant Obstetrics and Gynaecology, Aarhus University Hospital, Aarhus, Denmark
- A. Hamza, obstetrician, Consultant Obstetrics and Gynaecology, University Medical Center of Saarland, Homburg an der Saar, Germany
- M.A. Ledingham, obstetrician, Consultant Obstetrics and Gynaecology, the Queen Elizabeth Hospital Glasgow, UK
- B. Magowan, obstetrician, Consultant Obstetrics and Gynaecology, and previous Co-Chair UK RCOG Guidelines Committee, NHS Borders, Scotland, UK
Advisor committee (part of the committee since September 2023)
- E. Mestdagh, Director of Research and Development, AP University of Applied Sciences and Arts Antwerp, Belgium, Scientific advisor and guideline developer, KNOV, The Netherlands
- I. Wilsens, senior program coördinator development & innovation, KNOV, the Netherlands
- I. van Ee, patient representative, Dutch Patient Federation, the Netherlands
- P. Stenbäck, Midwife, MCHS, Program Director - Midwifery education. School of Business and Healthcare, Arcada University of Applied Sciences, Helsinki, Treasurer, board member EMA, European Midwifes Association, Belgium
- C. Matteo, European Midwives Association
Methodological support
- J. Tuijtelaars, advisor, Knowledge Institute of the Dutch Association of Medical Specialists
- J.H. van der Lee, senior advisor, Knowledge Institute of the Dutch Association of Medical Specialists
Belangenverklaringen
Declarations of interests
The Code for the prevention of improper influence due to conflicts of interest was followed.
The working group members have provided written statements about (financially supported) relations with commercial companies, organisations or institutions related to the subject matter of the guideline during the past three years. Furthermore, inquiries have been made regarding personal financial interests, interests due to personal relationships, interests related to reputation management, interest related to externally financed research and interests related to knowledge valorisation. The chair of the guideline development panel is informed about changes in interests during the development process. The declarations of interests are reconfirmed during the commentary phase. The declarations of interests can be requested at the administrative office of the Knowledge Institute of the Dutch Association of Medical Specialists and are summarised below.
|
Last name |
Principal position |
Ancillary position(s) |
Declared interests |
Action |
|
Duvekot (chair) |
Consultant Obstetrics and Gynaecology, Erasmus MC, Rotterdam |
Director 'Medisch Advies en Expertise Bureau Duvekot' - making expertises on medical calamities |
None |
None |
|
Dehaene |
Consultant Obstetrics and Gynaecology, Ghent University Hospital |
None |
None |
None |
|
Hamza |
Leading Consultant at the department of obstetrics and prenatal medicine Kantonspital Baden |
At the cantonal hospital I am involved in the clinical work, research and development. |
Inovolvement in research and development to improve maternofenal health.
|
None |
|
Ledingham |
Consultant in Maternal and Fetal Medicine, Queen Elizabeth Hospital, Glasgow |
Guideline developer for sign (scottisch intercollegiate guidelines group) |
None |
None |
|
Magowan |
Consultant Obstetrics and Gynaecology, and Co-Chair UK RCOG Guidelines Committee, NHS Borders, Scotland |
Previous Co-chair RCOG Guidelines committee |
None |
None |
|
Stenbäck |
Midwife, MCHS, Program Director - Midwifery education. School of Business and Healthcare. Arcada University of Applied Sciences, Helsinki. Treasurer, board member EMA, European Midwifes, Association, Belgium. |
Program director of Midwifery education, Arcada University of Applied Sciences, Helsinki. full time employment.
Treasurer of European Midwives Association, is a position of trust. EMA is a non-profit and non-governmental organisation of midwives, representing midwifery organisations and associations from the member states of the European Union (EU), the European Economic Area (EEA) and EU applicant countries and Council of Europe. |
None |
None |
|
Brismar Wendel |
MD, senior consulant, specialist in obstetrics and gynaecology. Department of Women's Health, Danderyd Hospital |
None |
None |
None |
|
Ayres de Campos |
Full Professor of Obstetrics and Gynecology. Medical School, University of Lisbon |
None |
None |
None |
|
Daskalakis |
Professor OB/GYN National and Kapodistrian University of Athens |
General obstetrics and Gynecology duties. |
None |
None |
|
Wilsens |
Senior program coördinator development & innovation, KNOV, the Netherlands |
None |
None |
None |
|
Glavind |
Senior Consultant in obstetrics, Aarhus University Hospital, Denmark |
Member of the Danish National Obstetrics Guideline Steering Comittee: Responsible for coordinating the process in developing, revising and approving Danish national guidelines in obstetrics including the planning of an annual national guideline meeting to discuss new and revised guidlines. This position is not remunerated. |
None |
None |
|
Kehl |
Delegate of the German Society of Perinatal Medicine and German Society of Gynecology and Obstetrics |
None |
None |
None |
|
Mestdagh |
Director of Research and Development - AP University of Applied Sciences and Arts Antwerp - Belgium |
Director of Research and development: |
None |
None |
|
Van Ee |
Dutch Patient federation – adviser patient perspective (fulltime) |
Psoriasis patiens Netherlands - coördinator patient participation and research (unpaid)
|
None |
None |
|
Kacerovsky |
Professor, University Hospital Hradec Kralove, Charles University, Faculty of Medicine in Hradec Kralove, Consultant in Maternal-Fetal Medicine, Hospital Most |
None |
None |
None |
Inbreng patiëntenperspectief
Representation of the patient perspective
Representatives of the Dutch Patient Federation provided review comments. The comments were discussed and where relevant incorporated by the guideline development panel.
This guideline accounts for everyone who gives birth, even though we speak of women and her/she in the guideline.
Implementatie
Implementation
Guideline implementation and practical applicability of the recommendations was taken into consideration during various stages of guideline development. Factors that may promote or hinder implementation of the guideline in daily practice were given specific attention.
The guideline is distributed digitally among all relevant professional groups. The guideline can also be downloaded from the Dutch guideline website: www.richtlijnendatabase.nl.
Werkwijze
Method
AGREE
This guideline has been developed conforming to the requirements of the report of Guidelines for Medical Specialists 3.0 by the advisory committee of the Quality Counsel. This report is based on the AGREE II instrument (Appraisal of Guidelines for Research & Evaluation II) (www.agreetrust.org)(Brouwers, 2010), a broadly accepted instrument in the international community and on the national quality standards for guidelines: “Guidelines for guidelines” (www.zorginstituutnederland.nl).
Identification of subject matter
Since this was a pilot project, the content of the questions and the support base in clinical practice was considered of less importance than the process of international collaboration and learning from each other. The panel made an inventory of clinical questions to be addressed based on their expert opinion and their knowledge of existing guidelines. The ‘Danish national clinical guideline concerning primiparous women with dystocia (lack of progress)’ published in 2015 was also used as a source of clinical questions.
Clinical questions and outcomes
The guideline development panel formulated definitive clinical questions and defined relevant outcome measures (both beneficial land harmful effects). The panel rated the outcome measures as critical, important and not important. Furthermore, where applicable, the panel defined relevant clinical differences.
Strategy for search and selection of literature
For the separate clinical questions, specific search terms were formulated and published scientific articles were searched for in (several) electronic databases. Furthermore, studies were scrutinized by cross-referencing for other included studies. The studies with potentially the highest quality of research were searched for first. The panel members selected literature in pairs (independently of each other) based on title and abstract. A second selection was performed based on full text. The databases, search terms and selection criteria are described in the modules containing the clinical questions.
No definitions were available at the time of performing the search. Therefore, definitions as used in the papers were used.
Quality assessment of individual studies
Individual studies were systematically assessed, based on methodological quality criteria that were determined prior to the search, so that risk of bias could be estimated. This is described in the “risk of bias” tables.
Summary of literature
The relevant research findings of all selected articles are shown in evidence tables. The most important findings in the literature are described in literature summaries. In case there were enough similarities between studies, the study data were pooled.
Grading quality of evidence and strength of recommendations
The strength of the conclusions of the scientific publications was determined using the GRADE-method. GRADE stands for Grading Recommendations Assessment, Development and Evaluation (see http://www.gradeworkinggroup.org/).
GRADE defines four gradations for the quality of scientific evidence: high, moderate, low or very low. These gradations provide information about the amount of certainty about the literature conclusions (http://www.guidelinedevelopment.org/handbook/).
The basic principles of the GRADE method are: formulating and prioritising clinical (patient) relevant outcome measures, a systematic review for each outcome measure, and appraisal of the evidence for each outcome measure based on the eight GRADE domains (domains for downgrading: risk of bias, inconsistency, indirectness, imprecision, and publication bias; domains for upgrading: dose-effect association, large effect, and residual plausible confounding).
GRADE distinguishes four levels for the quality of the scientific evidence: high, moderate, low and very low. These levels refer to the amount of certainty about the conclusion based on the literature, in particular the amount of certainty that the conclusion based on the literature adequately supports the recommendation (Schünemann, 2013; Hultcrantz, 2017).
|
GRADE |
Definition |
|
High |
|
|
Moderate |
|
|
Low |
|
|
Very low |
|
The limits of clinical decision making are very important in grading the evidence in guideline development according to the GRADE methodology (Hultcrantz, 2017). Exceedance of these limits would give rise to adaptation of the recommendation. All relevant outcome measures and considerations need to be taken into account to define the limits of clinical decision making. Therefore, the limits of clinical decision making are not one to one comparable to the minimal clinically relevant difference. In particular for interventions of low costs and without important drawbacks the limit of clinical decision making regarding the effectiveness of the intervention may be lower (i.e. closer to no effect) than the Minimal Clinically Important Difference (MCID) (Hultcrantz, 2017).
Considerations (evidence to decision)
Aspects such as expertise of working group members, patient preferences, costs, availability of facilities, and organisation of healthcare aspects are important to consider when formulating a recommendation. For each clinical question, these aspects are discussed in the paragraph Considerations, using a structured format based on the evidence-to-decision framework of the international GRADE Working Group (Alonso-Coello, 2016a; Alonso-Coello, 2016b). The evidence-to-decision framework is an integral part of the GRADE methodology.
Formulating recommendations
Recommendations provide an answer to the primary question and are based on the best scientific evidence available and the most important considerations. The level of scientific evidence and the importance given to considerations by the working group jointly determine the strength of the recommendation. In accordance with the GRADE method, a low level of evidence for conclusions in the systematic literature review does not rule out a strong recommendation, while a high level of evidence may be accompanied by weak recommendations. The strength of the recommendation is always determined by weighing all relevant arguments.
Knowledge gaps
During the development of this guideline, systematic searches were conducted for research contributing to answering the primary questions. For each primary question, the working group determined whether (additional) scientific research is desirable.
Commentary and authorisation phase
The draft guideline was subjected to commentaries by the scientific societies and patient organisations involved. The draft guideline was also submitted to members of the European Midwives Association (EMA). The comments were collected and discussed with the working group. The feedback was used to improve the guideline; afterwards the working group made the guideline definitive. The final version of the guideline was offered for authorization to the involved scientific societies and patient organisations and was authorized or approved, respectively.
Legal standing of guidelines
Guidelines are not legal prescriptions but contain evidence-based insights and recommendations that care providers should meet in order to provide high quality care. As these recommendations are primarily based on ‘general evidence for optimal care for the average patient’, care providers may deviate from the guideline based on their professional autonomy when they deem it necessary for individual cases. Deviating from the guideline may even be necessary in some situations. If care providers choose to deviate from the guideline, this should be done in consultation with the patient, where relevant. Deviation from the guideline should always be justified and documented.
Literature
Agoritsas T, Merglen A, Heen AF, Kristiansen A, Neumann I, Brito JP, Brignardello-Petersen R, Alexander PE, Rind DM, Vandvik PO, Guyatt GH. UpToDate adherence to GRADE criteria for strong recommendations: an analytical survey. BMJ Open 2017;7:e018593.
Alonso-Coello P, Schünemann HJ, Moberg J, Brignardello-Petersen R, Akl EA, Davoli M, Treweek S, Mustafa RA, Rada G, Rosenbaum S, Morelli A, Guyatt GH, Oxman AD; GRADE Working Group. GRADE Evidence to Decision (EtD) frameworks: a systematic and transparent approach to making well informed healthcare choices. 1: Introduction. BMJ 2016;353:i2016.
Alonso-Coello P, Oxman AD, Moberg J, Brignardello-Petersen R, Akl EA, Davoli M, Treweek S, Mustafa RA, Vandvik PO, Meerpohl J, Guyatt GH, Schünemann HJ; GRADE Working Group. GRADE Evidence to Decision (EtD) frameworks: a systematic and transparent approach to making well informed healthcare choices. 2: Clinical practice guidelines. BMJ. 2016 Jun 30;353:i2089.
Brouwers MC, Kho ME, Browman GP, Burgers JS, Cluzeau F, Feder G, Fervers B, Graham ID, Grimshaw J, Hanna SE, Littlejohns P, Makarski J, Zitzelsberger L; AGREE Next Steps Consortium. AGREE II: advancing guideline development, reporting and evaluation in health care. CMAJ. 2010 Dec 14;182(18):E839-42.
Hultcrantz M, Rind D, Akl EA, Treweek S, Mustafa RA, Iorio A, Alper BS, Meerpohl JJ, Murad MH, Ansari MT, Katikireddi SV, Östlund P, Tranæus S, Christensen R, Gartlehner G, Brozek J, Izcovich A, Schünemann H, Guyatt G. The GRADE Working Group clarifies the construct of certainty of evidence. J Clin Epidemiol. 2017 Jul;87:4-13.
Medisch Specialistische Richtlijnen 2.0 (2012). Adviescommissie Richtlijnen van de Raad Kwalitieit. http://richtlijnendatabase.nl/over_deze_site/over_richtlijnontwikkeling.html
Neumann I, Santesso N, Akl EA, Rind DM, Vandvik PO, Alonso-Coello P, Agoritsas T, Mustafa RA, Alexander PE, Schünemann H, Guyatt GH. A guide for health professionals to interpret and use recommendations in guidelines developed with the GRADE approach. J Clin Epidemiol. 2016 Apr;72:45-55. doi: 10.1016/j.jclinepi.2015.11.017. Epub 2016 Jan 6. Review. PubMed PMID: 26772609.
Schünemann H, Brożek J, Guyatt G, et al. GRADE handbook for grading quality of evidence and strength of recommendations. Updated October 2013. The GRADE Working Group, 2013. Available from http://gdt.guidelinedevelopment.org/central_prod/_design/client/handbook/handbook.html.
Schünemann HJ, Oxman AD, Brozek J, Glasziou P, Jaeschke R, Vist GE, Williams JW Jr, Kunz R, Craig J, Montori VM, Bossuyt P, Guyatt GH; GRADE Working Group. Grading quality of evidence and strength of recommendations for diagnostic tests and strategies. BMJ. 2008 May 17;336(7653):1106-10.
Schünemann, A Holger J [corrected to Schünemann, Holger J]. PubMed PMID: 18483053; PubMed Central PMCID: PMC2386626.
Wessels M, Hielkema L, van der Weijden T. How to identify existing literature on patients' knowledge, views, and values: the development of a validated search filter. J Med Libr Assoc. 2016 Oct;104(4):320-324.