Afdekken en fixeren van centraal veneuze katheters
Uitgangsvraag
Welke infectiepreventiemaatregelen dienen genomen te worden bij het afdekken en fixeren van een centraal veneuze katheter?
Aanbeveling
Alle typen centraal veneuze katheter
- Kies een materiaal voor het afdekken en fixeren van de katheter dat voldoet aan Europese wetgeving op het gebied van medische hulpmiddelen, waaronder de Verordening (EU) 2017/745, en de REACH-verordening (EG) 1907/2006.
Perifeer of centraal ingebrachte niet-getunnelde centraal veneuze katheter
- Fixeer de katheter bij voorkeur met een hechtingsvrij fixatiesysteem.
- Overweeg een alternatieve fixatiemethode wanneer op basis van patiënt karakteristieken de kans op dislocatie van de katheter groot is.
- Gebruik geen weefsellijm of tape om de katheter te fixeren.
- Dek de insteekplaats van de katheter bij voorkeur af met steriel polyurethaan afdekfolie.*
- Gebruik een steriel gaas met tape om de insteekplaats van de katheter af te dekken wanneer de patiënt overgevoelig is voor polyurethaan of veel transpireert.
- Overweeg het gebruik van steriel chloorhexidine gluconaat afdekfolie om de insteekplaats van de katheter af te dekken alleen wanneer de lokale incidentie van katheter-gerelateerde infecties hoger is dan op basis van vergelijkende (historisch of externe benchmark) surveillancegegevens verwacht, ondanks optimale naleving van de aanbevelingen in deze richtlijn.
- Vervang steriel polyurethaan afdekfolie en chloorhexidine gluconaat afdekfolie wanneer het loslaat en wanneer het zichtbaar verontreinigd of vochtig is.
- Vervang steriel gaas dagelijks.
- Vermijd langdurige blootstelling van steriel standaard polyurethaan afdekfolie aan water.
- Dek steriel gaas waterdicht af wanneer de patiënt gaat douchen.
* Standaard polyurethaan afdekfolie of polyurethaan afdekfolie met ‘border'.
Centraal ingebrachte getunnelde centraal veneuze katheter met vastgegroeide ‘cuff’
- Bespreek het al dan niet afdekken van de insteekplaats van de katheter met de patiënt (Samen Beslissen):
- afdekken is niet nodig vanuit infectiepreventieperspectief;
- afdekken kan een esthetisch voordeel zijn;
- afdekken kan de huid beschermen tegen wrijving door kleding;
- afdekken kan resulteren in irritatie van de huid door het afdekmateriaal;
- afdekken kan resulteren in het niet tijdig herkennen van vroege tekenen van infectie.
Poortkatheter
- Dek de insteekplaats van de katheter na aanprikken van het systeem bij voorkeur af met steriel standaard polyurethaan afdekfolie.
Overwegingen
Voor- en nadelen van de interventie en de kwaliteit van het bewijs
Er is literatuuronderzoek verricht naar de plaats van verschillende materialen en methoden voor het afdekken en fixeren van centraal veneuze katheters bij volwassen patiënten en kinderen (geen neonaten) in de medisch-specialistische zorg. Katheter-gerelateerde bloedbaaninfectie (KRBBI) en katheter-geassocieerde bloedbaaninfectie (KABBI) werden als cruciale uitkomstmaten gedefinieerd. Infecties van de insteekplaats, (trombo)flebitis, mortaliteit door alle oorzaken, KRBBI/KABBI-gerelateerde mortaliteit, huidirritatie of -schade, katheterfalen door alle oorzaken en katheter fixatie falen werden als belangrijke uitkomstmaten gedefinieerd. Een relatief risico kleiner dan 0.8 of groter dan 1.25 werd als klinisch relevant beschouwd voor alle uitkomstmaten, met uitzondering van mortaliteit. Voor mortaliteit werd een relatief risico kleiner dan 0.95 of groter dan 1.05 als klinisch relevant beschouwd.
In het literatuuronderzoek is geen onderscheid gemaakt tussen volwassen patiënten en kinderen, omdat de werkgroep voor het effect van verschillende materialen en methoden voor het afdekken en fixeren van centraal veneuze katheters op katheter-gerelateerde infecties geen verschil verwacht tussen volwassen patiënten en kinderen.
Materialen en methoden voor het afdekken van de katheter
Er werden vijf gerandomiseerde klinische studies geïncludeerd die steriel gaas en tape vergeleken met standaard polyurethaan (SPU) afdekfolie voor het afdekken van centraal veneuze katheters bij patiënten in het ziekenhuis.
- De cruciale uitkomstmaat KRBBI werd gerapporteerd, maar het effect van steriel gaas en tape kon niet worden geschat (1 studie, bewijskracht zeer laag).
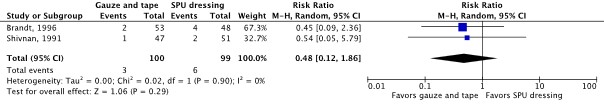
- Er was een klinisch relevant verschil voor de belangrijke uitkomstmaat infectie van de insteekplaats in het voordeel van steriel gaas en tape (RR 0.48; 95% CI 0.12 tot 1.86; 2 studies; bewijskracht zeer laag), een klinisch relevant verschil voor de belangrijke uitkomstmaat huidirritatie of -schade in het voordeel van SPU afdekfolie (RR 6.60; 95% CI 0.95 tot 45.75; 1 studie; bewijskracht laag), en een klinisch relevant verschil voor de belangrijke uitkomstmaat katheter fixatie falen in het voordeel van steriel gaas en tape (RR 0.76; 95% CI 0.25 tot 2.31; 2 studies; bewijskracht zeer laag).
- Er was een niet klinisch relevant verschil voor de belangrijke uitkomstmaten (trombo)flebitis (1 studie; bewijskracht zeer laag), en katheterfalen door alle oorzaken (1 studie; bewijskracht zeer laag).
- De andere belangrijke uitkomstmaten werden niet gerapporteerd.
De overall bewijskracht, gebaseerd op de cruciale uitkomstmaat KRBBI, was zeer laag.
We concluderen dat voor patiënten in het ziekenhuis met een centraal veneuze katheter het bewijs voor een effect van steriel gaas en tape op KRBBI zeer onzeker is; dat steriel gaas en tape met lage zekerheid resulteert in meer huidirritatie of -schade; dat het bewijs voor een effect van steriel gaas en tape op infectie van de insteekplaats, (trombo)flebitis, katheterfalen door alle oorzaken, en katheter fixatie falen zeer onzeker is; en dat er geen bewijs is gevonden voor een effect van steriel gaas en tape op de andere uitkomstmaten.
Er werden acht gerandomiseerde klinische studies geïncludeerd die chloorhexidine gluconaat (CHG) afdekfolie vergeleken met SPU afdekfolie voor het afdekken van centraal veneuze katheters bij patiënten in het ziekenhuis.
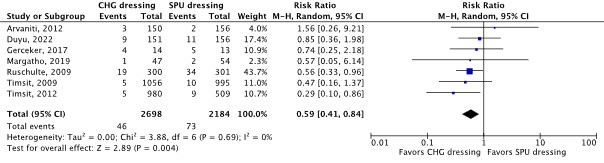
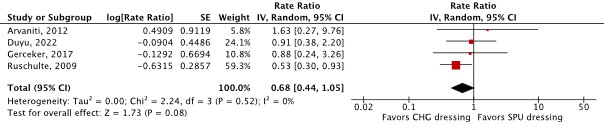
- Er was een klinisch relevant verschil voor de cruciale uitkomstmaat KRBBI in het voordeel van CHG afdekfolie (RR 0.59; 95% CI 0.41 tot 0.84; 7 studies; bewijskracht laag), een klinisch relevant verschil voor de cruciale uitkomstmaat KRBBI per 1,000 katheterdagen in het voordeel van CHG afdekfolie (RR 0.68; 95% CI 0.44 tot 1.05; 4 studies; bewijskracht laag), en een klinisch relevant verschil voor de belangrijke uitkomstmaat huidirritatie of -schade in het voordeel van SPU afdekfolie (RR 2.41; 95% CI 0.44 tot 13.23; 2 studies; bewijskracht zeer laag).
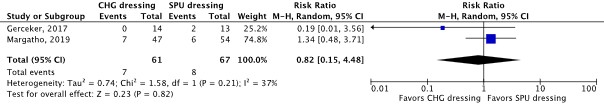
- Er was een niet klinisch relevant verschil voor de belangrijke uitkomstmaat infectie van de insteekplaats (2 studies; bewijskracht zeer laag).
- Voor de belangrijke uitkomstmaat mortaliteit door alle oorzaken waren de effecten tegengesteld (2 studies; bewijskracht zeer laag).
- De andere belangrijke uitkomstmaten werden niet gerapporteerd.
De overall bewijskracht, gebaseerd op de cruciale uitkomstmaten KRBBI en KRBBI per 1,000 katheterdagen, was laag.
We concluderen dat CHG afdekfolie voor patiënten in het ziekenhuis met een centraal veneuze katheter met lage zekerheid resulteert in minder KRBBI en minder KRBBI per 1,000 katheterdagen; dat het bewijs voor een effect van CHG afdekfolie op infectie van de insteekplaats, mortaliteit door alle oorzaken, en huidirritatie of -schade zeer onzeker is; en dat er geen bewijs is gevonden voor een effect van CHG afdekfolie op de andere uitkomstmaten.
Er werd één gerandomiseerde klinische studie geïncludeerd die polyhexamethyleen biguanide (PHMB) afdekfolie vergeleek met SPU afdekfolie voor het afdekken van centraal veneuze katheters bij patiënten in het ziekenhuis.
- De cruciale uitkomstmaat KABBI (1 studie) en de belangrijke uitkomstmaat infectie van de insteekplaats (1 studie) werden gerapporteerd, maar het effect van PHMB afdekfolie kon niet worden geschat (bewijskracht zeer laag).
- De andere belangrijk uitkomstmaten werden niet gerapporteerd.
De overall bewijskracht, gebaseerd op de cruciale uitkomstmaat KABBI, was zeer laag.
We concluderen dat voor patiënten in het ziekenhuis met een centraal veneuze katheter het bewijs voor een effect van PHMB afdekfolie op KABBI en infectie van de insteekplaats zeer onzeker is; en dat er geen bewijs is gevonden voor een effect van PHMB afdekfolie op de andere uitkomstmaten.
Materialen en methoden voor het fixeren van de katheter
Er werden geen studies gevonden die SPU afdekfolie vergeleken met een hechting en polyurethaan (PU) afdekfolie voor het fixeren (en afdekken) van centraal veneuze katheters bij patiënten in het ziekenhuis.
Er werden geen studies gevonden die polyurethaan afdekfolie met border (BPU) afdekfolie vergeleken met een hechting en PU afdekfolie bij patiënten in het ziekenhuis met een centraal veneuze katheter.
Er werden geen studies gevonden die geïntegreerd fixatie (IS) afdekfolie vergeleken met een hechting en PU afdekfolie voor het fixeren (en afdekken) van centraal veneuze katheters bij patiënten in het ziekenhuis.
Er werden vijf gerandomiseerde klinische studies geïncludeerd die een hechtingsvrij fixatie systeem (SSD) en PU afdekfolie vergeleken met een hechting en PU afdekfolie voor het fixeren (en afdekken) van centraal veneuze katheters bij patiënten in het ziekenhuis.
- Er was een klinisch relevant verschil voor de cruciale uitkomstmaat KRBBI in het voordeel van SSD en PU afdekfolie (RR 0.13; 95% CI 0.02 tot 0.98; 1 studie; bewijskracht laag), een klinisch relevant verschil voor de cruciale uitkomstmaat KRBBI poer 1,000 katheterdagen in het voordeel van SSD en PU afdekfolie (RR 0.13; 95% CI 0.02 tot 1.05; 1 studie; bewijskracht laag), een klinisch relevant verschil voor de belangrijke uitkomstmaat infectie van de insteekplaats in het voordeel van SSD en PU afdekfolie (RR 0.39; 95% CI 0.19 tot 0.80; 4 studies; bewijskracht laag), en een klinisch relevant verschil voor de belangrijke uitkomstmaat katheterfalen door alle oorzaken in het voordeel van hechting en PU dressing (RR 2.00; 95% CI 0.38 tot 10.47; 1 studie; bewijskracht zeer laag).
- Er was een niet klinisch relevant verschil voor de belangrijke uitkomstmaten huidirritatie of -schade (2 studies; bewijskracht zeer laag) en katheter fixatie falen (5 studies; bewijskracht zeer laag).
- De belangrijke uitkomstmaat mortaliteit door alle oorzaken werd gerapporteerd, maar het effect van SSD en PU afdekfolie kon niet worden geschat (2 studies; bewijskracht zeer laag).
- De andere belangrijke uitkomstmaten werden niet gerapporteerd.
De overall bewijskracht, gebaseerd op de cruciale uitkomstmaten KRBBI en KRBBI per 1,000 katheterdagen, was laag.
We concluderen dat voor patiënten in het ziekenhuis met een centraal veneuze katheter SSD en PU afdekfolie met lage zekerheid resulteert in minder KRBBI, minder KRBBI per 1,000 katheterdagen en minder infecties van de insteekplaats; dat het bewijs voor een effect van SSD en PU afdekfolie op mortaliteit door alle oorzaken, huidirritatie of -schade, katheterfalen door alle oorzaken, en katheter fixatie falen zeer onzeker is; en dat er geen bewijs is gevonden voor een effect van SSD en PU afdekfolie op de andere uitkomstmaten.
Er werden geen studies gevonden die een subcutaan verankeringssysteem en PU afdekfolie vergeleken met een hechting en PU afdekfolie voor het fixeren (en afdekken) van centraal veneuze katheters bij patiënten in het ziekenhuis.
Er werden twee gerandomiseerde klinische studies geïncludeerd die weefsellijm en PU afdekfolie vergeleken met een hechting en PU afdekfolie voor het fixeren (en afdekken) van centraal veneuze katheters bij patiënten in het ziekenhuis.
- De cruciale uitkomstmaten KRBBI (1 studie) en KRBBI per 1,000 katheterdagen (1 studie) werden gerapporteerd, maar het effect van weefsellijm en PU afdekfolie kon niet worden geschat (bewijskracht zeer laag).
- Er was een klinisch relevant verschil voor de belangrijke uitkomstmaat huidirritatie of -schade in het voordeel van weefsellijm en PU afdekfolie (RR 0.58; 0.09 tot 3.61; 2 studies; bewijskracht zeer laag), een klinisch relevant verschil voor de belangrijk uitkomstmaat katheterfalen door alle oorzaken in het voordeel van hechting en PU afdekfolie (RR 4.78; 0.94 tot 24.32; 2 studies; bewijskracht laag), en een klinisch relevant verschil voor de belangrijke uitkomstmaat katheter fixatie falen in het voordeel van hechting en PU afdekfolie (RR 4.78; 0.94 tot 24.32; 2 studies; bewijskracht laag).
- De belangrijke uitkomstmaten infectie van de insteekplaats (2 studies) en mortaliteit door alle oorzaken (1 studie) werden gerapporteerd, maar het effect van weefsellijm en PU afdekfolie kon niet worden geschat (bewijskracht zeer laag).
- De andere belangrijke uitkomstmaten werden niet gerapporteerd.
De overall bewijskracht, gebaseerd op de cruciale uitkomstmaat KRBBI, was zeer laag.
We concluderen dat voor patiënten in het ziekenhuis met een centraal veneuze katheter het bewijs voor een effect van weefsellijm en PU afdekfolie op KRBBI zeer onzeker is; dat weefsellijm en PU afdekfolie met lage zekerheid resulteert in meer katheterfalen door alle oorzaken en meer katheter fixatie falen; dat het bewijs voor een effect van weefsellijm en PU afdekfolie op infectie van de insteekplaats, mortaliteit door alle oorzaken, en huidirritatie of -schade zeer onzeker is; en dat er geen bewijs is gevonden voor een effect van weefsellijm en PU afdekfolie op de andere uitkomstmaten.
Er werden vier gerandomiseerde klinische studies geïncludeerd die weefsellijm en hechting en PU afdekfolie vergeleken met een hechting en PU afdekfolie voor het fixeren (en afdekken) van centraal veneuze katheters bij patiënten in het ziekenhuis.
- De cruciale uitkomstmaat KABBI (3 studies) en KABBI per 1,000 katheterdagen (1 studie) werden gerapporteerd, maar het effect van weefsellijm en hechting en PU afdekfolie kon niet worden geschat (bewijskracht zeer laag).
- Er was een klinisch relevant verschil voor de belangrijke uitkomstmaat katheterfalen door alle oorzaken in het voordeel van hechting en PU afdekfolie (RR 1.61; 95% CI 0.50 tot 5.18; 3 studies; bewijskracht zeer laag).
- Er was een niet klinisch relevant verschil voor de belangrijke uitkomstmaten huidirritatie of -schade (3 studies; bewijskracht zeer laag), en katheter fixatie falen (3 studies; bewijskracht zeer laag).
- De belangrijke uitkomstmaten infectie van de insteekplaats (3 studies) en mortaliteit door alle oorzaken (3 studies) werden gerapporteerd, maar het effect van weefsellijm en hechting en PU afdekfolie kon niet worden geschat (bewijskracht zeer laag).
- De andere belangrijke uitkomstmaten werden niet gerapporteerd.
De overall bewijskracht, gebaseerd op de cruciale uitkomstmaten KABBI en KABBI per 1,000 katheterdagen, was zeer laag.
We concluderen dat voor patiënten in het ziekenhuis met een centraal veneuze katheter het bewijs voor een effect van weefsellijm en PU afdekfolie op KABBI en KABBI per 1,000 katheterdagen zeer onzeker is; dat het bewijs voor een effect van weefsellijm en hechting en PU afdekfolie op infectie van de insteekplaats, mortaliteit door alle oorzaken, huidirritatie of -schade, katheterfalen door alle oorzaken, en katheter fixatie falen zeer onzeker is; en dat er geen bewijs is gevonden voor een effect van weefsellijm en hechting en PU afdekfolie op de andere uitkomstmaten.
Er werd één gerandomiseerde klinische studie geïncludeerd die tape en PU afdekfolie vergeleek met een hechting en PU afdekfolie voor het fixeren (en afdekken) van centraal veneuze katheters bij patiënten in het ziekenhuis.
- De cruciale uitkomstmaat KRBBI/KABBI werd niet gerapporteerd.
- Er was een klinisch relevant verschil voor de belangrijke uitkomstmaat katheter fixatie falen in het voordeel van hechting en PU afdekfolie (RR 5.84; 95% CI 1.40 tot 24.35; 1 studie; bewijskracht laag).
- De andere belangrijke uitkomstmaten werden niet gerapporteerd.
De overall bewijskracht, gebaseerd op de cruciale uitkomstmaat KRBBI/KABBI, kon niet worden bepaald.
We concluderen dat er voor patiënten in het ziekenhuis met een centraal veneuze katheter geen bewijs is gevonden voor een effect van tape en PU afdekfolie op KRBBI/KABBI; dat tape en PU afdekfolie met lage zekerheid resulteert in meer katheter fixatie falen; en dat er geen bewijs is gevonden voor een effect van tape en PU afdekfolie op de andere uitkomstmaten.
Er werd één gerandomiseerde klinische studie geïncludeerd die nietje en PU afdekfolie vergeleek met een hechting en PU afdekfolie voor het fixeren (en afdekken) van centraal veneuze katheters bij patiënten in het ziekenhuis.
- De cruciale uitkomstmaat KRBBI/KABSI werd niet gerapporteerd.
- De belangrijke uitkomstmaat katheter fixatie falen werd gerapporteerd, maar het effect van nietje en PU afdekfolie kon niet worden geschat (1 studie; bewijskracht zeer laag).
- De andere belangrijke uitkomstmaten werden niet gerapporteerd.
De overall bewijskracht, gebaseerd op de cruciale uitkomstmaat KRBBI/KABBI, kon niet worden bepaald.
We concluderen dat er voor patiënten in het ziekenhuis met een centraal veneuze katheter geen bewijs is gevonden voor een effect van nietje en PU afdekfolie op KRBBI/KABBI; dat het bewijs voor een effect van nietje en PU afdekfolie op katheter fixatie falen zeer onzeker is; en dat er geen bewijs is gevonden voor een effect van nietje en PU afdekfolie op de andere uitkomstmaten.
Overige effecten van de interventie
PU afdekfolie
PU afdekfolie (standaard of met ‘border’) is een steriele, semipermeabele, transparante folie die de centraal veneuze katheter afdekt en (mede) fixeert. Dit betekent dat de insteekplaats niet aanvullend hoeft te worden afgedekt als de patiënt (kortdurend) gaat douchen. Langdurige blootstelling van de folie aan water wordt afgeraden vanwege het risico op loslaten van de folie wanneer de huid verweekt. Bij douchen dient daarnaast rekening te worden gehouden met de temperatuurbestendigheid van de gebruikte materialen.
Het gebruik van PU afdekfolie heeft als voordeel dat de insteekplaats kan worden geïnspecteerd zonder het afdekmateriaal te verwijderen. Nadeel is dat sommige patiënten overgevoelig zijn voor polyurethaan, en dat semipermeabele transparante afdekfolies minder geschikt zijn voor patiënten die veel transpireren. SPU afdekfolie moeten worden vervangen als het loslaat, en wanneer het zichtbaar verontreinigd of vochtig is.
CHG afdekfolie
Het gebruik van CHG afdekfolie kan bijdragen aan het ontstaan van resistentie tegen CHG, maar kan volgens recent onderzoek ook leiden tot het ontstaan van kruisresistentie tegen klinisch belangrijke antibacteriële en antifungale middelen (Abbood, 2023; Horner, 2012; Kampf, 2018, Kampf, 2019A, Kampf, 2019B; Spettel, 2025). De klinische impact van CHG resistentie is nog onduidelijk. Datzelfde geldt voor de bijdrage van CHG gebruik aan de ontwikkeling van antimicrobiële resistentie. Echter, het mogelijk ontstaan van antimicrobiële resistentie als ongewenst effect van het gebruik van desinfectantia heeft recent geleid tot aanbevelingen om het gebruik van desinfectantia, waaronder CHG, te beperken tot die situaties waarin meerwaarde van het gebruik is aangetoond (Gezondheidsraad, 2016). Bij routinematig gebruik van CHG afdekfolies is alertheid op het ontstaan van antimicrobiële resistentie aangewezen.
Gezien een mogelijk effect van routinematig gebruik van CHG afdekfolie op het ontstaan van CHG resistentie en kruisresistentie tegen klinisch belangrijke antibacteriële en antifungale middelen adviseert de werkgroep om het gebruik van CHG afdekfolie alleen te overwegen wanneer de lokale incidentie van katheter-gerelateerde infecties hoger is dan op basis van vergelijkende surveillancegegevens verwacht, ondanks optimale naleving van de aanbevelingen in deze richtlijn. Surveillance gegevens kunnen worden vergeleken met eerdere (historische) gegevens van de eigen instelling dan wel met een externe regionale of landelijke benchmark. De meerwaarde van het gebruik van CHG afdekfolie in dergelijke situaties is mede afhankelijk van eventueel andere maatregelen die tegelijkertijd worden genomen.
CHG afdekfolie moeten worden vervangen als het loslaat, en wanneer het zichtbaar verontreinigd of vochtig is.
Steriel gaas en tape
Het gebruik van steriel gaas en tape om de katheter af te dekken heeft een plaats bij overgevoeligheid voor polyurethaan en bij patiënten die veel transpireren. Steriel gaas moet dagelijks worden vervangen en waterdicht worden afgedekt wanneer de patiënt gaat douchen.
Fixatiestrategie
Voor patiënten met motorische onrust en kinderen kan het wenselijk zijn om aanvullende of alternatieve fixatiemethoden te gebruiken om (partiele) dislocatie van de centraal veneuze katheter te voorkomen. Verlies van vasculaire toegang, onderbreking van vasoactieve medicatie of extravasatie van medicatie of parenterale voeding kunnen ernstige consequenties hebben. Daarnaast verhoogt dislocatie ook het infectierisico. De keuze voor een passende fixatiestrategie is in deze gevallen maatwerk en afhankelijk van patiëntkarakteristieken, omstandigheden en (lokaal) beschikbare materialen. Bij de keuze voor een fixatiestrategie is het van belang dat de insteekplaats geïnspecteerd kan worden, dat de perfusie distaal niet wordt belemmerd, en dat de gebruikte methode comfortabel is voor de patiënt en niet leidt tot drukplekken.
Vastgegroeide ‘cuff’
De insteekplaats van een centraal ingebrachte getunnelde centraal veneuze katheter met vastgegroeide ‘cuff’ hoeft vanuit infectiepreventieperspectief niet te worden afgedekt omdat de huid is genezen. Wel kunnen er andere redenen zijn om de insteekplaats af te dekken, zoals het beschermen van de huid tegen wrijving door kleding of het aan het zicht onttrekken van de insteekplaats om esthetische redenen. Echter, het afdekken van de insteekplaats kan ook nadelen hebben, zoals irritatie van de huid door het afdekmateriaal, en het niet tijdig herkennen van vroege tekenen van infectie door het aan het zicht onttrekken van de insteekplaats. Het advies is dan ook om het al dan niet afdekken van de insteekplaats van de katheter te bespreken met de patiënt (Samen Beslissen).
Poortkatheter
De insteekplaats van de poortkatheter hoeft niet te worden afgedekt, omdat er geen open wond is. Wel heeft het de voorkeur om de insteekplaats na aanprikken van het systeem af te dekken met standaard polyurethaan afdekfolie.
Internationale richtlijnen
De richtlijnen van Centers for Disease Prevention and Control (CDC) (O’Grady, 2017), International Nursing Society (INS) (No authors listed, 2021), Society for Healthcare Epidemiology of America, Infectious Disease Society of America and Association for Professionals in Infection Control and Epidemiology (SHEA/IDSA/APIC) (Buetti, 2022),en World Health Organization (WHO) (No authors listed, 2024) zijn geraadpleegd (Tabel 1).
CDC en INS bevelen het gebruik van steriel gaas of steriel transparant semipermeabel afdekfolie aan voor het afdekken van centraal veneuze katheters. WHO adviseert het gebruik van een occlusief afdekfolie voor perifeer ingebrachte centraal veneuze katheters. CDC, en INS adviseren het gebruik van CHG afdekfolie voor niet-getunnelde katheters bij volwassenen. INS adviseert CHG afdekfolie ook voor andere centraal veneuze katheters wanneer andere infectiepreventiemaatregelen niet effectief zijn. CDC en INS doen geen aanbeveling over het gebruik van CHG afdekfolie bij kinderen. SHEA/IDSA/APIC adviseren het gebruik van CHG afdekfolie voor volwassenen en kinderen (geen neonaten) voor alle centraal veneuze katheters, met de kanttekening dat de effectiviteit voor getunnelde katheters met genezen insteekplaats de effectiviteit niet is aangetoond. Zolang de wond lekt is het advies van CDC en INS om een steriel gaas te gebruiken. SHEA/IDSA/APIC doet geen aanbeveling over de indicaties voor het gebruik van steriel gaas.
CDC, INS en SHEA/IDSA/APIC adviseren om afdekmateriaal te vervangen wanneer het loslaat, en wanneer het zichtbaar verontreinigd of vochtig is. INS adviseert om transparant semipermeabel afdekfolie ook te vervangen wanneer de huid onder folie beschadigd is en steriel gaas wanneer inspectie van de huid on het gaas nodig is. CDC om de katheter en insteekplaats niet onder te dompelen in water, en de insteekplaats van de katheter en de connector waterdicht af te dekken wanneer de patiënt gaat douchen. INS adviseert steriel gaas tenminste iedere twee dagen te vervangen, en transparant semipermeabel afdekfolie tenminste iedere zeven dagen; CDC en SHEA/IDSA/APIC adviseren dit alleen voor niet-getunnelde centraal veneuze katheters. Voor getunnelde centraal veneuze katheters en poortkatheters adviseert CDC om transparant semipermeabel afdekfolie niet vaker dan eens per zeven dagen te vervangen, en te gebruiken totdat de insteekplaats is genezen. CDC en SHEA/IDSA/APIC doen geen aanbeveling over de noodzaak om getunnelde katheters met ‘cuff’ af te dekken; INS geeft aan dat afdekken van getunnelde katheters wellicht niet nodig is. WHO doet geen aanbeveling over het vervangen van het afdekmateriaal.
Het CDC adviseert om de katheter aanvullend te fixeren met een hechtingsvrij fixatie systeem; INS een geïntegreerd fixatie afdekfolie, een subcutaan verankeringssysteem, een hechtingsvrij fixatie systeem of weefsellijm. INS ontraadt het gebruik van een hechting, niet-steriel tape, of een rol verband voor het fixeren van de katheter. SHEA/IDSA/APIC en WHO doen geen aanbeveling over het aanvullend fixeren van de katheter.
Tabel 1. Internationale richtlijnen voor de preventie van katheter-gerelateerde infecties.
|
Richtlijn |
Aanbeveling(en) |
|
CDC (O’Grady, 2017) |
|
|
INS (No authors listed, 2021)1,2 |
|
|
SHEA/IDSA/APIC (Buetti, 2022) |
|
|
WHO (No authors listed, 2024) |
|
APIC = Association for Professionals in Infection Control; ASD = adhesive securement device (NL: hechtingsvrij fixatie systeem); CABSI = catheter-associated bloodstream infection (NL: katheter-geassocieerde bloedbaaninfectie); CDC = Centers for Disease Prevention and Control; CVAD = central venous access device (NL: centraal veneuze katheter); CRBSI = catheter-related bloodstream infection (NL: katheter-gerelateerde bloedbaaninfectie); CVC = central venous catheter (NL: centraal veneuze katheter); FDA = Food and Drug Administration; IDSA = Infectious Diseases Society of America ; INS = International Nursing Society; ISD = integrated securement device (NL: geïntegreerd fixatie afdekfolie); PICC = peripherally inserted central venous catheter (NL: perifeer ingebrachte centraal veneuze katheter); SASS = subcutaneous anchor securement system (NL: subcutaan verankeringssysteem); SHEA = Society for Healthcare Epidemiology of America; TA = tissue adhesive; TSM = transparent semipermeable membrane (NL: transparant semipermeabel afdekfolie); VAD = vascular access device (NL: intravasculaire katheter); WHO = World Health Organization
1 De INS-richtlijn is ontwikkeld met financiële ondersteuning van de industrie; ook waren auteurs niet vrij van persoonlijke financiële belangen.
2 Er wordt niet verwezen naar de in 2024 verschenen update van de INS-richtlijn, omdat deze niet publiek beschikbaar is.
Wettelijke kaders
Materialen voor het afdekken en fixeren van centraal veneuze katheters zijn medische hulpmiddelen en moeten als zodanig voldoen aan Europese wetgeving op het gebied van medische hulpmiddelen, waaronder de Verordening (EU) 2017/745, en de REACH-verordening (EG) 1907/2006.
Waarden en voorkeuren van patiënten (en evt. hun verzorgers)
Er is geen systematisch onderzoek verricht naar de waarden en voorkeuren van patiënten met betrekking tot het afdekken en fixeren van centraal veneuze katheters.
In het algemeen kan gezegd worden dat de preventie van katheter-gerelateerde infecties past binnen de huidige maatstaven voor passende zorg. Standaardisatie en naleving van aanbevelingen over het afdekken en fixeren van centraal veneuze katheters is voor patiënten belangrijk omdat dit de kans op infecties niet wegneemt, maar wel vermindert.
Bij de keuze van materialen en methoden voor het afdekken en fixeren van centraal veneuze katheters is het belangrijk rekening te houden met effecten op de mobiliteit van de patiënt en eventuele allergieën voor gebruikte materialen, zoals bijvoorbeeld polyurethaan overgevoeligheid.
Het gebruik van hechtingsvrije fixatiesystemen voor het fixeren van centraal veneuze katheters is naar verwachting prettiger voor de patiënt dan het gebruik van een hechting door de huid.
Kosten (middelenbeslag)
Er is geen systematisch literatuuronderzoek verricht naar de kosteneffectiviteit van verschillende materialen en methoden voor het afdekken en fixeren van centraal veneuze katheters.
In het algemeen kan gezegd worden dat katheter-gerelateerde infecties gepaard gaan met meer zorgkosten (Baier, 2020; Patel 2019; Pu, 2020; Ullman 2022). Het voorkómen van deze complicaties door standaardisatie en naleving van aanbevelingen over het afdekken en fixeren van centraal veneuze katheters resulteert naar verwachting in een vermindering van zorgkosten.
SPU afdekfolie is duurder in aanschaf dan steriel gaas en tape maar gaat langer mee, wat de extra materiaal kosten beperkt en personeelskosten vermindert. CHG afdekfolie is over het algemeen duurder in aanschaf dan SPU afdekfolie. Hechtingsvrije fixatiesystemen voor het fixeren van centraal veneuze katheters zijn naar verwachting duurder in aanschaf dan hechtingen.
Gelijkheid ((health) equity)
De werkgroep voorziet voor aanbevelingen over materialen en methoden voor het afdekken en fixeren van centraal veneuze katheters geen effect op de gezondheidsgelijkheid.
Aanvaardbaarheid
Ethische aanvaardbaarheid
De werkgroep voorziet voor aanbevelingen over materialen en methoden voor het afdekken en fixeren van centraal veneuze katheters geen ethische bezwaren.
Duurzaamheid
Er is geen systematisch literatuuronderzoek verricht naar duurzaamheid van verschillende materialen en methoden voor het afdekken en fixeren van centraal veneuze katheters.
Het is belangrijk een afweging te maken tussen de noodzaak van het gebruik van verschillende materialen en methoden voor het afdekken en fixeren van centraal veneuze katheters enerzijds en duurzaamheid anderzijds. Deze afweging moet vanuit infectiepreventieoogpunt verantwoord gebeuren en kan verschillen per zorgsetting. Uitgangspunt hierbij is dat duurzaamheid niet ten koste mag gaan van patiëntveiligheid.
In het algemeen kan gezegd worden dat de preventie van katheter-gerelateerde infecties leidt tot minder gebruik van medische hulpmiddelen, minder gebruik van antibiotica en de daarmee gepaard gaande ontwikkeling van antibioticaresistentie.
Eventuele voordelen van het gebruik van CHG-bevattende producten moeten worden afgewogen tegen het feit dat CHG moeilijk afbreekbaar is, waardoor het zich kan ophopen in het milieu en langere tijd schadelijk kan blijven. CHG is toxisch voor waterorganismen en kan niet-specifieke schade aanrichten aan microbiële populaties in het milieu. Daarnaast is het onduidelijk wat het effect is van CHG op het menselijk en dierlijk microbioom en op microbiomen in het milieu (Gezondheidsraad, 2016).
Haalbaarheid
De werkgroep voorziet voor aanbevelingen over materialen en methoden voor het afdekken en fixeren van centraal veneuze katheters geen belemmeringen voor de uitvoerbaarheid.
Aanbeveling – Afdekken en fixeren
Rationale van de aanbeveling: weging van argumenten voor en tegen de interventies
De aanbevelingen over het afdekken en fixeren van centraal veneuze katheters bij volwassen patiënten en kinderen (geen neonaten) in de medisch-specialistische zorg zijn gebaseerd op het systematisch literatuuronderzoek, de eerdere WIP-richtlijn Flebitis en bloedbaaninfecties door intraveneuze infuuskatheters, internationale richtlijnen, ondersteunende literatuur, en expert opinie. Er wordt geen onderscheid gemaakt tussen volwassen patiënten en kinderen.
Het is belangrijk dat centraal veneuze katheters beschermd worden tegen bacteriële besmetting vanuit de omgeving en op adequate wijze aan de huid worden bevestigd om (partiële) dislocatie van de katheter te voorkomen.
Materialen voor het afdekken en fixeren van centraal veneuze katheters zijn medische hulpmiddelen en moeten als zodanig voldoen aan Europese wetgeving op het gebied van medische hulpmiddelen, waaronder de Verordening (EU) 2017/745, en de REACH-verordening (EG) 1907/2006.
Perifeer of centraal ingebrachte niet-getunnelde centraal veneuze katheter
Er is onvoldoende bewijs gevonden voor een belangrijk effect van afdekken van de katheter met steriel gaas en tape op katheter-gerelateerde infecties vergeleken met afdekken met standaard polyurethaan afdekfolie. Wel laat wetenschappelijk bewijs van lage kwaliteit zien dat afdekken met steriel gaas en tape resulteert in meer huidirritatie of -schade.
Er is onvoldoende bewijs gevonden voor een belangrijk effect van afdekken van de katheter met polyhexamethyleen biguanide afdekfolie op katheter-gerelateerde infecties vergeleken met afdekken met standaard polyurethaan afdekfolie.
Wetenschappelijk bewijs van lage kwaliteit laat zien dat afdekken van de katheter met chloorhexidine gluconaat afdekfolie resulteert in minder katheter-gerelateerde bloedbaaninfecties, maar in meer huidirritatie of -schade dan afdekken met standaard polyurethaan afdekfolie. Routinematig gebruik van chloorhexidine gluconaat afdekfolie kan mogelijk bijdragen aan het ontstaan van chloorhexidine resistentie en kruisresistentie tegen klinisch belangrijke antibacteriële en antifungale middelen. Daarnaast is chloorhexidine gluconaat afdekfolie naar verwachting duurder dan standaard polyurethaan afdekfolie en de meerwaarde van het gebruik is mede afhankelijk van eventueel andere infectiepreventiemaatregelen die tegelijkertijd worden genomen.
Er zijn geen studies gevonden die het effect van fixeren van de katheter met alleen polyurethaan afdekfolie of geïntegreerd fixatie afdekfolie op katheter-gerelateerde infecties hebben vergeleken met fixeren met een hechting en polyurethaan afdekfolie.
Er is onvoldoende bewijs gevonden voor een belangrijk effect van fixeren van de katheter met tape op katheter-gerelateerde infecties vergeleken met fixeren met een hechting. Wel laat wetenschappelijk bewijs van lage kwaliteit zien dat het gebruik van tape resulteert in meer katheter fixatie falen.
Er is onvoldoende bewijs gevonden voor een belangrijk effect van fixeren van de katheter met een nietje op katheter-gerelateerde infecties vergeleken met fixeren met een hechting.
Er is onvoldoende bewijs gevonden voor een belangrijk effect van fixeren van de katheter met weefsellijm (met of zonder hechting) vergeleken met fixeren met een hechting. Wel laat wetenschappelijk bewijs van lage kwaliteit zien dat het gebruik van weefsellijm resulteert in minder katheterfalen door alle oorzaken en minder katheter fixatie falen.
Er zijn geen studies gevonden die het effect van fixeren van de katheter met een subcutaan verankeringssysteem op katheter-gerelateerde infecties hebben vergeleken met een hechting.
Wetenschappelijk bewijs van lage kwaliteit laat zien dat fixeren van de katheter met een hechtingsvrij fixatiesysteem resulteert in minder katheter-gerelateerde bloedbaaninfecties en minder infecties van de insteekplaats dan fixeren met een hechting. Het gebruik van hechtingsvrije fixatiesystemen voor het fixeren van de katheter is naar verwachting duurder dan het gebruik van een hechting door de huid, maar prettiger voor de patiënt.
De werkgroep adviseert daarom om centraal veneuze katheters bij voorkeur te fixeren met een hechtingsvrij fixatiesysteem en af te dekken met een polyurethaanfolie (standaard of met ‘border’). Afdekken met steriel gaas en tape wordt alleen aanbevolen wanneer de patiënt overgevoelig is voor polyurethaan of veel transpireert. Het gebruik van chloorhexidine gluconaat afdekfolie wordt alleen aanbevolen wanneer de lokale incidentie van katheter-gerelateerde infecties hoger is dan op basis van vergelijkende (historisch of externe benchmark) surveillancegegevens verwacht, ondanks optimale naleving van de aanbevelingen in deze richtlijn. Het gebruik van weefsellijm, tape en nietjes wordt ontraden.
Voor patiënten met motorische onrust en kinderen kan het wenselijk zijn om aanvullende of alternatieve fixatiemethoden te gebruiken om (partiele) dislocatie van de katheter te voorkomen.
De werkgroep adviseert om standaard polyurethaan afdekfolie en chloorhexidine gluconaat afdekfolie alleen te vervangen wanneer het loslaat, of wanneer het zichtbaar verontreinigd of vochtig is. Langdurige blootstelling van de folie aan water wordt afgeraden vanwege het risico op loslaten van de folie wanneer de huid verweekt. Steriel gaas moet dagelijks worden vervangen en waterdicht worden afgedekt wanneer de patiënt gaat douchen.
Centraal ingebrachte getunnelde centraal veneuze katheter met vastgegroeide ‘cuff’
Voor centraal ingebrachte getunnelde centraal veneuze katheter met vastgegroeide ‘cuff’ is het advies om het al dan niet afdekken van de insteekplaats van de katheter te bespreken met de patiënt (Samen Beslissen), en daarbij te benoemen dat afdekken niet nodig is vanuit infectiepreventieperspectief, dat afdekken enerzijds een esthetisch voordeel kan zijn en kan beschermen tegen wrijving door kleding, maar anderzijds juist kan resulteren in irritatie van de huid door het afdekmateriaal en in het niet tijdig herkennen van vroege tekenen van infectie.
Poortkatheter
De insteekplaats van de poortkatheter hoeft niet te worden afgedekt, omdat er geen open wond is. Wel heeft het de voorkeur om de insteekplaats na aanprikken van het systeem af te dekken met standaard polyurethaan afdekfolie.
Onderbouwing
Achtergrond
Een centraal veneuze katheter wordt ingebracht in een perifere of centrale vene en reikt tot in een centrale vene.
Een centraal veneuze katheter wordt gebruikt om (langer durend) toegang te verkrijgen tot de bloedbaan voor het toedienen van medicatie, vloeistoffen, bloedproducten en parenterale voeding, en in uitzonderlijk gevallen voor het afnemen van bloed.
Er zijn verschillende typen centraal veneuze katheters: 1) een centraal ingebrachte niet-getunnelde centraal veneuze katheter zonder ‘cuff’; 2) een centraal ingebrachte getunnelde centraal veneuze katheter met ‘cuff’; 3) een perifeer ingebrachte centraal veneuze katheter, ook wel bekend als ‘PICC’; en 4) een poortkatheter, ook wel bekend als ‘port-a-cath’. Centraal ingebrachte getunnelde centraal veneuze katheters zonder ‘cuff’ worden buiten beschouwing gelaten omdat deze in de Nederlandse praktijk niet worden toegepast.
Het gebruik van een centraal veneuze katheter kan gepaard gaan met infectieuze en niet-infectieuze complicaties, zoals een bloedbaaninfectie en trombose. Deze complicaties kunnen belastend zijn voor de patiënt en kunnen resulteren in een afname van de kwaliteit van leven en een toename in zorgkosten.
Het is belangrijk dat centraal veneuze katheters beschermd worden tegen bacteriële besmetting vanuit de omgeving en op adequate wijze aan de huid worden bevestigd om dislocatie van de katheter te voorkomen. Het afdekken en fixeren van centraal veneuze katheters voorkómt direct contact van de katheter met handen en andere niet-steriele materialen, zorgt voor een barrière voor de migratie van huidbacteriën van de huid rondom de insteekplaats naar het katheterkanaal en de kathetertip, en voorkómt (micro)bewegingen en het onbedoeld losraken van de katheter.
Er zijn veel verschillende materialen en methoden beschikbaar voor het afdekken en fixeren van centraal veneuze katheters, maar er is geen consensus over de plaats van de verschillende materialen en methoden bij de preventie van katheter-gerelateerde infecties.
Deze module beschrijft de plaats van verschillende materialen en methoden voor het afdekken en fixeren van centraal veneuze katheters bij de preventie van katheter-gerelateerde infecties voor volwassen patiënten en kinderen (geen neonaten) in de medisch-specialistische zorg. Centraal veneuze katheters voor hemodialyse vallen buiten de scope van deze richtlijn. Hechttechnieken en het fixeren van het toedieningssystemen vallen buiten de scope van deze module.
Conclusies / Summary of Findings
Comparison 1.1: Sterile gauze and tape versus SPU dressing
Table 1.1 Summary of findings - sterile gauze and tape versus SPU dressing
P: Patients with a central venous catheter
I: Sterile gauze and tape
C: SPU dressing
S: Hospital
|
Outcome |
Number of patients (studies) |
Relative effect (95% CI) |
Absolute effect estimate (95% CI)* |
Certainty of the evidence** |
Conclusions |
|
CRBSI (critical) |
72 (1 RCT)
|
RR not estimable
|
Sterile gauze and tape: not estimable SPU dressing: not estimable
RD not estimable |
Very low1 |
The evidence is very uncertain about the effect of sterile gauze and tape on CRBSI compared with SPU dressing in in-hospital patients with a central venous catheter.
(Giles, 2002) |
|
Insertion site infection (important) |
199 (2 RCTs) |
RR 0.48 (95% CI 0.12 to 1.86)
Clinically relevant In favor of sterile gauze and tape |
Sterile gauze and tape: 29 per 1,000 SPU dressing: 61 per 1,000
RD 32 fewer (95% CI 53 fewer to 52 more) |
Very low2 |
The evidence is very uncertain about the effect of sterile gauze and tape on insertion site infections compared with SPU dressing in in-hospital patients with a central venous catheter.
(Brandt, 1996; Shivnan, 1991) |
|
(Thrombo)phlebitis (important) |
25 (1 RCT) |
RR 0.92 (95% CI 0.06 to 13.18
Not clinically relevant |
Sterile gauze and tape: 77 per 1,000 SPU dressing: 83 per 1,000
RD 7 fewer (95% CI 73 fewer to 72 more) |
Very low2 |
The evidence is very uncertain about the effect of sterile gauze and tape on (thrombo)phlebitis compared with SPU dressing in in-hospital patients with a central venous catheter.
(Chico-Padrón, 2011) |
|
All-cause mortality (important) |
- |
- |
- |
No GRADE |
No evidence was found regarding the effect of sterile gauze and tape on all-cause mortality compared with SPU dressing in in-hospital patients with a central venous catheter. |
|
CRBSI-related mortality (important) |
- |
- |
- |
No GRADE |
No evidence was found regarding the effect of sterile gauze and tape on CRBSI-related mortality compared with SPU dressing in in-hospital patients with a central venous catheter. |
|
Skin irritation or damage (important) |
21 (1 RCT) |
RR 6.60 (95% CI 0.95 to 45.75)
Clinically relevant In favor of SPU dressing |
Sterile gauze and tape: 600 per 1,000 SPU dressing: 91 per 1,000
RD 509 fewer (95% CI 5 fewer to 4,068 more) |
Low3 |
Sterile gauze and tape may increase skin irritation or damage compared with SPU dressing in in-hospital patients with a central venous catheter.
(Pedrolo, 2011) |
|
Catheter failure for any reason (important) |
101 (1 RCT) |
RR 0.82 (95% CI 0.52 to 1.31)
Not clinically relevant |
Sterile gauze and tape: 376 per 1,000 SPU dressing: 458 per 1,000
RD 83 fewer (95% CI 220 fewer to 142 more) |
Very low2 |
The evidence is very uncertain about the effect of sterile gauze and tape on catheter failure for any reason compared with SPU dressing in in-hospital patients with a central venous catheter.
(Brandt, 1996) |
|
Catheter securement failure (important) |
126 (2 RCTs) |
RR 0.75 (95% CI 0.25 to 2.31)
Clinically relevant In favor of sterile gauze and tape |
Sterile gauze and tape: 57 per 1,000 SPU dressing: 76 per 1,000
RD 19 fewer (95% CI 57 fewer to 99 more) |
Very low2 |
The evidence is very uncertain about the effect of sterile gauze and tape on catheter securement failure compared with SPU dressing in in-hospital patients with a central venous catheter.
(Brandt, 1996; Chico-Padrón, 2011) |
CI = confidence interval; CRBSI = catheter-related bloodstream infection; RCT = randomized clinical trial; RD = risk difference; RR = risk ratio; SPU = standard polyurethane
* The risk in the sterile gauze and tape group was based on the assumed risk in the SPU dressing group and the relative effect of the intervention.
** The level of evidence for RCTs started at high.
1 Downgraded by three levels due to lack of blinding (risk of bias; -1), and zero events in both study groups with a total sample size <4,000 (imprecision; -2).
2 Downgraded by three levels due to lack of blinding (risk of bias; -1), and the 95% confidence interval of the risk ratio crossing both thresholds for clinical relevance (imprecision; -2).
3 Downgraded by two levels due to lack of blinding (risk of bias; -1), and the 95% confidence interval of the risk ratio crossing the upper threshold for clinical relevance (imprecision; -1).
Comparison 1.2: Chlorhexidine gluconate (CHG) dressing versus SPU dressing
Table 1.2 Summary of findings – CHG dressing versus SPU dressing
P: Patients with a central venous catheter
I: CHG dressing
C: SPU dressing
S: Hospital
|
Outcome |
Number of patients or catheter days (studies) |
Relative effect (95% CI) |
Absolute effect estimate (95% CI)* |
Certainty of the evidence** |
Conclusions |
|
CRBSI (critical) |
4,882 patients (7 RCTs) |
RR 0.59 (95% CI 0.41 to 0.84)
Clinically relevant In favor of CHG-impregnated dressing |
CHG-impregnated dressing: 20 per 1,000 SPU dressing: 33 per 1,000
RD 14 fewer per 1,000 (95% CI 20 fewer to 5 fewer) |
Low1
|
CHG dressing may reduce CRBSI compared with SPU dressing in in-hospital patients with a central venous catheter.
(Arvaniti, 2012; Duyu, 2022; Gerceker, 2017; Margatho, 2019; Ruschulte, 2009; Timsit, 2009; Timsit, 2012) |
|
CRBSI per 1,000 catheter days (critical) |
19,056 catheter days (4 RCTs) |
RR 0.68 (95% CI 0.44 to 1.05)
Clinically relevant In favor of CHG-impregnated dressing
|
CHG-impregnated dressing: 3 per 1,000 SPU dressing: 4 per 1,000
RD 1 fewer per 1,000 (95% CI 2 per 1,000 fewer to 0 per 1,000 fewer) |
Low2
|
CHG dressing may reduce CRBSI per 1,000 catheter days compared with SPU dressing in in-hospital patients with a central venous catheter.
(Arvaniti, 2012; Duyu, 2022; Gerceker, 2017; Margatho, 2019; Ruschulte, 2009; Timsit, 2009; Timsit, 2012) |
|
Insertion site infection (important) |
128 patients (2 RCTs) |
RR 0.82 (95% CI 0.15 to 4.48)
Not clinically relevant
|
CHG-impregnated dressing: 98 per 1,000 SPU dressing: 118 per 1,000
RD 21 fewer (95% CI 101 fewer to 416 more) |
Very low3 |
The evidence is very uncertain about the effect of CHG dressing on insertion site infections compared with SPU dressing in in-hospital patients with a central venous catheter.
(Gerceker, 2017; Margatho, 2019) |
|
(Thrombo)phlebitis (important) |
- |
- |
- |
No GRADE |
No evidence was found regarding the effect of CHG dressing on (thrombo)phlebitis compared with SPU dressing in in-hospital patients with a central venous catheter. |
|
All-cause mortality (important) |
407 patients (2 RCTs)
|
Arvaniti, 2012 RR 0.81 (95% CI 0.55 to 1.18)
Not clinically relevant
|
Arvaniti, 2012 CHG-impregnated dressing: 234 per 1,000 SPU dressing: 288 per 1,000
RD 55 fewer (95% CI 130 fewer to 52 more) |
Very low4
|
The evidence is very uncertain about the effect of CHG dressing on all-cause mortality compared with SPU dressing in in-hospital patients with a central venous catheter.
(Arvaniti, 2012; Margatho, 2019) |
|
Margatho, 2019 RR 1.64 (95% CI 1.10 to 2.44)
Clinically relevant In favor of SPU dressing |
Margatho, 2019 CHG-impregnated dressing: 638 per 1,000 SPU dressing: 389 per 1,000
RD 249 more (95% CI 39 more to 560 more) |
||||
|
CRBSI-related mortality (important) |
- |
- |
- |
No GRADE |
No evidence was found regarding the effect of CHG dressing on CRBSI-related mortality compared with SPU dressing in in-hospital patients with a central venous catheter. |
|
Skin irritation or damage (important) |
246 patients (2 RCTs) |
RR 2.41 (95% CI 0.44 to 13.23)
Clinically relevant In favor of SPU dressing |
CHG-impregnated dressing: 39 per 1,000 SPU dressing: 16 per 1,000
RD 23 more per 1,000 (95% CI 9 fewer to 96 more) |
Very low3 |
The evidence is very uncertain about the effect of CHG dressing on skin irritation or damage compared with SPU dressing in in-hospital patients with a central venous catheter.
(Levi, 2005; Margatho, 2019) |
|
Catheter failure for any reason (important) |
- |
- |
- |
No GRADE |
No evidence was found regarding the effect of CHG dressing on catheter failure for any reason compared with SPU dressing in in-hospital patients with a central venous catheter. |
|
Catheter securement failure (important) |
- |
- |
- |
No GRADE |
No evidence was found regarding the effect of CHG dressing on catheter securement failure compared with SPU dressing in in-hospital patients with a central venous catheter. |
CHG = chlorhexidine gluconate; CI = confidence interval; CRBSI = catheter-related bloodstream infection; RCT = randomized clinical trial; RD = risk difference; RR = risk ratio or rate ratio; SPU = standard polyurethane
* The risk in the CHG dressing group was based on the assumed risk in the SPU dressing group and the relative effect of the intervention.
** The level of evidence for RCTs started at high.
1 Downgraded by two levels due to lack of blinding, and use of catheters as unit of analysis (risk of bias; -1), and the 95% confidence interval of the risk ratio crossing the lower threshold for clinical relevance (imprecision; -1).
2 Downgraded by two levels due to lack of blinding, and use of catheters as unit of analysis (risk of bias; -1), and the 95% confidence interval of the rate ratio crossing the lower threshold for clinical relevance (imprecision; -1).
3 Downgraded by three levels due to lack of blinding (risk of bias; -1), and the 95% confidence interval of the risk ratio crossing both thresholds for clinical relevance (imprecision; -2).
4 Downgraded by three levels due to lack of blinding (risk of bias; -1), inconsistent results (inconsistency; -1), and the 95% confidence interval of the risk ratio crossing either the lower or the upper threshold for clinical relevance (imprecision; -1).
Comparison 1.3: Polyhexamethylene biguanide (PHMB) dressing versus SPU dressing
Table 1.3 Summary of findings – PHMB dressing versus SPU dressing
P: Patients with a central venous catheter
I: PHMB dressing
C: SPU dressing
S: Hospital
|
Outcome |
Number of patients (studies) |
Relative effect (95% CI) |
Absolute effect estimate (95% CI)* |
Certainty of the evidence** |
Conclusions |
|
CABSI (critical)1 |
80 (1 RCT) |
RR not estimable |
PHMB dressing: not estimable SPU dressing: not estimable
RD not estimable |
Very low2 |
The evidence is very uncertain about the effect of PHMB dressing on CABSI compared with SPU dressing in in-hospital patients with a central venous catheter.
(Pearse, 2022) |
|
Insertion site infection (important) |
80 (1 RCT) |
RR not estimable |
PHMB dressing: not estimable SPU dressing: not estimable
RD not estimable |
Very low2 |
The evidence is very uncertain about the effect of PHMB dressing on insertion site infection compared with SPU dressing in in-hospital patients with a central venous catheter.
(Pearse, 2022) |
|
(Thrombo)phlebitis (Important) |
- |
- |
- |
No GRADE |
No evidence was found regarding the effect of PHMB dressing on (thrombo)phlebitis compared with SPU dressing in in-hospital patients with a central venous catheter. |
|
All-cause mortality (important) |
- |
- |
- |
No GRADE |
No evidence was found regarding the effect of PHMB dressing on all-cause mortality compared with SPU dressing in in-hospital patients with a central venous catheter. |
|
CABSI-related mortality (important)1 |
- |
- |
- |
No GRADE |
No evidence was found regarding the effect of PHMB dressing on CABSI-related mortality compared with SPU dressing in in-hospital patients with a central venous catheter. |
|
Skin irritation or damage (important) |
- |
- |
- |
No GRADE |
No evidence was found regarding the effect of PHMB dressing on skin irritation or damage compared with SPU dressing in in-hospital patients with a central venous catheter. |
|
Catheter failure for any reason (important) |
- |
- |
- |
No GRADE |
No evidence was found regarding the effect of PHMB dressing on catheter failure for any reason compared with SPU dressing in in-hospital patients with a central venous catheter. |
|
Catheter securement failure (important) |
- |
- |
- |
No GRADE |
No evidence was found regarding the effect of PHMB dressing on catheter securement failure compared with SPU dressing in in-hospital patients with a central venous catheter. |
CABSI = catheter-associated bloodstream infection; CI = confidence interval; CRBSI = catheter-related bloodstream infection; PHMB = polyhexamethylene biguanide; RCT = randomized clinical trial; RD = risk difference; RR = risk ratio; SPU = standard polyurethane
* The risk in the PHMB dressing group was based on the assumed risk in the SPU dressing group and the relative effect of the intervention.
** The level of evidence for RCTs started at high.
1 None of the studies reported CRBSI as an outcome.
2 Downgraded by three levels due to lack of blinding (risk of bias; -1), and zero events in both study groups with a total sample size <4,000 (imprecision; -2).
Comparison 2.4: Sutureless securement device (SSD) and PU dressing versus suture and PU dressing
Table 2.4 Summary of findings - SSD and PU dressing versus suture and PU dressing
P: Patients with a central venous catheter
I: SSD and PU dressing
C: Suture and PU dressing
S: Hospital
|
Outcome |
Number of patients or catheter days (studies) |
Relative effect (95% CI) |
Absolute effect estimate (95% CI)* |
Certainty of the evidence** |
Conclusions |
|
CRBSI (critical) |
170 patients (1 RCT) |
RR 0.13 (95% CI 0.02 to 0.98)
Clinically relevant In favor of SSD and PU dressing
|
SSD and PU dressing: 12 per 1,000 Suture and PU dressing: 94 per 1,000
RD 82 fewer per 1,000 (95% CI 92 fewer to 2 fewer) |
Low1 |
SSD and PU dressing may reduce CRBSI compared with suture and PU dressing in in-hospital patients with a central venous catheter
(Yamamoto, 2002) |
|
CRBSI per 1,000 catheter days (critical) |
4,730 catheter days (1 RCT) |
RR 0.13 (95% CI 0.02 to 1.05)
Clinically relevant In favor of SSD and PU dressing
|
SSD and PU dressing: 0.4 per 1,000 Suture and PU dressing: 3 per 1,000
RD 2 fewer per 1,000 (95% CI 3 fewer to 0 more) |
Low2 |
SSD and PU dressing may reduce CRBSI per 1,000 catheter days compared with suture and PU dressing in in-hospital patients with a central venous catheter.
(Yamamoto, 2002) |
|
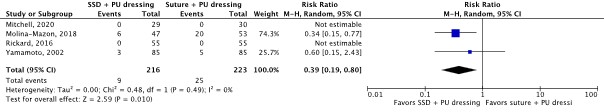
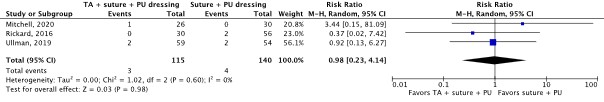
Insertion site infection (important) |
439 patients (4 RCTs) |
RR 0.39 (95% CI 0.19 to 0.80)
Clinically relevant In favor of SSD and PU dressing |
SSD and PU dressing: 44 per 1,000 Suture and PU dressing: 112 per 1,000
RD 68 fewer per 1,000 (95% CI 91 fewer to 22 fewer) |
Low1 |
SSD and PU dressing may reduce insertion site infection compared with suture and PU dressing in in-hospital patients with a central venous catheter.
(Mitchell, 2020; Molina-Mazon, 2018; Rickard, 2016; Yamamoto, 2002) |
|
(Thrombo)phlebitis (important) |
- |
- |
- |
No GRADE |
No evidence was found regarding the effect of SSD and PU dressing on phlebitis for any reason compared with suture and PU dressing in in-hospital patients with a central venous catheter. |
|
All-cause mortality (important) |
169 patients (2 RCTs) |
RR not estimable |
SSD and PU dressing: not estimable Suture and PU dressing: not estimable
RD not estimable |
Very low3 |
The evidence is very uncertain about the effect of SSD and PU dressing on CRBSI compared with suture and PU dressing in in-hospital patients with a central venous catheter.
(Mitchell, 2020; Rickard, 2016) |
|
CRBSI-related mortality (important) |
- |
- |
- |
No GRADE |
No evidence was found regarding the effect of SSD and PU dressing on CRBSI-related mortality compared with suture and PU dressing in in-hospital patients with a central venous catheter. |
|
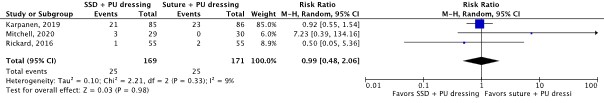
Skin irritation or damage (important) |
340 patients (2 RCTs) |
RR 0.99 (95% CI 0.48 to 2.06)
Not clinically relevant |
SSD and PU dressing: 145 per 1,000 Suture and PU dressing: 146 per 1,000
RD 1 fewer per 1,000 (95% CI 76 fewer to 155 more) |
Very low4 |
The evidence is very uncertain about the effect of SSD and PU dressing on skin irritation or damage compared with suture and PU dressing in in-hospital patients with a central venous catheter.
(Karpanen, 2019; Mitchell, 2020; Rickard, 2016) |
|
Catheter failure for any reason (important) |
110 patients (1 RCT) |
RR 2.00 (95% CI 0.38 to 10.47)
Clinically relevant In favor of suture and PU dressing |
SSD and PU dressing: 73 per 1,000 Suture and PU dressing: 36 per 1,000
RD 36 more per 1,000 (95% CI 23 fewer to 344 more) |
Very low4 |
The evidence is very uncertain about the effect of SSD and PU dressing on catheter failure for any reason compared with suture and PU dressing in in-hospital patients with a central venous catheter.
(Rickard, 2016) |
|
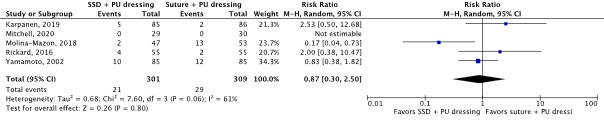
Catheter securement failure (important) |
610 patients (5 RCTs) |
RR 0.87 (95% CI 0.30 to 2.50)
Not clinically relevant |
SSD and PU dressing: 82 per 1,000 Suture and PU dressing: 94 per 1,000
RD 12 fewer per 1,000 (95% CI 66 fewer to 141 more) |
Very low5 |
The evidence is very uncertain about the effect of SSD and PU dressing on catheter securement failure compared with suture and PU dressing in in-hospital patients with a central venous catheter.
(Karpanen, 2019; Mitchell, 2020; Molina-Mazon, 2018; Rickard, 2016; Yamamoto, 2002) |
CI = confidence interval; CRBSI = catheter-related bloodstream infection; PU = polyurethane; RCT = randomized clinical trial; RD = risk difference; RR = risk ratio or rate ratio; SSD = sutureless securement device
* The risk in the SSD and PU dressing group was based on the assumed risk in the suture and PU dressing group and the relative effect of the intervention.
** The level of evidence for RCTs started at high.
1 Downgraded by two levels due to lack of blinding (risk of bias; -1), and the 95% confidence interval of the risk ratio crossing the lower threshold for clinical relevance (imprecision; -1).
2 Downgraded by two levels due to lack of blinding (risk of bias; -1), and the 95% confidence interval of the rate ratio crossing the lower threshold for clinical relevance (imprecision; -1).
3 Downgraded by three levels due to lack of blinding (risk of bias; -1), and zero events in both study groups with a total sample size <4,000 (imprecision; -2).
4 Downgraded by three levels due to lack of blinding (risk of bias; -1), and the 95% confidence interval of the risk ratio crossing both thresholds for clinical relevance (imprecision; -2).
5 Downgraded by three levels due to lack of blinding (risk of bias; -1), inconsistent results (inconsistency; -1), and the 95% confidence interval of the risk ratio crossing both thresholds for clinical relevance (imprecision; -1).
Comparison 2.6: Tissue adhesive (TA) and PU dressing versus suture and PU dressing
Table 2.6 Summary of findings - TA and PU dressing versus suture and PU dressing
P: Patients with a central venous catheter
I: TA and PU dressing
C: Suture and PU dressing
S: Hospital
|
Outcome |
Number of patients or catheter days (studies) |
Relative effect (95% CI) |
Absolute effect estimate (95% CI)* |
Certainty of the evidence** |
Conclusions |
|
CRBSI (critical) |
23 patients (1 RCT) |
RR not estimable
|
TA and PU dressing: not estimable Suture and PU dressing: not estimable
RD not estimable |
Very low1 |
The evidence is very uncertain about the effect of TA and PU dressing on CRBSI compared with suture and PU dressing in in-hospital patients with a central venous catheter.
(Ullman, 2017) |
|
CRBSI per 1,000 catheter days(critical) |
310 catheter days (1 RCT) |
RR not estimable
|
TA and PU dressing: not estimable Suture and PU dressing: not estimable
RD not estimable |
Very low1 |
The evidence is very uncertain about the effect of TA and PU dressing on CRBSI per 1,000 catheter days compared with suture and PU dressing in in-hospital patients with a central venous catheter.
(Ullman, 2017) |
|
Insertion site infection (important) |
101 patients (2 RCTs) |
RR not estimable |
TA and PU dressing: not estimable Suture and PU dressing: not estimable
RD not estimable |
Very low1 |
The evidence is very uncertain about the effect of TA and PU dressing on insertion site infection compared with suture and PU dressing in in-hospital patients with a central venous catheter.
(Rickard, 2016; Ullman, 2017) |
|
(Thrombo)phlebitis (important) |
- |
- |
- |
No GRADE |
No evidence was found regarding the effect of TA and SPU dressing on (thrombo)phlebitis compared with suture and PU dressing in in-hospital patients with a central venous catheter. |
|
All-cause mortality (important) |
78 patients (1 RCT) |
RR not estimable |
TA and PU dressing: not estimable Suture and PU dressing: not estimable
RD not estimable |
Very low2 |
The evidence is very uncertain about the effect of TA and PU dressing on all-cause mortality compared with suture and PU dressing in in-hospital patients with a central venous catheter.
(Rickard, 2016) |
|
CRBSI-related mortality (important) |
- |
- |
- |
No GRADE |
No evidence was found regarding the effect of TA and SPU dressing on CRBSI-related mortality compared with suture and PU dressing in in-hospital patients with a central venous catheter. |
|
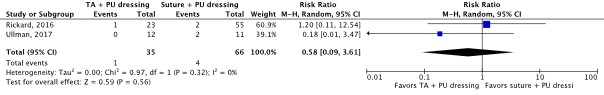
Skin irritation or damage (important) |
101 patients (2 RCTs) |
RR 0.58 (95% CI 0.09 to 3.61)
Clinically relevant In favor of TA and PU dressing |
TA and PU dressing: 35 per 1,000 Suture and PU dressing: 61 per 1,000
RD 25 fewer per 1,000 (95% CI 55 fewer to 158 more) |
Very low3 |
The evidence is very uncertain about the effect of TA and PU dressing on skin irritation or damage compared with suture and PU dressing in in-hospital patients with a central venous catheter.
(Rickard, 2016; Ullman, 2017) |
|
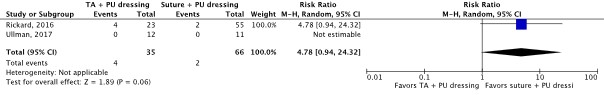
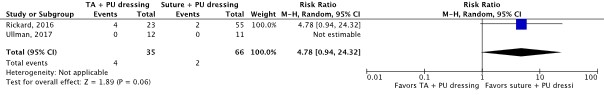
Catheter failure for any reason (important) |
110 patients (2 RCTs) |
RR 4.78 (95% CI 0.94 to 24.32)
Clinically relevant In favor of suture and PU dressing |
TA and PU dressing: 145 per 1,000 Suture and PU dressing: 30 per 1,000
RD 115 more per 1,000 (95% CI 2 fewer to 707 more) |
Low4 |
TA and PU dressing may increase catheter failure for any reason compared with suture and PU dressing in in-hospital patients with a central venous catheter.
(Rickard, 2016; Ullman, 2017) |
|
Catheter securement failure (important) |
110 patients (2 RCTs) |
RR 4.78 (95% CI 0.94 to 24.32)
Clinically relevant In favor of suture and PU dressing |
TA and PU dressing: 145 per 1,000 Suture and PU dressing: 30 per 1,000
RD 115 more per 1,000 (95% CI 2 fewer to 707 more) |
Low4 |
TA and PU dressing may increase catheter securement failure compared with suture and PU dressing in in-hospital patients with a central venous catheter.
(Rickard, 2016; Ullman, 2017) |
CI = confidence interval; CRBSI = catheter-related bloodstream infection; PU = polyurethane; RCT = randomized clinical trial; RD = risk difference; RR = risk ratio or rate ratio; SSD = sutureless securement device
* The risk in the TA and PU dressing group was based on the assumed risk in the suture and PU dressing group and the relative effect of the intervention.
** The level of evidence for RCTs started at high.
1 Downgraded by three levels due to lack of blinding (risk of bias; -1), and zero events in both study groups with a total sample size <4,000 (imprecision; -2).
2 Downgraded by three levels due to lack of blinding (risk of bias; -1), and zero events in both study groups with a total sample size <4,000 (imprecision; -2).
3 Downgraded by three levels due to lack of blinding (risk of bias; -1), and the 95% confidence interval of the risk ratio crossing both thresholds for clinical relevance (imprecision; -2).
4 Downgraded by two levels due to lack of blinding (risk of bias; -1), and the 95% confidence interval of the risk ratio crossing the upper threshold for clinical relevance (imprecision; -1).
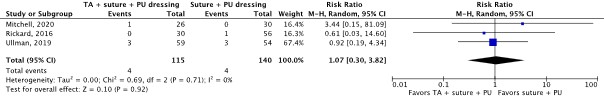
Comparison 2.7: TA, suture and PU dressing versus suture and PU dressing
Table 2.7 Summary of findings – TA, suture and PU dressing versus suture and PU dressing
P: Patients with a central venous catheter
I: TA, suture and PU dressing
C: Suture and PU dressing
S: Hospital
|
Outcome |
Number of patients or catheter days (studies) |
Relative effect (95% CI) |
Absolute effect estimate (95% CI)* |
Certainty of the evidence** |
Conclusions |
|
CABSI (critical)1 |
255 patients (3 RCTs) |
RR not estimable
|
TA, suture and PU dressing: not estimable Suture and PU dressing: 14 per 1,000
RD not estimable |
Very low2 |
The evidence is very uncertain about the effect of TA, suture and PU dressing on CABSI compared with suture and PU dressing in in-hospital patients with a central venous catheter.
(Mitchell, 2020; Rickard, 2016; Ullman, 2019) |
|
CABSI per 1,000 catheter days (critical)1 |
428 catheter days (1 RCT) |
RR not estimable
|
TA, suture and PU dressing: not estimable Suture and PU dressing: 4 per 1,000
RD not estimable |
Very low2 |
The evidence is very uncertain about the effect of TA, suture and PU dressing on CABSI per 1,000 catheter days compared with suture and PU dressing in in-hospital patients with a central venous catheter.
(Ullman, 2019) |
|
Insertion site infection (important) |
255 patients (3 RCTs) |
RR not estimable |
TA, suture and PU dressing: not estimable Suture and PU dressing: not estimable
RD not estimable |
Very low3 |
The evidence is very uncertain about the effect of TA, suture and PU dressing on insertion site infection compared with suture and PU dressing in in-hospital patients with a central venous catheter.
(Mitchell, 2020; Rickard, 2016; Ullman, 2019) |
|
(Thrombo)phlebitis (important) |
- |
- |
- |
No GRADE |
No evidence was found regarding the effect of TA, suture and SPU dressing on (thrombo)phlebitis compared with suture and PU dressing in in-hospital patients with a central venous catheter. |
|
All-cause mortality (important) |
255 patients (3 RCTs) |
RR not estimable |
TA, suture and PU dressing: not estimable Suture and PU dressing: 14 per 1,000
RD not estimable |
Very low2 |
The evidence is very uncertain about the effect of TA, suture and PU dressing on all-cause mortality compared with suture and PU dressing in in-hospital patients with a central venous catheter.
(Mitchell, 2020; Rickard, 2016; Ullman, 2019) |
|
CABSI-related mortality (important)1 |
- |
- |
- |
No GRADE |
No evidence was found regarding the effect of TA, suture and PU dressing on CABSI-related mortality compared with suture and PU dressing in in-hospital patients with a central venous catheter. |
|
Skin irritation or damage (important) |
255 patients (3 RCTs) |
RR 0.98 (95% CI 0.23 to 4.14)
Not clinically relevant |
TA, suture and PU dressing: 28 per 1,000 Suture and PU dressing: 29 per 1,000
RD 1 fewer per 1,000 (95% CI 22 fewer to 90 more) |
Very low4 |
The evidence is very uncertain about the effect of TA, suture and PU dressing on skin irritation or damage compared with suture and PU dressing in in-hospital patients with a central venous catheter.
(Mitchell, 2020; Rickard, 2016; Ullman, 2019) |
|
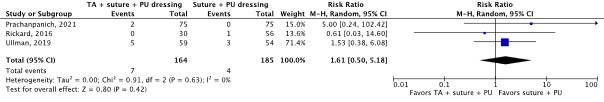
Catheter failure for any reason (important) |
349 patients (3 RCTs) |
RR 1.61 (95% CI 0.50 to 5.18)
Clinically relevant In favor of suture and PU dressing |
TA, suture and PU dressing: 35 per 1,000 Suture and PU dressing: 22 per 1,000
RD 13 more per 1,000 (95% CI 11 fewer to 90 more) |
Very low4 |
The evidence is very uncertain about the effect of TA, suture and PU dressing on catheter failure for any reason compared with suture and PU dressing in in-hospital patients with a central venous catheter.
(Prachanpanich, 2021; Rickard, 2016; Ullman, 2019) |
|
Catheter securement failure (important) |
255 patients (3 RCTs) |
RR 1.07 (95% CI 0.30 to 3.82)
Not clinically relevant |
TA, suture and PU dressing: 31 per 1,000 Suture and PU dressing: 29 per 1,000
RD 2 more per 1,000 (95% CI 20 fewer to 80 more) |
Very low4 |
The evidence is very uncertain about the effect of TA, suture and PU dressing on catheter failure for any reason compared with suture and PU dressing in in-hospital patients with a central venous catheter.
(Mitchell, 2020; Rickard, 2016; Ullman, 2019) |
CABSI = catheter-associated bloodstream infection; CI = confidence interval; CRBSI = catheter-related bloodstream infection; PU = polyurethane; RCT = randomized clinical trial; RD = risk difference; RR = risk ratio or rate ratio; TA = tissue adhesive
* The risk in the TA, suture and PU dressing group was based on the assumed risk in the suture and PU dressing group and the relative effect of the intervention.
** The level of evidence for RCTs started at high.
1 None of the studies reported CRBSI as an outcome.
2 Downgraded by three levels due to lack of blinding (risk of bias; -1), and zero events in one study group with a total sample size <4,000 (imprecision; -2).
3 Downgraded by three levels due to lack of blinding (risk of bias; -1), and zero events in both study groups with a total sample size <4,000 (imprecision; -2).
4 Downgraded by three levels due to lack of blinding (risk of bias; -1), and the 95% confidence interval of the risk ratio crossing both thresholds for clinical relevance (imprecision; -2).
Comparison 2.8: Tape and PU dressing versus suture and PU dressing
Table 2.8 Summary of findings - tape and PU dressing versus suture and PU dressing
P: Patients with a central venous catheter
I: Tape and PU dressing
C: Suture and PU dressing
S: Hospital
|
Outcome |
Number of patients (studies) |
Relative effect (95% CI) |
Absolute effect estimate (95% CI)* |
Certainty of the evidence** |
Conclusions |
|
CRBSI/CABSI (critical) |
- |
- |
- |
No GRADE |
No evidence was found regarding the effect of tape and PU dressing on CRBSI/CABSI compared with suture and PU dressing in in-hospital patients with a central venous catheter. |
|
Insertion site infection (important) |
- |
- |
- |
No GRADE |
No evidence was found regarding the effect of tape and PU dressing on insertion site infection compared with suture and PU dressing in in-hospital patients with a central venous catheter. |
|
(Thrombo)phlebitis (important) |
- |
- |
- |
No GRADE |
No evidence was found regarding the effect of tape and PU dressing on (thrombo)phlebitis compared with suture and PU dressing in in-hospital patients with a central venous catheter. |
|
All-cause mortality (important) |
- |
- |
- |
No GRADE |
No evidence was found regarding the effect of tape and PU dressing on all-cause mortality compared with suture and PU dressing in in-hospital patients with a central venous catheter. |
|
CRBSI/CABSI-related mortality (important) |
- |
- |
- |
No GRADE |
No evidence was found regarding the effect of tape and PU dressing on CRBSI/CABSI-related mortality compared with suture and PU dressing in in-hospital patients with a central venous catheter. |
|
Skin irritation or damage (important) |
- |
- |
- |
No GRADE |
No evidence was found regarding the effect of tape and PU dressing on skin irritation or damage compared with suture and PU dressing in in-hospital patients with a central venous catheter. |
|
Catheter failure for any reason (important) |
- |
- |
- |
No GRADE |
No evidence was found regarding the effect of tape and PU dressing on catheter failure for any reason compared with suture and PU dressing in in-hospital patients with a central venous catheter. |
|
Catheter securement failure (important) |
66 (1 RCT) |
RR 5.84 (95% CI 1.40 to 24.35)
Clinically relevant In favor of suture and PU dressing |
Tape and PU dressing: 344 per 1,000 Suture and PU dressing: 59 per 1,000
RD 285 more per 1,000 (95% CI 24 more to 1,374 more) |
Low1 |
Tape and PU dressing may increase catheter securement failure compared with suture and PU dressing in in-hospital patients with a central venous catheter.
(Graf, 2006) |
CABSI = catheter-associated bloodstream infection; CI = confidence interval; CRBSI = catheter-related bloodstream infection; PU = polyurethane; RCT = randomized clinical trial; RD = risk difference; RR = risk ratio
* The risk in the tape and PU dressing group was based on the assumed risk in the suture and PU dressing group and the relative effect of the intervention.
** The level of evidence for RCTs started at high.
1 Downgraded by two levels due to lack of blinding (risk of bias; -1), and the optimal information size not met (imprecision; -1).
Comparison 2.9: Staple and PU dressing versus suture and PU dressing
Table 2.9 Summary of findings - staple and PU dressing versus suture and PU dressing
P: Patients with a central venous catheter
I: Staple and PU dressing
C: Suture and PU dressing
S: Hospital
|
Outcome |
Number of patients (studies) |
Relative effect (95% CI) |
Absolute effect estimate (95% CI)* |
Certainty of the evidence** |
Conclusions |
|
CRBSI/CABSI (critical) |
- |
- |
- |
No GRADE |
No evidence was found regarding the effect of staple and PU dressing on CRBSI/CABSI compared with suture and PU dressing in in-hospital patients with a central venous catheter. |
|
Insertion site infection (important) |
- |
- |
- |
No GRADE |
No evidence was found regarding the effect of staple and PU dressing on insertion site infection compared with suture and PU dressing in in-hospital patients with a central venous catheter. |
|
(Thrombo)phlebitis (important) |
- |
- |
- |
No GRADE |
No evidence was found regarding the effect of staple and PU dressing on (thrombo)phlebitis compared with suture and PU dressing in in-hospital patients with a central venous catheter. |
|
All-cause mortality (important) |
- |
- |
- |
No GRADE |
No evidence was found regarding the effect of staple and PU dressing on all-cause mortality compared with suture and PU dressing in in-hospital patients with a central venous catheter. |
|
CRBSI/CABSI-related mortality (important) |
- |
- |
- |
No GRADE |
No evidence was found regarding the effect of staple and PU dressing on CRBSI/CABSI-related mortality compared with suture and PU dressing in in-hospital patients with a central venous catheter. |
|
Skin irritation or damage (important) |
- |
- |
- |
No GRADE |
No evidence was found regarding the effect of staple and PU dressing on skin irritation or damage compared with suture and PU dressing in in-hospital patients with a central venous catheter. |
|
Catheter failure for any reason (important) |
- |
- |
- |
No GRADE |
No evidence was found regarding the effect of staple and PU dressing on catheter failure for any reason compared with suture and PU dressing in in-hospital patients with a central venous catheter. |
|
Catheter securement failure (important) |
20 (1 RCT) |
RR not estimable
|
Staple and PU dressing: 33 per 1,000 Suture and PU dressing: not estimable
RD not estimable |
Very low1 |
The evidence is very uncertain about the effect of staple and PU dressing on catheter securement failure compared with suture and PU dressing in in-hospital patients with a central venous catheter.
(Vinjirayer, 2004) |
CABSI = catheter-associated bloodstream infection; CI = confidence interval; CRBSI = catheter-related bloodstream infection; PU = polyurethane; RCT = randomized clinical trial; RD = risk difference; RR = risk ratio
* The risk in the staple and PU dressing group was based on the assumed risk in the suture and PU dressing group and the relative effect of the intervention.
** The level of evidence for RCTs started at high.
1 Downgraded by three levels due to lack of blinding (risk of bias; -1), and zero events in one study group with a total sample size <4,000 (imprecision; -2).
Samenvatting literatuur
Summary of literature – description of studies
One SR, describing 46 studies, and two studies that were not described in this SR were included in the literature summary. The quality assessment of the SR is summarized in the Quality assessment table.
The review by Xu (2024) is a systematic review on the effectiveness of dressings and securement devices to prevent central venous catheter-associated complications, and updates a previous Cochrane review (Ullman, 2015A). The Cochrane Wounds Trials Register, the Cochrane Central Register of Controlled Trials, Ovid MEDLINE, Ovid Embase, EBSCO CINAHL, and multiple clinical trial registries were searched for RCTs from inception to November 2022. Criteria for inclusion of studies were: 1) RCT, irrespective of language or publication status; 2) the study included any person of any age with any type of central venous catheters in any healthcare or community setting; 3) the study compared any central venous catheter dressings (including, but not limited to sterile gauze and tape, new and old generation standard polyurethane (SPU) dressing, bordered polyurethane (BPU) dressing, hydrocolloid dressing, highly-adhesive transparent dressing, absorbent dressing, integrated securement dressing, chlorhexidine gluconate (CHG) dressing, medication-impregnated dressing, and no dressing) or securement devices (including, but not limited to sutureless securement device, tissue adhesive, subcutaneous catheter securement system, sutures, staples, and no securement (in addition to the primary dressing); 4) the study reported at least one prespecified outcome (catheter-related bloodstream infection, catheter tip colonization, entry/exit-site infection, skin colonization, skin irritation, catheter security, dressing condition/durability, or mortality). In total, 46 studies were included in the review. The Cochrane Risk of Bias Assessment Tool was used to assess the risk of bias for the following domains: random sequence generation, allocation concealment, blinding of participants and healthcare workers, blinding of outcome assessors, incomplete outcome data, selective outcome reporting, and other bias (Higgins, 2022).
Twenty-two of 46 studies were considered eligible to answer the search question of the current module. Twenty-four studies were excluded; two were quasi-randomized controlled trials (Conly, 1989; Gilardi, 2021), three were performed in dialysis patients (de Barros, 2009; Hagerstrom, 1994; Le Corre, 2003), two were performed in neonates (Garland, 2001; Hill, 2010), one had hydrocolloid dressing (not used in Dutch hospitals) as the intervention (Nikoletti, 1999); one compared old and new generation SPU dressings (Wille, 1993); one had BPU as the comparator (Biehl, 2016); two had sterile gauze and tape as the comparator (Düzkaya, 2016; Pedrolo, 2014A); one had no dressing as the comparator (Chambers, 2005); four did not have suture and SPU dressing as the comparator (Chan, 2017; Goossens, 2018; Kleidon, 2017; Webster, 2017); three did not report any of the critical or important outcome measures (Carrer, 2005; Olson, 2004; Roberts, 1998); three were not published in Dutch or English (Fang, 2011; Gu, 2013; Li, 2020), and one did not report extractable outcome data (Pedrolo, 2014B).
Important study characteristics of the individual studies, including those not described in the SR, are summarized in the Characteristics of included studies table. The assessment of the risk of bias for the individual studies is summarized in the Risk of bias table.
Summary of literature - results
PICOS 1: Dressings
The literature summary is not presented separately for adults and children because the working group expects the effect of different dressings for central venous catheters on catheter-related infections not to differ between adults and children.
Comparison 1.1: Sterile gauze and tape versus SPU dressing
P: Patients with a central venous catheter
I: Sterile gauze and tape
C: SPU dressing
S: Hospital
Five RCTs compared sterile gauze and tape with SPU dressing in adult in-hospital patients with a central venous catheter (Brandt, 1996; Chico-Padron, 2011; Giles, 2002; Pedrolo, 2011; Shivnan, 1991). Included patients had a centrally inserted non-tunneled (Giles, 2002; Pedrolo, 2011; Shivnan, 1991) or tunneled (Brandt, 1996) central venous catheter, or a peripherally inserted central venous catheter (Chico-Padrón, 2011). The methods used for the securement of centrally inserted non-tunneled central venous catheters (Giles, 2002; Pedrolo, 2011; Shivnan, 1991) and peripherally inserted central venous catheters (Chico-Padrón, 2011) were not described.
1. CRBSI (critical)
One RCT reported CRBSI as an outcome (Giles, 2002). The incidence of CRBSI was 0/39 (not estimable) in the sterile gauze and tape group, compared to 0/33 (not estimable) in the SPU dressing group. The risk ratio could not be estimated.
2. Insertion site infection (important)
Two RCTs reported insertion site infection as an outcome (Brandt, 1996; Shivnan, 1991). Infections were laboratory-confirmed in the study by Shivnan (1991). The results were pooled in a meta-analysis (Figure 1.1.2). The pooled incidence of insertion site infection was 3/100 (3.0%) in the sterile gauze and tape group, compared to 6/99 (6.1%) in the SPU dressing group. This resulted in a pooled 52% relative risk reduction in favor of sterile gauze and tape (RRpooled 0.48; 95% CI 0.12 to 1.86). The relative risk reduction was considered clinically relevant.
Figure 1.1.2 Forest plot of insertion site infection for gauze and tape versus SPU dressing in in-hospital patients with a central venous catheter. Pooled risk ratio, random effects model.
3. (Thrombo)phlebitis (important)
One RCT reported (thrombo)phlebitis as an outcome (Chico-Padrón, 2011). The incidence of (thrombo)phlebitis was 1/13 (7.7%) in the sterile gauze and tape group, compared to 1/12 (8.3%) in the SPU dressing group. This resulted in an 8% relative risk reduction in favor of sterile gauze and tape (RR 0.92; 95% CI 0.06 to 13.18). The relative risk increase was considered not clinically relevant.
4. All-cause mortality (important)
None of the studies reported all-cause mortality as an outcome.
5. CRBSI-related mortality (important)
None of the studies reported CRBSI-related mortality as an outcome.
6. Skin irritation or damage (important)
One RCT reported skin irritation or damage as an outcome (Pedrolo, 2011). The incidence of skin irritation or damage was 6/10 (60.0%) in the sterile gauze and tape group, compared to 1/11 (9.1%) in the SPU dressing group. This resulted in a 560% relative risk increase in favor of SPU dressing (RR 6.60; 95% CI 0.95 to 45.75). The relative risk increase was considered clinically relevant.
7. Catheter failure for any reason (important)
One RCT reported catheter failure for any reason as an outcome (Brandt, 1996). The incidence of catheter failure for any reason was 20/53 (37.7%) in the sterile gauze and tape group, compared to 22/48 (45.8%) in the SPU dressing group. This resulted in an 18% relative risk reduction in favor of sterile gauze and tape (RR 0.82; 95% CI 0.52 to 1.31). The relative risk reduction was considered not clinically relevant.
8. Catheter securement failure (important)
Two RCTs reported catheter securement failure as an outcome (Brandt, 1996; Chico-Padrón, 2011). The results were pooled in a meta-analysis (Figure 1.1.8). The pooled incidence of catheter securement failure was 5/66 (7.6%) in the sterile gauze and tape group, compared to 6/60 (10.0%) in the SPU dressing group. This resulted in a pooled 25% relative risk reduction in favor of sterile gauze and tape (RRpooled 0.75; 95% CI 0.25 to 2.31). The relative risk reduction was considered clinically relevant.
Figure 1.1.8 Forest plot of catheter securement failure for gauze and tape versus SPU dressing in in-hospital patients with a central venous catheter. Pooled risk ratio, random effects model.
Comparison 1.2: Chlorhexidine gluconate (CHG) dressing versus SPU dressing
P: Patients with a central venous catheter
I: CHG dressing
C: SPU dressing
S: Hospital
Eight RCTs compared CHG dressing with SPU dressing in in-hospital patients with a central venous catheter (Arvaniti, 2012; Duyu, 2022; Gerceker, 2017; Levi, 2005; Margatho, 2019; Ruschulte, 2009; Timsit, 2009; Timsit, 2012). Five RCTs included adults (Arvaniti, 2012; Margatho, 2019; Ruschulte, 2009; Timsit, 2009; Timsit, 2012), and three RCTs children (Duyu, 2022; Gerceker, 2017; Levi, 2005). Included patients had a centrally inserted non-tunneled central venous catheter (Arvaniti, 2012; Duyu, 2022; Levi, 2005; Margatho, 2019; Ruschulte, 2009; Timsit, 2009; Timsit, 2012), or tunneled (Gerceker, 2017) central venous catheter. For most RCTs, the methods used for catheter securement were not described, except for one RCT that used clips and sutures (Ruschulte, 2009).
1. CRBSI (critical)
Seven RCTs reported CRBSI as an outcome (Arvaniti, 2012; Duyu, 2022; Gerceker, 2017; Margatho, 2019; Ruschulte, 2009; Timsit, 2009; Timsit, 2012). The results were pooled in a meta-analysis (Figure 1.2.1). The pooled incidence of CRBSI was 46/2,698 (1.7%) in the CHG dressing group, compared to 73/2,184 (3.3%) in the SPU dressing group. This resulted in a pooled 41% relative risk reduction in favor of CHG dressing (RRpooled 0.59; 95% CI 0.41 to 0.84). The relative risk reduction was considered clinically relevant.
Figure 1.2.1 Forest plot of CRBSI for CHG dressing versus SPU dressing in in-hospital patients with a central venous catheter. Pooled risk ratio, random effects model.
2. CRBSI per 1,000 catheter days (critical)
Four RCTs reported CRBSI per 1,000 catheter days as an outcome (Arvaniti, 2012; Duyu 2022; Gerceker, 2017; Ruschulte, 2009). The results were pooled in a meta-analysis (Figure 1.2.2). The pooled incidence rate of CRBSI was 35/9,414 (3.7 per 1,000 catheter days) in the CHG-impregnated dressing group, compared to 52/9,642 (5.4 per 1,000 catheter days) in the SPU dressing group. This resulted in a pooled 32% relative rate reduction in favor of CHG-impregnated dressing (RRpooled 0.68; 95% CI 0.44 to 1.05). The relative rate reduction was considered clinically relevant.
Figure 1.2.2 Forest plot of CRBSI per 1,000 catheter days for CHG dressing versus SPU dressing in in-hospital patients with a central venous catheter. Pooled rate ratio, random effects model.
3. Insertion site infection (important)
Two RCTs reported insertion site infection, not laboratory-confirmed, as an outcome (Gerceker, 2017; Margatho, 2019). The results were pooled in a meta-analysis (Figure 1.2.3). The pooled incidence of insertion site infection was 7/61 (11.5%) in the CHG-impregnated dressing group, compared to 8/67 (11.9%) in the SPU dressing group. This resulted in a pooled 18% relative risk reduction in favor of CHG-impregnated dressing (RRpooled 0.82; 95% CI 0.15 to 4.48). The relative risk reduction was considered not clinically relevant.
Figure 1.2.3 Forest plot of insertion site infection for CHG dressing versus SPU dressing in in-hospital patients with a central venous catheter. Pooled rate ratio, random effects model.
4. (Thrombo)phlebitis (important)
None of the studies reported (thrombo)phlebitis as an outcome.
5. All-cause mortality (important)
Two RCTs reported all-cause mortality as an outcome (Arvaniti, 2012; Margatho, 2019). Results were inconsistent and, therefore, not pooled in a meta-analysis. In the study by Arvaniti (2012), the incidence of all-cause mortality was 35/150 (23.3%) in the CHG-impregnated dressing group, compared to 45/156 (28.9%) in the SPU dressing group. This resulted in a 19% relative risk reduction in favor of CHG-impregnated dressing (RR 0.81; 95% CI 0.55 to 1.18). In the study by Margatho (2019), the incidence of all-cause mortality was 30/47 (63.8%) in the CHG-impregnated dressing group, compared to 21/54 (38.9%) in the SPU dressing group. This resulted in a 64% relative risk increase in favor of SPU dressing (RR 1.64; 95% CI 1.10 to 2.44).
6. CRBSI-related mortality (important)
None of the studies reported CRBSI-related mortality as an outcome.
7. Skin irritation or damage (important)
Two RCTs reported skin irritation or damage as an outcome (Levy, 2005; Margatho, 2019). The results were pooled in a meta-analysis (Figure 1.2.7). The pooled incidence of skin irritation or damage was 5/121 (4.1%) in the CHG-impregnated dressing group, compared to 2/125 (1.6%) in the SPU dressing group. This resulted in a pooled 141% relative risk increase in favor of SPU dressing (RRpooled 2.41; 95% CI 0.44 to 13.23). The relative risk increase was considered clinically relevant.
Figure 1.2.7 Forest plot of skin irritation or damage for CHG dressing versus SPU dressing in in-hospital patients with a central venous catheter. Pooled risk ratio, random effects model.
8. Catheter failure for any reason (important)
None of the studies reported catheter failure for any reason as an outcome.
9. Catheter securement failure (important)
None of the studies reported catheter securement failure for any reason as an outcome.
Comparison 1.3: Polyhexamethylene biguanide (PHMB) dressing versus SPU dressing
P: Patients with a central venous catheter
I: PHMB dressing
C: SPU dressing
S: Hospital
One RCT compared PHMB dressing with SPU dressing in adult in-hospital patients with a central venous catheter (Pearse, 2022). The type of central venous catheter was not further specified. A foam disk securement device was used for securement.
1. CABSI (critical)
One RCT reported CABSI as an outcome (Pearse, 2022). The incidence of CABSI was 0/37 (not estimable) in the PHMB dressing group, compared to 0/43 (not estimable) in the SPU dressing group. The risk ratio could not be estimated.
2. Insertion site infection (important)
One RCT reported insertion site infection, not laboratory-confirmed, as an outcome (Pearse, 2022). The incidence of CABSI was 0/37 (not estimable) in the PHMB dressing group, compared to 0/43 (not estimable) in the SPU dressing group. The risk ratio could not be estimated.
3. (Thrombo)phlebitis (important)
None of the studies reported (thrombo)phlebitis as an outcome.
4. All-cause mortality (important)
None of the studies reported all-cause mortality as an outcome.
5. CABSI-related mortality (important)
None of the studies reported CABSI-related mortality as an outcome.
6. Skin irritation or damage (important)
None of the studies reported skin irritation or damage as an outcome.
7. Catheter failure for any reason (important)
None of the studies reported catheter failure for any reason as an outcome.
8. Catheter securement failure (important)
None of the studies reported catheter securement failure.
PICOS 2: Securement methods
The literature summary is not presented separately for adults and children because the working group expects the effect of different securement methods for central venous catheters on catheter-related infections not to differ between adults and children.
Comparison 2.1: SPU dressing versus suture and polyurethane (PU) dressing
P: Patients with a central venous catheter
I: SPU dressing
C: Suture and PU dressing
S: Hospital
None of the studies compared SPU dressing with suture and PU dressing in in-hospital patients with a central venous catheter.
Comparison 2.2: BPU dressing versus suture and PU dressing
P: Patients with a central venous catheter
I: BPU dressing
C: Suture and PU dressing
H: Hospital
None of the studies compared BPU dressing with suture and PU dressing in in-hospital patients with a central venous catheter.
Comparison 2.3: Integrated securement (IS) dressing versus suture and PU dressing
P: Patients with a central venous catheter
I: IS dressing
C: Suture and PU dressing
H: Hospital
None of the studies compared IS dressing with suture and PU dressing in in-hospital patients with a central venous catheter.
Comparison 2.4: Sutureless securement device (SSD) and PU dressing versus suture and PU dressing
P: Patients with a central venous catheter
I: SSD and PU dressing
C: Suture and PU dressing
H: Hospital
Five RCTs compared SSD and SPU dressing with suture and PU dressing in adult in-hospital patients with a central venous catheter (Karpanen, 2019; Mitchell, 2020; Molina-Mazon, 2018; Rickard, 2016; Yamamoto, 2002). Included patients had a centrally inserted non-tunneled central venous catheter (Karpanen, 2019; Mitchell, 2020; Molina-Mazon, 2018; Rickard, 2016), or a peripherally inserted central venous catheter (Yamamoto, 2002). Two RCTs used SPU dressing in both groups (Molina-Mazon, 2018; Yamamoto, 2002), one RCT used BPU dressing in both groups (Karpanen, 2019), one RCT used SPU dressing in the SSD group and BPU in the suture group, and one RCT used CHG dressing (PU dressing with CHG gelpad) in both groups (Mitchell, 2020).
1. CRBSI (critical)
One RCT reported CRBSI as an outcome (Yamamoto, 2002). The incidence of CRBSI was 1/85 (1.2%) in the SSD and PU dressing group, compared to 8/85 (9.4%) in the suture and PU dressing group. This resulted in an 87% relative risk reduction in favor of SSD and PU dressing (RR 0.13; 95% CI 0.02 to 0.98). The relative risk reduction was considered clinically relevant.
2. CRBSI per 1,000 catheter days (critical)
One RCT reported CRBSI per 1,000 catheter days as an outcome (Yamamoto, 2002). The incidence rate of CRBSI was 1/2,796 (0.4 per 1,000 catheter days in the SSD and PU dressing group, compared to 8/2,934 (2.7 per 1,000 catheter days) in the suture and PU dressing group. This resulted in an 87% relative rate reduction in favor of SSD and PU dressing (RR 0.13; 95% CI 0.02 to 1.05). The relative rate reduction was considered clinically relevant.
3. Insertion site infection (important)
Four RCTs reported insertion site infection as an outcome (Mitchell, 2020; Molina-Mazon, 2018; Rickard, 2016; Yamamoto, 2002). Infections were laboratory-confirmed in the study by Mitchell (2020). The results were pooled in a meta-analysis (Figure 2.4.3). The pooled incidence of insertion site infection was 9/216 (4.2%) in the SSD and PU dressing group, compared to 25/223 (11.2%) in the suture and PU dressing group. This resulted in a pooled 61% relative risk reduction in favor of SDD and PU dressing (RRpooled 0.39; 95% CI 0.19 to 0.80). The relative risk reduction was considered clinically relevant.
Figure 2.4.3 Forest plot of insertion site infection for SSD and PU dressing versus suture and PU dressing in in-hospital patients with a central venous catheter. Pooled risk ratio, random effects model.
4. (Thrombo)phlebitis (important)
None of the studies reported (thrombo)phlebitis as an outcome.
5. All-cause mortality (important)
Two RCTs reported all-cause mortality as an outcome (Mitchell, 2020; Rickard, 2016). The results were pooled in a meta-analysis. The pooled incidence of all-cause mortality was 4/84 (4.8%) in the SSD and PU dressing group, compared to 0/85 (not estimable) in the suture and PU dressing group. The risk ratio could not be estimated.
6. CRBSI-related mortality (important)
None of the studies reported CRBSI-related mortality as an outcome.
7. Skin irritation or damage (important)
Three RCTs reported skin irritation or damage as an outcome (Karpanen, 2019; Mitchell, 2020; Rickard, 2016). The results were pooled in a meta-analysis (Figure 2.4.7). The pooled incidence of skin irritation or damage was 25/169 (14.8%) in the SSD and PU dressing group, compared to 25/171 (14.6%) in the suture and PU dressing group. This resulted in a pooled 1% relative risk reduction in favor of SDD and PU dressing (RRpooled 0.99; 95% CI 0.48 to 2.06). The relative risk reduction was considered not clinically relevant.
Figure 2.4.7 Forest plot of skin irritation or damage for SSD and PU dressing versus suture and PU dressing in in-hospital patients with a central venous catheter. Pooled risk ratio, random effects model.
8. Catheter failure for any reason (important)
One RCT reported catheter failure for any reason as an outcome in (Rickard, 2016). The incidence of catheter failure for any reason was 4/55 (7.3%) in the SSD and PU dressing group, compared to 2/55 (3.6%) in the suture and PU dressing group. This resulted in a 100% relative risk increase in favor of suture and PU dressing (RR 2.00; 95% CI 0.38 to 10.47). The relative risk increase was considered clinically relevant.
9. Catheter securement failure (important)
Five RCTs reported catheter securement failure as an outcome (Karpanen, 2019; Mitchell, 2020; Molina-Mazon, 2018; Rickard, 2016; Yamamoto, 2002). The results were pooled in a meta-analysis (Figure 2.4.9). The pooled incidence of catheter securement failure was 21/301 (7.0%) in the SSD and PU dressing group, compared to 29/309 (9.4%) in the suture and PU dressing group. This resulted in a pooled 13% relative risk reduction in favor of SDD and PU dressing (RRpooled 0.87; 95% CI 0.30 to 2.50). The relative risk reduction was considered not clinically relevant.
Figure 2.4.9 Forest plot of catheter securement failure for SSD and PU dressing versus suture and PU dressing in in-hospital patients with a central venous catheter. Pooled risk ratio, random effects model.
Comparison 2.5: Subcutaneous anchor securement system (SASS) and SPU dressing versus suture and SPU dressing
P: Patients with a central venous catheter
I: SASS and SPU dressing
C: Suture and SPU dressing
S: Hospital
None of the studies compared SASS and SPU dressing with suture and SPU dressing in in-hospital patients with a central venous catheter.
Comparison 2.6: Tissue adhesive (TA) and PU dressing versus suture and PU dressing
P: Patients with a central venous catheter
I: TA and PU dressing
C: Suture and PU dressing
S: Hospital
Two RCTs compared TA and PU dressing with suture and PU dressing in in-hospital patients with a central venous catheter (Rickard, 2016; Ullman, 2017). One RCT included adults (Rickard, 2016), and one RCT included children (Ullman, 2017). Included patients had a centrally inserted non-tunneled (Rickard, 2016) or tunneled (Ullman, 2017) central venous catheter. One RCT used BPU dressing in both groups (Ullman, 2017), and one RCT used SPU dressing in the TA group and BPU in the suture group (Rickard, 2016).
1. CRBSI (critical)
One RCT reported CRBSI as an outcome (Ullman, 2017). The incidence of CRBSI was 0/12 (not estimable) in the TA and PU dressing group, compared to 0/11 (not estimable) in the suture and PU dressing group. The risk ratio could not be estimated.
2. CRBSI per 1,000 catheter days (critical)
One RCT reported CRBSI per 1,000 catheter days as an outcome (Ullman, 2017). The incidence rate of CRBSI was 0/148 (not estimable) in the TA and PU dressing group, compared to 0/162 (not estimable) in the suture and PU dressing group. The rate ratio could not be estimated.
3. Insertion site infection (important)
Two RCTs reported insertion site infection, not laboratory-confirmed, as an outcome (Rickard, 2016; Ullman, 2017). The results were pooled in a meta-analysis. The pooled incidence of insertion site infection was 0/35 (not estimable) in the TA and PU dressing group, compared to 0/66 (not estimable) in the suture and PU dressing group. The risk ratio could not be estimated.
4. (Thrombo)phlebitis (important)
None of the studies reported (thrombo)phlebitis as an outcome.
5. All-cause mortality (important)
One RCT reported all-cause mortality as an outcome (Rickard, 2016). The incidence of all-cause mortality was 0/23 (not estimable) in the TA and PU dressing group, compared to 0/55 (not estimable) in the suture and PU dressing group. The risk ratio could not be estimated.
6. CRBSI-related mortality (important)
None of the studies reported CRBSI-related mortality as an outcome.
7. Skin irritation or damage (important)
Two RCTs reported skin irritation or damage as an outcome (Rickard, 2016; Ullman, 2017). The results were pooled in a meta-analysis (Figure 2.6.7). The pooled incidence of skin irritation or damage was 1/35 (2.9%) in the TA and PU dressing group, compared to 4/66 (6.1%) in the suture and PU dressing group. This resulted in a pooled 42% relative risk reduction in favor of TA and PU dressing (RRpooled 0.58; 95% CI 0.09 to 3.61). The relative risk reduction was considered clinically relevant.
Figure 2.6.7 Forest plot of skin irritation or damage for TA and PU dressing versus suture and PU dressing in in-hospital patients with a central venous catheter. Pooled risk ratio, random effects model.
8. Catheter failure for any reason (important)
Two RCTs reported catheter failure for any reason as an outcome (Rickard, 2016; Ullman, 2017). The results were pooled in a meta-analysis (Figure 2.6.8). The pooled incidence of catheter failure for any reason was 4/35 (11.4%) in the TA and PU dressing group, compared to 2/66 (3.0%) in the suture and PU dressing group. This resulted in a pooled 378% relative risk increase in favor of suture and PU dressing (RRpooled 4.78; 95% CI 0.94 to 24.32). The relative risk increase was considered clinically relevant.
Figure 2.6.8 Forest plot of catheter failure for any reason for TA and PU dressing versus suture and PU dressing in in-hospital patients with a central venous catheter. Pooled risk ratio, random effects model.
9. Catheter securement failure (important)
Two RCTs reported catheter securement failure as an outcome (Rickard, 2016; Ullman, 2017). The results were pooled in a meta-analysis (Figure 2.6.9). The pooled incidence of catheter securement failure was 4/35 (11.4%) in the TA and PU dressing group, compared to 2/66 (3.0%) in the suture and PU dressing group. This resulted in a pooled 378% relative risk increase in favor of suture and PU dressing (RRpooled 4.78; 95% CI 0.94 to 24.32). The relative risk increase was considered clinically relevant.
Figure 2.6.9 Forest plot of catheter securement failure for TA and PU dressing versus suture and PU dressing in in-hospital patients with a central venous catheter. Pooled risk ratio, random effects model.
Comparison 2.7: TA, suture and PU dressing versus suture and PU dressing
P: Patients with a central venous catheter
I: TA, suture and PU dressing
C: Suture and PU dressing
S: Hospital
Four RCTs compared TA, suture and SPU dressing with suture and PU dressing in in-hospital patients with a central venous catheter (Mitchell, 2020; Prachanpanich, 2021; Rickard, 2016; Ullman, 2019). Three RCTs included adults (Mitchell, 2020; Prachanpanich, 2021; Rickard, 2016), and one RCT included children (Ullman, 2019). All included patients had a centrally inserted non-tunneled central venous catheter. One RCT used SPU dressing in both groups (Prachanpanich, 2021), one RCT used BPU dressing in both groups (Ullmann, 2019), one RCT used SPU dressing in the TA group and BPU in the suture group, and one RCT used CHG dressing (PU dressing with CHG gelpad) in both groups (Mitchell, 2020).
1. CABSI (critical)
Three RCTs reported CABSI as an outcome (Mitchell, 2020; Rickard, 2016; Ullman, 2019). The incidence of CABSI was 0/115 (not estimable) in the TA, suture and PU dressing group, compared to 2/140 (1.4%) in the suture and PU dressing group. The risk ratio could not be estimated.
2. CABSI per 1,000 catheter days (critical)
One RCT reported CABSI per 1,000 catheter days as an outcome (Ullman, 2019). The incidence rate of CABSI was 0/200 (not estimable) in the TA, suture and PU dressing group, compared to 1/228 (4 per 1,000 catheter days) in the suture and PU dressing group. The rate ratio could not be estimated.
3. Insertion site infection (important)
Three RCTs reported insertion site infection as an outcome (Mitchell, 2020; Rickard, 2016; Ullman, 2019). Infections were laboratory-confirmed in two RCTs (Mitchell, 2020; Ullman, 2019); infections were not laboratory-confirmed in one RCT (Rickard, 2016). The results were pooled in a meta-analysis. The pooled incidence of insertion site infection was 0/115 (not estimable) in the TA, suture and PU dressing group, compared to 0/140 (not estimable) in the suture and PU dressing group. The risk ratio could not be estimated.
4. (Thrombo)phlebitis (important)
None of the studies reported (thrombo)phlebitis as an outcome.
5. All-cause mortality (important)
Three RCTs reported all-cause mortality as an outcome (Mitchell, 2020; Rickard, 2016; Ullman, 2019). The incidence of all-cause mortality was 0/115 (not estimable) in the TA, suture and PU dressing group, compared to 2/140 (1.4%) in the suture and PU dressing group. The risk ratio could not be estimated.
6. CABSI-related mortality (important)
None of the studies reported CABSI-related mortality as an outcome.
7. Skin irritation or damage (important)
Three RCTs reported skin irritation or damage as an outcome (Mitchel, 2020; Rickard, 2016; Ullman, 2019). The results were pooled in a meta-analysis (Figure 2.7.7). The pooled incidence of insertion site infection was 3/115 (2.6%) in the TA, suture and PU dressing group, compared to 4/140 (2.9%) in the suture and PU dressing group. This resulted in a pooled 2% relative risk reduction in favor of TA, suture and PU dressing (RRpooled 0.98; 95% CI 0.23 to 4.14). The relative risk reduction was considered not clinically relevant.
Figure 2.7.7 Forest plot of skin irritation or damage for TA, suture and PU dressing versus suture and PU dressing in in-hospital patients with a central venous catheter. Pooled risk ratio, random effects model.
8. Catheter failure for any reason (important)
Three RCTs reported catheter failure for any reason as an outcome (Prachanpanich, 2021; Rickard, 2016; Ullman, 2019). The results were pooled in a meta-analysis (Figure 2.7.8). The pooled incidence of catheter failure for any reason was 7/164 (4.3%) in the TA, suture and PU dressing group, compared to 4/185 (2.2%) in the suture and PU dressing group. This resulted in a pooled 61% relative risk increase in favor of suture and PU dressing (RRpooled 1.61; 95% CI 0.50 to 5.18). The relative risk increase was considered clinically relevant.
Figure 2.7.8 Forest plot of catheter failure for any reason for TA, suture and PU dressing versus suture and PU dressing in in-hospital patients with a central venous catheter. Pooled risk ratio, random effects model.
9. Catheter securement failure (important)
Three RCTs reported catheter securement failure as an outcome (Mitchell, 2020; Rickard, 2016; Ullman, 2019). The results were pooled in a meta-analysis (Figure 2.7.9). The pooled incidence of catheter securement failure was 4/115 (3.5%) in the TA, suture and PU dressing group, compared to 4/140 (2.9%) in the suture and PU dressing group. This resulted in a pooled 7% relative risk increase in favor of suture and PU dressing (RRpooled 1.07; 95% CI 0.30 to 3.82). The relative risk increase was considered not clinically relevant.
Figure 2.7.9 Forest plot of catheter securement failure for TA, suture and PU dressing versus suture and PU dressing in in-hospital patients with a central venous catheter. Pooled risk ratio, random effects model.
Comparison 2.8: Tape and PU dressing versus suture and PU dressing
P: Patients with a central venous catheter
I: Tape and PU dressing
C: Suture and PU dressing
S: Hospital
One RCT compared tape and PU dressing with suture and PU dressing in pediatric in-hospital patients with a central venous catheter (Graf, 2006). All included patients had a peripherally inserted central venous catheter. SPU dressing was used in both groups. Whether the tape used was sterile or non-sterile tape was not reported.
1. CRBSI/CABSI (critical)
None of the studies reported CRBSI/CABSI as an outcome.
2. Insertion site infection (important)
None of the studies reported insertion site infection as an outcome.
3. (Thrombo)phlebitis (important)
None of the studies reported (thrombo)phlebitis as an outcome.
4. All-cause mortality (important)
None of the studies reported all-cause mortality as an outcome.
5. CRBSI/CABSI-related mortality (important)
None of the studies reported CRBSI-related mortality as an outcome.
6. Skin irritation or damage (important)
None of the studies reported skin irritation or damage as an outcome.
7. Catheter failure for any reason (important)
None of the studies reported catheter failure for any reason as an outcome.
8. Catheter securement failure (important)
One RCT reported catheter securement failure as an outcome (Graf, 2006). The incidence of catheter securement failure was 11/32 (34.4%) in the tape and PU dressing group, compared to 2/34 (5.9%) in the suture and PU dressing group. This resulted in a 484% relative risk increase in favor of suture and PU dressing (RR 5.84; 95% CI 1.40 to 24.35). The relative risk increase was considered clinically relevant.
Comparison 2.9: Staple and PU dressing versus suture and PU dressing
P: Patients with a central venous catheter
I: Staple and PU dressing
C: Suture and PU dressing
S: Hospital
One RCT compared staple and PU dressing with suture and PU dressing in adult in-hospital patients with a central venous catheter (Vinjirayer, 2004). All included patients had a centrally inserted non-tunneled central venous catheter. Identical PU dressing was used in both groups.
1. CRBSI/CABSI (critical)
None of the studies reported CRBSI/CABSI as an outcome.
2. Insertion site infection (important)
None of the studies reported insertion site infection as an outcome.
3. (Thrombo)phlebitis (important)
None of the studies reported (thrombo)phlebitis as an outcome.
4. All-cause mortality (important)
None of the studies reported all-cause mortality as an outcome.
5. CRBSI/CABSI-related mortality (important)
None of the studies reported CRBSI-related mortality as an outcome.
6. Skin irritation or damage (important)
None of the studies reported skin irritation or damage as an outcome.
7. Catheter failure for any reason (important)
None of the studies reported catheter failure for any reason as an outcome.
8. Catheter securement failure (important)
One RCT reported catheter securement failure as an outcome (Vinjirayer, 2004). The incidence of catheter securement failure was 3/10 (3.3%) in the staple and PU dressing group, compared to 0/10 (not estimable) in the suture and PU dressing group. The risk ratio could not be estimated.
Zoeken en selecteren
A systematic review of the literature was performed to answer the following (search)questions:
Search question 1 - Dressings
What is the effect of different dressings on catheter-related infections for in-hospital patients with a central venous catheter?
Search question 2 – Securement methods
What is the effect of different securement methods on catheter-related infections for patients in the hospital with a central venous catheter?
Table 1. PICOS 1 - Dressings
|
Patients |
Patients (non-neonates) with a central venous catheter |
|
Intervention |
CHG dressing Other medication-impregnated dressing Other non-medication-impregnated dressing Sterile gauze and tape |
|
Control |
SPU dressing |
|
Outcomes* |
CRBSI CABSI** Insertion site infection (Thrombo)phlebitis All-cause mortality CRBSI/CABSI-related mortality Skin irritation or damage Catheter failure for any reason (e.g., phlebitis, infiltration, accidental removal, occlusion) |
|
Setting |
Hospital |
|
Other selection criteria |
Study design: systematic review, randomized controlled trial, or comparative observational study |
CABSI = catheter-associated bloodstream infection; CHG = chlorhexidine gluconate; CRBSI = catheter-related bloodstream infection; SPU = standard polyurethane
* Per patient and, if reported, per 1,000 catheter days
**If CRBSI is not reported in any of the included studies
Table 2. PICOS 2 – Securement methods
|
Patients |
Patients (non-neonates) with a central venous catheter |
|
Intervention |
Any securement method other than suture and PU dressing* |
|
Control |
Suture and PU dressing* |
|
Outcomes** |
CRBSI CABSI*** Insertion site infection (Thrombo)phlebitis All-cause mortality CRBSI/CABSI-related mortality Catheter failure for any reason (e.g., phlebitis, infiltration, accidental removal, dislocation, occlusion) Catheter securement failure |
|
Setting |
Hospital |
|
Other selection criteria |
Study design: systematic review, randomized controlled trial, or comparative observational study |
CABSI = catheter-associated bloodstream infection; CRBSI = catheter-related bloodstream infection; PU = polyurethane
* Standard polyurethane (SPU) dressing or bordered polyurethane (BPU) dressing
** Per patient and, if reported, per 1,000 catheter days
***If CRBSI is not reported in any of the included studies
Comparators
The guideline panel decided to use the dressings and securement methods recommended in the previous WIP-guideline Flebitis en bloedbaaninfecties door intraveneuze infuuskatheters as comparators.
Relevant outcome measures
The guideline panel considered catheter-related bloodstream infection (CRBSI) and catheter-associated bloodstream infection (CABSI)* as critical outcome measures for decision-making; and insertion site infection, (thrombo)phlebitis, all-cause mortality, CRBSI (CABSI)-related mortality, insertion failure, and catheter failure for any reason as important outcome measures for decision-making.
* If CRBSI is not reported in any of the included studies
For CRBSI and CABSI, the guideline panel decided to use the definitions as described previously (Maki, 2006; Mermel, 2009; O’Grady, 2011). CRBSI was defined as a primary bloodstream infection with laboratory confirmation of the catheter as the source of the infection. CABSI was defined as a laboratory-confirmed primary bloodstream infection with the catheter in situ or within 48 hours of removal. For all other outcome measures, the guideline panel decided to use the definitions used by the authors of the individual studies.
The guideline panel defined the following thresholds for clinical relevance:
- Mortality: risk ratios of 0.95 and 1.05 (RR<0.95 or RR>1.05)
- Other dichotomous outcomes: risk ratios of 0.8 and 1.25 (RR<0.8 or RR>1.25)
Methods
The literature search for this search question was combined with the similar search question for peripheral venous catheters (see module Afdekken en fixeren van perifeer veneuze katheters) and peripheral arterial catheters (See module Afdekken en fixeren van perifeer arteriële katheters).
Embase.com and Ovid/Medline databases were searched with relevant search terms from 2000 until April 12, 2024. See Verantwoording for the detailed search strategy. The systematic literature search resulted in 942 hits for search question 1 and 495 hits for search question 2.
Studies were selected based on the following eligibility criteria:
- Systematic review (SR) of randomized controlled trials (RCT) (at least two databases searched, detailed search strategy with search date, in- and exclusion criteria, description of individual study results, risk of bias assessment per study), or RCT;
- SR describes at least one RCT that is not described in another selected SR;
- The research question includes all elements of the PICOS;
- The study population is not limited to hemodialysis patients;
- Full paper (i.e., no conference abstract, editorial, letter, or note);
- Full text is available;
- Full text is written in English or Dutch.
The reference lists of excluded systematic reviews were checked for studies that fulfilled the eligibility criteria and were not retrieved in the systematic literature search.
Based on title and abstract screening, 117 studies were initially selected for search question 1 and 61 for search question 2. Deduplication resulted in 138 unique studies. After reading the full text, 116 studies were excluded (Table of excluded studies), and 22 were included. Reference checking of excluded systematic reviews did not result in additional eligible studies.
Referenties
- •Systematic reviews
- Xu H, Hyun A, Mihala G, Rickard CM, Cooke ML, Lin F, Mitchell M, Ullman AJ. The effectiveness of dressings and securement devices to prevent central venous catheter-associated complications: A systematic review and meta-analysis. Int J Nurs Stud. 2024 Jan;149:104620. doi: 10.1016/j.ijnurstu.2023.104620. Epub 2023 Oct 9. PMID: 37879273.
- •Randomized controlled trials
- Arvaniti K, Lathyris D, Clouva-Molyvdas P, Haidich AB, Mouloudi E, Synnefaki E, Koulourida V, Georgopoulos D, Gerogianni N, Nakos G, Matamis D; Catheter-Related Infections in ICU (CRI-ICU) Group. Comparison of Oligon catheters and chlorhexidine-impregnated sponges with standard multilumen central venous catheters for prevention of associated colonization and infections in intensive care unit patients: a multicenter, randomized, controlled study. Crit Care Med. 2012 Feb;40(2):420-9. doi: 10.1097/CCM.0b013e31822f0d4b. PMID: 21926583.
- Brandt B, DePalma J, Irwin M, Shogan J, Lucke JF. Comparison of central venous catheter dressings in bone marrow transplant recipients. Oncol Nurs Forum. 1996 Jun;23(5):829-36. PMID: 8792352.
- Chico-Padrón RM, Carrión-García L, Delle-Vedove-Rosales L, González-Vargas CS, Marrero-Perera M, Medina-Chico S, Rodríguez-Díaz MP, Alonso-Díaz M, Jiménez-Sosa A. Comparative safety and costs of transparent versus gauze wound dressings in intravenous catheterization. J Nurs Care Qual. 2011 Oct-Dec;26(4):371-6. doi: 10.1097/NCQ.0b013e318210741b. PMID: 21407090.
- Duyu M, Karakaya Z, Yazici P, Yavuz S, Yersel NM, Tascilar MO, Firat N, Bozkurt O, Caglar Mocan Y. Comparison of chlorhexidine impregnated dressing and standard dressing for the prevention of central-line associated blood stream infection and colonization in critically ill pediatric patients: A randomized controlled trial. Pediatr Int. 2022 Jan;64(1):e15011. doi: 10.1111/ped.15011. PMID: 34610185.
- Gerçeker GÖ, Yardımcı F, Aydınok Y. Randomized controlled trial of care bundles with chlorhexidine dressing and advanced dressings to prevent catheter-related bloodstream infections in pediatric hematology-oncology patients. Eur J Oncol Nurs. 2017 Jun;28:14-20. doi: 10.1016/j.ejon.2017.02.008. Epub 2017 Mar 4. PMID: 28478850.
- Giles Y, Aksoy M, Tezelman S. What really affects the incidence of central venous catheter-related infections for short-term catheterization? Acta Chir Belg. 2002 Aug;102(4):256-8. doi: 10.1080/00015458.2002.11679308. PMID: 12244905.
- Graf JM, Newman CD, McPherson ML. Sutured securement of peripherally inserted central catheters yields fewer complications in pediatric patients. JPEN J Parenter Enteral Nutr. 2006 Nov-Dec;30(6):532-5. doi: 10.1177/0148607106030006532. PMID: 17047181.
- Karpanen TJ, Casey AL, Whitehouse T, Timsit JF, Mimoz O, Palomar M, Elliott TSJ. A clinical evaluation of two central venous catheter stabilization systems. Ann Intensive Care. 2019 Apr 17;9(1):49. doi: 10.1186/s13613-019-0519-6. PMID: 30997586; PMCID: PMC6470223.
- Levy I, Katz J, Solter E, Samra Z, Vidne B, Birk E, Ashkenazi S, Dagan O. Chlorhexidine-impregnated dressing for prevention of colonization of central venous catheters in infants and children: a randomized controlled study. Pediatr Infect Dis J. 2005 Aug;24(8):676-9. doi: 10.1097/01.inf.0000172934.98865.14. PMID: 16094219.
- Margatho AS, Ciol MA, Hoffman JM, Dos Reis PED, Furuya RK, Lima DAFS, Basile-Filho A, Silveira RCCP. Chlorhexidine-impregnated gel dressing compared with transparent polyurethane dressing in the prevention of catheter-related infections in critically ill adult patients: A pilot randomised controlled trial. Aust Crit Care. 2019 Nov;32(6):471-478. doi: 10.1016/j.aucc.2018.11.001. Epub 2018 Dec 15. PMID: 30558968.
- Mitchell ML, Ullman AJ, Takashima M, Davis C, Mihala G, Powell M, Gibson V, Zhang L, Bauer M, Geoffrey Playford E, Rickard CM. Central venous access device Securement and dressing effectiveness: The CASCADE pilot randomised controlled trial in the adult intensive care. Aust Crit Care. 2020 Sep;33(5):441-451. doi: 10.1016/j.aucc.2019.10.002. Epub 2019 Nov 19. PMID: 31757717.
- Molina-Mazón CS, Martín-Cerezo X, Domene-Nieves de la Vega G, Asensio-Flores S, Adamuz-Tomás J. Comparative study on fixation of central venous catheter by suture versus adhesive device. Enferm Intensiva (Engl Ed). 2018 Jul-Sep;29(3):103-112. English, Spanish. doi: 10.1016/j.enfi.2017.10.004. Epub 2018 Mar 27. PMID: 29602709.
- Pearse I, Marsh N, Rickard CM, Ullman AJ, Larsen E, Pelecanos A, McGuinness N, Irvine L, Rapchuk IL, Ziegenfuss M, Corley A. Polyhexamethylene biguanide discs versus unmedicated dressings for prevention of central venous catheter-associated infection in the intensive care unit: A pilot randomised controlled trial to assess protocol safety and feasibility. Aust Crit Care. 2022 Sep;35(5):512-519. doi: 10.1016/j.aucc.2021.05.015. Epub 2021 Jul 25. PMID: 34321181.
- Pedrolo E, Danski MTR, Mingorance P, Lazzari LSMD, Johann DA. Clinical controlled trial on central venous catheter dressings. Acta Paulista Enfermagem. 2011;24:278-83. doi:10.1590/S0103-21002011000200019.
- Prachanpanich N, Morakul S, Kiatmongkolkul N. Effectiveness of securing central venous catheters with topical tissue adhesive in patients undergoing cardiac surgery: a randomized controlled pilot study. BMC Anesthesiol. 2021 Mar 8;21(1):70. doi: 10.1186/s12871-021-01282-0. PMID: 33685394; PMCID: PMC7938567.
- Rickard CM, Edwards M, Spooner AJ, Mihala G, Marsh N, Best J, Wendt T, Rapchuk I, Gabriel S, Thomson B, Corley A, Fraser JF. A 4-arm randomized controlled pilot trial of innovative solutions for jugular central venous access device securement in 221 cardiac surgical patients. J Crit Care. 2016 Dec;36:35-42. doi: 10.1016/j.jcrc.2016.06.006. Epub 2016 Jun 21. PMID: 27546745.
- Ruschulte H, Franke M, Gastmeier P, Zenz S, Mahr KH, Buchholz S, Hertenstein B, Hecker H, Piepenbrock S. Prevention of central venous catheter related infections with chlorhexidine gluconate impregnated wound dressings: a randomized controlled trial. Ann Hematol. 2009 Mar;88(3):267-72. doi: 10.1007/s00277-008-0568-7. Epub 2008 Aug 5. PMID: 18679683.
- Shivnan JC, McGuire D, Freedman S, Sharkazy E, Bosserman G, Larson E, Grouleff P. A comparison of transparent adherent and dry sterile gauze dressings for long-term central catheters in patients undergoing bone marrow transplant. Oncol Nurs Forum. 1991 Nov-Dec;18(8):1349-56. PMID: 1762975.
- Timsit JF, Schwebel C, Bouadma L, Geffroy A, Garrouste-Orgeas M, Pease S, Herault MC, Haouache H, Calvino-Gunther S, Gestin B, Armand-Lefevre L, Leflon V, Chaplain C, Benali A, Francais A, Adrie C, Zahar JR, Thuong M, Arrault X, Croize J, Lucet JC; Dressing Study Group. Chlorhexidine-impregnated sponges and less frequent dressing changes for prevention of catheter-related infections in critically ill adults: a randomized controlled trial. JAMA. 2009 Mar 25;301(12):1231-41. doi: 10.1001/jama.2009.376. PMID: 19318651.
- Timsit JF, Mimoz O, Mourvillier B, Souweine B, Garrouste-Orgeas M, Alfandari S, Plantefeve G, Bronchard R, Troche G, Gauzit R, Antona M, Canet E, Bohe J, Lepape A, Vesin A, Arrault X, Schwebel C, Adrie C, Zahar JR, Ruckly S, Tournegros C, Lucet JC. Randomized controlled trial of chlorhexidine dressing and highly adhesive dressing for preventing catheter-related infections in critically ill adults. Am J Respir Crit Care Med. 2012 Dec 15;186(12):1272-8. doi: 10.1164/rccm.201206-1038OC. Epub 2012 Oct 4. PMID: 23043083.
- Ullman AJ, Kleidon T, Gibson V, McBride CA, Mihala G, Cooke M, Rickard CM. Innovative dressing and securement of tunneled central venous access devices in pediatrics: a pilot randomized controlled trial. BMC Cancer. 2017 Aug 30;17(1):595. doi: 10.1186/s12885-017-3606-9. PMID: 28854967; PMCID: PMC5577834.
- Ullman AJ, Long D, Williams T, Pearson K, Mihala G, Mattke AC, Macfarlane F, Rickard CM. Innovation in Central Venous Access Device Security: A Pilot Randomized Controlled Trial in Pediatric Critical Care. Pediatr Crit Care Med. 2019 Oct;20(10):e480-e488. doi: 10.1097/PCC.0000000000002059. PMID: 31274778.
- Vinjirayer A, Jefferson P, Ball DR. Securing central venous catheters: a comparison of sutures with staples. Emerg Med J. 2004 Sep;21(5):582-3. doi: 10.1136/emj.2003.009860. PMID: 15333537; PMCID: PMC1726413.
- Yamamoto AJ, Solomon JA, Soulen MC, Tang J, Parkinson K, Lin R, Schears GJ. Sutureless securement device reduces complications of peripherally inserted central venous catheters. J Vasc Interv Radiol. 2002 Jan;13(1):77-81. doi: 10.1016/s1051-0443(07)60012-8. PMID: 11788698.
- •Other
- Abbood HM, Hijazi K, Gould IM. Chlorhexidine Resistance or Cross-Resistance, That Is the Question. Antibiotics (Basel). 2023 Apr 22;12(5):798. doi: 10.3390/antibiotics12050798. PMID: 37237701; PMCID: PMC10215778.
- Baier C, Linke L, Eder M, Schwab F, Chaberny IF, Vonberg RP, Ebadi E. Incidence, risk factors and healthcare costs of central line-associated nosocomial bloodstream infections in hematologic and oncologic patients. PLoS One. 2020 Jan 24;15(1):e0227772. doi: 10.1371/journal.pone.0227772. PMID: 31978169; PMCID: PMC6980604.
- Buetti N, Marschall J, Drees M, Fakih MG, Hadaway L, Maragakis LL, Monsees E, Novosad S, O'Grady NP, Rupp ME, Wolf J, Yokoe D, Mermel LA. Strategies to prevent central line-associated bloodstream infections in acute-care hospitals: 2022 Update. Infect Control Hosp Epidemiol. 2022 May;43(5):553-569. doi: 10.1017/ice.2022.87. Epub 2022 Apr 19. PMID: 35437133; PMCID: PMC9096710.
- Gezondheidsraad. Zorgvuldig omgaan met desinfectantia. 2016. (https://zoek.officielebekendmakingen.nl/blg-795009.pdf, accessed 7 September 2023).
- Higgins JP, Altman DG, Sterne JAC. Assessing risk of bias in included studies. In: Higgins JPT, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, Welch VA (Eds.). Cochrane Handbook for Systematic Reviews of Interventions Version 6.3. Cochrane (updated February 2022). 2022
- Horner C, Mawer D, Wilcox M. Reduced susceptibility to chlorhexidine in staphylococci: is it increasing and does it matter? J Antimicrob Chemother. 2012 Nov;67(11):2547-59. doi: 10.1093/jac/dks284. Epub 2012 Jul 24. PMID: 22833635.
- Kampf G. Biocidal Agents Used for Disinfection Can Enhance Antibiotic Resistance in Gram-Negative Species. Antibiotics (Basel). 2018 Dec 14;7(4):110. doi: 10.3390/antibiotics7040110. PMID: 30558235; PMCID: PMC6316403.
- Kampf G. Adaptive bacterial response to low level chlorhexidine exposure and its implications for hand hygiene. Microb Cell. 2019 Mar 7;6(7):307-320. doi: 10.15698/mic2019.07.683. PMID: 31294043; PMCID: PMC6600115. A
- Kampf G. Antibiotic Resistance Can Be Enhanced in Gram-Positive Species by Some Biocidal Agents Used for Disinfection. Antibiotics (Basel). 2019 Feb 1;8(1):13. doi: 10.3390/antibiotics8010013. PMID: 30717270; PMCID: PMC6466599. B
- Maki DG, Kluger DM, Crnich CJ. The risk of bloodstream infection in adults with different intravascular devices: a systematic review of 200 published prospective studies. Mayo Clin Proc. 2006 Sep;81(9):1159-71. doi: 10.4065/81.9.1159. PMID: 16970212.
- Mermel LA, Allon M, Bouza E, Craven DE, Flynn P, O'Grady NP, Raad II, Rijnders BJ, Sherertz RJ, Warren DK. Clinical practice guidelines for the diagnosis and management of intravascular catheter-related infection: 2009 Update by the Infectious Diseases Society of America. Clin Infect Dis. 2009 Jul 1;49(1):1-45. doi: 10.1086/599376. Erratum in: Clin Infect Dis. 2010 Apr 1;50(7):1079. Dosage error in article text. Erratum in: Clin Infect Dis. 2010 Feb 1;50(3):457. PMID: 19489710; PMCID: PMC4039170.
- O'Grady NP, Alexander M, Burns LA, Dellinger EP, Garland J, Heard SO, Lipsett PA, Masur H, Mermel LA, Pearson ML, Raad II, Randolph AG, Rupp ME, Saint S; Healthcare Infection Control Practices Advisory Committee (HICPAC). Guidelines for the prevention of intravascular catheter-related infections. Clin Infect Dis. 2011 May;52(9):e162-93. doi: 10.1093/cid/cir257. Epub 2011 Apr 1. PMID: 21460264; PMCID: PMC3106269.
- Patel AR, Patel AR, Singh S, Singh S, Khawaja I. Central Line Catheters and Associated Complications: A Review. Cureus. 2019 May 22;11(5):e4717. doi: 10.7759/cureus.4717. PMID: 31355077; PMCID: PMC6650175.
- Pu YL, Li ZS, Zhi XX, Shi YA, Meng AF, Cheng F, Ali A, Li C, Fang H, Wang C. Complications and Costs of Peripherally Inserted Central Venous Catheters Compared With Implantable Port Catheters for Cancer Patients: A Meta-analysis. Cancer Nurs. 2020 Nov/Dec;43(6):455-467. doi: 10.1097/NCC.0000000000000742. PMID: 31464692.
- Spettel K, Bumberger D, Kriz R, Frank S, Loy M, Galazka S, Suchomel M, Lagler H, Makristathis A, Willinger B. In vitro long-term exposure to chlorhexidine or triclosan induces cross-resistance against azoles in Nakaseomyces glabratus. Antimicrob Resist Infect Control. 2025 Jan 23;14(1):2. doi: 10.1186/s13756-024-01511-4. PMID: 39849551; PMCID: PMC11755926.
- Ullman AJ, Cooke ML, Mitchell M, Lin F, New K, Long DA, Mihala G, Rickard CM. Dressings and securement devices for central venous catheters (CVC). Cochrane Database Syst Rev. 2015 Sep 10;2015(9):CD010367. doi: 10.1002/14651858.CD010367.pub2. PMID: 26358142; PMCID: PMC6457749. A
- Ullman AJ, Gibson V, Takashima MD, Kleidon TM, Schults J, Saiyed M, Cattanach P, Paterson R, Cooke M, Rickard CM, Byrnes J, Chopra V. Pediatric central venous access devices: practice, performance, and costs. Pediatr Res. 2022 Nov;92(5):1381-1390. doi: 10.1038/s41390-022-01977-1. Epub 2022 Feb 8. PMID: 35136199; PMCID: PMC9700519.
- No authors listed. 2021 Infusion Therapy Standards of Practice Updates. J Infus Nurs. 2021 Jul-Aug 01;44(4):189-190. doi: 10.1097/NAN.0000000000000436. PMID: 34197345.
- No authors listed. Guidelines for the prevention of bloodstream infections and other infections associated with the use of intravascular catheters: Part 1: peripheral catheters [Internet]. Geneva: World Health Organization; 2024. PMID: 38810002.
Evidence tabellen
Table of quality assessment for systematic reviews
|
Study |
Appropriate and clearly focused question? |
Comprehensive and systematic literature search?
|
Description of included and excluded studies? |
Description of relevant characteristics of included studies? |
Appropriate adjustment for potential confounders in observational studies? |
Assessment of scientific quality of included studies? |
Enough similarities between studies to make combining them reasonable? |
Potential risk of publication bias taken into account? |
Potential conflicts of interest reported? |
|
First author, year |
Yes/no/unclear |
Yes/no/unclear |
Yes/no/unclear |
Yes/no/unclear |
Yes/no/unclear/ not applicable |
Yes/no/unclear |
Yes/no/unclear |
Yes/no/unclear |
Yes/no/unclear |
|
Xu, 2024 |
Yes |
Yes |
Yes |
Yes |
Not applicable |
Yes |
Yes |
Yes |
Yes |
Characteristics of included studies table
|
Author, year Country Single- or multicenter
|
Dressing / securement device |
N patients (baseline); catheters; catheter days Population Ward / procedure Device type Follow-up |
Outcomes |
Remarks |
|
Studies included in SR (Xu, 2024) – randomized controlled trials |
|
|
|
|
|
Arvaniti, 2012
Greece
Multicenter |
G1: SPU dressing + CHG disk (Biopatch)* G2: SPU dressing*
* after catheter stabilization |
T: 306; 306; 2,202
Adults
ICU (trauma, medical, surgery)
CVCa
Until catheter removal or transfer from ICU |
CRBSI All-cause mortality |
G3: not included in review due to co-intervention (silver-impregnated CVC) |
|
Brandt, 1996
United States
Single-center |
G1: SPU dressing (Opsite IV3000)* G2: Sterile gauze and tape*
* Catheter securement not described |
T: 101; 101; NR
Adults
BMT unit / autologous BMT
Tunneled CVC
Until catheter removal or discharge |
Insertion site infection Catheter failure Catheter securement failure
|
- |
|
Duyu, 2022
Turkey
Single-center |
G1: PU dressing with CHG gelpad (3M Tegaderm CHG IV)* G2: SPU dressing (3M Tegaderm)*
* Catheter securement not described |
T: 307; 307; 4,993
Children
PICU
Non-tunneled CVC
Until 48 hours after PICU discharge |
CRBSI
|
- |
|
Gerceker, 2017
Turkey
Single-center |
G1: PU dressing with CHG gelpad and securement tape (3M Tegaderm CHG IV Securement) G2: BPU dressing (3M Tegaderm IV Advanced Securement) |
T: 27; 27; 2,230
Children
Hematology-oncology
Tunneled CVC Implantable port CVC
NR |
CRBSI Insertion site infection |
- |
|
Giles, 2002
Turkey
Single-center |
G1: SPU dressing* G2: Sterile gauze and tape*
* Catheter securement not described |
T: 72; 72; NR
Adults
General surgery
CVCa
Until catheter removal |
CRBSI
|
- |
|
Graf, 2006
United States
Single-center |
G1: Suture + SPU dressing (3M Tegaderm) G2: Tape* + SPU dressing (3M Tegaderm)
* Sterile or non-sterile tape not described
|
T: 66; 66; NR
Children
PICU / antibiotic, nutrition, or IV fluid therapy
PICC
Until catheter removal |
Catheter securement failure |
‘- |
|
Karpanen, 2019
United Kingdom, Spain, France
Multicenter |
G1: Suture + BPU dressing (3M Tegaderm IV Advanced Securement) G2: SSD (3M PICC/CVC Securement Device) + BPU dressing (3M Tegaderm IV Advanced Securement) |
T: 171; 171; NR
Adults
ICU
Non-tunneled CVC
Until catheter removal or discharge |
Skin irritation or damage Catheter securement failure
|
- |
|
Levy, 2005
Israel
Single-center |
G1: SPU dressing + CHG disk (Biopatch)* G2: SPU dressing*
* Catheter securement not described |
T: 145; NR; NR
Children
PCCU
CVCa
NR |
Skin irritation or damage
|
- |
|
Margatho, 2019
Brazil
Single-center |
G1: SPU dressing (3M Tegaderm)* G2: PU dressing with CHG gelpad (3M Tegaderm CHG IV)*
* Catheter securement not described |
T: 101; 101; NR
Adults
ICU
CVCa
Until catheter removal |
CRBSI Insertion site infection All-cause mortality Skin irritation or damage
|
Pilot RCT |
|
Mitchel, 2020
Australia
Single-center |
G1: Suture + PU dressing with CHG gelpad (3M Tegaderm CHG IV) G2: Suture + PU dressing with CHG gelpad (3M Tegaderm CHG IV) + TA (Histoacryl) G3: SSD (StatLock) + PU dressing with CHG gelpad (3M Tegaderm CHG IV) G4: Sutures + IS dressing (SorbaView SHIELD) + CHG disk (Biopatch) |
T: 121; 121; 735
Adults
ICU
Non-tunneled CVC
Until catheter removal |
CABSI Insertion site infection All-cause mortality Skin irritation or damage Catheter securement failure
|
Pilot RCT |
|
Molina-Mazon, 2018
Spain
Single-center |
G1: Suture + SPU dressing (3M Tegaderm) G2: SSD + SPU dressing (3M PICC/CVC Securement Device) |
T: 100; 100; NR
Adults
CCU
CVCa
Until catheter removal |
Insertion site infection Catheter securement failure |
Pilot RCT |
|
Pearse, 2022
Australia
Single-center |
G1: FSD (Easi-V) + SPU dressing (Opsite IV3000) G2: FSD (Easi-V) + SPU dressing (Opsite IV3000) + PHMB disk (Kendall antimicrobial foam disk) |
T: 86*; 80; NR
Adults
ICU
CVCa
Until 48 hours after catheter removal
* CVC insertion cancelled: G1: 0; G2: 6 |
CABSI Insertion site infection |
- |
|
Pedrolo, 2011
Brazil
Single-center |
G1: SPU dressing (3M Tegaderm)* G2: Sterile gauze and tape*
* Catheter securement not described
|
T: 21; 21; NR
Adults
ICU
Non-tunneled CVC
Until catheter removal |
Skin irritation or damage |
- |
|
Rickard, 2016
Australia
Single-center |
G1: Suture + BPU dressing (3M Tegaderm IV transparent film with border) G2: Suture + PAL dressing (Opsite Post-op Visible) G3: SSD (Grip-Lok CVC) + SPU dressing (Opsite IV3000) G4: TA (Histoacryl) + SPU dressing (Opsite IV3000) G4: Suture + TA (Histoacryl) + SPU dressing (Opsite IV3000) |
T: 221; 221; NR
Adults
Cardiac surgery
CVCa
Until 48 hours after catheter removal |
CABSI Insertion site infection All-cause mortality Skin irritation or damage Catheter failure Catheter securement failure |
Pilot RCT
Randomization to G4 was ceased after CVC dislodgement in 3 of 24 patients. Instead G5 was created for the remaining 30 patients. |
|
Ruschulte, 2009
Germany
Single-center |
G1: SPU dressing + CHG disk (Biopatch)* G2: SPU dressing*
* Catheter securement with clip and suture |
T: 601; 601; 9,731
Adults
Hematology and oncology / chemotherapy
CVCa
NR |
CRBSI
|
- |
|
Shivnan, 1991
United States
Single-center |
G1: SPU dressing (3M Tegaderm)* G2: Sterile gauze and tape*
* Catheter securement not described |
T: 103; 103; NR
Adults
BMT unit / autologous or allogenic BMT
CVCa
NR |
Insertion site infection |
Missing data: 5 |
|
Timsit, 2009
France
Multicenter |
G1: SPU dressing (3M Tegaderm)* G2: SPU dressing + CHG disk (Biopatch)*
* Catheter securement not described
|
T: 1,653*; 3,778**; 28,931 CVC: NR; 2,051; NR AC: NR; 1,727; NR
Adults
ICU
CVC*** or PAC
Until 48 hours after ICU discharge
* CVC/PAC insertion cancelled: G1: 9; G2: 8 ** Multiple catheters per patient *** Tunneled: G1: 5; G2: 1 |
CRBSI
|
Unit of analysis: catheters
Published manuscript describes overall outcomes; data per device type provided by authors
CVC group includes 6 (out of 2,051) tunneled CVCs |
|
Timsit, 2012
France
Multicenter |
G1: SPU dressing (3M Tegaderm)* G2: PU dressing with CHG gelpad and securement tape (3M Tegaderm CHG IV Securement) G3: Highly adhesive transparent dressing (3M HP Transparent)
* Catheter securement not described |
T: 1,879; 4,163*; 34,339 CVC: NR; 1,962; NR AC: NR; 2,201; NR
Adults
ICU
CVCa PAC
Until 48 hours after ICU discharge
* Multiple catheters per patient |
CRBSI
|
Unit of analysis: catheters
Published manuscript describes overall outcomes; data per device type provided by authors (G1 and G2 only) |
|
Ullman, 2017
Australia
Multicenter |
G1: Suture + BPU dressing (3M Tegaderm IV transparent film with border) G2: Suture + SSD (StatLock or GripLok) + BPU dressing (3M Tegaderm IV transparent film with border) G3: TA (Histoacryl) + BPU dressing (3M Tegaderm IV transparent film with border) G4: Suture + IS dressing (SorbaView SHIELD) |
T: 48; 48; NR
Children
Hematology Oncology
Tunneled CVC
Until 4 weeks after insertion or catheter removal or discharge |
CRBSI Insertion site infection Skin irritation or damage Catheter failure Catheter securement failure |
Pilot RCT
|
|
Ullman, 2019
Australia
Single-center |
G1: Suture + BPU dressing (3M Tegaderm IV border) + CHG disk (Biopatch) G2: TA + suture + BPU dressing (3M Tegaderm IV transparent film with border) + CHG disk (Biopatch) G3: Suture + IS dressing (SorbaView SHIELD) + CHG disk |
T: 169; 169; 702
Children
Anesthesia PICU
Non-tunneled CVC
Until 4 weeks after insertion or catheter removal or discharge |
CABSI Insertion site infection All-cause mortality Skin irritation or damage Catheter failure Catheter securement failure |
Pilot RCT
|
|
Vinjirayer, 2004
United Kingdom
Single-center |
G1: Staple + transparent dressing G2: Suture + transparent dressing |
T: 20; 20; NR
Adults
Surgical ward
CVCa
Until 3 days after surgery or catheter removal |
Catheter securement failure |
- |
|
Yamamoto, 2002
United States
Single-center |
G1: Suture + SPU dressing G2: SSD (StatLock) + SPU dressing |
T: 170; 170; 5,730
Adults
Hospital (not further specified)
PICC
Until catheter removal |
CRBSI Insertion site infection Catheter securement failure |
- |
|
Additional studies - randomized controlled trials |
|
|
|
|
|
Chico-Padrón, 2011
Spain
Single-center |
G1: Transparent dressing* G2: Gauze and tape*
* Catheter securement not described |
T: 50; 50; NR
Adults
CCU
PVC or PICC
Until complication, removal or discharge from CCU |
Phlebitis Catheter securement failure |
- |
|
Prachanpanich, 2021
Thailand
Single-center |
G1: Suture + SPU dressing (3M Tegaderm) G2: TA + suture + SPU dressing (3M Tegaderm) |
T: 150; 150; NR
Adults
CCU / cardiac surgery
CVCa
Until 3 days after surgery |
Catheter failure |
- |
BMT = bone marrow transplantation; CABSI = catheter-associated bloodstream infection; CCU = cardio care unit; CHG = chlorhexidine gluconate; CRBSI = catheter-related bloodstream infection; CVC = central venous catheter; ED = emergency department; FSD: foam securement device; G = group; IS = integrated securement; ICU = intensive care unit; NR = not reported; PAC = peripheral arterial catheter; PAL=polyurethane with absorbent lattice pad; PCCU: pediatric cardio care unit; PICC = peripherally inserted central venous catheter; PICU = pediatric intensive care unit; PHMB = polyhexamethylene biguanide; PU = polyurethane; PVC = peripheral venous catheter; RCT = randomized controlled trial; SPU = standard polyurethane; SR = systematic review; SSD = sutureless securement device; T = total number; TA = tissue adhesive
a CVCs were assumed to be non-tunneled, unless specified otherwise.
Risk of bias table
Based on the Cochrane risk of bias assessment tool (Higgins, 2022) that was used in the systematic review by Xu (2024).
Randomized controlled trials
|
Author, year |
Random sequence generation |
Allocation concealment |
Blinding of participants and HCWs |
Blinding of outcome assessment |
Incomplete outcome data |
Selective reporting |
Other bias |
Overall risk of bias |
|
Arvaniti, 2012 |
Low |
Unclear |
HighC1 |
Low |
Low |
Unclear |
Unclear |
High (all outcomes) |
|
Brandt, 1996 |
Unclear |
Unclear |
HighC1 |
Unclear |
Low |
Unclear |
Unclear |
High (all outcomes) |
|
Chico-Padrón, 2011 |
Low |
Unclear |
HighC1 |
Unclear |
Low |
Low |
Unclear |
High (all outcomes) |
|
Duyu, 2022 |
Low |
Low |
HighC1 |
Low |
Low |
Unclear |
Low |
High (all outcomes) |
|
Gerceker, 2017 |
Low |
Low |
HighC1 |
Unclear |
Low |
Unclear |
Low |
High (all outcomes) |
|
Giles, 2002 |
Unclear |
Unclear |
HighC1 |
Unclear |
Low |
Unclear |
Unclear |
High (all outcomes) |
|
Graf, 2006 |
Unclear |
Unclear |
HighC1 |
Unclear |
Low |
Unclear |
Low |
High (all outcomes) |
|
Karpanen, 2019 |
Unclear |
Unclear |
HighC1 |
HighD1 |
Low |
Low |
Low |
High (all outcomes) |
|
Levy, 2005 |
Low |
Unclear |
HighC1 |
Low |
Low |
Unclear |
Unclear |
High (all outcomes) |
|
Margatho, 2019 |
Low |
Low |
HighC1 |
Unclear |
Low |
Low |
Unclear |
High (all outcomes) |
|
Mitchell, 2020 |
Low |
Low |
HighC1 |
Unclear |
Low |
Low |
Unclear |
High (all outcomes) |
|
Molina-Mazon, 2018 |
Low |
Unclear |
HighC1 |
Unclear |
Low |
Unclear |
Low |
High (all outcomes) |
|
Pearse, 2022 |
Low |
Low |
HighC1 |
Low |
Low |
Low |
Unclear |
High (all outcomes) |
|
Pedrolo, 2011 |
Unclear |
Unclear |
HighC1 |
Unclear |
Low |
Unclear |
Unclear |
High (all outcomes) |
|
Prachanpanich, 2021 |
Low |
Low |
HighC1 |
HighD1 |
Low |
Unclear |
Low |
High (all outcomes) |
|
Rickard, 2016 |
Low |
Low |
HighC1 |
Unclear |
Low |
Low |
Low |
High (all outcomes) |
|
Ruschulte, 2009 |
Unclear |
Unclear |
HighC1 |
Low |
Low |
Unclear |
Unclear |
High (all outcomes) |
|
Shivnan, 1991 |
Unclear |
Unclear |
HighC1 |
Unclear |
Low |
Unclear |
HighG1 |
High (all outcomes) |
|
Timsit, 2009 |
Low |
Unclear |
HighC1 |
Low |
Low |
Unclear |
HighG2 |
High (all outcomes) |
|
Timsit, 2012 |
Low |
Unclear |
HighC1 |
Low |
Low |
Unclear |
HighG1, G2 |
High (all outcomes) |
|
Ullman, 2017 |
Low |
Low |
HighC1 |
Unclear |
Low |
Low |
HighG1 |
High (all outcomes) |
|
Ullman, 2019 |
Low |
Low |
HighC1 |
Unclear |
Low |
Low |
Unclear |
High (all outcomes) |
|
Vinjirayer, 2004 |
Unclear |
Unclear |
HighC1 |
Unclear |
Low |
Unclear |
Unclear |
High (all outcomes) |
|
Yamamoto, 2002 |
Unclear |
Low |
HighC1 |
Unclear |
Low |
Unclear |
HighG1 |
High (all outcomes) |
C1 Blinding of participants and personnel was not achievable due to visibility of the intervention
D1 Outcome assessors were not blinded
G1 Funding by industry, no bias expected
G2 Catheters are used as the unit of analysis (not independent)
Tables of excluded studies
Systematic search – full text assessment
|
Reference |
Reason for exclusion |
|
American Society of Anesthesiologists Task Force on Central Venous Access; Rupp SM, Apfelbaum JL, Blitt C, Caplan RA, Connis RT, Domino KB, Fleisher LA, Grant S, Mark JB, Morray JP, Nickinovich DG, Tung A. Practice guidelines for central venous access: a report by the American Society of Anesthesiologists Task Force on Central Venous Access. Anesthesiology. 2012 Mar;116(3):539-73. doi: 10.1097/ALN.0b013e31823c9569. PMID: 22307320. |
Narrative review |
|
Arora RS, Roberts R, Eden TO, Pizer B. Interventions other than anticoagulants and systemic antibiotics for prevention of central venous catheter-related infections in children with cancer. Cochrane Database Syst Rev. 2010 Dec 8;(12):CD007785. doi: 10.1002/14651858.CD007785.pub2. PMID: 21154387. |
I/C didn't meet PICOS
|
|
Atay S, Yilmaz Kurt F. Effectiveness of transparent film dressing for peripheral intravenous catheter. J Vasc Access. 2021 Jan;22(1):135-140. doi: 10.1177/1129729820927238. Epub 2020 Jun 12. PMID: 32529940. |
Quasi-randomized controlled trial |
|
Azzopardi A, Trapani J. Chlorhexidine-based versus non-chlorhexidine dressings to prevent catheter-related bloodstream infections: An evidence-based review. Nurs Crit Care. 2024 Jan;29(1):191-195. doi: 10.1111/nicc.12879. Epub 2022 Dec 28. PMID: 36579384. |
Narrative review |
|
Bahl A, Gibson SM, Jankowski D, Chen NW. Short peripheral intravenous catheter securement with cyanoacrylate glue compared to conventional dressing: A randomized controlled trial. J Vasc Access. 2023 Jan;24(1):52-63. doi: 10.1177/11297298211024037. Epub 2021 Jun 11. PMID: 34112019. |
P didn’t meet PICOS |
|
Bausone-Gazda D, Lefaiver CA, Walters SA. A randomized controlled trial to compare the complications of 2 peripheral intravenous catheter-stabilization systems. J Infus Nurs. 2010 Nov-Dec;33(6):371-84. doi: 10.1097/NAN.0b013e3181f85be2. PMID: 21079465. |
I/C didn't meet PICOS Co-intervention dependent on study group
|
|
Benligül EM, Bektaś M. Effectiveness of chlorhexidine-impregnated central venous catheter dressing for preventing catheter-related bloodstream infections in pediatric patients: A systematic review and meta-analysis study. J Pediatr Infect Dis 2022;18:116-126. doi: 10.1055/s-0043-1764479. |
Selection criteria for systematic reviews were were not fulfilled |
|
Biehl LM, Huth A, Panse J, Krämer C, Hentrich M, Engelhardt M, Schäfer-Eckart K, Kofla G, Kiehl M, Wendtner CM, Karthaus M, Ullmann AJ, Hellmich M, Christ H, Vehreschild MJ. A randomized trial on chlorhexidine dressings for the prevention of catheter-related bloodstream infections in neutropenic patients. Ann Oncol. 2016 Oct;27(10):1916-22. doi: 10.1093/annonc/mdw275. Epub 2016 Jul 25. PMID: 27456299. |
I/C didn’t meet PICOS
|
|
Blanchard D, Bourgeois S. Securement and Dressing Devices for Central Venous Catheters. Am J Nurs. 2016 Jun;116(6):49. doi: 10.1097/01.NAJ.0000484232.62740.cb. PMID: 27227864. |
Summary of systematic review (Ullman, 2015A) |
|
Buetti N, Timsit JF. Management and Prevention of Central Venous Catheter-Related Infections in the ICU. Semin Respir Crit Care Med. 2019 Aug;40(4):508-523. doi: 10.1055/s-0039-1693705. Epub 2019 Oct 4. PMID: 31585477. |
Narrative review |
|
Buetti N, Tabah A, Timsit JF, Zingg W. What is new in catheter use and catheter infection prevention in the ICU. Curr Opin Crit Care. 2020 Oct;26(5):459-465. doi: 10.1097/MCC.0000000000000754. PMID: 32773616. |
Narrative review |
|
Bugden S, Shean K, Scott M, Mihala G, Clark S, Johnstone C, Fraser JF, Rickard CM. Skin Glue Reduces the Failure Rate of Emergency Department-Inserted Peripheral Intravenous Catheters: A Randomized Controlled Trial. Ann Emerg Med. 2016 Aug;68(2):196-201. doi: 10.1016/j.annemergmed.2015.11.026. Epub 2015 Dec 31. PMID: 26747220. |
P didn’t meet PICOS |
|
Büyükyılmaz F, Şahiner NC, Cağlar S, Eren H. Effectiveness of an Intravenous Protection Device in Pediatric Patients on Catheter Dwell Time and Phlebitis Score. Asian Nurs Res (Korean Soc Nurs Sci). 2019 Oct;13(4):236-241. doi: 10.1016/j.anr.2019.09.001. Epub 2019 Sep 26. PMID: 31562930. |
I/C didn’t meet PICOS |
|
Card S, Piersa A, Kaplon A, Vanneman MW, Dalia A, Weaver B, Hao D. Infectious Risk of Arterial Lines: A Narrative Review. J Cardiothorac Vasc Anesth. 2023 Oct;37(10):2050-2056. doi: 10.1053/j.jvca.2023.06.037. Epub 2023 Jun 30. PMID: 37500369. |
Narrative review |
|
Carrer S, Bocchi A, Bortolotti M, Braga N, Gilli G, Candini M, Tartari S. Effect of different sterile barrier precautions and central venous catheter dressing on the skin colonization around the insertion site. Minerva Anestesiol. 2005 May;71(5):197-206. English, Italian. PMID: 15834348. |
O didn’t meet PICOS |
|
Chambers ST, Sanders J, Patton WN, Ganly P, Birch M, Crump JA, Spearing RL. Reduction of exit-site infections of tunnelled intravascular catheters among neutropenic patients by sustained-release chlorhexidine dressings: results from a prospective randomized controlled trial. J Hosp Infect. 2005 Sep;61(1):53-61. doi: 10.1016/j.jhin.2005.01.023. PMID: 16002181. |
I/C didn’t meet PICOS |
|
Chan RJ, Northfield S, Larsen E, Mihala G, Ullman A, Hancock P, Marsh N, Gavin N, Wyld D, Allworth A, Russell E, Choudhury MA, Flynn J, Rickard CM. Central venous Access device SeCurement And Dressing Effectiveness for peripherally inserted central catheters in adult acute hospital patients (CASCADE): a pilot randomised controlled trial. Trials. 2017 Oct 4;18(1):458. doi: 10.1186/s13063-017-2207-x. PMID: 28978332; PMCID: PMC5628427. |
I/C didn’t meet PICOS |
|
Charters B, Foster K, Lawton B, Lee L, Byrnes J, Mihala G, Cassidy C, Schults J, Kleidon TM, McCaffery R, Van K, Funk V, Ullman A. Novel Peripheral Intravenous Catheter Securement for Children and Catheter Failure Reduction: A Randomized Clinical Trial. JAMA Pediatr. 2024 Apr 1:e240167. doi: 10.1001/jamapediatrics.2024.0167. Epub ahead of print. PMID: 38558161; PMCID: PMC10985620. |
I/C didn’t meet PICOS |
|
Cicalini S, Palmieri F, Petrosillo N. Clinical review: new technologies for prevention of intravascular catheter-related infections. Crit Care. 2004 Jun;8(3):157-62. doi: 10.1186/cc2380. Epub 2003 Sep 29. PMID: 15153233; PMCID: PMC468883. |
Narrative review |
|
Corley A, Ullman AJ, Marsh N, Emily N L, Mihala G, Harris PNA, Rickard CM. SECUREment bundles to prevent peripheral intravenous catheter failure-the SECURE-PIVC trial: study protocol for a pilot randomized controlled trial. Br J Nurs. 2020 Oct 22;29(19):S40-S46. doi: 10.12968/bjon.2020.29.19.S40. PMID: 33104433. |
Study protocol |
|
Corley A, Ullman AJ, Marsh N, Genzel J, Larsen EN, Young E, Booker C, Harris PNA, Rickard CM. A pilot randomized controlled trial of securement bundles to reduce peripheral intravenous catheter failure. Heart Lung. 2023 Jan-Feb;57:45-53. doi: 10.1016/j.hrtlng.2022.07.015. Epub 2022 Aug 27. PMID: 36041346. |
I/C didn’t meet PICOS |
|
Corley A, Marsh N, Ullman AJ, Rickard CM. Peripheral intravenous catheter securement: An integrative review of contemporary literature around medical adhesive tapes and supplementary securement products. J Clin Nurs. 2023 May;32(9-10):1841-1857. doi: 10.1111/jocn.16237. Epub 2022 Feb 3. PMID: 35118759. |
Selection criteria for systematic reviews were not fulfilled |
|
Crnich CJ, Maki DG. The promise of novel technology for the prevention of intravascular device-related bloodstream infection. I. Pathogenesis and short-term devices. Clin Infect Dis. 2002 May 1;34(9):1232-42. doi: 10.1086/339863. Epub 2002 Apr 2. PMID: 11941550. |
Narrative review |
|
Dang FP, Li HJ, Tian JH. Comparative efficacy of 13 antimicrobial dressings and different securement devices in reducing catheter-related bloodstream infections: A Bayesian network meta-analysis. Medicine (Baltimore). 2019 Apr;98(14):e14940. doi: 10.1097/MD.0000000000014940. PMID: 30946317; PMCID: PMC6455863. |
Selection criteria for systematic reviews were not fulfilled |
|
Daniels KR, Frei CR. Antimicrobial-impregnated discs for prevention of intravenous catheter-related infections. Am J Infect Dis. 2012 Febr;8(1):50-59. |
Selection criteria for systematic reviews were not fulfilled |
|
de Campos Pereira Silveira RC, Dos Reis PED, Ferreira EB, Braga FTMM, Galvão CM, Clark AM. Dressings for the central venous catheter to prevent infection in patients undergoing hematopoietic stem cell transplantation: a systematic review and meta-analysis. Support Care Cancer. 2020 Feb;28(2):425-438. doi: 10.1007/s00520-019-05065-9. Epub 2019 Sep 6. PMID: 31493134. |
Selection criteria for systematic reviews were not fulfilled |
|
de Jonge RC, Polderman KH, Gemke RJ. Central venous catheter use in the pediatric patient: mechanical and infectious complications. Pediatr Crit Care Med. 2005 May;6(3):329-39. doi: 10.1097/01.PCC.0000161074.94315.0A. PMID: 15857534. |
Narrative review |
|
Düzkaya DS, Sahiner NC, Uysal G, Yakut T, Çitak A. Chlorhexidine-Impregnated Dressings and Prevention of Catheter-Associated Bloodstream Infections in a Pediatric Intensive Care Unit. Crit Care Nurse. 2016 Dec;36(6):e1-e7. doi: 10.4037/ccn2016561. PMID: 27908954. |
I/C didn’t meet PICOS |
|
Edwards M, Rickard CM, Rapchuk I, Corley A, Marsh N, Spooner AJ, Mihala G, Fraser JF. A pilot trial of bordered polyurethane dressings, tissue adhesive and sutureless devices compared with standard polyurethane dressings for securing short-term arterial catheters. Crit Care Resusc. 2014 Sep;16(3):175-83. PMID: 25161019. |
P didn’t meet PICOS |
|
Elliott T. Intravascular catheter-related sepsis--novel methods of prevention. Intensive Care Med. 2000;26 Suppl 1:S45-50. doi: 10.1007/s001340051118. PMID: 10786958. |
Narrative review I/C didn’t meet PICOS |
|
Elliott TS. The prevention of central venous catheter-related sepsis. J Chemother. 2001 Nov;13 Spec No 1(1):234-8. doi: 10.1179/joc.2001.13.Supplement-2.234. PMID: 11936372. |
Narrative review |
|
Fohlen A, Briant AR, Dutheil JJ, Le Pennec V, Pelage JP, Parienti JJ. Complications of peripherally inserted central catheters in adult hospitalized patients and outpatients in the KTFIXPICC study: A randomized controlled trial evaluating a fixation device KT FIX Plussystem. Am J Infect Control. 2022 Aug;50(8):916-921. doi: 10.1016/j.ajic.2021.12.014. Epub 2021 Dec 29. PMID: 34973357. |
Outcomes werenot reported by setting |
|
Fontenoy C, Kamel SO. Silver in the medical devices/equipments: Marketing or real clinical interest?. Le Pharmacien Hospitalier. 2011;46(1):e1-e11. doi: 10.1016/j.phhp.2010.11.001. |
Narrative review |
|
Frey AM, Schears GJ. Why are we stuck on tape and suture? A review of catheter securement devices. J Infus Nurs. 2006 Jan-Feb;29(1):34-8. doi: 10.1097/00129804-200601000-00007. PMID: 16428999. |
Narrative review |
|
Gallieni M. Transparent film for intravascular catheter exit-site dressings. J Vasc Access. 2004 Apr-Jun;5(2):69-75. doi: 10.1177/112972980400500205. PMID: 16596544. |
Narrative review |
|
Gillies D, O'Riordan E, Carr D, O'Brien I, Frost J, Gunning R. Central venous catheter dressings: a systematic review. J Adv Nurs. 2003 Dec;44(6):623-32. doi: 10.1046/j.0309-2402.2003.02852.x. PMID: 14651685. |
Selection criteria for systematic reviews were not fulfilled |
|
Goossens GA, Grumiaux N, Janssens C, Jérôme M, Fieuws S, Moons P, Stas M, Maleux G. SecurAstaP trial: securement with SecurAcath versus StatLock for peripherally inserted central catheters, a randomised open trial. BMJ Open. 2018 Feb 24;8(2):e016058. doi: 10.1136/bmjopen-2017-016058. PMID: 29478011; PMCID: PMC5855473. |
I/C didn’t meet PICOS |
|
Gravante F, Lombardi A, Gagliardi AM, Pucci A, Latina R. Dressings and Securement Devices of Peripheral Arterial Catheters in Intensive Care Units and Operating Theaters: A Systematic Review. Dimens Crit Care Nurs. 2020 Sep/Oct;39(5):242-250. doi: 10.1097/DCC.0000000000000433. PMID: 32740194. |
P didn’t meet PICOS |
|
Guanche-Sicilia A, Sánchez-Gómez MB, Castro-Peraza ME, Rodríguez-Gómez JÁ, Gómez-Salgado J, Duarte-Clíments G. Prevention and Treatment of Phlebitis Secondary to the Insertion of a Peripheral Venous Catheter: A Scoping Review from a Nursing Perspective. Healthcare (Basel). 2021 May 19;9(5):611. doi: 10.3390/healthcare9050611. PMID: 34069674; PMCID: PMC8160666. |
Selection criteria for systematic reviews were not fulfilled |
|
Günther SC, Schwebel C, Hamidfar-Roy R, Bonadona A, Lugosi M, Ara-Somohano C, Minet C, Potton L, Cartier JC, Vésin A, Chautemps M, Styfalova L, Ruckly S, Timsit JF. Complications of intravascular catheters in ICU: definitions, incidence and severity. A randomized controlled trial comparing usual transparent dressings versus new-generation dressings (the ADVANCED study). Intensive Care Med. 2016 Nov;42(11):1753-1765. doi: 10.1007/s00134-016-4582-2. Epub 2016 Oct 12. PMID: 27734108. |
Outcomes were not reported by device type |
|
Healy C, Baldwin I, Currey J, Driscoll A. A randomised controlled trial to determine the effectiveness of a radial arterial catheter dressing. Crit Care Resusc. 2018 Mar;20(1):61-67. PMID: 29458323. |
I/C didn’t meet PICOS |
|
Heard SO. Catheter-related infection: diagnosis, prevention and treatment. Ann Acad Med Singap. 2001 Jul;30(4):419-29. PMID: 11503552. |
Narrative review |
|
Hina HR, McDowell JRS. Minimising central line-associated bloodstream infection rate in inserting central venous catheters in the adult intensive care units. J Clin Nurs. 2017 Dec;26(23-24):3962-3973. doi: 10.1111/jocn.13824. Epub 2017 Jul 5. PMID: 28334482. |
I/C didn’t meet PICOS |
|
Huang EY, Chen C, Abdullah F, Aspelund G, Barnhart DC, Calkins CM, Cowles RA, Downard CD, Goldin AB, Lee SL, St Peter SD, Arca MJ; 2011 American Pediatric Surgical Association Outcomes and Clinical Trials Committee. Strategies for the prevention of central venous catheter infections: an American Pediatric Surgical Association Outcomes and Clinical Trials Committee systematic review. J Pediatr Surg. 2011 Oct;46(10):2000-11. doi: 10.1016/j.jpedsurg.2011.06.017. PMID: 22008341. |
Selection criteria for systematic reviews were not fulfilled |
|
Janum S, Zingg W, Classen V, Afshari A. Bench-to-bedside review: Challenges of diagnosis, care and prevention of central catheter-related bloodstream infections in children. Crit Care. 2013 Aug 28;17(4):238. doi: 10.1186/cc12730. PMID: 24041298; PMCID: PMC4057411. |
Narrative review |
|
Jia L, Bi D, Zheng H, Chen C, Lin Y, Yang C, Zhang W, Wang Y. Analgesic Pump Tubing Securement to Prevent Dislodgement of Peripheral Vein Indwelling Catheter. J Perianesth Nurs. 2021 Oct;36(5):480-486.e1. doi: 10.1016/j.jopan.2020.08.003. Epub 2021 Jun 25. PMID: 34183256. |
I/C didn’t meet PICOS |
|
Jitrungruengnij N, Anugulruengkitt S, Rattananupong T, Prinyawat M, Jantarabenjakul W, Wacharachaisurapol N, Chatsuwan T, Janewongwirot P, Suchartlikitwong P, Tawan M, Kanchanabutr P, Pancharoen C, Puthanakit T. Efficacy of chlorhexidine patches on central line-associated bloodstream infections in children. Pediatr Int. 2020 Jul;62(7):789-796. doi: 10.1111/ped.14200. Epub 2020 Jul 9. PMID: 32065485. |
I/C didn’t meet PICOS |
|
Kleidon TM, Ullman AJ, Gibson V, Chaseling B, Schoutrop J, Mihala G, Rickard CM. A Pilot Randomized Controlled Trial of Novel Dressing and Securement Techniques in 101 Pediatric Patients. J Vasc Interv Radiol. 2017 Nov;28(11):1548-1556.e1. doi: 10.1016/j.jvir.2017.07.012. Epub 2017 Sep 19. PMID: 28893464. |
I/C didn’t meet PICOS |
|
Kleidon TM, Rickard CM, Gibson V, Mihala G, Schults JA, Xu HG, Bauer MJ, Marsh N, Larsen EN, Cattanach P, Ullman AJ. Smile - Secure my intravenous line effectively: A pilot randomised controlled trial of peripheral intravenous catheter securement in paediatrics. J Tissue Viability. 2020 May;29(2):82-90. doi: 10.1016/j.jtv.2020.03.006. Epub 2020 Mar 26. PMID: 32249091. |
I/C didn’t meet PICOS |
|
Larsen EN, Corley A, Mitchell M, Lye I, Powell M, Tom S, Mihala G, Ullman AJ, Gibson V, Marsh N, Kleidon TM, Rapchuk IL, Rickard CM. A pilot randomised controlled trial of dressing and securement methods to prevent arterial catheter failure in intensive care. Aust Crit Care. 2021 Jan;34(1):38-46. doi: 10.1016/j.aucc.2020.05.004. Epub 2020 Jul 19. PMID: 32698986. |
P didn’t meet PICOS |
|
Lee OK, Johnston L. A systematic review for effective management of central venous catheters and catheter sites in acute care paediatric patients. Worldviews Evid Based Nurs. 2005;2(1):4-13; discussion 14-5. doi: 10.1111/j.1524-475X.2005.04085.x. PMID: 17040550. |
Selection criteria for systematic reviews were not fulfilled P didn’t meet PICOS (for relevant studies) |
|
Lešnik A, Gorenjak M, Žumer S, Zorčič V, Mišanović Ž, Majhenič M, Podstenšek A, Toplak M, Fekonja U, Markota A. Tissue adhesives for peripheral intravenous catheter securement: A prospective randomized controlled pilot trial. Am J Emerg Med. 2021 Jun;44:128-131. doi: 10.1016/j.ajem.2021.01.088. Epub 2021 Feb 1. PMID: 33610080. |
I/C didn’t meet PICOS |
|
Lorente L. Prevention of catheter-related infection: Which catheter, which access and which insertion technique should be chosen? Reanimation 2013;22:S409-S416. doi: 10.1007/s13546-012-0538-x. |
Narrative review |
|
Loveday HP, Wilson JA, Prieto J, Wilcox MH. epic3: revised recommendation for intravenous catheter and catheter site care. J Hosp Infect. 2016 Apr;92(4):346-8. doi: 10.1016/j.jhin.2015.11.011. Epub 2015 Dec 15. PMID: 26979872. |
Narrative review |
|
Luo X, Guo Y, Yu H, Li S, Yin X. Effectiveness, safety and comfort of StatLock securement for peripherally-inserted central catheters: A systematic review and meta-analysis. Nurs Health Sci. 2017 Dec;19(4):403-413. doi: 10.1111/nhs.12361. Epub 2017 Jul 21. PMID: 28730735. |
Selection criteria for systematic reviews were not fulfilled Full texts of included studies were not available |
|
Machado AF, Pedreira Mda L, Chaud MN. Adverse events related to the use of peripheral intravenous catheters in children according to dressing regimens. Rev Lat Am Enfermagem. 2008 May-Jun;16(3):362-7. English, Portuguese, Spanish. doi: 10.1590/s0104-11692008000300005. PMID: 18695807. |
P didn’t meet PICOS |
|
Macmillan T, Pennington M, Summers JA, Goddard K, Zala D, Herz N, Peacock JL, Keevil S, Chalkidou A. SecurAcath for Securing Peripherally Inserted Central Catheters: A NICE Medical Technology Guidance. Appl Health Econ Health Policy. 2018 Dec;16(6):779-791. doi: 10.1007/s40258-018-0427-1. PMID: 30123950; PMCID: PMC6244619. |
Selection criteria for systematic reviews were not fulfilled |
|
Malek AE, Raad II. Preventing catheter-related infections in cancer patients: a review of current strategies. Expert Rev Anti Infect Ther. 2020 Jun;18(6):531-538. doi: 10.1080/14787210.2020.1750367. Epub 2020 Apr 12. PMID: 32237923. |
Narrative review |
|
Marschall J, Mermel LA, Fakih M, Hadaway L, Kallen A, O'Grady NP, Pettis AM, Rupp ME, Sandora T, Maragakis LL, Yokoe DS; Society for Healthcare Epidemiology of America. Strategies to prevent central line-associated bloodstream infections in acute care hospitals: 2014 update. Infect Control Hosp Epidemiol. 2014 Jul;35(7):753-71. doi: 10.1086/676533. PMID: 24915204. |
Narrative review |
|
Marsh N, Webster J, Mihala G, Rickard CM. Devices and dressings to secure peripheral venous catheters to prevent complications. Cochrane Database Syst Rev. 2015 Jun 12;2015(6):CD011070. doi: 10.1002/14651858.CD011070.pub2. PMID: 26068958; PMCID: PMC10686038. A |
P didn’t meet PICOS |
|
Marsh N, Webster J, Flynn J, Mihala G, Hewer B, Fraser J, Rickard CM. Securement methods for peripheral venous catheters to prevent failure: a randomised controlled pilot trial. J Vasc Access. 2015 May-Jun;16(3):237-44. doi: 10.5301/jva.5000348. Epub 2015 Feb 4. PMID: 25656258. B |
P didn’t meet PICOS |
|
Marsh N, Webster J, Mihala G, Rickard CM. Devices and dressings to secure peripheral venous catheters: A Cochrane systematic review and meta-analysis. Int J Nurs Stud. 2017 Feb;67:12-19. doi: 10.1016/j.ijnurstu.2016.11.007. Epub 2016 Nov 16. PMID: 27889585. |
Duplicate publication (Marsh, 2015) |
|
Marsh N, Larsen E, Genzel J, Mihala G, Ullman AJ, Kleidon T, Cadigan S, Rickard CM. A novel integrated dressing to secure peripheral intravenous catheters in an adult acute hospital: a pilot randomised controlled trial. Trials. 2018 Oct 30;19(1):596. doi: 10.1186/s13063-018-2985-9. PMID: 30376880; PMCID: PMC6208178. |
I/C didn’t meet PICOS |
|
Mermel LA. Prevention of intravascular catheter-related infections. Ann Intern Med. 2000 Mar 7;132(5):391-402. doi: 10.7326/0003-4819-132-5-200003070-00009. Erratum in: Ann Intern Med 2000 Sep 5;133(5):395. PMID: 10691590. |
Narrative review |
|
Miller DL, O'Grady NP; Society of Interventional Radiology. Guidelines for the prevention of intravascular catheter-related infections: recommendations relevant to interventional radiology for venous catheter placement and maintenance. J Vasc Interv Radiol. 2012 Aug;23(8):997-1007. doi: 10.1016/j.jvir.2012.04.023. PMID: 22840801; PMCID: PMC3725970. |
Summary of CDC guideline 2011 |
|
Miller SE, Maragakis LL. Central line-associated bloodstream infection prevention. Curr Opin Infect Dis. 2012 Aug;25(4):412-22. doi: 10.1097/QCO.0b013e328355e4da. PMID: 22766647. |
Narrative review |
|
Mimoz O, Chopra V, Timsit JF. What's new in catheter-related infection: skin cleansing and skin antisepsis. Intensive Care Med. 2016 Nov;42(11):1784-1786. doi: 10.1007/s00134-016-4244-4. Epub 2016 Feb 3. PMID: 26846517. |
Opinion paper |
|
Needham R, Strehle EM. Evaluation of dressings used with local anaesthetic cream and for peripheral venous cannulation. Paediatr Nurs. 2008 Oct;20(8):34-6. doi: 10.7748/paed2008.10.20.8.34.c8266. PMID: 18980039. |
Quasi-randomized controlled trial I/C didn’t meet PICOS |
|
O'Grady NP, Alexander M, Dellinger EP, Gerberding JL, Heard SO, Maki DG, Masur H, McCormick RD, Mermel LA, Pearson ML, Raad II, Randolph A, Weinstein RA; Healthcare Infection Control Practices Advisory Committee. Guidelines for the prevention of intravascular catheter-related infections. Am J Infect Control. 2002 Dec;30(8):476-89. doi: 10.1067/mic.2002.129427. PMID: 12461511. |
Replaced by CDC guideline 2011 (update 2017) |
|
O'Grady NP, Alexander M, Dellinger EP, Gerberding JL, Heard SO, Maki DG, Masur H, McCormick RD, Mermel LA, Pearson ML, Raad II, Randolph A, Weinstein RA; Healthcare Infection Control Practices Advisory Committee. Guidelines for the prevention of intravascular catheter-related infections. Clin Infect Dis. 2002;35(11):1281-1307. doi: 10.1086/344188. |
Replaced by CDC guideline 2011 (update 2017) |
|
O'Grady NP, Alexander M, Dellinger EP, Gerberding JL, Heard SO, Maki DG, Masur H, McCormick RD, Mermel LA, Pearson ML, Raad II, Randolph A, Weinstein RA; Healthcare Infection Control Practices Advisory Committee. Guidelines for the prevention of intravascular catheter-related infections. Infect Control Hosp Epidemiol. 2002 Dec;23(12):759-69. doi: 10.1086/502007. PMID: 12517020. |
Replaced by CDC guideline 2011 (update 2017) |
|
O'Grady NP. Applying the science to the prevention of catheter-related infections. J Crit Care. 2002 Jun;17(2):114-21. doi: 10.1053/jcrc.2002.34366. PMID: 12096374. |
Narrative review |
|
O'Horo JC, Maki DG, Krupp AE, Safdar N. Arterial catheters as a source of bloodstream infection: a systematic review and meta-analysis. Crit Care Med. 2014 Jun;42(6):1334-9. doi: 10.1097/CCM.0000000000000166. PMID: 24413576. |
Selection criteria for systematic reviews were not fulfilled |
|
Olson K, Rennie RP, Hanson J, Ryan M, Gilpin J, Falsetti M, Heffner T, Gaudet S. Evaluation of a no-dressing intervention for tunneled central venous catheter exit sites. J Infus Nurs. 2004 Jan-Feb;27(1):37-44. doi: 10.1097/00129804-200401000-00006. PMID: 14734986. |
O didn’t meet PICOS |
|
Özkula U, Özhasenekler A, Kurtoğlu Çelik G, Tanrıverdi F, Pamukçu Günaydın G, Ergin M, Yıldırım Ç, Gökhan Ş. Tissue adhesives to secure peripheral intravenous catheters: A randomized controlled trial in patients over 65 years. Turk J Emerg Med. 2018 Aug 23;19(1):12-15. doi: 10.1016/j.tjem.2018.08.003. PMID: 30793059; PMCID: PMC6370908. |
P didn’t meet PICOS |
|
Padilla-Nula F, Bergua-Lorente A, Farrero-Mena J, Escolà-Nogués A, Llauradó-Mateu M, Serret-Nuevo C, Bellon F. Effectiveness of cyanoacrylate glue in the fixation of midline catheters and peripherally inserted central catheters in hospitalised adult patients: Randomised clinical trial (CIANO-ETI). SAGE Open Med. 2023 Apr 28;11:20503121231170743. doi: 10.1177/20503121231170743. PMID: 37152837; PMCID: PMC10155010. |
Outcomes were not reported by device type |
|
Patel PK, Gupta A, Vaughn VM, Mann JD, Ameling JM, Meddings J. Review of Strategies to Reduce Central Line-Associated Bloodstream Infection (CLABSI) and Catheter-Associated Urinary Tract Infection (CAUTI) in Adult ICUs. J Hosp Med. 2018 Feb 1;13(2):105-116. doi: 10.12788/jhm.2856. Epub 2017 Nov 8. PMID: 29154382. |
Narrative review |
|
Pedrolo E, Danski MT, Vayego SA. Chlorhexidine and gauze and tape dressings for central venous catheters: a randomized clinical trial. Rev Lat Am Enfermagem. 2014 Oct;22(5):764-71. doi: 10.1590/0104-1169.3443.2478. PMID: 25493671; PMCID: PMC4292677. A |
I/C didn’t meet PICOS |
|
Pinelli F, Pittiruti M, Van Boxtel T, Barone G, Biffi R, Capozzoli G, Crocoli A, Elli S, Elisei D, Fabiani A, Garrino C, Graziano U, Montagnani L, Prato AP, Scoppettuolo G, Zadra N, Zanaboni C, Zerla P, Konstantinou E, Jones M, Rosay H, Simcock L, Stas M, Pepe G. GAVeCeLT-WoCoVA Consensus on subcutaneously anchored securement devices for the securement of venous catheters: Current evidence and recommendations for future research. J Vasc Access. 2021 Sep;22(5):716-725. doi: 10.1177/1129729820924568. Epub 2020 Jul 2. PMID: 32613887. |
Selection criteria for systematic reviews were not fulfilled |
|
Pittiruti M, Scoppettuolo G, Dolcetti L, Celentano D, Emoli A, Marche B, Musarò A. Clinical experience of a subcutaneously anchored sutureless system for securing central venous catheters. Br J Nurs. 2019 Jan 24;28(2):S4-S14. doi: 10.12968/bjon.2019.28.2.S4. PMID: 30673323. |
Observational study |
|
Pratt RJ, Pellowe CM, Wilson JA, Loveday HP, Harper PJ, Jones SR, McDougall C, Wilcox MH. epic2: National evidence-based guidelines for preventing healthcare-associated infections in NHS hospitals in England. J Hosp Infect. 2007 Feb;65 Suppl 1:S1-64. doi: 10.1016/S0195-6701(07)60002-4. PMID: 17307562; PMCID: PMC7134414. |
Narrative review |
|
Puig-Asensio M, Marra AR, Childs CA, Kukla ME, Perencevich EN, Schweizer ML. Effectiveness of chlorhexidine dressings to prevent catheter-related bloodstream infections. Does one size fit all? A systematic literature review and meta-analysis. Infect Control Hosp Epidemiol. 2020 Dec;41(12):1388-1395. doi: 10.1017/ice.2020.356. Epub 2020 Sep 16. PMID: 32935659. |
Selection criteria for systematic reviews were not fulfilled |
|
Rai M, Dexter T. Intravascular catheters and sepsis. Care of the Critically Ill. 2001;17(1):21-25. |
Narrative review |
|
Reynolds H, Taraporewalla K, Tower M, Mihala G, Tuffaha HW, Fraser JF, Rickard CM. Novel technologies can provide effective dressing and securement for peripheral arterial catheters: A pilot randomised controlled trial in the operating theatre and the intensive care unit. Aust Crit Care. 2015 Aug;28(3):140-8. doi: 10.1016/j.aucc.2014.12.001. Epub 2015 Jan 9. PMID: 25583412. |
P didn’t meet PICOS |
|
Richardson D. Vascular access nursing practice, standards of care, and strategies to prevent infection: A review of skin cleansing agents and dressing materials (part 1 of a 3-part series). JAVA - J Association Vasc Access. 2006;11(4):215-221. doi: 10.2309/java.11-4-14. |
Narrative review |
|
Rickard CM, Marsh N, Webster J, Runnegar N, Larsen E, McGrail MR, Fullerton F, Bettington E, Whitty JA, Choudhury MA, Tuffaha H, Corley A, McMillan DJ, Fraser JF, Marshall AP, Playford EG. Dressings and securements for the prevention of peripheral intravenous catheter failure in adults (SAVE): a pragmatic, randomised controlled, superiority trial. Lancet. 2018 Aug 4;392(10145):419-430. doi: 10.1016/S0140-6736(18)31380-1. Epub 2018 Jul 26. PMID: 30057103. |
P didn’t meet PICOS |
|
Rosado V, Romanelli RM, Camargos PA. Risk factors and preventive measures for catheter-related bloodstream infections. J Pediatr (Rio J). 2011 Nov-Dec;87(6):469-77. doi: 10.2223/JPED.2134. PMID: 22170387. |
Narrative review |
|
Royer T. Improving short peripheral IV outcomes: a clinical trial of two securement methods. J Association Vasc Access. 2003;8;45-49. doi: 10.2309/1552-8855-8.4.45. |
Narrative review |
|
Rubinson L, Diette GB. Best practices for insertion of central venous catheters in intensive-care units to prevent catheter-related bloodstream infections. J Lab Clin Med. 2004 Jan;143(1):5-13. doi: 10.1016/j.lab.2003.07.003. PMID: 14749680. |
Narrative review |
|
Safdar N, Kluger DM, Maki DG. A review of risk factors for catheter-related bloodstream infection caused by percutaneously inserted, noncuffed central venous catheters: implications for preventive strategies. Medicine (Baltimore). 2002 Nov;81(6):466-79. doi: 10.1097/00005792-200211000-00007. PMID: 12441903. |
Selection criteria for systematic reviews were not fulfilled |
|
Safdar N, O'Horo JC, Maki DG. Arterial catheter-related bloodstream infection: incidence, pathogenesis, risk factors and prevention. J Hosp Infect. 2013 Nov;85(3):189-95. doi: 10.1016/j.jhin.2013.06.018. Epub 2013 Sep 23. PMID: 24070632. |
Description of results of two non-published RCTs |
|
Safdar N, O'Horo JC, Ghufran A, Bearden A, Didier ME, Chateau D, Maki DG. Chlorhexidine-impregnated dressing for prevention of catheter-related bloodstream infection: a meta-analysis. Crit Care Med. 2014 Jul;42(7):1703-13. doi: 10.1097/CCM.0000000000000319. PMID: 24674924; PMCID: PMC4258905. |
Selection criteria for systematic reviews were not fulfilled |
|
San Martín Rodríguez L, Henríquez Azcona A, Tina Majuelo P. Comparación de un apósito transparente y otro de gasa. Mantenimiento de vías venosas [Comparison of a transparent dressing with gauze. Venous blood vessel maintenance]. Rev Enferm. 2002 Feb;25(2):12-6. Spanish. PMID: 13677776. |
Foreign language (Spanish) |
|
Schears GJ. Summary of product trials for 10, 164 patients: comparing an intravenous stabilizing device to tape. J Infus Nurs. 2006 Jul-Aug;29(4):225-31. doi: 10.1097/00129804-200607000-00009. Erratum in: J Infus Nurs. 2006 Sep-Oct;29(5):242. PMID: 16858255. |
Narrative review Full text was not available |
|
Schiffer CA, Mangu PB, Wade JC, Camp-Sorrell D, Cope DG, El-Rayes BF, Gorman M, Ligibel J, Mansfield P, Levine M. Central venous catheter care for the patient with cancer: American Society of Clinical Oncology clinical practice guideline. J Clin Oncol. 2013 Apr 1;31(10):1357-70. doi: 10.1200/JCO.2012.45.5733. Epub 2013 Mar 4. PMID: 23460705. |
Archived / supplementary data (search strategy, evidence tables and risk of bias tables) were not available |
|
Smith B, Royer TI. New standards for improving peripheral i.v. catheter securement. Nursing. 2007 Mar;37(3):72-4. doi: 10.1097/01.nurse.0000261859.31596.3e. PMID: 17546791. |
Observational study |
|
Sreeramoju P. Preventing healthcare-associated infections: beyond best practice. Am J Med Sci. 2013 Mar;345(3):239-44. doi: 10.1097/MAJ.0b013e31824435e6. PMID: 22270397. |
Narrative review |
|
Storti M. L’impiego di cerotto inpoliuretano nella medicazionedel CVC riduce maggiormenteil rischio di infezione rispettoalla medicazione con garzasterile? [Does the use of a polyurethane patch in the CVC dressing further reduce the risk of infection compared with sterile gauze dressing?]. Infez Med. 2011 Jun;19(2):67-73. Italian. PMID: 21753246. |
Foreign language (Italian) |
|
Theaker C. Infection control issues in central venous catheter care. Intensive Crit Care Nurs. 2005 Apr;21(2):99-109. doi: 10.1016/j.iccn.2004.10.007. Epub 2004 Dec 8. PMID: 15778074. |
Narrative review |
|
Timsit JF. Diagnosis and prevention of catheter-related infections. Curr Opin Crit Care. 2007 Oct;13(5):563-71. doi: 10.1097/MCC.0b013e3282efa03f. PMID: 17762237. |
Narrative review Full text was not available |
|
Timsit JF, Baleine J, Bernard L, Calvino-Gunther S, Darmon M, Dellamonica J, Desruennes E, Leone M, Lepape A, Leroy O, Lucet JC, Merchaoui Z, Mimoz O, Misset B, Parienti JJ, Quenot JP, Roch A, Schmidt M, Slama M, Souweine B, Zahar JR, Zingg W, Bodet-Contentin L, Maxime V. Expert consensus-based clinical practice guidelines management of intravascular catheters in the intensive care unit. Ann Intensive Care. 2020 Sep 7;10(1):118. doi: 10.1186/s13613-020-00713-4. PMID: 32894389; PMCID: PMC7477021. |
Selection criteria for systematic reviews were not fulfilled |
|
Ullman AJ, Cooke ML, Mitchell M, Lin F, New K, Long DA, Mihala G, Rickard CM. Dressings and securement devices for central venous catheters (CVC). Cochrane Database Syst Rev. 2015 Sep 10;2015(9):CD010367. doi: 10.1002/14651858.CD010367.pub2. PMID: 26358142; PMCID: PMC6457749. A |
Updated (Xu, 2024)
|
|
Ullman AJ, Cooke M, Rickard CM. Examining the role of securement and dressing products to prevent central venous access device failure: A narrative review. J Association Vasc Access 2015;20(2):99-110. B |
Narrative review Full text was not available |
|
Ullman AJ, Cooke ML, Mitchell M, Lin F, New K, Long DA, Mihala G, Rickard CM. Dressing and securement for central venous access devices (CVADs): A Cochrane systematic review. Int J Nurs Stud. 2016 Jul;59:177-96. doi: 10.1016/j.ijnurstu.2016.04.003. Epub 2016 Apr 24. PMID: 27222463. |
Duplicate publication (Ullman, 2015A) |
|
Walz JM, Memtsoudis SG, Heard SO. Prevention of central venous catheter bloodstream infections. J Intensive Care Med. 2010 May-Jun;25(3):131-8. doi: 10.1177/0885066609358952. Epub 2010 Jan 19. PMID: 20089527. |
Narrative review |
|
Webster J, Gillies D, O'Riordan E, Sherriff KL, Rickard CM. WITHDRAWN: Gauze and tape and transparent polyurethane dressings for central venous catheters. Cochrane Database Syst Rev. 2016 May 4;2016(5):CD003827. doi: 10.1002/14651858.CD003827.pub3. PMID: 27144903; PMCID: PMC10680418. |
Withdrawn / topic covered elsewhere (Ullman, 2015A) |
|
Webster J, Larsen E, Marsh N, Choudhury A, Harris P, Rickard CM. Chlorhexidine gluconate or polyhexamethylene biguanide disc dressing to reduce the incidence of central-line-associated bloodstream infection: a feasibility randomized controlled trial (the CLABSI trial). J Hosp Infect. 2017 Jul;96(3):223-228. doi: 10.1016/j.jhin.2017.04.009. Epub 2017 Apr 16. PMID: 28526171. |
I/C didn’t meet PICOS |
|
Wei L, Li Y, Li X, Bian L, Wen Z, Li M. Chlorhexidine-impregnated dressing for the prophylaxis of central venous catheter-related complications: a systematic review and meta-analysis. BMC Infect Dis. 2019 May 16;19(1):429. doi: 10.1186/s12879-019-4029-9. PMID: 31096918; PMCID: PMC6524337. |
Included RCTs were all described in Xu, 2024 |
|
Yang Q, Lei S. Alginate Dressing Application in Hemostasis After Using Seldinger Peripherally Inserted Central Venous Catheter in Tumor Patients. Indian J Hematol Blood Transfus. 2015 Dec;31(4):434-8. doi: 10.1007/s12288-014-0490-1. Epub 2014 Dec 28. PMID: 26306067; PMCID: PMC4542767. |
Quasi-randomized controlled trial |
|
Yu K, Lu M, Meng Y, Zhao Y, Li Z. Chlorhexidine gluconate transparent dressing does not decrease central line-associated bloodstream infection in critically ill patients: A randomized controlled trial. Int J Nurs Pract. 2019 Dec;25(6):e12776. doi: 10.1111/ijn.12776. Epub 2019 Oct 20. PMID: 31631496; PMCID: PMC9285536. |
O didn’t meet PICOS |
|
Zeidman A, Ram E, Bank Y, Stein GY, Horowitz A, Dresnik Z. The efficacy and safety of MICROMAT as an intravenous site infection protector. Microbial ecology in health and disease. 2004;16(4):211-213. doi: 10.1080/08910600410025428. |
Outcomes were not reported by device type |
|
Zhang XW, Huang W, Wei HY. Modified fixation of indwelling intravenous catheter in management of exudate. Eur Rev Med Pharmacol Sci. 2017 Jul;21(3 Suppl):93-96. PMID: 28745783. |
P was not sufficiently described |
|
Zhang S, Lingle BS, Phelps S. A Revolutionary, Proven Solution to Vascular Access Concerns: A Review of the Advantageous Properties and Benefits of Catheter Securement Cyanoacrylate Adhesives. J Infus Nurs. 2022 May-Jun 01;45(3):154-164. doi: 10.1097/NAN.0000000000000467. PMID: 35537003. |
Selection criteria for systematic reviews were not fulfilled |
|
Zingg W, Barton A, Bitmead J, Eggimann P, Pujol M, Simon A, Tatzel J. Best practice in the use of peripheral venous catheters: A scoping review and expert consensus. Infect Prev Pract. 2023 Feb 3;5(2):100271. doi: 10.1016/j.infpip.2023.100271. PMID: 36910422; PMCID: PMC9995289. |
Expert consensus paper |
|
(No authors listed). Managing bloodstream infections associated with intravascular catheters. Drug Ther Bull. 2001 Oct;39(10):75-80. doi: 10.1136/dtb.2001.391075. PMID: 11706519. |
Narrative review |
|
(No authors listed). Preventing catheter-related infectious complications. J Support Oncol. 2007 Feb;5(2):70-1. PMID: 17348360. |
Full text was not available |
C = comparator; I = intervention; O = outcome; P = population; RCT = randomized controlled trial; S = setting.
In- and excluded systematic reviews – reference lists
|
Ali SMM, Koul SS, Memon MI, Pasha TMU, Afghan S, Tahir F. Comparison of chlorhexidine based dressing versus simple occlusive dressing in preventing catheter related bloodstream infections at medical ICU in a resource constraint settings. Intensive Care Med Exp. 2015 Oct 1;3(Suppl 1):A812. doi: 10.1186/2197-425X-3-S1-A812. PMCID: PMC4798442. |
Conference abstract |
|
Andersen PT, Herlevsen P, Schaumburg H. A comparative study of 'Op-site' and 'Nobecutan gauze' dressings for central venous line care. J Hosp Infect. 1986 Mar;7(2):161-8. doi: 10.1016/0195-6701(86)90059-9. PMID: 2871079. |
Observational study |
|
Arpa Y, Aygün H, YalçInbaş Y, San D, Ulukol A. Comparison of catheter related infection rates in pediatric cardiovascular surgery patients with use of transparent cover and transparent cover saturated with chlorhexidin gluconate in central catheter care. Turkish J Res Dev Nurs. 2013;15:57–67. |
Observational study |
|
Aslan N, Yıldızdaş D, Menemencioğlu A, Korkmaz F, Horoz ÖÖ, Gündeşlioğlu ÖÖ. Çocuk yoğun bakım ünitemizde kateter ilişkili kan akımı enfeksiyonunun önlenmesi açısından standart bakım örtüsü ve klorheksidin glukonat içeren bakım örtüsünün karşılaştırılması. J Pediatr Emerg Intensive Care Med. 2020;7:24-29. doi: 10.4274/cayd.galenos.2019.75547. |
Foreign language (Turkish) Observational study Includes hemodialysis (results not reported by patient type) |
|
Conly JM, Grieves K, Peters B. A prospective, randomized study comparing transparent and dry gauze dressings for central venous catheters. J Infect Dis. 1989 Feb;159(2):310-9. doi: 10.1093/infdis/159.2.310. PMID: 2644372. |
Quasi-randomized controlled trial |
|
Corley A. SECUREment bundles for Peripheral IntraVenous Catheters in general medical and surgical patients: a feasibility trial. 2019. http://www.who.int/trialsearch/Trial2.aspx?TrialID=ACTRN 12619000026123. |
Full text was not available (page cannot be found) |
|
Craven DE, Lichtenberg DA, Kunches LM, McDonough AT, Gonzalez MI, Heeren TC, McCabe WR. A randomized study comparing a transparent polyurethane dressing to a dry gauze dressing for peripheral intravenous catheter sites. Infect Control. 1985 Sep;6(9):361-6. doi: 10.1017/s0195941700063323. PMID: 3850859. |
Quasi-randomized controlled trial |
|
Crawford AG, Fuhr JP Jr, Rao B. Cost-benefit analysis of chlorhexidine gluconate dressing in the prevention of catheter-related bloodstream infections. Infect Control Hosp Epidemiol. 2004 Aug;25(8):668-74. doi: 10.1086/502459. PMID: 15357159. |
Cost-effectiveness study (no original efficacy data) |
|
de Barros L, Arenas V, Bettencourt A, Diccini S, Fram D, Belasco A, et al. Evaluation of two types of dressings used on central venous catheters for hemodialysis. Acta Paulista de Enfermagem. 2009;22(Especial - Nefrologia):481-486. doi: 10.1590/S0103-21002009000800004. |
P didn’t meet PICOS |
|
Ergul AB, Gokcek I, Ozcan A, Cetin S, Gultekin N, Torun YA. Use of a chlorhexidine-impregnated dressing reduced catheter-related bloodstream infections caused by Gram-positive microorganisms. Pak J Med Sci. 2018 Mar-Apr;34(2):347-351. doi: 10.12669/pjms.342.14810. PMID: 29805406; PMCID: PMC5954377. |
Observational study |
|
Fang F, Zhang HY, Wang F, Yang W, Zhang JL. Comparison of three different fixiation methods for peripherally inserted central catheter. Chin J Clin Nutr. 2011;19:119–123. |
Foreign language (Chinese) |
|
Forni C, D'Alessandro F, Gambino O, Amodeo A, Pignotti E, Zanotti E, Tremosini M, Trofa C, Sabattini T, Matino F, Genco R, Schiavone M, Bombino C, Mini S, Rocchegiani L, Notarnicola T, Capezzali D, Boschi R, Loro L. [Effectiveness of the transparent sterile dressing vs standard to fix the peripheral venous catheter (PVC) on the incidence of phlebitis. A randomized controlled trial]. Assist Inferm Ric. 2012 Apr-Jun;31(2):63-9. Italian. doi: 10.1702/1131.12467. PMID: 22825293. |
Foreign language (Italian) |
|
Garland JS, Alex CP, Mueller CD, Otten D, Shivpuri C, Harris MC, Naples M, Pellegrini J, Buck RK, McAuliffe TL, Goldmann DA, Maki DG. A randomized trial comparing povidone-iodine to a chlorhexidine gluconate-impregnated dressing for prevention of central venous catheter infections in neonates. Pediatrics. 2001 Jun;107(6):1431-6. doi: 10.1542/peds.107.6.1431. PMID: 11389271. |
P didn’t meet PICOS |
|
Gilardi E, Piano A, Chellini P, Fiori B, Dolcetti L, Pittiruti M, Scoppettuolo G. Reduction of bacterial colonization at the exit site of peripherally inserted central catheters: A comparison between chlorhexidine-releasing sponge dressings and cyano-acrylate. J Vasc Access. 2021 Jul;22(4):597-601. doi: 10.1177/1129729820954743. Epub 2020 Sep 4. PMID: 32883154. |
Quasi-randomized controlled trial |
|
Gu Y, Yuan J, Hu J, et al. Comparison of the effects and costs of three kinds of application in venous central venous maintenance. Chin J Nurs. 2013;48:510-513. |
Full text was not available Foreign language (Chinese) |
|
Hagerstrom M, Matthleson K, Thuessen C. A randomised dressing method for haemodialysis patients with permanent central venous catheters. 4th European Conference on Advances in Wound Management proceedings 1994:abstract 197. |
Conference abstract P didn’t meet PICOS |
|
He LJ. A comparative study of the StatLock device and tape for peripherally inserted central catheter. Chin J Clin Nutr. 2012;20:126-127. |
Full text was not available Foreign language (Chinese) |
|
Hill ML, Baldwin L, Slaughter JC, Walsh WF, Weitkamp JH. A silver-alginate-coated dressing to reduce peripherally inserted central catheter (PICC) infections in NICU patients: a pilot randomized controlled trial. J Perinatol. 2010 Jul;30(7):469-73. doi: 10.1038/jp.2009.190. Epub 2009 Dec 10. PMID: 20010613. |
P didn’t meet PICOS |
|
Khattak AZ, Ross R, Ngo T, Shoemaker CT. A randomized controlled evaluation of absorption of silver with the use of silver alginate (Algidex) patches in very low birth weight (VLBW) infants with central lines. J Perinatol. 2010 May;30(5):337-42. doi: 10.1038/jp.2009.169. Epub 2009 Nov 26. PMID: 19940856. |
P didn’t meet PICOS |
|
Le Corre I, Delorme M, Cournoyer S. A prospective, randomized trial comparing a transparent dressing and a dry gauze on the exit site of long term central venous catheters of hemodialysis patients. J Vasc Access. 2003 Apr-Jun;4(2):56-61. PMID: 17642061. |
P didn’t meet PICOS |
|
Li J, Li N, Fu W, Feng JK, Zhang QF. [Influence of silver ion dressing on central venous catheter-related infection in severe burn patients]. Zhonghua Shao Shang Za Zhi. 2020 Aug 20;36(8):698-703. Chinese. doi: 10.3760/cma.j.cn501120-20190519-00246. PMID: 32829608. |
Foreign language (Chinese) |
|
Lin J, Ma S. The effectiveness of StatLock device in securing peripherally inserted central catheter. J Nursing Sci. 2010;25:13-14. |
Full text was not available (title incorrect) Foreign language (Chinese) |
|
Liu LJ. A comparative study of different securement technique for peripherally inserted central catheter. Chin Health Nutr. 2012;6:159-160. (geen NLM) |
Full text was not available Foreign language (Chinese) |
|
Livesley J, Richardson S. Securing methods for peripheral cannulae. Nurs Stand. 1993 Apr 21-27;7(31):31-4. doi: 10.7748/ns.7.31.31.s41. PMID: 8512837. |
I/C didn’t meet PICOS |
|
Lucet JC, Bouadma L, Zahar JR, Schwebel C, Geffroy A, Pease S, Herault MC, Haouache H, Adrie C, Thuong M, Français A, Garrouste-Orgeas M, Timsit JF. Infectious risk associated with arterial catheters compared with central venous catheters. Crit Care Med. 2010 Apr;38(4):1030-5. doi: 10.1097/CCM.0b013e3181d4502e. PMID: 20154601. |
Observational study |
|
Madeo M, Martin CR, Turner C, Kirkby V, Thompson DR. A randomized trial comparing Arglaes (a transparent dressing containing silver ions) to Tegaderm (a transparent polyurethane dressing) for dressing peripheral arterial catheters and central vascular catheters. Intensive Crit Care Nurs. 1998 Aug;14(4):187-91. doi: 10.1016/s0964-3397(98)80512-0. PMID: 9849245. |
Quasi-randomized controlled trial |
|
Maki D, Mermel L, Kluger D, et al. The efficacy of a chlorhexidine impregnated sponge (Biopatch) for the prevention of intravascular catheter-related infection-a prospective randomized controlled multicenter study. Abstracts of the 40th Interscience Conference on Antimicrobial Agents and Chemotherapy. 2000:422. |
Conference abstract Outcomes were not reported by device type |
|
Maunoury F, Motrunich A, Palka-Santini M, Bernatchez SF, Ruckly S, Timsit JF. Cost-Effectiveness Analysis of a Transparent Antimicrobial Dressing for Managing Central Venous and Arterial Catheters in Intensive Care Units. PLoS One. 2015 Jun 18;10(6):e0130439. doi: 10.1371/journal.pone.0130439. PMID: 26086783; PMCID: PMC4472776. |
Cost-effectiveness study (no original efficacy data) |
|
Mi WP, Wang XJ, Liu HY. A randomized controlled trial to compare two securement methods of central venous catheter. Chin Nursing Research, 2011;25:1558-1559. |
Full text was not available Foreign language (Chinese) |
|
Mushahar L, Mei LW, Yusuf WS, Sivathasan S, Kamaruddin N, Idzham NJ. Exit-Site Dressing and Infection in Peritoneal Dialysis: A Randomized Controlled Pilot Trial. Perit Dial Int. 2016 Mar-Apr;36(2):135-9. doi: 10.3747/pdi.2014.00195. Epub 2015 Sep 15. PMID: 26374836; PMCID: PMC4803357. |
P didn’t meet PICOS |
|
Neufeld M. A randomized control trial of the effectiveness of opsite wound versus IV3000 in maintaining an occlusive central line dressing. Master’s thesis. The School of Graduate Studies, McMaster University, Canada. 1991. |
Master thesis |
|
Nikoletti S, Leslie G, Gandossi S, Coombs G, Wilson R. A prospective, randomized, controlled trial comparing transparent polyurethane and hydrocolloid dressings for central venous catheters. Am J Infect Control. 1999 Dec;27(6):488-96. doi: 10.1016/s0196-6553(99)70026-x. PMID: 10586152. |
I/C didn’t meet PICOS |
|
O'Horo SK, Corson D, Baum RA. Efficacy of biopatch™ in reducing catheter related infections in cuffed, tunneled central venous catheters. J Vasc Interventional Radiol 2013;24(4):S127. |
Conference abstract |
|
Pedrolo E, Santos MC, de Oliveira GLR, Mingorance P, Reichembach Danski MT, Boostel R. Chlorhexidine-impregnated dressing for central venous catheter: pilot clinical trial. Revista Enfermagem UERJ. 2014;22(6):760-4. doi: 10.12957/reuerj.2014.5547. B |
Reporting of outcome data was insufficient for data extraction |
|
Pivkina AI, Gusarov VG, Blot SI, Zhivotneva IV, Pasko NV, Zamyatin MN. Effect of an acrylic terpolymer barrier film beneath transparent catheter dressings on skin integrity, risk of dressing disruption, catheter colonisation and infection. Intensive Crit Care Nurs. 2018 Jun;46:17-23. doi: 10.1016/j.iccn.2017.11.002. Epub 2018 Mar 23. PMID: 29576395. |
I/C didn’t meet PICOS |
|
Powell C, Regan C, Fabri PJ, Ruberg RL. Evaluation of Opsite catheter dressings for parenteral nutrition: a prospective, randomized study. JPEN J Parenter Enteral Nutr. 1982 Jan-Feb;6(1):43-6. doi: 10.1177/014860718200600143. PMID: 6804654. |
Quasi-randomized controlled trial |
|
Qian YL, Meng Y, Lu QM. The effects of StatLock for PICCs securement. Chin Nursing Research. 2014;28:2506-2507. |
Full text was not available Foreign language (Chinese) |
|
Qin CY. The effect of StatLock on the fixation of the peripherally inserted central catheters. Modern Clin Nursing. 2011;10:20-21. |
Full text was not available Foreign language (Chinese) |
|
Reynolds MG, Tebbs SE, Elliott TS. Do dressings with increased permeability reduce the incidence of central venous catheter related sepsis? Intensive Crit Care Nurs. 1997 Feb;13(1):26-9. doi: 10.1016/s0964-3397(97)80688-x. PMID: 9095879. |
Quasi-randomized controlled trial |
|
Righetti M, Palmieri N, Bracchi O, Prencipe M, Bruschetta E, Colombo F, Brenna I, Stefani F, Amar K, Scalia A, Conte F. Tegaderm™ CHG dressing significantly improves catheter-related infection rate in hemodialysis patients. J Vasc Access. 2016 Sep 21;17(5):417-422. doi: 10.5301/jva.5000596. Epub 2016 Aug 1. PMID: 27516139. |
P didn’t meet PICOS |
|
Roberts B, Cheung D. Biopatch--a new concept in antimicrobial dressings for invasive devices. Aust Crit Care. 1998 Mar;11(1):16-9. doi: 10.1016/s1036-7314(98)70426-6. PMID: 9708081. |
O didn’t meet PICOS |
|
Schears GJ, Liebig C, Frey AM, Chiavacci R, Harris K, Lin RJ, Costarion AT. StatLock catheter securement device significantly reduces central venous catheter complications. Patient Safety Initiative 2000. Spotlight on Solutions: Compendium of successful Practices 2001;1:18–37. National Patient Safety Foundation. (geen NLM) |
Full text was not available |
|
Scheithauer S, Lewalter K, Schröder J, Koch A, Häfner H, Krizanovic V, Nowicki K, Hilgers RD, Lemmen SW. Reduction of central venous line-associated bloodstream infection rates by using a chlorhexidine-containing dressing. Infection. 2014 Feb;42(1):155-9. doi: 10.1007/s15010-013-0519-7. Epub 2013 Aug 9. PMID: 23934685. |
Observational study |
|
Schwebel C, Lucet JC, Vesin A, Arrault X, Calvino-Gunther S, Bouadma L, Timsit JF. Economic evaluation of chlorhexidine-impregnated sponges for preventing catheter-related infections in critically ill adults in the Dressing Study. Crit Care Med. 2012 Jan;40(1):11-7. doi: 10.1097/CCM.0b013e31822f0604. PMID: 21926570. |
Cost-effectiveness study (no original efficacy data) |
|
Stephenson C. The advantages of a precision-engineered securement device for fixation of arterial pressure-monitoring catheters. J Association Vasc Access 2005;10(3):130-132. doi: 10.2309/java.10-3-8. |
Observational study |
|
Thokala P, Arrowsmith M, Poku E, Martyn-St James M, Anderson J, Foster S, Elliott T, Whitehouse T. Economic impact of Tegaderm chlorhexidine gluconate (CHG) dressing in critically ill patients. J Infect Prev. 2016 Sep;17(5):216-223. doi: 10.1177/1757177416657162. Epub 2016 Jul 13. PMID: 27582899; PMCID: PMC4994702. |
Cost-effectiveness study (no original efficacy data) |
|
Tripepi-Bova KA, Woods KD, Loach MC. A comparison of transparent polyurethane and dry gauze dressings for peripheral i.v. catheter sites: rates of phlebitis, infiltration, and dislodgment by patients. Am J Crit Care. 1997 Sep;6(5):377-81. PMID: 9283675. |
P didn’t meet PICOS |
|
Wang ZW. A randomized controlled trial to compare three stabilization systems of peripherally inserted central venous catheter. Master thesis. Beijing, China: Peking Union Medical College Hospital. 2012. |
Master thesis. |
|
Wang HY. The effectiveness of StatLock device for PICCs securement. Chin J Traumatol Disability Medicine. 2013;21:130-131. |
Full text was not available Foreign language (Chinese) |
|
Wang HX, Xie SY, Wang H, Chu HK. The Effects of Chlorhexidine Dressing on Health Care-Associated Infection in Hospitalized Patients: A Meta-Analysis. Iran J Public Health. 2019 May;48(5):796-807. PMID: 31523635; PMCID: PMC6717407. |
Included RCTs were all described in Xu, 2024 |
|
Wille JC, Blussé van Oud Albas A, Thewessen EA. A comparison of two transparent film-type dressings in central venous therapy. J Hosp Infect. 1993 Feb;23(2):113-21. doi: 10.1016/0195-6701(93)90015-r. PMID: 8097215. |
I/C didn’t meet PICOS |
|
Ye X, Rupnow M, Bastide P, Lafuma A, Ovington L, Jarvis WR. Economic impact of use of chlorhexidine-impregnated sponge dressing for prevention of central line-associated infections in the United States. Am J Infect Control. 2011 Oct;39(8):647-654. doi: 10.1016/j.ajic.2010.11.008. Epub 2011 Jun 8. PMID: 21641681. |
Cost-effectiveness study (no original efficacy data) |
|
Young GP, Alexeyeff M, Russell DM, Thomas RJ. Catheter sepsis during parenteral nutrition: the safety of long-term OpSite dressings. JPEN J Parenter Enteral Nutr. 1988 Jul-Aug;12(4):365-70. doi: 10.1177/0148607188012004365. PMID: 3138446. |
Clinical controlled trial |
|
Zhan F, Zhang HY, Wang F, Yang W, Zhang JL. Comparison of three different fixation methods for peripherally inserted central catheter. Chin J Clin Nutritrition. 2011;19:119-123. |
Full text was not available Foreign language (Chinese) |
|
Zhang JJ, Zhou QY, Zhang YY. The application of StatLock device in PICCs securement. J Nursing Education. 2011;26:1409-1410. |
Full text was not available Foreign language (Chinese) |
C = comparator; I = intervention; O = outcome; P = population; RCT = randomized controlled trial; S = setting.
Verantwoording
Beoordelingsdatum en geldigheid
Publicatiedatum : 12-02-2026
Beoordeeld op geldigheid : 12-02-2026
Algemene gegevens
De ontwikkeling/herziening van deze richtlijnmodule werd ondersteund door het Kennisinstituut van de Federatie Medisch Specialisten (www.demedischspecialist.nl/kennisinstituut) en werd gefinancierd door het ministerie van VWS. De financier heeft geen enkele invloed gehad op de inhoud van de richtlijnmodule.
Samenstelling werkgroep
Werkgroep
- S. (Selma) Bons, Nederlandse Vereniging voor Anesthesiologie (NVA), voorzitter
- Dr. M. (Michelle) Gompelman, Nederlandse Internisten Vereniging (NIV), Nederlandse Vereniging van Internist-Infectiologen (NVII)
- R. (Renze) Jongstra, Verpleegkundigen & Verzorgenden Nederland (V&VN)
- H. (Heidy) Koene, Vereniging voor Hygiëne & Infectiepreventie in de Gezondheidszorg (VHIG)
- M.H.H. (Marc) Königs, Nederlandse Vereniging voor Intensive Care (NVIC)
- Dr. B.J. (Bart) Laan (AIOS), Nederlandse Internisten Vereniging (NIV), Nederlandse Vereniging van Internist-Infectiologen (NVII)
- K. (Kelly) Niggebrugge-Mentink, Nederlandse Vereniging van Ziekenhuisapothekers (NVZA)
- Dr. J.H. (Jan) van Zeijl, Nederlandse Vereniging voor Medische Microbiologie (NVMM)
Klankbordgroep
- Dr. J.R.A. (Jeetindra) Balak, Nederlandse Internisten Vereniging (NIV), Nederlandse Federatie voor Nefrologie (NFN)
- Werkgroep richtlijn Centraal veneuze toegang (NVvH)
- Werkgroep leidraad Infusietechnologie (NVKF)
Met ondersteuning van
- Mw. A. (Alies) Oost, informatiespecialist, Kennisinstituut van Federatie Medisch Specialisten
- Dr. H. (Haitske) Graveland, senior adviseur, Kennisinstituut van Federatie Medisch Specialisten
- Dr. M.F.Q. (Marjolein) Kluijtmans-van den Bergh, senior adviseur, Kennisinstituut van Federatie Medisch Specialisten
Belangenverklaringen
De Code ter voorkoming van oneigenlijke beïnvloeding door belangenverstrengeling is gevolgd. Alle werkgroepleden hebben schriftelijk verklaard of zij in de laatste drie jaar directe financiële belangen (betrekking bij een commercieel bedrijf, persoonlijke financiële belangen, onderzoeksfinanciering) of indirecte belangen (persoonlijke relaties, reputatiemanagement) hebben gehad. Gedurende de ontwikkeling of herziening van een module worden wijzigingen in belangen aan de voorzitter doorgegeven. De belangenverklaring wordt opnieuw bevestigd tijdens de commentaarfase.
Een overzicht van de belangen van werkgroepleden en het oordeel over het omgaan met eventuele belangen vindt u in onderstaande tabel. De ondertekende belangenverklaringen zijn op te vragen bij het secretariaat van het Kennisinstituut van de Federatie Medisch Specialisten.
|
Werkgroeplid |
Functie |
Nevenfuncties |
Gemelde belangen |
Ondernomen actie |
|
S. (Selma) Bons |
Anesthesioloog, Prinses Maxima Centrum voor kinderoncologie, Utrecht |
Lid werkgroep SRI richtlijnen
Lid bestuur sectie kinderanesthesiologie NVA
Lid SRI |
Geen |
Geen restrictie |
|
Dr. M. (Michelle) Gompelman |
Internist-infectioloog, Elkerliek Ziekenhuis, Helmond |
Lid werkgroep FMS-richtlijn Centraal veneuze toegang
|
Geen |
Geen restrictie |
|
A. (Renze) Jongstra |
Intensive care verpleegkundige |
Vicevoorzitter V&VN afdeling IC (onbetaald)
|
Geen |
Geen restrictie |
|
H. (Heidy) Koene |
Deskundige infectiepreventie, Erasmus MC, Rotterdam |
Geen |
Geen |
Geen restrictie |
|
M.H.H. (Marc) Königs |
Intensivist, Maxima Medisch Centrum. Eindhoven/Veldhoven (100%) |
Duikerarts / duikclubarts
Waarnemer intensivist Aruba en via IC (max. 200 uur/jaar) In het verleden voordrachten op congressen voor 3M en BD (betaald) - ESAIC ( Milaan / München) - WOCOVA ( Athene) - Nordic congres for vasculary acces |
Geen |
Geen restrictie (adviseurschap 3M onbetaald) |
|
Dr. B.J. (Bart) Laan |
AIOS Interne Geneeskunde, Amsterdam UMC, Amsterdam |
Geen |
Geen |
Geen restrictie |
|
K. (Kelly) Niggebrugge-Mentink |
Ziekenhuisapotheker, Hagaziekenhuis, Den Haag |
Gastdocent Fontys Hogeschool (verpleegkundig specialistenopleiding); betaald Gastspreker congres Stichting Vascular Infusion Technology; onbetaald |
Geen |
Geen restrictie |
|
Dr. J.H. (Jan) van Zeijl |
Arts-microbioloog, Certe Medische Diagnostiek & Advies, afdeling Medische Microbiologie (tot april 2024)
Waarnemend arts-microbioloog, Certe Medische Diagnostiek & Advies, afdeling Medische Microbiologie (september t/m december 2025) |
Bestuurslid (sinds januari 2024 voorzitter) Vasculitis Stichting met portefeuille vrijwilligersbeleid en Zorg, Research en Belangenbehartiging; onbetaald |
Geen |
Geen restrictie |
|
Klankbordgroeplid |
Functie |
Nevenfuncties |
Gemelde belangen |
Ondernomen actie |
|
Dr. J.R.A. (Jeetindra) Balak |
Internist, Leids UMC, Leiden |
Geen |
Geen |
Geen restrictie |
Inbreng patiëntenperspectief
Er werd aandacht besteed aan het patiëntenperspectief door het uitnodigen van Patiëntenfederatie Nederland (PFNL) voor de schriftelijke knelpunteninventarisatie. De verkregen input is meegenomen bij het opstellen van de uitgangsvragen, de keuze voor de uitkomstmaten en bij het opstellen van de overwegingen. De conceptrichtlijn is tevens voor commentaar voorgelegd aan PFNL en de eventueel aangeleverde commentaren zijn bekeken en verwerkt.
Kwalitatieve raming van mogelijke financiële gevolgen in het kader van de Wkkgz
Bij de richtlijn is conform de Wet kwaliteit, klachten en geschillen zorg (Wkkgz) een kwalitatieve raming uitgevoerd of de aanbevelingen mogelijk leiden tot substantiële financiële gevolgen. Bij het uitvoeren van deze beoordeling zijn richtlijnmodules op verschillende domeinen getoetst (zie het stroomschema op de Richtlijnendatabase).
Uit de kwalitatieve raming blijkt dat er geen substantiële financiële gevolgen zijn, zie onderstaande tabel.
|
Module |
Uitkomst raming |
Toelichting |
|
Afdekken en fixeren van centraal veneuze katheters |
Geen substantiële financiële gevolgen |
Hoewel uit de toetsing volgt dat de aanbevelingen breed toepasbaar zijn (>40.000 patiënten), volgt ook uit de toetsing dat het overgrote deel (±90%) van de zorgaanbieders en zorgverleners al aan de norm voldoet. Er worden daarom geen substantiële financiële gevolgen verwacht. |
Werkwijze
AGREE
Deze richtlijnmodule is opgesteld conform de eisen vermeld in het rapport Medisch Specialistische Richtlijnen 3.0 van de adviescommissie Richtlijnen van de Raad Kwaliteit. Dit rapport is gebaseerd op het AGREE II-instrument (Appraisal of Guidelines for Research & Evaluation II; Brouwers, 2010).
Knelpuntenanalyse en uitgangsvragen
Tijdens de voorbereidende fase inventariseerde de werkgroep de knelpunten met betrekking tot infectiepreventiemaatregelen rondom intravasculaire katheters. De werkgroep beoordeelde de aanbeveling(en) uit de eerdere WIP-richtlijn Arteriële katheters en de WIP-richtlijn Flebitis en bloedbaaninfecties door intraveneuze infuuskatheters. Tevens zijn er in de schriftelijke knelpunteninventarisatie knelpunten aangedragen door Inspectie Gezondheidszorg en Jeugd (IGJ), Landelijke Vereniging van Operatieassistenten (LVO), Nederlandse Vereniging van Anesthesiemedewerkers (NVAM), Nederlandse Vereniging van Revalidatieartsen (VRA), Nederlandse Vereniging van Ziekenhuizen (NVZ), Nederlandse Vereniging voor Heelkunde (NVvH), Nederlandse Vereniging voor Medische Microbiologie (NVMM), Rijksinstituut voor Volksgezondheid en Milieu (RIVM), Stichting Kind en Ziekenhuis, Vereniging voor Hygiëne & Infectiepreventie in de Gezondheidszorg (VHIG), Verpleegkundigen & Verzorgenden Nederland (V&VN). Een verslag hiervan is opgenomen onder Verslag schriftelijke knelpunteninventarisatie.
Op basis van de uitkomsten van de knelpuntenanalyse zijn door de werkgroep concept uitgangsvragen opgesteld en definitief vastgesteld.
Uitkomstmaten
Na het opstellen van de zoekvragen behorende bij de uitgangsvragen inventariseerde de werkgroep welke uitkomstmaten voor de patiënt relevant zijn, waarbij zowel naar gewenste als ongewenste effecten werd gekeken. Hierbij werd een maximum van acht uitkomstmaten gehanteerd. De werkgroep waardeerde deze uitkomstmaten volgens hun relatieve belang bij de besluitvorming rondom aanbevelingen, als cruciaal (kritiek voor de besluitvorming), belangrijk (maar niet cruciaal) en onbelangrijk. Ook definieerde de werkgroep tenminste voor de cruciale uitkomstmaten welke verschillen zij klinisch (patiënt) relevant vonden.
Methode literatuursamenvatting
Een uitgebreide beschrijving van de strategie voor zoeken en selecteren van literatuur is te vinden onder Zoekverantwoording. Indien mogelijk werden de gegevens uit verschillende studies gepoold in een random-effects-model. Review Manager 5.4 werd gebruikt voor de statistische analyses. De beoordeling van de wetenschappelijke bewijskracht wordt hieronder toegelicht.
Beoordelen van de kracht van het wetenschappelijke bewijs
De kracht van het wetenschappelijke bewijs werd bepaald volgens de GRADE-methode. GRADE staat voor ‘Grading Recommendations Assessment, Development and Evaluation’ (zie https://www.gradeworkinggroup.org/). De basisprincipes van de GRADE-methodiek zijn: het benoemen en prioriteren van de klinisch (patiënt) relevante uitkomstmaten, een systematische review per uitkomstmaat, en een beoordeling van de bewijskracht per uitkomstmaat op basis van de acht GRADE-domeinen (domeinen voor downgraden: risk of bias, inconsistentie, indirectheid, imprecisie, en publicatiebias; domeinen voor upgraden: dosis-effect relatie, groot effect, en residuele plausibele confounding).
GRADE onderscheidt vier gradaties voor de kwaliteit van het wetenschappelijk bewijs: hoog, redelijk, laag en zeer laag. Deze gradaties verwijzen naar de mate van zekerheid die er bestaat over de literatuurconclusie, in het bijzonder de mate van zekerheid dat de literatuurconclusie de aanbeveling adequaat ondersteunt (Schünemann, 2013; Hultcrantz, 2017).
|
GRADE |
Definitie |
|
Hoog |
|
|
Redelijk |
|
|
Laag |
|
|
Zeer laag |
|
Bij het beoordelen (graderen) van de kracht van het wetenschappelijk bewijs in richtlijnen volgens de GRADE-methodiek spelen grenzen voor klinische besluitvorming een belangrijke rol (Hultcrantz, 2017). Dit zijn de grenzen die bij overschrijding aanleiding zouden geven tot een aanpassing van de aanbeveling. Om de grenzen voor klinische besluitvorming te bepalen moeten alle relevante uitkomstmaten en overwegingen worden meegewogen. De grenzen voor klinische besluitvorming zijn daarmee niet één op één vergelijkbaar met het minimaal klinisch relevant verschil (Minimal Clinically Important Difference, MCID). Met name in situaties waarin een interventie geen belangrijke nadelen heeft en de kosten relatief laag zijn, kan de grens voor klinische besluitvorming met betrekking tot de effectiviteit van de interventie bij een lagere waarde (dichter bij het nul effect) liggen dan de MCID (Hultcrantz, 2017).
Overwegingen (van bewijs naar aanbeveling)
Om te komen tot een aanbeveling zijn naast (de kwaliteit van) het wetenschappelijke bewijs ook andere aspecten belangrijk en worden meegewogen, zoals aanvullende argumenten uit bijvoorbeeld de biomechanica of fysiologie, waarden en voorkeuren van patiënten, kosten (middelenbeslag), duurzaamheid, aanvaardbaarheid, haalbaarheid en implementatie. Deze aspecten zijn systematisch vermeld en beoordeeld (gewogen) onder het kopje Overwegingen en kunnen (mede) gebaseerd zijn op expert opinion. Hierbij is gebruik gemaakt van een gestructureerd format gebaseerd op het evidence-to-decision framework van de internationale GRADE Working Group (Alonso-Coello, 2016A; Alonso-Coello 2016B). Dit evidence-to-decision framework is een integraal onderdeel van de GRADE-methodiek.
Formuleren van aanbevelingen
De aanbevelingen geven antwoord op de uitgangsvraag en zijn gebaseerd op het beschikbare wetenschappelijke bewijs en de belangrijkste overwegingen, en een weging van de gunstige en ongunstige effecten van de relevante interventies. De kracht van het wetenschappelijk bewijs en het gewicht dat door de werkgroep wordt toegekend aan de overwegingen, bepalen samen de sterkte van de aanbeveling. Conform de GRADE-methodiek sluit een lage bewijskracht van conclusies in de systematische literatuuranalyse een sterke aanbeveling niet a priori uit, en zijn bij een hoge bewijskracht ook zwakke aanbevelingen mogelijk (Agoritsas, 2017; Neumann, 2016). De sterkte van de aanbeveling wordt altijd bepaald door weging van alle relevante argumenten tezamen. De werkgroep heeft bij elke aanbeveling opgenomen hoe zij tot de richting en sterkte van de aanbeveling is gekomen.
In de GRADE-methodiek wordt onderscheid gemaakt tussen sterke en zwakke (of conditionele) aanbevelingen. De sterkte van een aanbeveling verwijst naar de mate van zekerheid dat de voordelen van de interventie opwegen tegen de nadelen (of vice versa), gezien over het hele spectrum van patiënten waarvoor de aanbeveling is bedoeld. De sterkte van een aanbeveling heeft duidelijke implicaties voor patiënten, behandelaars en beleidsmakers (zie onderstaande tabel). Een aanbeveling is geen dictaat, zelfs een sterke aanbeveling gebaseerd op bewijs van hoge kwaliteit (GRADE gradering HOOG) zal niet altijd van toepassing zijn, onder alle mogelijke omstandigheden en voor elke individuele patiënt.
|
Implicaties van sterke en zwakke aanbevelingen voor verschillende richtlijngebruikers |
||
|
|
Sterke aanbeveling |
Zwakke (conditionele) aanbeveling |
|
Voor patiënten |
De meeste patiënten zouden de aanbevolen interventie of aanpak kiezen en slechts een klein aantal niet. |
Een aanzienlijk deel van de patiënten zouden de aanbevolen interventie of aanpak kiezen, maar veel patiënten ook niet. |
|
Voor behandelaars |
De meeste patiënten zouden de aanbevolen interventie of aanpak moeten ontvangen. |
Er zijn meerdere geschikte interventies of aanpakken. De patiënt moet worden ondersteund bij de keuze voor de interventie of aanpak die het beste aansluit bij zijn of haar waarden en voorkeuren. |
|
Voor beleidsmakers |
De aanbevolen interventie of aanpak kan worden gezien als standaardbeleid. |
Beleidsbepaling vereist uitvoerige discussie met betrokkenheid van veel stakeholders. Er is een grotere kans op lokale beleidsverschillen. |
Commentaar- en autorisatiefase
De conceptrichtlijnmodule werd aan de betrokken (wetenschappelijke) verenigingen en (patiënt) organisaties voorgelegd ter commentaar. De commentaren werden verzameld en besproken met de werkgroep. Naar aanleiding van de commentaren werd de conceptrichtlijnmodule aangepast en definitief vastgesteld door de werkgroep. De definitieve richtlijnmodule werd aan de deelnemende (wetenschappelijke) verenigingen en (patiënt) organisaties voorgelegd voor autorisatie en door hen geautoriseerd dan wel geaccordeerd.
Literatuur
Agoritsas T, Merglen A, Heen AF, Kristiansen A, Neumann I, Brito JP, Brignardello-Petersen R, Alexander PE, Rind DM, Vandvik PO, Guyatt GH. UpToDate adherence to GRADE criteria for strong recommendations: an analytical survey. BMJ Open. 2017 Nov 16;7(11):e018593. doi: 10.1136/bmjopen-2017-018593. PubMed PMID: 29150475; PubMed Central PMCID: PMC5701989.
Alonso-Coello P, Schünemann HJ, Moberg J, Brignardello-Petersen R, Akl EA, Davoli M, Treweek S, Mustafa RA, Rada G, Rosenbaum S, Morelli A, Guyatt GH, Oxman AD; GRADE Working Group. GRADE Evidence to Decision (EtD) frameworks: a systematic and transparent approach to making well informed healthcare choices. 1: Introduction. BMJ. 2016 Jun 28;353:i2016. doi: 10.1136/bmj.i2016. PubMed PMID: 27353417. A
Alonso-Coello P, Oxman AD, Moberg J, Brignardello-Petersen R, Akl EA, Davoli M, Treweek S, Mustafa RA, Vandvik PO, Meerpohl J, Guyatt GH, Schünemann HJ; GRADE Working Group. GRADE Evidence to Decision (EtD) frameworks: a systematic and transparent approach to making well informed healthcare choices. 2: Clinical practice guidelines. BMJ. 2016 Jun 30;353:i2089. doi: 10.1136/bmj.i2089. PubMed PMID: 27365494. B
Brouwers MC, Kho ME, Browman GP, Burgers JS, Cluzeau F, Feder G, Fervers B, Graham ID, Grimshaw J, Hanna SE, Littlejohns P, Makarski J, Zitzelsberger L; AGREE Next Steps Consortium. AGREE II: advancing guideline development, reporting and evaluation in health care. CMAJ. 2010 Dec 14;182(18):E839-42. doi: 10.1503/cmaj.090449. Epub 2010 Jul 5. Review. PubMed PMID: 20603348; PubMed Central PMCID: PMC3001530.
Hultcrantz M, Rind D, Akl EA, Treweek S, Mustafa RA, Iorio A, Alper BS, Meerpohl JJ, Murad MH, Ansari MT, Katikireddi SV, Östlund P, Tranæus S, Christensen R, Gartlehner G, Brozek J, Izcovich A, Schünemann H, Guyatt G. The GRADE Working Group clarifies the construct of certainty of evidence. J Clin Epidemiol. 2017 Jul;87:4-13. doi: 10.1016/j.jclinepi.2017.05.006. Epub 2017 May 18. PubMed PMID: 28529184; PubMed Central PMCID: PMC6542664.
Medisch Specialistische Richtlijnen 3.0 (2023). Adviescommissie Richtlijnen van de Raad Kwaliteit. Available from: https://richtlijnendatabase.nl/uploaded/docs/FMS_MedSpecRicht_2023__v04.pdf?u=1bULOR
Neumann I, Santesso N, Akl EA, Rind DM, Vandvik PO, Alonso-Coello P, Agoritsas T, Mustafa RA, Alexander PE, Schünemann H, Guyatt GH. A guide for health professionals to interpret and use recommendations in guidelines developed with the GRADE approach. J Clin Epidemiol. 2016 Apr;72:45-55. doi: 10.1016/j.jclinepi.2015.11.017. Epub 2016 Jan 6. PMID: 26772609.
Schünemann H, Brożek J, Guyatt G, et al. GRADE handbook for grading quality of evidence and strength of recommendations. Updated October 2013. The GRADE Working Group, 2013. Available from: https://gdt.gradepro.org/app/handbook/handbook.html.
Zoekverantwoording
Search question 1
|
Richtlijn Intravasculaire katheters |
|
|
Uitgangsvraag: Welke infectiepreventiemaatregelen dienen genomen te worden bij het afdekken en fixeren van een perifeer veneuze katheter, een centraal veneuze katheter en een perifeer arteriële katheter? Zoekvraag: What is the effect of chlorhexidine gluconate-impregnated dressings on catheter-related infectious complications compared with dressings not impregnated with chlorhexidine-gluconate in patients requiring a peripheral venous catheter, central venous catheter or arterial catheter? |
|
|
Database(s): Embase.com, Ovid/Medline |
Datum: 22 mei 2024 |
|
Periode: vanaf 2000 |
Talen: geen restrictie |
|
Literatuurspecialist: Alies Oost |
Rayyan review: https://rayyan.ai/reviews/995577 |
|
BMI-zoekblokken: voor verschillende opdrachten wordt (deels) gebruik gemaakt van de zoekblokken van BMI-Online https://blocks.bmi-online.nl/ Deduplication: voor het ontdubbelen is gebruik gemaakt van http://dedupendnote.nl:9777/ |
|
|
Toelichting: Voor deze vraag is gezocht op de elementen:
|
|
Zoekopbrengst
|
|
EMBASE |
OVID/MEDLINE |
Ontdubbeld |
|
SR |
167 |
98 |
172 |
|
RCT |
382 |
237 |
426 |
|
Observationele studies |
333 |
153 |
344 |
|
Totaal |
882 |
488 |
942* |
*in Rayyan
Zoekstrategie
Embase.com
|
No. |
Query |
Results |
|
No. |
Query |
Results |
|
#1 |
'central venous catheter'/exp OR 'central venous catheterization'/exp OR 'peripherally inserted central venous catheter'/exp OR 'peripheral venous catheter'/exp OR 'artery catheter'/exp OR 'artery catheterization'/exp OR ((peripheral* NEAR/4 (catheter* OR cannula* OR line* OR access* OR infusion OR iv OR ivs)):ti,ab,kw) OR pivc:ti,ab,kw OR pivcs:ti,ab,kw OR 'venous access':ti,ab,kw OR ((('central intravenous' OR 'central vein*' OR 'central venous' OR 'central intravascular') NEAR/3 (catheter* OR device* OR line* OR infusion OR iv OR ivs OR access OR cannula*)):ti,ab,kw) OR hickman*:ti,ab,kw OR broviac:ti,ab,kw OR picc*:ti,ab,kw OR 'pic line*':ti,ab,kw OR 'central line*':ti,ab,kw OR 'central catheter*':ti,ab,kw OR cvc:ti,ab,kw OR cvcs:ti,ab,kw OR cvad*:ti,ab,kw OR 'swan ganz':ti,ab,kw OR (((arterial OR artery) NEAR/3 (catheter* OR cannula* OR line* OR access)):ti,ab,kw) |
138258 |
|
#2 |
'chlorhexidine gluconate'/exp OR 'chlorhexidine'/exp OR chlorhexidin*:ti,ab,kw OR clorhexidin*:ti,ab,kw OR chlorohexidin*:ti,ab,kw OR chg:ti,ab,kw OR hibiclens:ti,ab,kw OR peridex:ti,ab,kw OR periogard:ti,ab,kw OR nolvasan:ti,ab,kw OR novalsan:ti,ab,kw OR tubulicid:ti,ab,kw OR 'central line dressing'/exp OR 'antimicrobial dressing'/exp OR 'occlusive dressing'/exp OR 'hydrocolloid dressing'/exp OR (((antimicrobial OR 'anti microbial' OR disinfect* OR 'dis infect*' OR antibacterial OR 'anti bacterial' OR antibiotic OR antiinfective* OR 'anti infective*' OR antiseptic OR iodine OR povidone OR inadine OR iodium OR jodium OR medadine OR hydrocolloid* OR polyurethane* OR silver OR occlusive) NEAR/3 (dressing* OR gauze* OR patch* OR sponge*)):ti,ab,kw) OR (((allevyn OR mepilex OR melgisorb OR exufiber OR aquacel) NEAR/2 ag):ti,ab,kw) OR dressing*:ti OR ((gauze NEAR/3 tape):ti,ab,kw) OR (('gauze dressing'/exp OR 'transparent dressing'/exp OR 'dressing'/exp OR 'sponge'/exp OR 'patch'/exp) AND ('sulfadiazine silver'/exp OR 'sulfathiazole silver'/exp OR 'silver'/exp OR 'iodine'/exp OR 'povidone iodine'/exp OR 'polyurethan'/exp OR 'topical antiinfective agent'/exp)) |
51518 |
|
#3 |
#1 AND #2 NOT ('conference abstract'/it OR 'editorial'/it OR 'letter'/it OR 'note'/it) NOT (('animal'/exp OR 'animal experiment'/exp OR 'animal model'/exp OR 'nonhuman'/exp) NOT 'human'/exp) AND [2000-2024]/py |
1303 |
|
#4 |
'meta analysis'/exp OR 'meta analysis (topic)'/exp OR metaanaly*:ti,ab OR 'meta analy*':ti,ab OR metanaly*:ti,ab OR 'systematic review'/de OR 'cochrane database of systematic reviews'/jt OR prisma:ti,ab OR prospero:ti,ab OR (((systemati* OR scoping OR umbrella OR 'structured literature') NEAR/3 (review* OR overview*)):ti,ab) OR ((systemic* NEAR/1 review*):ti,ab) OR (((systemati* OR literature OR database* OR 'data base*') NEAR/10 search*):ti,ab) OR (((structured OR comprehensive* OR systemic*) NEAR/3 search*):ti,ab) OR (((literature NEAR/3 review*):ti,ab) AND (search*:ti,ab OR database*:ti,ab OR 'data base*':ti,ab)) OR (('data extraction':ti,ab OR 'data source*':ti,ab) AND 'study selection':ti,ab) OR ('search strategy':ti,ab AND 'selection criteria':ti,ab) OR ('data source*':ti,ab AND 'data synthesis':ti,ab) OR medline:ab OR pubmed:ab OR embase:ab OR cochrane:ab OR (((critical OR rapid) NEAR/2 (review* OR overview* OR synthes*)):ti) OR ((((critical* OR rapid*) NEAR/3 (review* OR overview* OR synthes*)):ab) AND (search*:ab OR database*:ab OR 'data base*':ab)) OR metasynthes*:ti,ab OR 'meta synthes*':ti,ab |
1030581 |
|
#5 |
'clinical trial'/exp OR 'randomization'/exp OR 'single blind procedure'/exp OR 'double blind procedure'/exp OR 'crossover procedure'/exp OR 'placebo'/exp OR 'prospective study'/exp OR rct:ab,ti OR random*:ab,ti OR 'single blind':ab,ti OR 'randomised controlled trial':ab,ti OR 'randomized controlled trial'/exp OR placebo*:ab,ti |
4037216 |
|
#6 |
'major clinical study'/de OR 'clinical study'/de OR 'case control study'/de OR 'family study'/de OR 'longitudinal study'/de OR 'retrospective study'/de OR 'prospective study'/de OR 'comparative study'/de OR 'cohort analysis'/de OR ((cohort NEAR/1 (study OR studies)):ab,ti) OR (('case control' NEAR/1 (study OR studies)):ab,ti) OR (('follow up' NEAR/1 (study OR studies)):ab,ti) OR (observational NEAR/1 (study OR studies)) OR ((epidemiologic NEAR/1 (study OR studies)):ab,ti) OR (('cross sectional' NEAR/1 (study OR studies)):ab,ti) |
8239542 |
|
#7 |
'case control study'/de OR 'comparative study'/exp OR 'control group'/de OR 'controlled study'/de OR 'controlled clinical trial'/de OR 'crossover procedure'/de OR 'double blind procedure'/de OR 'phase 2 clinical trial'/de OR 'phase 3 clinical trial'/de OR 'phase 4 clinical trial'/de OR 'pretest posttest design'/de OR 'pretest posttest control group design'/de OR 'quasi experimental study'/de OR 'single blind procedure'/de OR 'triple blind procedure'/de OR (((control OR controlled) NEAR/6 trial):ti,ab,kw) OR (((control OR controlled) NEAR/6 (study OR studies)):ti,ab,kw) OR (((control OR controlled) NEAR/1 active):ti,ab,kw) OR 'open label*':ti,ab,kw OR (((double OR two OR three OR multi OR trial) NEAR/1 (arm OR arms)):ti,ab,kw) OR ((allocat* NEAR/10 (arm OR arms)):ti,ab,kw) OR placebo*:ti,ab,kw OR 'sham-control*':ti,ab,kw OR (((single OR double OR triple OR assessor) NEAR/1 (blind* OR masked)):ti,ab,kw) OR nonrandom*:ti,ab,kw OR 'non-random*':ti,ab,kw OR 'quasi-experiment*':ti,ab,kw OR crossover:ti,ab,kw OR 'cross over':ti,ab,kw OR 'parallel group*':ti,ab,kw OR 'factorial trial':ti,ab,kw OR ((phase NEAR/5 (study OR trial)):ti,ab,kw) OR ((case* NEAR/6 (matched OR control*)):ti,ab,kw) OR ((match* NEAR/6 (pair OR pairs OR cohort* OR control* OR group* OR healthy OR age OR sex OR gender OR patient* OR subject* OR participant*)):ti,ab,kw) OR ((propensity NEAR/6 (scor* OR match*)):ti,ab,kw) OR versus:ti OR vs:ti OR compar*:ti OR ((compar* NEAR/1 study):ti,ab,kw) OR (('major clinical study'/de OR 'clinical study'/de OR 'cohort analysis'/de OR 'observational study'/de OR 'cross-sectional study'/de OR 'multicenter study'/de OR 'correlational study'/de OR 'follow up'/de OR cohort*:ti,ab,kw OR 'follow up':ti,ab,kw OR followup:ti,ab,kw OR longitudinal*:ti,ab,kw OR prospective*:ti,ab,kw OR retrospective*:ti,ab,kw OR observational*:ti,ab,kw OR 'cross sectional*':ti,ab,kw OR cross?ectional*:ti,ab,kw OR multicent*:ti,ab,kw OR 'multi-cent*':ti,ab,kw OR consecutive*:ti,ab,kw) AND (group:ti,ab,kw OR groups:ti,ab,kw OR subgroup*:ti,ab,kw OR versus:ti,ab,kw OR vs:ti,ab,kw OR compar*:ti,ab,kw OR 'odds ratio*':ab OR 'relative odds':ab OR 'risk ratio*':ab OR 'relative risk*':ab OR 'rate ratio':ab OR aor:ab OR arr:ab OR rrr:ab OR ((('or' OR 'rr') NEAR/6 ci):ab))) |
15109487 |
|
#8 |
#3 AND #4 - SR |
167 |
|
#9 |
#3 AND #5 NOT #8 - RCT |
382 |
|
#10 |
#3 AND (#6 OR #7) NOT (#8 OR #9) - observationeel |
333 |
|
#11 |
#8 OR #9 OR #10 |
882 |
Ovid/Medline
|
# |
Searches |
Results |
|
1 |
exp Central Venous Catheters/ or exp Catheterization, Central Venous/ or exp Catheterization, Peripheral/ or Vascular Access Devices/ or (peripheral* adj4 (catheter* or cannula* or line* or access* or infusion or iv or ivs)).ti,ab,kf. or pivc.ti,ab,kf. or pivcs.ti,ab,kf. or 'venous access'.ti,ab,kf. or (('central intravenous' or 'central vein*' or 'central venous' or 'central intravascular') adj3 (catheter* or device* or line* or infusion or iv or ivs or access or cannula*)).ti,ab,kf. or hickman*.ti,ab,kf. or broviac.ti,ab,kf. or picc*.ti,ab,kf. or 'pic line*'.ti,ab,kf. or 'central line*'.ti,ab,kf. or 'central catheter*'.ti,ab,kf. or cvc.ti,ab,kf. or cvcs.ti,ab,kf. or cvad*.ti,ab,kf. or 'swan ganz'.ti,ab,kf. or ((arterial or artery) adj3 (catheter* or cannula* or line* or access)).ti,ab,kf. |
78338 |
|
2 |
exp Chlorhexidine/ or chlorhexidin*.ti,ab,kf. or clorhexidin*.ti,ab,kf. or chlorohexidin*.ti,ab,kf. or chg.ti,ab,kf. or Hibiclens.ti,ab,kf. or Peridex.ti,ab,kf. or Periogard.ti,ab,kf. or nolvasan.ti,ab,kf. or novalsan.ti,ab,kf. or tubulicid.ti,ab,kf. or exp Occlusive Dressings/ or exp Bandages, Hydrocolloid/ or ((antimicrobial or 'anti microbial' or disinfect* or 'dis infect*' or antibacterial or 'anti bacterial' or antibiotic or antiinfective* or 'anti infective*' or antiseptic or iodine or povidone or inadine or iodium or jodium or medadine or hydrocolloid* or polyurethane* or silver or occlusive) adj3 (dressing* or gauze* or patch* or sponge*)).ti,ab,kf. or ((Allevyn or mepilex or melgisorb or exufiber or aquacel) adj2 Ag).ti,ab,kf. or dressing*.ti. or (gauze adj3 tape).ti,ab,kf. or ((exp Transdermal Patch/ or exp Tissue Adhesives/ or Bandages/) and (exp Silver Sulfadiazine/ or exp Silver/ or exp Iodine/ or exp Povidone-Iodine/ or exp Polyurethanes/ or exp Anti-Infective Agents, Local/)) |
34007 |
|
3 |
(1 and 2) not (comment/ or editorial/ or letter/) not ((exp animals/ or exp models, animal/) not humans/) |
862 |
|
4 |
limit 3 to yr="2000 -Current" |
727 |
|
5 |
meta-analysis/ or meta-analysis as topic/ or (metaanaly* or meta-analy* or metanaly*).ti,ab,kf. or systematic review/ or cochrane.jw. or (prisma or prospero).ti,ab,kf. or ((systemati* or scoping or umbrella or "structured literature") adj3 (review* or overview*)).ti,ab,kf. or (systemic* adj1 review*).ti,ab,kf. or ((systemati* or literature or database* or data-base*) adj10 search*).ti,ab,kf. or ((structured or comprehensive* or systemic*) adj3 search*).ti,ab,kf. or ((literature adj3 review*) and (search* or database* or data-base*)).ti,ab,kf. or (("data extraction" or "data source*") and "study selection").ti,ab,kf. or ("search strategy" and "selection criteria").ti,ab,kf. or ("data source*" and "data synthesis").ti,ab,kf. or (medline or pubmed or embase or cochrane).ab. or ((critical or rapid) adj2 (review* or overview* or synthes*)).ti. or (((critical* or rapid*) adj3 (review* or overview* or synthes*)) and (search* or database* or data-base*)).ab. or (metasynthes* or meta-synthes*).ti,ab,kf. |
747918 |
|
6 |
exp clinical trial/ or randomized controlled trial/ or exp clinical trials as topic/ or randomized controlled trials as topic/ or Random Allocation/ or Double-Blind Method/ or Single-Blind Method/ or (clinical trial, phase i or clinical trial, phase ii or clinical trial, phase iii or clinical trial, phase iv or controlled clinical trial or randomized controlled trial or multicenter study or clinical trial).pt. or random*.ti,ab. or (clinic* adj trial*).tw. or ((singl* or doubl* or treb* or tripl*) adj (blind$3 or mask$3)).tw. or Placebos/ or placebo*.tw. |
2729122 |
|
7 |
Epidemiologic studies/ or case control studies/ or exp cohort studies/ or Controlled Before-After Studies/ or Case control.tw. or cohort.tw. or Cohort analy$.tw. or (Follow up adj (study or studies)).tw. or (observational adj (study or studies)).tw. or Longitudinal.tw. or Retrospective*.tw. or prospective*.tw. or consecutive*.tw. or Cross sectional.tw. or Cross-sectional studies/ or historically controlled study/ or interrupted time series analysis/ [Onder exp cohort studies vallen ook longitudinale, prospectieve en retrospectieve studies] |
4731772 |
|
8 |
Case-control Studies/ or clinical trial, phase ii/ or clinical trial, phase iii/ or clinical trial, phase iv/ or comparative study/ or control groups/ or controlled before-after studies/ or controlled clinical trial/ or double-blind method/ or historically controlled study/ or matched-pair analysis/ or single-blind method/ or (((control or controlled) adj6 (study or studies or trial)) or (compar* adj (study or studies)) or ((control or controlled) adj1 active) or "open label*" or ((double or two or three or multi or trial) adj (arm or arms)) or (allocat* adj10 (arm or arms)) or placebo* or "sham-control*" or ((single or double or triple or assessor) adj1 (blind* or masked)) or nonrandom* or "non-random*" or "quasi-experiment*" or "parallel group*" or "factorial trial" or "pretest posttest" or (phase adj5 (study or trial)) or (case* adj6 (matched or control*)) or (match* adj6 (pair or pairs or cohort* or control* or group* or healthy or age or sex or gender or patient* or subject* or participant*)) or (propensity adj6 (scor* or match*))).ti,ab,kf. or (confounding adj6 adjust*).ti,ab. or (versus or vs or compar*).ti. or ((exp cohort studies/ or epidemiologic studies/ or multicenter study/ or observational study/ or seroepidemiologic studies/ or (cohort* or 'follow up' or followup or longitudinal* or prospective* or retrospective* or observational* or multicent* or 'multi-cent*' or consecutive*).ti,ab,kf.) and ((group or groups or subgroup* or versus or vs or compar*).ti,ab,kf. or ('odds ratio*' or 'relative odds' or 'risk ratio*' or 'relative risk*' or aor or arr or rrr).ab. or (("OR" or "RR") adj6 CI).ab.)) |
5695688 |
|
9 |
4 and 5 - SR |
98 |
|
10 |
(4 and 6) not 9 - RCT |
237 |
|
11 |
(4 and (7 or 8)) not (9 or 10) - observationeel |
153 |
|
12 |
9 or 10 or 11 |
488 |
Search question 2
|
Richtlijn Intravasculaire katheters |
|
|
Uitgangsvraag: Welke infectiepreventiemaatregelen dienen genomen te worden bij het afdekken en fixeren van een perifeer veneuze katheter, een centraal veneuze katheter en een perifeer arteriële katheter? Zoekvraag: What is the effect of different catheter fixation methods on catheter-related infectious complications in patients requiring a peripheral venous catheter, central venous catheter or an arterial catheter? |
|
|
Database(s): Embase.com, Ovid/Medline |
Datum: 24 april 2024 |
|
Periode: vanaf 2000 |
Talen: geen restrictie |
|
Literatuurspecialist: Alies Oost |
Rayyan review: https://rayyan.ai/reviews/995596 |
|
BMI-zoekblokken: voor verschillende opdrachten wordt (deels) gebruik gemaakt van de zoekblokken van BMI-Online https://blocks.bmi-online.nl/ Deduplication: voor het ontdubbelen is gebruik gemaakt van http://dedupendnote.nl:9777/ |
|
|
Toelichting: Voor deze vraag is gezocht op de elementen:
De sleutelartikelen worden gevonden met deze search, m.u.v. het artikel van Javeri et al. (Indian Journal of Critical Care Medicine, 2020), deze bevat in titel/ abstract/ trefwoord geen term voor catheter fixation. |
|
Zoekopbrengst
|
|
EMBASE |
OVID/MEDLINE |
Ontdubbeld |
|
SR |
49 |
28 |
45 |
|
RCT |
164 |
105 |
195 |
|
Observationele studies |
224 |
134 |
255 |
|
Totaal |
437 |
267 |
495* |
*in Rayyan
Zoekstrategie
Embase.com
|
No. |
Query |
Results |
|
#1 |
'central venous catheter'/exp OR 'central venous catheterization'/exp OR 'peripherally inserted central venous catheter'/exp OR 'peripheral venous catheter'/exp OR 'artery catheter'/exp OR 'artery catheterization'/exp OR ((peripheral* NEAR/4 (catheter* OR cannula* OR line* OR access* OR infusion OR iv OR ivs)):ti,ab,kw) OR pivc:ti,ab,kw OR pivcs:ti,ab,kw OR 'venous access':ti,ab,kw OR 'vascular access':ti,ab,kw OR ((('central intravenous' OR 'central vein*' OR 'central venous') NEAR/3 (catheter* OR device* OR line* OR infusion OR iv OR ivs OR access OR cannula*)):ti,ab,kw) OR hickman*:ti,ab,kw OR broviac:ti,ab,kw OR picc*:ti,ab,kw OR 'pic line*':ti,ab,kw OR 'central line*':ti,ab,kw OR 'central catheter*':ti,ab,kw OR cvc:ti,ab,kw OR cvcs:ti,ab,kw OR cvad*:ti,ab,kw OR 'swan ganz':ti,ab,kw OR (((arterial OR artery) NEAR/3 (catheter* OR cannula* OR line* OR access)):ti,ab,kw) |
152358 |
|
#2 |
'adhesive agent'/exp OR 'cyanoacrylate'/exp OR 'sutureless technique'/exp OR 'catheter holder'/de OR (((secur* OR fixat*) NEAR/3 (catheter* OR 'central catheter*' OR 'central line*' OR 'central intravenous' OR 'central vein*' OR 'central venous' OR cvc OR cvcs OR picc OR 'arterial line*' OR 'arterial catheter*' OR device* OR product* OR system* OR method* OR glue OR adhesive*)):ti,ab,kw) OR ((catheter* NEAR/3 (stabilization OR stabilisation)):ti,ab,kw) OR sutureless:ti,ab,kw OR 'suture less':ti,ab,kw OR suturless:ti,ab,kw OR 'suture free':ti,ab,kw OR sasd:ti,ab,kw OR sass:ti,ab,kw OR ((gauze NEAR/3 tape):ti,ab,kw) OR 'catheter holder*':ti,ab,kw OR securacath:ti,ab,kw OR statlock:ti,ab,kw OR 'grip lok':ti,ab,kw OR griplok:ti,ab,kw OR ((cyanoacrylate NEAR/3 (glue OR adhesive OR secur* OR fixat*)):ti,ab,kw) |
150790 |
|
#3 |
#1 AND #2 NOT ('conference abstract'/it OR 'editorial'/it OR 'letter'/it OR 'note'/it) NOT (('animal'/exp OR 'animal experiment'/exp OR 'animal model'/exp OR 'nonhuman'/exp) NOT 'human'/exp) |
943 |
|
#4 |
#3 AND [2000-2024]/py |
845 |
|
#5 |
'meta analysis'/exp OR 'meta analysis (topic)'/exp OR metaanaly*:ti,ab OR 'meta analy*':ti,ab OR metanaly*:ti,ab OR 'systematic review'/de OR 'cochrane database of systematic reviews'/jt OR prisma:ti,ab OR prospero:ti,ab OR (((systemati* OR scoping OR umbrella OR 'structured literature') NEAR/3 (review* OR overview*)):ti,ab) OR ((systemic* NEAR/1 review*):ti,ab) OR (((systemati* OR literature OR database* OR 'data base*') NEAR/10 search*):ti,ab) OR (((structured OR comprehensive* OR systemic*) NEAR/3 search*):ti,ab) OR (((literature NEAR/3 review*):ti,ab) AND (search*:ti,ab OR database*:ti,ab OR 'data base*':ti,ab)) OR (('data extraction':ti,ab OR 'data source*':ti,ab) AND 'study selection':ti,ab) OR ('search strategy':ti,ab AND 'selection criteria':ti,ab) OR ('data source*':ti,ab AND 'data synthesis':ti,ab) OR medline:ab OR pubmed:ab OR embase:ab OR cochrane:ab OR (((critical OR rapid) NEAR/2 (review* OR overview* OR synthes*)):ti) OR ((((critical* OR rapid*) NEAR/3 (review* OR overview* OR synthes*)):ab) AND (search*:ab OR database*:ab OR 'data base*':ab)) OR metasynthes*:ti,ab OR 'meta synthes*':ti,ab |
1021882 |
|
#6 |
'clinical trial'/exp OR 'randomization'/exp OR 'single blind procedure'/exp OR 'double blind procedure'/exp OR 'crossover procedure'/exp OR 'placebo'/exp OR 'prospective study'/exp OR rct:ab,ti OR random*:ab,ti OR 'single blind':ab,ti OR 'randomised controlled trial':ab,ti OR 'randomized controlled trial'/exp OR placebo*:ab,ti |
4017003 |
|
#7 |
'major clinical study'/de OR 'clinical study'/de OR 'case control study'/de OR 'family study'/de OR 'longitudinal study'/de OR 'retrospective study'/de OR 'prospective study'/de OR 'comparative study'/de OR 'cohort analysis'/de OR ((cohort NEAR/1 (study OR studies)):ab,ti) OR (('case control' NEAR/1 (study OR studies)):ab,ti) OR (('follow up' NEAR/1 (study OR studies)):ab,ti) OR (observational NEAR/1 (study OR studies)) OR ((epidemiologic NEAR/1 (study OR studies)):ab,ti) OR (('cross sectional' NEAR/1 (study OR studies)):ab,ti) |
8186968 |
|
#8 |
'case control study'/de OR 'comparative study'/exp OR 'control group'/de OR 'controlled study'/de OR 'controlled clinical trial'/de OR 'crossover procedure'/de OR 'double blind procedure'/de OR 'phase 2 clinical trial'/de OR 'phase 3 clinical trial'/de OR 'phase 4 clinical trial'/de OR 'pretest posttest design'/de OR 'pretest posttest control group design'/de OR 'quasi experimental study'/de OR 'single blind procedure'/de OR 'triple blind procedure'/de OR (((control OR controlled) NEAR/6 trial):ti,ab,kw) OR (((control OR controlled) NEAR/6 (study OR studies)):ti,ab,kw) OR (((control OR controlled) NEAR/1 active):ti,ab,kw) OR 'open label*':ti,ab,kw OR (((double OR two OR three OR multi OR trial) NEAR/1 (arm OR arms)):ti,ab,kw) OR ((allocat* NEAR/10 (arm OR arms)):ti,ab,kw) OR placebo*:ti,ab,kw OR 'sham-control*':ti,ab,kw OR (((single OR double OR triple OR assessor) NEAR/1 (blind* OR masked)):ti,ab,kw) OR nonrandom*:ti,ab,kw OR 'non-random*':ti,ab,kw OR 'quasi-experiment*':ti,ab,kw OR crossover:ti,ab,kw OR 'cross over':ti,ab,kw OR 'parallel group*':ti,ab,kw OR 'factorial trial':ti,ab,kw OR ((phase NEAR/5 (study OR trial)):ti,ab,kw) OR ((case* NEAR/6 (matched OR control*)):ti,ab,kw) OR ((match* NEAR/6 (pair OR pairs OR cohort* OR control* OR group* OR healthy OR age OR sex OR gender OR patient* OR subject* OR participant*)):ti,ab,kw) OR ((propensity NEAR/6 (scor* OR match*)):ti,ab,kw) OR versus:ti OR vs:ti OR compar*:ti OR ((compar* NEAR/1 study):ti,ab,kw) OR (('major clinical study'/de OR 'clinical study'/de OR 'cohort analysis'/de OR 'observational study'/de OR 'cross-sectional study'/de OR 'multicenter study'/de OR 'correlational study'/de OR 'follow up'/de OR cohort*:ti,ab,kw OR 'follow up':ti,ab,kw OR followup:ti,ab,kw OR longitudinal*:ti,ab,kw OR prospective*:ti,ab,kw OR retrospective*:ti,ab,kw OR observational*:ti,ab,kw OR 'cross sectional*':ti,ab,kw OR cross?ectional*:ti,ab,kw OR multicent*:ti,ab,kw OR 'multi-cent*':ti,ab,kw OR consecutive*:ti,ab,kw) AND (group:ti,ab,kw OR groups:ti,ab,kw OR subgroup*:ti,ab,kw OR versus:ti,ab,kw OR vs:ti,ab,kw OR compar*:ti,ab,kw OR 'odds ratio*':ab OR 'relative odds':ab OR 'risk ratio*':ab OR 'relative risk*':ab OR 'rate ratio':ab OR aor:ab OR arr:ab OR rrr:ab OR ((('or' OR 'rr') NEAR/6 ci):ab))) |
15017556 |
|
#9 |
#4 AND #5 - SR |
49 |
|
#10 |
#4 AND #6 NOT #9 - RCT |
164 |
|
#11 |
#4 AND (#7 OR #8) NOT (#9 OR #10) - observationeel |
224 |
|
#12 |
#9 OR #10 OR #11 |
437 |
Ovid/Medline
|
# |
Searches |
Results |
|
1 |
exp Central Venous Catheters/ or exp Catheterization, Central Venous/ or exp Catheterization, Peripheral/ or Vascular Access Devices/ or (peripheral* adj4 (catheter* or cannula* or line* or access* or infusion or iv or ivs)).ti,ab,kf. or pivc.ti,ab,kf. or pivcs.ti,ab,kf. or 'venous access'.ti,ab,kf. or 'vascular access'.ti,ab,kf. or (('central intravenous' or 'central vein*' or 'central venous') adj3 (catheter* or device* or line* or infusion or iv or ivs or access or cannula*)).ti,ab,kf. or hickman*.ti,ab,kf. or broviac.ti,ab,kf. or picc*.ti,ab,kf. or 'pic line*'.ti,ab,kf. or 'central line*'.ti,ab,kf. or 'central catheter*'.ti,ab,kf. or cvc.ti,ab,kf. or cvcs.ti,ab,kf. or cvad*.ti,ab,kf. or 'swan ganz'.ti,ab,kf. or ((arterial or artery) adj3 (catheter* or cannula* or line* or access)).ti,ab,kf. |
86174 |
|
2 |
exp Adhesives/ or exp Tissue Adhesives/ or exp Cyanoacrylates/ or ((secur* or fixat*) adj3 (catheter* or 'central catheter*' or 'central line*' or 'central intravenous' or 'central vein*' or 'central venous' or cvc or cvcs or picc or 'arterial line*' or 'arterial catheter*' or device* or product* or system* or method* or glue or adhesive*)).ti,ab,kf. or (catheter* adj3 (stabilization or stabilisation)).ti,ab,kf. or sutureless.ti,ab,kf. or 'suture less'.ti,ab,kf. or suturless.ti,ab,kf. or 'suture free'.ti,ab,kf. or sasd.ti,ab,kf. or sass.ti,ab,kf. or (gauze adj3 tape).ti,ab,kf. or 'catheter holder*'.ti,ab,kf. or securacath.ti,ab,kf. or statlock.ti,ab,kf. or 'grip lok'.ti,ab,kf. or griplok.ti,ab,kf. or (cyanoacrylate adj3 (glue or adhesive or secur* or fixat*)).ti,ab,kf. |
53703 |
|
3 |
(1 and 2) not (comment/ or editorial/ or letter/) not ((exp animals/ or exp models, animal/) not humans/) |
528 |
|
4 |
limit 3 to yr="2000 -Current" |
435 |
|
5 |
meta-analysis/ or meta-analysis as topic/ or (metaanaly* or meta-analy* or metanaly*).ti,ab,kf. or systematic review/ or cochrane.jw. or (prisma or prospero).ti,ab,kf. or ((systemati* or scoping or umbrella or "structured literature") adj3 (review* or overview*)).ti,ab,kf. or (systemic* adj1 review*).ti,ab,kf. or ((systemati* or literature or database* or data-base*) adj10 search*).ti,ab,kf. or ((structured or comprehensive* or systemic*) adj3 search*).ti,ab,kf. or ((literature adj3 review*) and (search* or database* or data-base*)).ti,ab,kf. or (("data extraction" or "data source*") and "study selection").ti,ab,kf. or ("search strategy" and "selection criteria").ti,ab,kf. or ("data source*" and "data synthesis").ti,ab,kf. or (medline or pubmed or embase or cochrane).ab. or ((critical or rapid) adj2 (review* or overview* or synthes*)).ti. or (((critical* or rapid*) adj3 (review* or overview* or synthes*)) and (search* or database* or data-base*)).ab. or (metasynthes* or meta-synthes*).ti,ab,kf. |
741328 |
|
6 |
exp clinical trial/ or randomized controlled trial/ or exp clinical trials as topic/ or randomized controlled trials as topic/ or Random Allocation/ or Double-Blind Method/ or Single-Blind Method/ or (clinical trial, phase i or clinical trial, phase ii or clinical trial, phase iii or clinical trial, phase iv or controlled clinical trial or randomized controlled trial or multicenter study or clinical trial).pt. or random*.ti,ab. or (clinic* adj trial*).tw. or ((singl* or doubl* or treb* or tripl*) adj (blind$3 or mask$3)).tw. or Placebos/ or placebo*.tw. |
2716593 |
|
7 |
Epidemiologic studies/ or case control studies/ or exp cohort studies/ or Controlled Before-After Studies/ or Case control.tw. or cohort.tw. or Cohort analy$.tw. or (Follow up adj (study or studies)).tw. or (observational adj (study or studies)).tw. or Longitudinal.tw. or Retrospective*.tw. or prospective*.tw. or consecutive*.tw. or Cross sectional.tw. or Cross-sectional studies/ or historically controlled study/ or interrupted time series analysis/ [Onder exp cohort studies vallen ook longitudinale, prospectieve en retrospectieve studies] |
4707287 |
|
8 |
Case-control Studies/ or clinical trial, phase ii/ or clinical trial, phase iii/ or clinical trial, phase iv/ or comparative study/ or control groups/ or controlled before-after studies/ or controlled clinical trial/ or double-blind method/ or historically controlled study/ or matched-pair analysis/ or single-blind method/ or (((control or controlled) adj6 (study or studies or trial)) or (compar* adj (study or studies)) or ((control or controlled) adj1 active) or "open label*" or ((double or two or three or multi or trial) adj (arm or arms)) or (allocat* adj10 (arm or arms)) or placebo* or "sham-control*" or ((single or double or triple or assessor) adj1 (blind* or masked)) or nonrandom* or "non-random*" or "quasi-experiment*" or "parallel group*" or "factorial trial" or "pretest posttest" or (phase adj5 (study or trial)) or (case* adj6 (matched or control*)) or (match* adj6 (pair or pairs or cohort* or control* or group* or healthy or age or sex or gender or patient* or subject* or participant*)) or (propensity adj6 (scor* or match*))).ti,ab,kf. or (confounding adj6 adjust*).ti,ab. or (versus or vs or compar*).ti. or ((exp cohort studies/ or epidemiologic studies/ or multicenter study/ or observational study/ or seroepidemiologic studies/ or (cohort* or 'follow up' or followup or longitudinal* or prospective* or retrospective* or observational* or multicent* or 'multi-cent*' or consecutive*).ti,ab,kf.) and ((group or groups or subgroup* or versus or vs or compar*).ti,ab,kf. or ('odds ratio*' or 'relative odds' or 'risk ratio*' or 'relative risk*' or aor or arr or rrr).ab. or (("OR" or "RR") adj6 CI).ab.)) |
5672837 |
|
9 |
4 and 5 - SR |
28 |
|
10 |
(4 and 6) not 9 - RCT |
105 |
|
11 |
(4 and (7 or 8)) not (9 or 10) - observationeel |
134 |
|
12 |
9 or 10 or 11 |
267 |