Maligniteit geassocieerde trombose
Uitgangsvraag
Wat is het optimale antistollingsbeleid bij patiënten met een actieve maligniteit en een acute VTE, voor de initiële periode (eerste 3-6 maanden) en voor de langere termijn (na 6 maanden)?
Aanbeveling
Behandel patiënten met een maligniteit en een acute VTE met een therapeutische dosis DOAC (apixaban, edoxaban, rivaroxaban) of LMWH, volgens de doseringsvoorschriften vermeld in het Farmacotherapeutisch kompas.
Behandel patiënten met acute VTE en een niet-gereseceerde maligniteit van de tractus digestivus (inclusief lever-, galweg- en pancreastumoren) of tractus urogenitalis bij voorkeur met LMWH. Overweeg apixaban als alternatief.
Overweeg patiënten met een maligniteit en VTE met een indicatie voor langdurige behandeling met therapeutische antistolling, na de eerste zes maanden te behandelen met een gereduceerde dosis apixaban (2dd 2,5 mg), tenzij er contra-indicaties zijn voor een DOAC.
Overwegingen
Balans tussen gewenste en ongewenste effecten
Uit de analyse van de literatuur blijkt dat initiële behandeling met directe orale anticoagulantia (DOACs) bij patiënten met een acute maligniteit geassocieerde veneuze trombo-embolie (VTE) waarschijnlijk leidt tot een klinisch relevant lager risico op recidief VTE (cruciale uitkomst), vergeleken met een initiële behandeling met laag-moleculair-gewicht heparine (LMWH). Er is mogelijk geen klinisch relevant verschil in majeure bloedingen (cruciale uitkomst) tussen een behandeling met DOAC of een behandeling met LMWH. Voor mortaliteit (cruciale uitkomst) geldt dat er ook geen klinisch relevant verschil is tussen de twee behandelingen. Initiële behandeling met DOACs leidt waarschijnlijk tot een relevant hoger risico op klinisch relevante niet-majeure bloedingen (belangrijke uitkomst), vergeleken met een LMWH-behandeling. Voor de belangrijke uitkomst net clinical benefit geldt dat het bewijs te onzeker is om een uitspraak te kunnen doen over het effect van behandeling met DOACs vergeleken met LMWH gezien alleen 1 studie dit heeft onderzocht.
Hoewel in de meta-analyse het risico op majeure bloedingen niet klinisch relevant verschilde tussen behandeling met DOAC en LMWH bij acute maligniteit geassocieerde VTE , zijn er wel signalen uit de individuele studies dat sommige DOAC meer majeure bloedingen geven in specifieke situaties. Met name patiënten met een (in-situ) tumor in de tractus digestivus of tractus urogenitalis bleken in sommige studies vaker majeure bloedingen te krijgen. Om die reden werd bijvoorbeeld de eerste van die twee categorieën halverwege de SELECT-D studie niet meer geïncludeerd in de studie (Young, 2018). De vorige versie van de module gaf dan ook het advies patiënten met een maligniteit van de tractus digestivus (inclusief lever- en pancreastumoren) en de tractus urogenitalis bij voorkeur met LMWH te behandelen. De studies die in deze update van de module extra zijn meegenomen, met name de Caravaggio studie waarin apixaban is gebruikt en patiënten met gastro-intestinale en urogenitale maligniteiten wel zijn meegenomen, suggereerden een vergelijkbaar risico op majeure bloedingen bij behandeling met apixaban ten opzichte van LMWH. Hoewel de subgroep analyses te klein zijn om hier een harde uitspraak over te doen, beschouwt de richtlijncommissie apixaban als een redelijk alternatief voor LMWH in deze setting.
In de beschikbare studies over acute maligniteit geassocieerde VTE met DOAC en LMWH zijn sommige types patiënten (bijvoorbeeld met ernstige nierinsufficiëntie, trombocytopenie <100x10^9/l) of soorten maligniteiten (intracerebrale tumoren, hematologische maligniteiten zoals acute leukemie of na stamceltransplantatie) niet goed vertegenwoordigd. Het is niet duidelijk in hoeverre de gevonden resultaten en aanbevelingen daarom ook van toepassing zijn voor deze patiëntengroepen en type maligniteiten.
Interacties tussen DOACs en chemotherapeutica of andere comedicatie zijn maar in beperkte mate onderzocht. De opname en klaring van DOACs is afhankelijk van P-glycoproteïne en CYP450 enzymen, die allen kunnen worden geremd of versterkt door verschillende chemotherapeutica, immuuntherapie of andere medicatie zoals -azolen of anti-epileptica. Er zijn intrinsieke verschillen tussen de drie beschikbare DOACs, maar de klinische impact hiervan is niet bekend en niet vergeleken tussen de verschillende DOACs. De farmacologisch meest ‘optimale’ DOAC zou dus per patiënt, per soort maligniteit en per type chemotherapeuticum of co-medicatie kunnen verschillen. Het is van belang om samen met de patiënt af te wegen welke DOAC het meest passend is.
We hebben geen literatuur search gedaan naar de optimale duur van antistollingsbehandeling bij patiënten met een acute VTE en een actieve maligniteit. De beschreven adviezen zijn gebaseerd op de bestaande richtlijn Antitrombotisch beleid (2016), de ASH-guidelines voor behandeling van kanker-geassocieerde VTE (2021) en de behandelduur die is gehanteerd in de beoordeelde RCTs voor initiële behandeling van acute kanker-geassocieerde VTE. De duur van de antistollingsbehandeling bij patiënten met een acute VTE en een actieve maligniteit is tenminste zes maanden. Patiënten met een hoog bloedingsrisico dienen ten minste drie maanden behandeld te worden. Zolang er sprake is van actieve maligniteit dan wel chemotherapie of adjuvante behandeling kan worden overwogen om deze behandeling te verlengen.
Twee studies (EVE-trial en API-CAT-trial) hebben onderzocht of voor verlengde behandeling van kanker-geassocieerde VTE na zes maanden dosisreductie van apixaban (naar 2dd 2.5 mg) mogelijk is (McBane, 2025; Mahé, 2025). Deze studies lieten zien dat gereduceerde dosis apixaban non-inferieur was aan volledige dosering apixaban voor recidief VTE, maar wel resulteerde in een lager risico op de combinatie van ernstige en klinisch relevante niet-ernstige bloedingen. Derhalve kan, na een initiële behandeling met LMWH of DOAC van tenminste zes maanden, worden overwogen om over te gaan op gereduceerde dosis apixaban (2dd 2.5 mg). Voor de andere DOACs is dit niet onderzocht.
Bij voortgezette behandeling is het aan te bevelen regelmatig, en in ieder geval jaarlijks, een individuele afweging te maken tussen het verlagen van het recidief tromboserisico en het bloedingsrisico tijdens behandeling met antistolling.
Kwaliteit van bewijs
De overall kwaliteit van bewijs is laag. Dit betekent dat we onzeker zijn over het gevonden geschatte effect van de cruciale uitkomstmaten. Het is relevant om te benoemen dat de bewijskracht varieerde van laag (majeure bloeding) tot redelijk (recidief VTE) en hoog (mortaliteit).
Er is afgewaardeerd vanwege (zeer) ernstige:
- Imprecisie: onnauwkeurigheid, omdat het betrouwbaarheidsinterval de grens/beide grenzen van klinische relevantie overschrijdt.
Waarden en voorkeuren van patiënten (en eventueel hun naasten/verzorgers)
De uitkomst van de kankerbehandeling (bijvoorbeeld progressievrije overleving en mortaliteit) en kwaliteit van leven zijn voor de meeste patiënten met kanker doorgaans het belangrijkst. De keus DOAC of LMWH lijkt geen invloed te hebben op overleving. Wel lijken DOACs geassocieerd met een lager risico op recidief trombo-embolie, maar met meer niet-majeure klinische relevante bloedingen, met grofweg dezelfde ‘number needed to treat/harm’. Hoewel maar 1 studie onderzoek heeft gedaan naar ‘net clinical benefit’, lijken de voordelen en nadelen van DOACs ten opzichte van LMWH tegen elkaar op te wegen voor behandeling van acute maligniteit geassocieerde VTE. Er is weinig onderzoek gedaan naar het effect van subcutane injecties versus orale medicatie op de kwaliteit van leven, maar de weinig beschikbare literatuur suggereert dat patiënten een voorkeur zullen hebben voor orale medicatie (Hutchinson, 2019). In de CANVAS-studie waren er geen relevante verschillen in patiënt-gerapporteerde kwaliteit van leven of beleving van de antistollingsbehandeling tussen patiënten behandeld met een DOAC of LMWH; wel was de therapie adherentie beter in patiënten die een DOAC kregen (Schrag, 2023).
Kostenaspecten
De interventie levert minder kosten op ten opzichte van de controle behandeling vanwege de lagere kosten van (generieke) DOACs ten opzichte van LMWH. Dit zou de keuze voor DOACs kunnen laten doorslaan in positieve wijze.
Gelijkheid ((health) equity/equitable)
De interventie leidt niet tot een toename van gezondheidsgelijkheid.
Aanvaardbaarheid
Ethische aanvaardbaarheid
Zowel DOACs als LMWH zijn aanvaardbaar voor alle betrokkenen.
Duurzaamheid
LMWH zijn minder duurzaam vanwege het restmateriaal (injectiespuit).
Haalbaarheid
Behandeling met zowel DOAC als LMWH is haalbaar. Het kunnen slikken van orale medicatie is een voorwaarde voor DOACs, terwijl het (zelfstandig) kunnen toedienen van subcutane injecties een voorwaarde is voor LMWH.
Rationale van de aanbeveling: weging van argumenten voor en tegen de interventies
Een behandeling met DOACs vs LMWH leidt niet tot een klinisch relevant verschil in mortaliteit. Daarnaast leidt een behandeling met een DOAC mogelijk tot minder recidief VTE, maar zonder verschil in majeure bloedingen t.o.v. behandeling met LMWH. Wel werden er meer klinisch relevante niet-majeure bloedingen gezien in de patiënten die een DOAC kregen. Om deze reden ziet de werkgroep ruimte om patiënten met een maligniteit geassocieerde acute VTE te behandelen met een DOAC of LMWH.
Eindoordeel:
Sterke aanbeveling voor Doen.
Onderbouwing
In patients with cancer and acute venous thromboembolism (VTE), low-molecular-weight heparin (LMWH) at therapeutic doses was long considered the preferred treatment. This recommendation was based on several randomized trials comparing LMWH with vitamin K antagonists (VKAs), which demonstrated superior efficacy of LMWH in preventing recurrent VTE (Kahale, 2018). Over the past two decades, direct oral anticoagulants (DOACs) were introduced. Phase 3 trials comparing DOACs with VKAs in patients with acute VTE showed similar risks of recurrent VTE and bleeding, with a potential advantage of DOACs in reducing intracranial hemorrhage (van der Hulle, 2014; Posch, 2015; van Es, 2014). However, these studies included few patients with active cancer. Subsequently, several subsequent phase 3 trials have been conducted to evaluate the efficacy and safety of DOACs compared with LMWH specifically in patients with active cancer and acute VTE.
Summary of Findings
Initial anticoagulant treatment in patients with acute cancer-associated venous thromboembolism (VTE)
Population: Adults with active cancer and objectively confirmed acute deep vein thrombosis (DVT) and/or pulmonary embolism (PE)
Intervention: Direct oral anticoagulant (DOAC) – Rivaroxaban, edoxaban, apixaban
Comparator: Low-molecular-weight heparin (LMWH) – Dalteparin
|
Duration of treatment |
Outcome
|
Study results and measurements |
Absolute effect estimates |
Certainty of the Evidence (Quality of evidence) |
Conclusions |
|
|
3-6 months |
|
LMWH |
DOAC |
|
||
|
Recurrent VTE (critical) |
RR 0.66 (95%CI 0.50 to 0.87) |
8 per 100 |
6 per 100 |
⨁⨁⨁◯ Moderate1
|
Initial treatment with DOAC likely reduces the risk of a recurrent VTE when compared with LMWH in patients with acute cancer- associated VTE. . |
|
|
Major bleeding (critical)
|
RR 1.22 (95%CI 0.74 to 2.00) |
4 per 100 |
5 per 100 |
⨁⨁◯◯ Low2 |
Initial treatment with DOAC may result in little to no difference in major bleeding events when compared with LMWH in patients with acute cancer-associated VTE. |
|
|
Mortality (critical) |
RR 1.02 (95%CI 0.87 to 1.19) |
29 per 100 |
29 per 100 |
⨁⨁⨁⨁ High |
Initial treatment with DOAC results in little to no difference in mortality when compared with LMWH in patients with acute cancer-associated VTE. |
|
|
CRNMB (important) |
RR 1.60 (95%CI 1.13 to 2.28) |
7 per 100 |
12 per 100 |
⨁⨁⨁◯ Moderate1
|
Initial treatment with DOAC likely increases the risk of a clinically relevant non-major bleeding when compared with LMWH in patients with acute cancer-associated VTE. |
|
|
Net clinical benefit (important) |
HR 0.97 (95%CI 0.70 to 1.36) |
14 per 100 |
13 per 100 |
⨁◯◯◯ Very low3 |
The evidence is very uncertain about the effect of initial treatment with DOAC on net clinical benefit when compared with LMWH in patients with acute cancer-associated VTE. |
|
|
> 6 months |
|
|
LMWH |
DOAC |
|
|
|
All outcomes |
No trial could be included on the treatment duration longer than six months. |
|||||
HR: hazard ratio; RR: risk ratio; CRNMB: clinically relevant non-major bleeding.
1 Downgraded because of imprecision (CI crosses one boundary of clinically important difference)
2 Downgraded because of imprecision (CI crosses both boundaries of clinically important difference)
3 Downgraded because of risk of bias (industry sponsorship heavily involved in the trial) and imprecision (CI crosses both boundaries of clinically important difference)
Description of studies
A total of six studies were included in the analysis of the literature. Important study characteristics and results are summarized in Table 2. Of these studies, six investigated the initial treatment of cancer-associated VTE and none the long-term treatment of cancer-associated VTE (beyond the initial six months following diagnosis). In the trial of Raskob (2018), participants could continue the treatment beyond 6 months; however, this was up to the treating physician. The results on long-term treatment were therefore not included. The assessment of the risk of bias is summarized in the risk of bias tables (under the tab ‘Evidence tabellen’).
Young (2018) conducted the SELECT-D-trial, a multicenter, randomized, open label phase 3 pilot trial in the United Kingdom, comparing rivaroxaban with dalteparin in patients with active cancer and symptomatic or incidental PE, or symptomatic lower extremity proximal DVT. Patients were allocated to six months of rivaroxaban (15 mg twice daily for three weeks, then 20 mg once daily for a total of six months) or dalteparin (200 IU/kg daily during month one, then 150 IU/kg daily for months 2-6). The primary outcome was VTE recurrence over six months, and secondary safety outcomes were major bleeding and CRNMB. Exclusion criteria included high bleeding risk, platelet count <75 x 10⁹/L, creatinine clearance <30 mL/min, or life expectancy <3 months. Due to the high incidence of major bleeding, patients with malignant tumors of the esophagus or gastroesophageal junction were excluded from enrollment as a precautionary measure from the midpoint of the trial onwards. The trial was extended for a follow-up of 12 months. However, in this continuation of the SELECT-D trial, participants were newly randomized to rivaroxaban or placebo which was not the comparison of interest. Therefore, the 12 months publication of the SELECT-D trial was excluded (Marshall, 2020; see also exclusion table).
Raskob (2018) conducted the Hokusai VTE Cancer trial, a multinational, randomized, open label, noninferiority phase 3 trial in patients with active cancer and newly diagnosed acute symptomatic or incidental VTE. Participants were randomized to receive either edoxaban (60 mg once daily after at least five days of initial LMWH), or subcutaneous dalteparin (200 IU/kg for one month followed by 150 IU/kg daily). Patients with severe renal dysfunction, high bleeding risk, or brain tumors were excluded. In both groups, treatment was continued for at least six months, with a possible extension up to 12 months. Primary outcome was a composite of recurrence VTE or major bleeding. Secondary outcomes were the individual components recurrent VTE and major bleeding, and all-cause mortality.
McBane (2020) conducted the ADAM VTE-trial, a multicenter, open label, randomized trial in the United States. Adult patients, 18 years or older, with histologically confirmed active cancer and acute newly diagnosed VTE (symptomatic or incidental PE, proximal or distal upper or lower extremity DVT, splanchnic or cerebral vein thrombosis) were enrolled. Patients were randomized to receive apixaban (10 mg twice daily for seven days followed by 5 mg twice daily) or subcutaneous dalteparin (200 IU/kg for 1 month, followed by 150 IU/kg daily) for a total of six months. Exclusion criteria included ECOG PS >2, platelet count <50 x10⁹/L, or high bleeding risk. The primary safety outcome was any episode of major bleeding, secondary outcomes were any VTE recurrence, and a composite of major bleeding plus CRNMB.
Agnelli (2020) conducted the CARAVAGGIO-trial, a multinational, randomized, open label, noninferiority trial. Adult patients with active cancer and objectively confirmed newly diagnosed symptomatic or incidental proximal lower-limb DVT or PE (symptomatic or incidental) were enrolled. Participants were randomized to receive apixaban (10 mg twice daily for seven days followed by 5 mg twice daily) or dalteparin (200 IU/kg for one month followed by 150 IU/kg daily), for six months. Patients with brain tumors, platelet count <50 x10⁹/L, or high bleeding risk were excluded. Primary outcome was objectively confirmed recurrent VTE during the six-month trial period. The principal safety outcome was major bleeding.
Planquette (2022) conducted the CASTA-DIVA-trial, a multicenter, open-label, randomized controlled noninferiority trial performed in France. The study included adult patients with active cancer and objectively confirmed newly diagnosed VTE (symptomatic or incidental lower-limb DVT or inferior vena cava thrombosis, or PE, or both). Patients were randomized to receive rivaroxaban (15 mg twice daily for three weeks followed by 20 mg once daily) or dalteparin (200 IU/kg for one month then 150 IU/kg once daily) for a period of three months. Patients with ECOG performance status >2, significant thrombocytopenia (<75 x10⁹/L), or severe renal insufficiency were excluded. The primary efficacy outcome was a composite of recurrent VTE and worsening of pulmonary vascular or venous obstruction at three months. Secondary outcomes included symptomatic recurrences of PE or DVT, major bleeding events, a composite of major and CRNMB events, and all-cause mortality. Recruitment was stopped prematurely after 158 patients had been randomized, due to a slower-than-anticipated recruitment rate.
Schrag (2023) performed the CANVAS-trial, a multicenter, open label, noninferiority randomized comparative effectiveness study conducted across 67 oncology practices in the United States. Adult patients with active cancer and a new diagnosis of VTE (including symptomatic or asymptomatic PE or DVT) were eligible. Patients were randomized to receive either a DOAC (rivaroxaban, apixaban, or edoxaban, per physician’s choice) or LMWH. There were minimal exclusion criteria to reflect real-world practice. Outcomes were recurrence of nonfatal VTE, major bleeding, CRNMB, minor bleeding, mortality at six months, and health-related quality of life and participants’ perceptions of the burdens or benefits of anticoagulant treatment.
Table 2. Characteristics of included studies
|
Study |
Participants |
Comparison |
Follow-up for primary outcome |
Outcome measures |
Comments |
Risk of bias (per outcome measure)* |
|
Young, 2018 (SELECT-D) |
N=406
Male sex: 116 (57%) Metastatic disease: 58%
Control: 203 Median age: 67 (34-87) years Male sex: 98 (48%) Metastatic disease: 58%
|
Rivaroxaban (15 mg twice daily for 3 weeks followed by 20 mg once daily) |
6 months |
Primary: Recurrent VTE
Secondary: Major bleeding and CRNMB |
Pilot trial; open-label; industry-funded; Inclusion of esophagus or gastroesophageal junction tumors was stopped halfway due to high bleeding incidences |
Some concerns (all outcomes) |
|
Raskob, 2018 (Hokusai VTE Cancer) |
N=1050 (analyzed = 1046)
Mean age: 64.3 (11.) Male sex:: 277 (53.1%) Metastatic disease: 52.5%
Male sex: 263 (50.2%) Metastatic disease: 53.4% |
Edoxaban (60 mg once daily after at least 5 days LMWH) |
12 months |
Primary: Composite of recurrent VTE or major bleeding
Secondary: Recurrent VTE CRNMB Major or CRNMB Mortality Event-free survival |
Open-label; industry-funded
|
High risk of bias (all outcomes) |
|
McBane, 2020 (ADAM VTE) |
N=300 (treated = 187)
Mean age: 64.4 (11.3) years Male sex: 72 (48%) Metastatic disease: 65.3%
Male sex: 73 (48.7%) Metastatic disease: 66%
|
Apixaban (10 mg twice daily for 7 days followed by 5 mg twice daily) vs. |
6 months |
Primary: Major bleeding
Secondary: Recurrent VTE or arterial thromboembolism Major plus CRNMB |
Open-label; small trial; industry-funded |
Low risk of bias (all outcomes) |
|
Agnelli, 2020 (CARAVAGGIO) |
N=1155
Mean age: 67.2 (11.3) years Male sex: 292 (50.7%) Metastatic disease: 67.5%
Male sex: 276 (47.7%) Metastatic disease: 68.4% |
Apixaban (10 mg twice daily for 7 days followed by 5 mg twice daily) vs. |
6 months |
Primary: Recurrent VTE
Principal safety: Major bleeding Secondary: Recurrent VTE or major bleeding CRNMB Mortality Event-free survival |
Open-label; noninferiority trial; partially industry-funded |
Low risk of bias (all outcomes) |
|
Planquette, 2022 (CASTA-DIVA) |
N=158
Mean age: 68.6 (62.9-77.8) years Male sex: 37 (50%) Metastatic disease: 76.8%
Control: 84 Male sex: 40 (47.6%) Metastatic disease: 75.6%
|
Rivaroxaban (15 mg twice daily for 3 weeks followed by 20 mg once daily) vs. Dalteparin (200 IU/kg once daily for 1 month followed by 150 IU/kg once daily) |
3 months |
Primary: Recurrent VTE
Secondary: Major bleeding Mortality Symptomatic recurrent PE or DVT |
Noninferiority criteria not met due to small sample |
Low risk of bias (all outcomes) |
|
Schrag, 2023 (CANVAS) |
N=671
Median age: 64 (56-70) years Male sex: 149 (45%) Metastatic disease: 70%
Male sex: 111 (36%) Metastatic disease: 68%
|
DOAC (rivaroxaban (N=122; 37.0%), apixaban (N=193; 58.5%), dabigatran (N=9; 2.7%) or edoxaban (N=6; 1.8%), per physician’s choice) |
6 months |
Primary: Recurrent nonfatal VTE
Secondary: |
Open-label; noninferiority trial; partially third party-funded |
Low risk of bias (all outcomes) |
*For further details, see risk of bias table in the appendix
Results
The effect estimates were differently reported across studies; some studies reported a hazard ratio, others only the event numbers. In order to use all the available information, risk ratios (RR) were calculated based on the numbers provided in the studies.
Treatment duration 3-6 months
Recurrent VTE (critical)
Six studies (Young, 2018; Raskob, 2018; McBane, 2020; Agnelli, 2020; Planquette, 2022; Schrag, 2023) reported on the incidence of recurrent venous thromboembolism (VTE) in patients with active cancer receiving direct oral anticoagulants (DOACs) compared with low-molecular-weight heparin (LMWH) for initial treatment of acute VTE. The studies evaluated rivaroxaban, edoxaban, apixaban, or various DOACs per physician’s discretion.
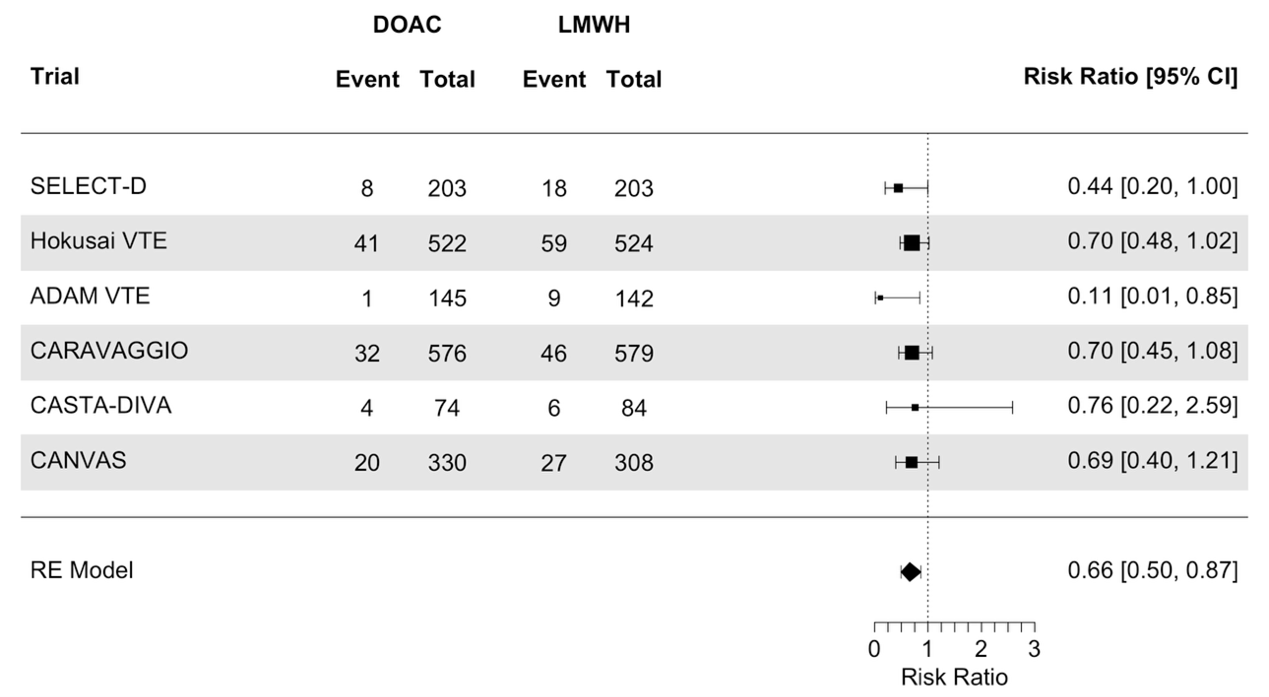
The results were estimated with RRs. The number of events and number of randomized participants was used to calculate the RRs. The pooled analysis showed that the risk of a recurrent VTE decreased (clinically relevant) with the use of a DOAC compared with LMWH (RR 0.66; 95%CI 0.50 to 0.87; Figure 1).
Figure 1. Meta-analysis on the effect of initial treatment of acute VTE in patients with active cancer with DOAC compared to treatment with LMWH, on the outcome recurrent VTE
DOAC: direct oral anticoagulants, LMWH: low molecular weight heparin
Major bleeding (critical)
Six studies (Young, 2018; Raskob, 2018; McBane, 2020; Agnelli, 2020; Planquette, 2022; Schrag, 2023) reported on the incidence of major bleeding in patients with active cancer receiving DOACs compared with LMWH for initial treatment of acute VTE. One study (McBane, 2020; ADAM-VTE trial) reported no major bleeding events in DOAC-group and two events in the LMWH-group. No effect estimate was calculated for this study.
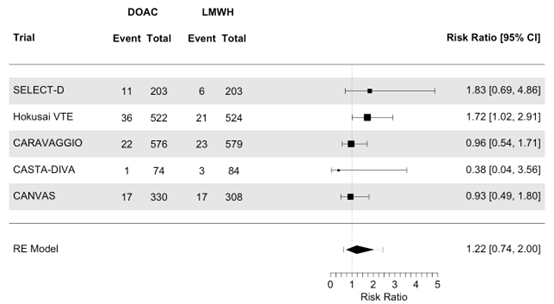
The results were estimated with RRs. The number of events and number of randomized participants was used to calculate the RRs. The pooled analysis showed that the risk of a major bleeding was similar (no clinically relevant difference) with the use of a DOAC compared with LMWH (RR 1.22; 95%CI 0.74 to 2.00; Figure 2).
Figure 2. Meta-analysis on the effect of initial treatment of acute VTE in patients with active cancer with DOAC compared to treatment with LMWH, on the outcome major bleeding
DOAC: direct oral anticoagulants, LMWH: low molecular weight heparin
Mortality (critical)
Six studies (Young, 2018; Raskob, 2018; McBane, 2020; Agnelli, 2020; Planquette, 2022; Schrag, 2023) reported on the incidence of all-cause mortality in patients with active cancer receiving DOACs compared to LMWH for initial treatment of acute VTE.
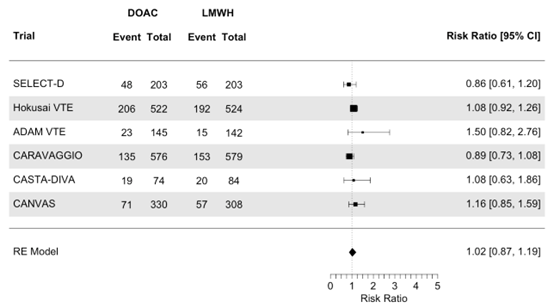
The results were estimated with RRs. The number of events and number of randomized participants was used to calculate the RRs. The pooled analysis showed that the risk of mortality was similar (no clinically relevant difference) with the use of a DOAC compared with LMWH (RR 1.02; 95%CI 0.87 to 1.19, Figure 3).
Figure 3. Meta-analysis on the effect of initial treatment of acute VTE in patients with active cancer with DOAC compared to treatment with LMWH, on the outcome mortality
DOAC: direct oral anticoagulants, LMWH: low molecular weight heparin
Clinically relevant non-major bleeding (CRNMB) (important)
Six studies (Young, 2018; Raskob, 2018; McBane, 2020; Agnelli, 2020; Planquette, 2022; Schrag, 2023) reported on the incidence of clinically relevant non-major bleeding (CRNMB) in patients with active cancer receiving DOACs compared with LMWH for initial treatment of acute VTE.
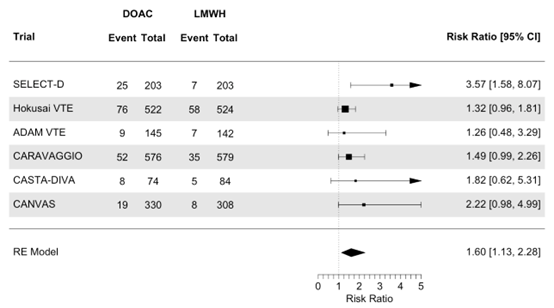
The results were estimated with RRs. The number of events and number of randomized participants was used to calculate the RRs. The pooled analysis showed that the risk of a CRNMB was increased (clinically relevant) with the use of a DOAC compared with LMWH (RR 1.60; 95%CI 1.13 to 2.28, Figure 4).
Figure 4. Meta-analysis on the effect of initial treatment of acute VTE in patients with active cancer with DOAC compared to treatment with LMWH, on the outcome clinically relevant non-major bleeding
DOAC: direct oral anticoagulants, LMWH: low molecular weight heparin
Net clinical benefit (important)
One study (Raskob, 2018) reported a net clinical benefit (a composite of recurrent VTE or major bleeding) in patients with active cancer receiving edoxaban compared with dalteparin for initial treatment of acute VTE. The reported HR was 0.97 (95%CI 0.70 to 1.36), which is not a clinically relevant difference.
Treatment duration > 6 months
No trial was included on the effect of treatment with DOACs beyond the initial six months, compared to LMWH-treatment in patients with acute cancer-associated VTE.
A systematic review of the literature was performed to answer the following question(s):
What is the efficacy and safety of direct oral anticoagulants (DOACs) compared to low-molecular-weight heparin (LMWH) in patients with cancer-associated VTE (i.e., deep vein thrombosis (DVT) of the leg and/or pulmonary embolism (PE)), for (a) the initial treatment period (first 3–6 months), and (b) the long-term treatment period (beyond 6 months)?
Table 1. PICO
| Patients | Adults with acute cancer-associated venous thromboembolism (VTE, i.e. DVT of the leg and/or PE) |
| Intervention | Anticoagulant therapy with direct oral anticoagulants (DOACs) |
| Control | Anticoagulant therapy with low-molecular-weight heparin (LMWH) |
| Outcomes | Mortality, recurrent VTE, major bleeding, net clinical benefit, clinically relevant non-major bleeding (CRNMB) |
| Other selection criteria | Study design: systematic reviews and randomized controlled trials |
| Planned analyses |
Analyses will be separated by the duration of treatment (3-6 months versus > 6 months) If the pooled analysis shows indications of heterogeneity, a subgroup analysis by type of DOAC will be performed |
Relevant outcome measures
The guideline panel considered mortality, recurrent VTE and major bleeding as critical outcome measures for decision making; and net clinical benefit and CRNMB as important outcome measures for decision making.
A priori, the guideline panel did not define the outcome measures listed above but used the definitions used in the studies.
The definition of active malignancy differs in the literature and between studies, but roughly was as follows:
- a diagnosis of (recurrent) cancer (excluding basal cell carcinoma or squamous cell carcinoma of the skin) in the past 6 months;
- up to 6 months after the last anti-cancer treatment;
- the presence of a known chronic malignant condition (such as chronic myeloid/ lymphoid leukemia or multiple myeloma) or
- the presence of known metastatic disease, regardless of whether the patient is in a (terminal) palliative phase.
The guideline panel defined a relative risk of ≤ 0.80 or ≥ 1.25 as the threshold for a minimal clinically important difference across all outcome measures. These criteria were applied to both hazard ratios (HR) and odds ratios (OR).
Search and select (Methods)
In 2020 the module Treatment of cancer-associated thrombosis was updated. A systematic literature update was performed by a medical information specialist searching the following bibliographic databases on 23 April 2018 for systematic reviews and RCTs: Medline (via PubMed), Embase (via Ovid), Web of Science, The Cochrane Library en Academic Search Premier. Initially, 28 studies were selected based on title and abstract screening. After reading the full text, 25 studies were excluded (see the exclusion table under the tab ‘Evidence tabellen’), and three studies were included: two RCTs (Raskob, 2018; Young, 2018) and one systematic literature review (Li, 2018). Dose-finding studies (phase 2 trials) and studies on primary prophylaxis of VTE in cancer patients were excluded. Since the review of Li (2018) only included the studies of Raskob (2018) and Young (2018) and some additional non-randomized studies, the literature analysis focused on the studies of Raskob (2018) and Young (2018).
In this 2024 update, a systematic literature update was performed by a medical information specialist using the following bibliographic databases: Embase.com and Ovid/Medline. Both databases were searched from January 2018 to 17 October 2024 for systematic reviews and RCTs. Systematic searches were completed using a combination of controlled vocabulary/subject headings (e.g., Emtree-terms, MeSH) wherever they were available and natural language keywords. Duplicates were removed using EndNote software. After deduplication a total of 1232 records were imported for title/abstract screening. Initially, 27 studies were selected based on title and abstract screening. After reading the full text, 23 studies were excluded (see the exclusion table under the tab ‘Evidence tabellen’), and four additional studies were included (McBane, 2020; Agnelli, 2020; Planquette, 2022 and Schrag, 2023). Dose-finding studies (phase 2 trials), comparisons between two doses of DOACs and studies on primary prophylaxis of VTE in cancer patients were excluded.
- Agnelli G, Becattini C, Meyer G, Muñoz A, Huisman MV, Connors JM, Cohen AT, Lee AYY, Gallus AS, Johnson M, Porreca E, Bauersachs R. Apixaban for the Treatment of Venous Thromboembolism Associated with Cancer. N Engl J Med. 2020 Apr 23;382(17):1599-1607. doi: 10.1056/NEJMoa1915103. PMID: 32320557.
- van Es N, Coppens M, Schulman S, Middeldorp S, Büller HR. Direct oral anticoagulants compared with vitamin K antagonists for acute venous thromboembolism: evidence from phase 3 trials. Blood. 2014 Sep 18;124(12):1968-75. doi: 10.1182/blood-2014-04-571232. Epub 2014 Jun 24. PMID: 24963045.
- van der Hulle T, den Exter PL, Kooiman J, van der Hoeven JJ, Huisman MV, Klok FA. Meta-analysis of the efficacy and safety of new oral anticoagulants in patients with cancer-associated acute venous thromboembolism. J Thromb Haemost. 2014 Jul;12(7):1116-20. doi: 10.1111/jth.12605. Epub 2014 Jun 19. PMID: 24819040.
- Hutchinson A, Rees S, Young A, Maraveyas A, Date K, Johnson MJ. Oral anticoagulation is preferable to injected, but only if it is safe and effective: An interview study of patient and carer experience of oral and injected anticoagulant therapy for cancer-associated thrombosis in the select-d trial. Palliat Med. 2019 May;33(5):510-517. doi: 10.1177/0269216318815377. Epub 2018 Nov 29. PMID: 30488789; PMCID: PMC6506899.
- Kahale LA, Tsolakian IG, Hakoum MB, Matar CF, Barba M, Yosuico VE, Terrenato I, Sperati F, Schünemann H, Akl EA. Anticoagulation for people with cancer and central venous catheters. Cochrane Database Syst Rev. 2018 Jun 1;6(6):CD006468. doi: 10.1002/14651858.CD006468.pub6. PMID: 29856471; PMCID: PMC6389340.
- Mahé I, Carrier M, Mayeur D, Chidiac J, Vicaut E, Falvo N, Sanchez O, Grange C, Monreal M, López-Núñez JJ, Otero-Candelera R, Le Gal G, Yeo E, Righini M, Robert-Ebadi H, Huisman MV, Klok FA, Westerweel P, Agnelli G, Becattini C, Bamias A, Syrigos K, Szmit S, Torbicki A, Verhamme P, Maraveyas A, Cohen AT, Ay C, Chapelle C, Meyer G, Couturaud F, Mismetti P, Girard P, Bertoletti L, Laporte S; API-CAT Investigators. Extended Reduced-Dose Apixaban for Cancer-Associated Venous Thromboembolism. N Engl J Med. 2025 Apr 10;392(14):1363-1373. doi: 10.1056/NEJMoa2416112. Epub 2025 Mar 29. PMID: 40162636.
- McBane RD, Wysokinski WE, Le-Rademacher JG, Nahhas GJ, Ashrani AA, Tafur A, McCrae KR, Milhem M, Kolesar J, Wieben ED, Khorana AA. Apixaban and Dalteparin in Active Malignancy–Associated Venous Thromboembolism: The ADAM VTE Trial. J Thromb Haemost. 2020 Nov;18(11):4115-4123. doi: 10.1111/jth.15002. PMID: 32686279.
- McBane RD 2nd, Loprinzi CL, Zemla T, Tafur A, Sanfilippo K, Liu JJ, Garcia DA, Heun J, Gundabolu K, Onitilo AA, Perepu U, Drescher MR, Henkin S, Houghton D, Ashrani A, Billett H, McCue SA, Lee MK, Le-Rademacher JG, Wysokinski WE; EVE trial investigators. Extending venous thromboembolism secondary prevention with apixaban in cancer patients. The EVE trial. J Thromb Haemost. 2024 Jun;22(6):1704-1714. doi: 10.1016/j.jtha.2024.03.011. Epub 2024 Mar 25. Erratum in: J Thromb Haemost. 2025 Feb;23(2):756. doi: 10.1016/j.jtha.2024.12.011. PMID: 38537780.
- Planquette B, Faille D, Doubine S, Canet E, Lemarié C, Doubovetzky M, Chatellier G, Sanchez O. Rivaroxaban vs Dalteparin in Cancer-Associated Thromboembolism: A Randomized Trial. Chest. 2022 Jun;161(6):1571-1580. doi: 10.1016/j.chest.2021.11.018. PMID: 34875463.
- Posch F, Königsbrügge O, Zielinski C, Pabinger I, Ay C. Treatment of venous thromboembolism in patients with cancer: A network meta-analysis comparing efficacy and safety of anticoagulants. Thromb Res. 2015 Sep;136(3):582-9. doi: 10.1016/j.thromres.2015.07.011. Epub 2015 Jul 17. PMID: 26210891; PMCID: PMC7311195.
- Raskob GE, van Es N, Verhamme P, Carrier M, Di Nisio M, Garcia D, Grosso MA, Kakkar AK, Kovacs MJ, Mercuri MF, Segers A, Shi M, Wang TF, Weitz JI, Büller HR; Hokusai VTE Cancer Investigators. Edoxaban for the Treatment of Cancer-Associated Venous Thromboembolism. N Engl J Med. 2018 Feb 15;378(7):615-624. doi: 10.1056/NEJMoa1711948. PMID: 29231094.
- Schrag D, Lin GA, Goldhaber SZ, Sanoff HK, Bhatia S, Hudis C, Meara E, Shah N, Rivest A, Trivedi A, Finkelstein DM. Direct Oral Anticoagulants vs Low-Molecular-Weight Heparin and Recurrent VTE in Patients with Cancer: A Randomized Clinical Trial. JAMA. 2023 Aug 1;330(5):425–434. doi:10.1001/jama.2023.12515. PMID: 37523463.
- Young AM, Marshall A, Thirlwall J, Chapman O, Lokare A, Hill C, Hale D, Dunn JA, Lyman GH, Hutchinson C, MacCallum P, Kakkar A, Hobbs FDR, Petrou S, Dale J, Poole CJ, Maraveyas A, Levine M. Comparison of an Oral Factor Xa Inhibitor With Low Molecular Weight Heparin in Patients With Cancer With Venous Thromboembolism: Results of a Randomized Trial (SELECT-D). J Clin Oncol. 2018 Jul 10;36(20):2017-2023. doi: 10.1200/JCO.2018.78.8034. PMID: 29746227.
Risk of Bias tables
|
Study reference
|
Was the allocation sequence adequately generated? |
Was the allocation adequately concealed? |
Blinding: Was knowledge of the allocated interventions adequately prevented? Were patients/healthcare providers/data collectors/outcome assessors/data analysts blinded? Were patients blinded? |
Was loss to follow-up (missing outcome data) infrequent? |
Are reports of the study free of selective outcome reporting?
|
Was the study apparently free of other problems that could put it at a risk of bias? |
Overall risk of bias
|
|
Young, 2018 |
Definitely yes – “Consenting patients were randomly assigned at a one-to-one ratio using a computer-based minimization algorithm with stratification by stage of disease, baseline platelet count, type of VTE, and risk of clotting by tumor type.” |
Definitely yes – Secure allocation process used across sites. |
Probably yes – Open-label design
“Trial staff, participants, and investigators were not blinded to treatment allocation.” An independent committee of experienced clinicians unaware of treatment allocation adjudicated all the outcomes.
|
Definitely yes |
Definitely yes |
Probably no
Although the trial was sponsored by the industry, the grant was unrestricted and the sponsor had no conduct in the design, data analysis, etc. However, after a safety analysis showing a nonsignificant difference in major bleeding, the enrollment of patients with esophagus or gastroesophageal cancer was stopped. |
Some concerns - for all outcomes
Due to the exclusion of some patients halfway through the study. |
|
Raskob, 2018 |
Definitely yes – “Randomization was performed with the use of an interactive Web-based system, with stratification according to whether risk factors for bleeding were present and whether the patient met the criteria to receive a lower dose of edoxaban.” |
Definitely yes – Allocation concealment via web system. |
Definitely yes – Blinded outcome adjudication committee ensured unbiased assessment. |
Definitely yes |
Definitely yes |
Definitely no
The trial was sponsored by industry and was heavily involved in every aspect of the trial. |
High Risk of bias – all outcome
Due to industry sponsor being heavily involved in every aspect of the trial. |
|
McBane, 2020 |
Definitely yes – Randomized controlled trial with clear procedure. Stratification factors used for randomization include cancer stage and cancer specific risk of venous thromboembolism using the Khorana score. |
Probably yes – Randomization mentioned, but concealment method unclear. |
Probably yes – Open-label design with no blinding for bleeding outcomes. Major bleeding and thrombotic events were centrally adjudicated without knowledge of the patient treatment assignment. Although CRNMB was not blinded adjudicated, there is a clear definition for this outcome. |
Definitely yes |
Definitely yes |
Probably yes
The trial received a grant from the industry. Most likely the sponsor had no influence on the design, etc. because the authors stated: “The primary investigator, in collaboration with the Academic and Community Cancer Research United (ACCRU) research consortium, was responsible for the trial design, protocol development, data collection, statistical analysis, data interpretation, manuscript preparation, and trial oversight.” |
Low Risk of bias - for all outcomes |
|
Agnelli, 2020 |
Definitely yes – Computer-generated random sequence with stratification stratified according to the type of venous thromboembolism (symptomatic or incidental) and timing of the cancer diagnosis (active or historical). |
Definitely yes – Centralized randomization via interactive web system ensured concealment. |
Probably yes – Outcomes assessed by blinded committee, but open-label design may influence patient behavior. Comment by authors on blinding: “double-blinding would have required daily subcutaneous injection of placebo in the apixaban recipients. Ethically, the use of daily injectable placebo and the related inconvenience for patients that already need complex treatment for cancer are difficult to justify.”
|
Definitely yes – Low loss to follow-up; outcomes assessed for nearly all randomized patients. |
Definitely yes |
Definitely yes
Although the trial was sponsored by the industry, the grant was unrestricted and the sponsor had no conduct in the design, data analysis, etc.
|
Low Risk of bias - for all outcomes |
|
Planquette, 2022 |
Definitely yes – “Randomization was performed centrally through an interactive web response system (CleanWeb) and was stratified according to center and disease stage (localized or locally advanced vs metastatic).” |
Definitely yes – Allocation sequence concealed through electronic system. |
Definitely yes – Blinded central adjudication committee assessed outcomes. |
Definitely yes |
Definitely yes |
Definitely yes
Trial was supported by an unrestricted grant from the industry. |
Low Risk of bias - for all outcomes |
|
Schrag, 2023 |
Definitely yes – Randomization process and blocking per site clearly described. |
Definitely yes – Concealment ensured through central assignment. |
Definitely yes - Treating physician was blinded to group assignment. Outcomes were adjudicated blinded to group assignment for all outcomes. |
Definitely yes |
Definitely yes |
Definitely yes – Non-industry sponsor was not involved in the design, analysis, etc. |
Low Risk of bias - for all outcomes |
Table of excluded studies 2024
|
Reference |
Reason for exclusion |
|
Ageno W, Vedovati MC, Cohen A, Huisman M, Bauersachs R, Gussoni G, Becattini C, Agnelli G. Bleeding with Apixaban and Dalteparin in Patients with Cancer-Associated Venous Thromboembolism: Results from the Caravaggio Study. Thromb Haemost. 2021 May;121(5):616-624. doi: 10.1055/s-0040-1720975. Epub 2020 Nov 17. PMID: 33202447. |
Post-hoc analysis of original RCT (Agnelli 2020) |
|
Agnelli G, Muñoz A, Franco L, Mahé I, Brenner B, Connors JM, Gussoni G, Hamulyak EN, Lambert C, Suero MR, Bauersachs R, Torbicki A, Becattini C. Apixaban and Dalteparin for the Treatment of Venous Thromboembolism in Patients with Different Sites of Cancer. Thromb Haemost. 2022 May;122(5):796-807. doi: 10.1055/s-0041-1735194. Epub 2021 Sep 16. PMID: 34530482. |
Post-hoc analysis of original RCT (Agnelli 2020) |
|
Al-Mansouri L, AL-Obaidi FR, Altaweel RB, Soto-Perez-de-Celis E, Al-Farhan H, Abdullah NA, Alrubaiy L. Direct oral anticoagulants for the prevention and treatment of venous thromboembolic events in cancer patients: a meta-analysis of randomized controlled trials. J Cancer Prev Curr Res. 2024;20(1):100–105. Published 2023 May 19. |
Not a primary study
|
|
Al-Tourah L, Mithoowani S, Lim W, Ikesaka R. The incidence of major bleeding in adult patients with urogenital and gynecological cancer being treated with direct oral anticoagulants (DOACs): a systematic review. J Thromb Thrombolysis. 2024 Apr;57(4):630-637. doi: 10.1007/s11239-024-02956-5. Epub 2024 Mar 1. PMID: 38429468. |
Not a primary study
|
|
Alikhan R, Gomez K, Maraveyas A, Noble S, Young A, Thomas M; British Society for Haematology. Cancer-associated venous thrombosis in adults (second edition): A British Society for Haematology Guideline. Br J Haematol. 2024 Jul;205(1):71-87. doi: 10.1111/bjh.19414. Epub 2024 Apr 25. PMID: 38664942. |
Not a primary study, guideline
|
|
Bosch FTM, Mulder FI, Huisman MV, Zwicker JI, Di Nisio M, Carrier M, Segers A, Verhamme P, Middeldorp S, Weitz JI, Grosso MA, Duggal A, Büller HR, Wang TF, Garcia D, Kamphuisen PW, Raskob GE, van Es N. Risk factors for gastrointestinal bleeding in patients with gastrointestinal cancer using edoxaban. J Thromb Haemost. 2021 Dec;19(12):3008-3017. doi: 10.1111/jth.15516. Epub 2021 Sep 12. PMID: 34455706; PMCID: PMC9292167. |
Did not adhere to PICO |
|
Chatani R, Yamashita Y, Morimoto T, Muraoka N, Umetsu M, Nishimoto Y, Takada T, Ogihara Y, Nishikawa T, Ikeda N, Otsui K, Sueta D, Tsubata Y, Shoji M, Shikama A, Hosoi Y, Tanabe Y, Tsukahara K, Nakanishi N, Kim K, Ikeda S, Mushiake K, Kadota K, Ono K, Kimura T. Edoxaban for 12 vs. 3 months in cancer-associated isolated distal deep vein thrombosis according to different doses: insights from the ONCO DVT study. Eur Heart J Cardiovasc Pharmacother. 2024 Aug 14;10(5):422-431. doi: 10.1093/ehjcvp/pvae028. PMID: 38650055; PMCID: PMC11323369. |
Post-hoc analysis of Yamashita 2023
|
|
Chaudhury A, Balakrishnan A, Thai C, Holmstrom B, Nanjappa S, Ma Z, Jaglal MV. The Efficacy and Safety of Rivaroxaban and Dalteparin in the Treatment of Cancer Associated Venous Thrombosis. Indian J Hematol Blood Transfus. 2018 Jul;34(3):530-534. doi: 10.1007/s12288-017-0895-8. Epub 2017 Oct 27. PMID: 30127566; PMCID: PMC6081306. |
Did not adhere to PICO |
|
Cohen AT, Creeper KJ, Alikhan R, Er C, Connors JM, Huisman MV, Munoz A, Vescovo G, Bauersachs R, Ageno W, Agnelli G, Becattini C. Early Time Courses of Recurrent Venous Thromboembolism and Bleeding during Apixaban or Dalteparin Therapy for Patients with Cancer. Thromb Haemost. 2024 Jul;124(7):676-683. doi: 10.1055/s-0043-1778642. Epub 2024 Jan 9. PMID: 38196077. |
Secondary publication of Agnelli (2020)
|
|
Di Nisio M, van Es N, Carrier M, Wang TF, Garcia D, Segers A, Weitz J, Buller H, Raskob G. Extended treatment with edoxaban in cancer patients with venous thromboembolism: A post-hoc analysis of the Hokusai-VTE Cancer study. J Thromb Haemost. 2019 Nov;17(11):1866-1874. doi: 10.1111/jth.14561. Epub 2019 Jul 25. PMID: 31271705. |
Secondary publication of Raskob (2018) |
|
Grifoni E, Baroncelli A, Pinto G, Cosentino E, Micheletti I, Signorini I, Panigada G, Landini G, Masotti L. Efficacy and Safety of Edoxaban in Cancer-Associated Venous Thromboembolism: A Real World Retrospective Study. TH Open. 2022 Mar 1;6(2):e99-e106. doi: 10.1055/a-1783-9744. PMID: 35707622; PMCID: PMC9113858. |
Did not adhere to PICO (not a randomized controlled trial) |
|
Kraaijpoel N, Di Nisio M, Mulder FI, van Es N, Beyer-Westendorf J, Carrier M, Garcia D, Grosso M, Kakkar AK, Mercuri MF, Middeldorp S, Hernandez CR, Santamaria A, Schwocho L, Segers A, Verhamme P, Wang TF, Weitz JI, Zhang G, Zwicker JI, Büller HR, Raskob GE. Clinical Impact of Bleeding in Cancer-Associated Venous Thromboembolism: Results from the Hokusai VTE Cancer Study. Thromb Haemost. 2018 Aug;118(8):1439-1449. doi: 10.1055/s-0038-1667001. Epub 2018 Jul 30. PMID: 30060256. |
Secondary publication of Raskob (2018) |
|
Mahe I, Muñoz A, Cohen A, Raskob G, Mahé I, Bauersachs R, Vedovati MC, Brenner B, Connors JM, Gussoni G, Huisman MV, Suero MR, Torbicki A, Agnelli G, Becattini C. Reduced-dose versus full-dose apixaban in cancer patients with venous thromboembolism: the API-CAT non-inferiority randomized trial. Lancet Haematol. 2025 Feb;10(2):e99-e109. doi: 10.1016/S2352-3026(24)00012-7. PMID: 38435012. |
Dose comparison trial for extended VTE treatment with apixaban |
|
Marshall A, Campbell C, Hill C, Hale D, Thirlwall J, Parpia S, Maraveyas A, Poole C, Levine M, Young AM. Treatment of cancer-associated venous thromboembolism: 12-month outcomes of the placebo versus rivaroxaban randomization of the SELECT-D Trial (SELECT-D: 12m). J Thromb Haemost. 2020 May;18(5):905-915. doi: 10.1111/jth.14740. PMID: 32087556. |
Compared with placebo, not LMWH |
|
McBane RD, Tafur A, Le-Rademacher JG, Fashanu OE, Khorana AA, Lyman GH, Milhem M, McCrae KR, Wysokinski WE, Ashrani AA. Extending venous thromboembolism secondary prevention with apixaban in cancer patients: The EVE randomized clinical trial. J Thromb Haemost. 2024 Jan;22(1):97-108. doi: 10.1111/jth.16585. PMID: 37816545. |
Dose comparison trial for extended VTE treatment with apixaban |
|
Moik F, Colling M, Mahé I, Jara-Palomares L, Pabinger I, Ay C. Extended anticoagulation treatment for cancer-associated thrombosis-Rates of recurrence and bleeding beyond 6 months: A systematic review. J Thromb Haemost. 2022 Mar;20(3):619-634. doi: 10.1111/jth.15599. Epub 2021 Dec 8. PMID: 34816583; PMCID: PMC9299994. |
Not a primary study |
|
Mokadem ME, Hassan A, Algaby AZ. Efficacy and safety of apixaban in patients with active malignancy and acute deep venous thrombosis. Vascular. 2021 Oct;29(5):745-750. doi: 10.1177/1708538120971148. Epub 2020 Nov 5. PMID: 33153401. |
Not randomized or comparative |
|
Palumbo JS, Lensing AWA, Brandão LR, Hooimeijer HL, Kenet G, van Ommen H, Pap AF, Majumder M, Kubitza D, Thelen K, Willmann S, Prins MH, Monagle P, Male C. Anticoagulation in pediatric cancer-associated venous thromboembolism: a subgroup analysis of EINSTEIN-Jr. Blood Adv. 2022 Nov 22;6(22):5821-5828. doi: 10.1182/bloodadvances.2022008160. PMID: 36006613; PMCID: PMC9641171. |
Pediatric population, not adult
|
|
Pina E, Antonio M, Peris J, Rosselló E, Domènech P, Peñafiel J, Tebe C. Bemiparin as a long-term treatment for venous thrombosis in cancer patients: the ELEBAMA study. Clin Transl Oncol. 2020 Apr;22(4):616-620. doi: 10.1007/s12094-019-02159-y. Epub 2019 Jun 19. PMID: 31218649. |
Did not adhere to PICO
|
|
Ullah F, Song J, Rojas Hernandez CM, Kroll MH, Escalante CP, Toale KM. Safety and Effectiveness of Direct Oral Anticoagulants for the Treatment of Gastrointestinal Cancer-Associated Venous Thromboembolism. Oncologist. 2023 Nov 2;28(11):e1005-e1016. doi: 10.1093/oncolo/oyad148. PMID: 37310796; PMCID: PMC10628557. |
Wrong study design
|
|
Tia JM, Casapao JC, Liangco W, Gaspar-Trinidad E. Treatment efficacy and risk of bleeding among cancer patients treated for venous thromboembolism with dabigatran compared to warfarin. Phil J Intern Med. 2018;56:252–257. |
Did not adhere to PICO |
|
Yamashita Y, Morimoto T, Amano H, Hasegawa K, Okabe H, Tsubata Y, Nishimoto Y, Umetsu M, Takase T, Muraoka N, Ikeda S, Fukuda K. Edoxaban for 12 Months Versus 3 Months in Patients with Cancer with Isolated Distal Deep Vein Thrombosis (ONCO DVT Study): An Open-Label, Multicenter, Randomized Clinical Trial. JAMA Intern Med. 2023 Oct 1;183(10):1056-1064. doi: 10.1001/jamainternmed.2023.3764. PMID: 37627168. |
Comparison between two durations of treatment with edoxaban. |
Table of excluded studies 2018
|
Author, year |
Reason for exclusion |
|
Van der Hulle 2014 |
Meta-analysis DOAC versus VKA |
|
Carrier 2014 |
No comparison between DOAC and LMWH |
|
Di Minno 2017 |
Meta-analysis DOAC versus VKA |
|
Brunetti 2017 |
Meta-analysis DOAC versus VKA |
|
Posch 2015 |
Meta-analysis DOAC versus VKA |
|
Vedovati 2015 |
Meta-analysis DOAC versus VKA |
|
Larsen 2014 |
Meta-analysis DOAC versus VKA |
|
Van En 2014 |
Meta-analysis DOAC versus VKA |
|
Schulman 2016 |
Meta-analysis DOAC versus VKA |
|
Agnelli 2015 |
Non-randomized study |
|
Bott-Kitslaar 2016 |
Non-randomized study |
|
Davies 2018 |
Non-randomized study |
|
Mancuso 2016 |
Non-randomized study |
|
Mantha 2017 |
Non-randomized study |
|
Raskob 2016 |
Non-randomized study |
|
Schulman 2015 |
Non-randomized study |
|
Simmons 2018 |
Non-randomized study |
|
Alzghari 2017 |
Non-randomized study |
|
Streiff 2018 |
Non-randomized study |
|
Ross 2017 |
Non-randomized study |
|
Xiang 2018 |
Non-randomized study |
|
Hirsh 2018 |
Editorial |
|
Lee 2018 |
Narrative review |
|
Noble 2016a |
Narrative review |
|
Noble 2016b |
Narrative review |
Beoordelingsdatum en geldigheid
Publicatiedatum : 01-05-2026
Beoordeeld op geldigheid : 01-05-2026
Samenstelling werkgroep
Voor het ontwikkelen van de richtlijnmodule is in 2022 een multidisciplinaire cluster ingesteld. Het cluster Antitrombotisch beleid bestaat uit meerdere richtlijnen, zie hier voor de actuele clusterindeling. De stuurgroep bewaakt het proces van modulair onderhoud binnen het cluster. De expertisegroepsleden geven hun expertise in, indien nodig. De volgende personen uit het cluster zijn betrokken geweest bij de herziening van deze module:
Clusterstuurgroepleden
- Prof. dr. M.V. (Menno) Huisman, internist-vasculaire geneeskunde, LUMC, NIV (voorzitter)
- Dr. M.J.H.A. (Marieke) Kruip, internist-hematoloog, Erasmus MC, NIV, NVVH (Nederlandse Vereniging voor Hematologie)
- Dr. S.H. (Steven) Renes, anesthesioloog, Radboudumc, NVA
- Dr. L.M. (Linda) de Heer, cardiothoracaal chirurg, UMC Utrecht, NVT
- Dr. A.K. Stroobants, klinisch chemicus, Radboudumc, NVKC
- Drs. E. (Egbert) Krug, traumachirurg, LUMC, NVvH
- Dr. H.B. (Harmen) Ettema, orthopedisch chirurg, Isala, NOV
- Dr. B. (Banne) Nemeth, aios orthopedie, LUMC, NOV
- Dr. M.E. (Maarten) Tushuizen, maag-darm-leverarts, LUMC, NVMDL
- Dr. C.H. (Heleen) van Ommen, kinderarts-hematoloog, Erasmus MC, NVK
- Dr. E.J. (Esther) Nossent, longarts, Amsterdam UMC, NVALT
- Dr. N. (Nakisa) Khorsand, ziekenhuisapotheker, OLVG, NVZA
- Dr. S.A. (Sonja) de Munnik, klinisch geneticus, Radboudumc, VKGN
Betrokken clusterexpertisegroepleden
- Dr. M.N. (Mandy) Lauw, internist-hematoloog, Erasmus MC, NIV, NVVH (Nederlandse Vereniging voor Hematologie)
- Prof. Dr. F.A. (Erik) Klok, internist-vasculaire geneeskunde, LUMC, NIV
Meelezende clusterexpertisegroepleden
- Dr. J. (Josien) van Es, longarts, Amsterdam UMC, NVALT
Met ondersteuning van
- H. (Hanneke) Olthuis, adviseur, Kennisinstituut van de Federatie Medisch Specialisten (tot 1 februari 2026)
- Dr. B.H. (Bernardine) Stegeman, senior adviseur, Kennisinstituut van de Federatie Medisch Specialisten
- E. (Esther) van der Bijl, Informatiespecialist, Kennisinstituut van de Federatie Medisch Specialisten
Belangenverklaringen
Een overzicht van de belangen van de clusterleden en het oordeel over het omgaan met eventuele belangen vindt u in onderstaande tabel. De ondertekende belangenverklaringen zijn op te vragen bij het secretariaat van het Kennisinstituut van de Federatie Medisch Specialisten via secretariaat@kennisinstituut.nl.
Tabel Gemelde (neven)functies en belangen stuurgroepleden en betrokken clusterexpertisegroepleden
|
Naam |
Hoofdfunctie |
Nevenwerkzaamheden |
Persoonlijke financiële belangen |
Persoonlijke relaties |
Extern gefinancierd onderzoek |
Intellectuele belangen en reputatie |
Overige belangen |
Restrictie |
|
Stuurgroepleden |
||||||||
|
Huisman (vz) |
internist LUMC Leiden |
Voorzitter nederlands kennisplatform antistollingszorg onbetaald; |
geen |
geen |
Hartstichting, COVID en herseninfarct, projectleider ja; |
nee |
nvt |
Geen restricties |
|
Ettema |
Orthopedisch chirurg Isala Zwolle |
Geen |
Geen |
Geen |
Geen |
Geen |
Geen |
Geen restricties (in 2025 geen herbevestiging ontvangen) |
|
de Heer |
Cardio-thoracaal chirurg |
Geen |
Geen |
Geen |
Geen |
Geen |
Geen |
Geen restricties |
|
Stroobants |
Klinisch chemicus |
Bestuur vakvereniging VHL (secretaris, onbetaald), RvT ECAT (betaald, aan RadboudUMC) |
Dienstverband RadboudUMC |
n.v.t. |
n.v.t. |
Geen |
n.v.t. |
Geen restricties |
|
Tushuizen |
MDL-arts LUMC |
lid Horizonscan Geneesmiddelen, onbetaald |
Geen |
Geen |
TNO, markers voor fibrose, rol als projectleider |
Geen |
Geen |
Geen restricties |
|
Khorsand |
Ziekenhuisapotheker OLVG |
Voorzitter SIG hematologie NVZA (onbetaald); Voorzitter programma commissie congres NVZA (onbetaald) |
Geen |
Geen |
Geen |
Geen |
Geen |
Geen restricties |
|
Kruip |
Hematoloog Erasmus MC (0.3 FTE) |
Voorzitter Federatie Nederlandse trombosediensten, onbetaald (afgerond); RvC Lenticure, onbetaald; Lid guideline committee European Hematology Association, onbetaald |
Geen |
Geen |
ZonMw en Trombosestichting NL, Trombose bij Covid 19 (afgerond); ZonMw, VITT na Coronavaccinatie (afgerond); FNT, coronavaccinatie bij VKA (afgerond); Trombosestichting NL - Antiplatelet Therapy and menstrual blood loss; PARASOL study (Projectleider JA); Horizon Europe - SERENITY: Improved supportive, palliative, survivorship and end-of-life care of cancer patients (Local PI); Zorginstituut, Recognition and registration of low health literary in patients with diabetes to enable shared decision making (Projectleider NEE); ZonMW, Bloedstollend veilig: Safety-II education focused on anticoagulants in master of Medicine (Projectleider NEE); Citrienfonds, Inclusieve zorg voor mensen met geldzorgen in digitale tijden (projectleider NEE) |
Geen |
Sprekersvergoeding, betaald aan het Erasmus MC van Sobi, Roche en BMS (afgerond) |
Geen restricties (onderwerp van commercieel gefinancieerd onderzoek valt buiten bestek van deze cyclus) |
|
Renes |
Anesthesioloog-pijnspecialist, RadboudUMC (betaald) |
Kwaliteitsvisitatie NVA (vacatiegeld) |
geen |
geen |
Geen |
geen |
geen |
Geen restricties |
|
van Ommen |
Hoofd afd. Kinderhematologie & kinderoncologie Erasmus MC Sophia Kinderziekenhuis |
Geen |
geen |
Geen |
Fase 3 trial effectiviteit van edoxaban voor behandeling van trombose bij kinderen, financier Daiichi Sankyo, local PI en steeringCie (afgesloten in 2022); Microscopic evaluation of clots in ECMO systems, Octapharma, rol als PI; IPTN ThromPED registry (international observational registry/study of children with thrombosis, financier BI, BMS en INVENT, rol als chair IPTN |
Geen |
Adviseur voor verschillende farmaceuten voor ontwikkeling van onderzoek van antistollingsmiddelen of antidota bij kinderen zoals Asundexian (Bayer BV), andexanet (Astra Zeneca) |
Geen restricties (onderwerp van commercieel gefinancierd onderzoek valt buiten bestek van deze cyclus, adviesrollen neergelegd gedurende de richtlijnontwikkeling.) |
|
Krug |
Chirurg, LUMC |
Geen |
Geen |
Geen |
Geen |
Geen |
Geen |
Geen restricties |
|
de Munnik |
Klinisch geneticus Radboudumc |
Voorzitter commissie kwaliteit VKGN, |
Geen |
Geen |
Geen |
Geen |
Geen |
Geen restricties |
|
Nossent |
Longarts |
NVALT Bronkhorst Colloquium(onbetaald); NVALT CCO Pulmonary Vascular Diseases and ILD; chair (betaald); Scientific Committtee PHA Europe (onbetaald); ERS/ESC taskforce redefining pulmonary arterial hypertension with cardiopulmonary comorbidities; secretary (onbetaald); PHAROS TF PH-CLD (onbetaald); Medical advisory board: MSD. (betaald). |
Geen. |
Geen |
E.J. Nossent has received research grants from: |
Geen |
Geen |
Geen restricties (studies en betreffen onderwerpen die niet relevant zijn in deze cyclus) |
|
Betrokken clusterexpertisegroepleden |
||||||||
|
Klok |
Internist, LUMC Leiden, betaald (1 FTE) Universiteit van Mainz (onbetaald) |
voorzitter NVIVG Diverse richtlijncommissies en werkgroepen van ESC en ERS (onbetaald)
|
Geen |
Geen |
• Bayer, Leo Pharma en BSCI (toelichting: Ik heb recent een project voorgezeten om met een internationale werkgroep een standaard set van uitkomsten (ICHOM) te maken voor VTE. Dit project werd financieel ondersteund door Bayer (40k EUR), BSCI (40k EUR) en Leo Pharma (40k EUR). Dit betrof ‘ unrestricted’ grants, waarbij deze sponsoren op geen enkele manier invloed hadden op de inhoud van het projetct, dat in 2020 begon en nu is afgerond), ) |
Geen |
Geen |
Geen restricties (onderwerp van commercieel gefinancierd onderzoek valt buiten bestek van deze cyclus) |
|
Lauw |
Internist-hematoloog, Erasmus MC Rotterdam, afdeling hematologie |
Scientific editor HemaSphere, betaald, EHA Faculty CRTH, betaald Editorial board member van diverse jounrals in hematologi en trombose, onbetaald |
nvt |
nvt |
1. ZONMW- Prevention of venous 4. ISTH, Splanchnic Venous Thrombosis Outcomes in Patients with Myeloproliferative Neoplasms (SSC grant, projectleidersrol) 5. INVENT-VTE, ADAPTiON-study, Apixaban Dose Adjustment in Patient with Thrombocytopenia in Oncology, rol als projectleider |
Nvt |
Sprekersvergoedingen, betaald aan het Erasmus MC, van: Inari, MEDtalks, Viatris, Amgen, AbbVie |
Restrictie t.a.v. besluitvorming over tromboseprofylaxe bij patiënten met ALL en behandeling van VTE bij patiënten met hematologische maligniteiten. |
Inbreng patiëntenperspectief
Kwalitatieve raming van mogelijke financiële gevolgen in het kader van de Wkkgz
Bij de richtlijnmodule voerden de clusterleden conform de Wet kwaliteit, klachten en geschillen zorg (Wkkgz) een kwalitatieve raming uit om te beoordelen of de aanbevelingen mogelijk leiden tot substantiële financiële gevolgen. Bij het uitvoeren van deze beoordeling is de richtlijnmodule op verschillende domeinen getoetst (zie het stroomschema bij Werkwijze).
|
Module |
Uitkomst raming |
Toelichting |
|
Maligniteit geassocieerde trombose |
geen financiële gevolgen |
Hoewel uit de toetsing volgt dat de aanbeveling(en) breed toepasbaar zijn (5.000-40.000 patiënten), volgt ook uit de toetsing dat het overgrote deel (±90%) van de zorgaanbieders en zorgverleners al aan de norm voldoet. Er worden daarom geen substantiële financiële gevolgen verwacht. |
Werkwijze
Voor meer details over de gebruikte richtlijnmethodologie verwijzen wij u naar de Werkwijze. Relevante informatie voor de ontwikkeling/herziening van deze richtlijnmodule is hieronder weergegeven.
Zoekverantwoording
Zoekstrategie – 17 oktober 2024
Embase.com
|
No. |
Query |
Results |
|
#1 |
'venous thromboembolism'/exp OR 'deep vein thrombosis'/exp OR 'lung embolism'/exp OR (((venous OR vein* OR lung* OR pulmon*) NEAR/3 (embol* OR microembol* OR thromboembol*)):ti,ab,kw) OR vte:ti,ab,kw OR dvt:ti,ab,kw OR ((deep* NEAR/3 (thromb* OR 'blood clot*' OR embol*)):ti,ab,kw) |
266601 |
|
#2 |
'neoplasm'/exp OR adenoma*:ti,ab,kw OR anticarcinogen*:ti,ab,kw OR blastoma*:ti,ab,kw OR cancer*:ti,ab,kw OR carcinogen*:ti,ab,kw OR carcinom*:ti,ab,kw OR carcinosarcom*:ti,ab,kw OR chordoma*:ti,ab,kw OR germinoma*:ti,ab,kw OR gonadoblastom*:ti,ab,kw OR hepatoblastom*:ti,ab,kw OR ((hodgkin* NEXT/1 disease*):ti,ab,kw) OR leukemi*:ti,ab,kw OR lymphangioma*:ti,ab,kw OR lymphangiomyoma*:ti,ab,kw OR lymphangiosarcom*:ti,ab,kw OR lymphom*:ti,ab,kw OR malign*:ti,ab,kw OR melanom*:ti,ab,kw OR meningiom*:ti,ab,kw OR mesenchymom*:ti,ab,kw OR mesonephrom*:ti,ab,kw OR metasta*:ti,ab,kw OR neoplas*:ti,ab,kw OR neuroma*:ti,ab,kw OR nsclc:ti,ab,kw OR oncogen*:ti,ab,kw OR oncolog*:ti,ab,kw OR paraneoplastic*:ti,ab,kw OR plasmacytom*:ti,ab,kw OR precancer*:ti,ab,kw OR sarcoma*:ti,ab,kw OR teratocarcinom*:ti,ab,kw OR teratom*:ti,ab,kw OR tumor*:ti,ab,kw OR tumour*:ti,ab,kw |
7825666 |
|
#3 |
'thrombosis prevention'/exp OR 'anticoagulant agent'/exp OR 'anticoagulation'/exp OR 'anticoagulant therapy'/exp OR 'apixaban'/exp OR 'dabigatran'/exp OR 'rivaroxaban'/exp OR 'edoxaban'/exp OR 'blood clotting factor 10a inhibitor'/exp OR thromboprophyla*:ti,ab,kw OR (((thrombo* OR antithrombo* OR vte) NEAR/3 (prophylaxis OR prophylactic OR prevention)):ti,ab,kw) OR 'anti coagulant*':ti,ab,kw OR 'anticoagulant*':ti,ab,kw OR 'anticoagulat*':ti,ab,kw OR 'anti coagulat*':ti,ab,kw OR 'antithrombotic*':ti,ab,kw OR 'anti thrombotic*':ti,ab,kw OR 'antithrombos*':ti,ab,kw OR 'anti thrombos*':ti,ab,kw OR 'antithrombocytic*':ti,ab,kw OR 'anti thrombocytic*':ti,ab,kw OR 'antiplatelet agent*':ti,ab,kw OR 'antiplatelet drug*':ti,ab,kw OR 'platelet aggregation inhibitor*':ti,ab,kw OR 'platelet inhibitor*':ti,ab,kw OR 'platelet antagonist*':ti,ab,kw OR 'thrombocyte aggregation inhibiting agent*':ti,ab,kw OR 'thrombocyte aggregation inhibitor*':ti,ab,kw OR 'direct oral anticoagulant agent'/exp OR 'direct oral anticoagulant'/exp OR doac*:ti,ab,kw OR oac:ti,ab,kw OR oacs:ti,ab,kw OR 'aboxoma':ti,ab,kw OR 'apixaban':ti,ab,kw OR 'apixaben':ti,ab,kw OR 'bms 562247':ti,ab,kw OR 'bms 562247 01':ti,ab,kw OR 'bms562247':ti,ab,kw OR 'eliques':ti,ab,kw OR 'eliquis':ti,ab,kw OR 'lunast':ti,ab,kw OR 'pf 0465257':ti,ab,kw OR 'pf0465257':ti,ab,kw OR 'tah 3311':ti,ab,kw OR 'tah 3341':ti,ab,kw OR 'tah3311':ti,ab,kw OR 'tah3341':ti,ab,kw OR 'bibr 953':ti,ab,kw OR 'bibr953':ti,ab,kw OR 'dabigatran':ti,ab,kw OR 'aban':ti,ab,kw OR 'assubex':ti,ab,kw OR 'ast 8294':ti,ab,kw OR 'ast8294':ti,ab,kw OR 'bay 59 7939':ti,ab,kw OR 'bay 597939':ti,ab,kw OR 'bay59 7939':ti,ab,kw OR 'bay597939':ti,ab,kw OR 'bs 112':ti,ab,kw OR 'bs112':ti,ab,kw OR 'dst 8294':ti,ab,kw OR 'dst8294':ti,ab,kw OR 'jnj 39039039':ti,ab,kw OR 'jnj39039039':ti,ab,kw OR 'kriva':ti,ab,kw OR 'naxat':ti,ab,kw OR 'rivaro':ti,ab,kw OR 'rivarolto':ti,ab,kw OR 'rivaroxaban':ti,ab,kw OR 'rivaxa':ti,ab,kw OR 'throsaben':ti,ab,kw OR 'xanirva':ti,ab,kw OR 'xarelto':ti,ab,kw OR 'xerdoxo':ti,ab,kw OR 'xindus':ti,ab,kw OR 'du 176':ti,ab,kw OR 'du 176b':ti,ab,kw OR 'du176':ti,ab,kw OR 'du176b':ti,ab,kw OR 'edoxaban':ti,ab,kw OR 'edoxaban tosilate':ti,ab,kw OR 'edoxaban tosylate':ti,ab,kw OR 'endoxaban':ti,ab,kw OR 'lixiana':ti,ab,kw OR 'roteas':ti,ab,kw OR 'savaysa':ti,ab,kw OR ((('factor 10a' OR 'factor xa') NEAR/3 'inhibit*'):ti,ab,kw) |
959177 |
|
#4 |
#1 AND #2 AND #3 |
26130 |
|
#5 |
#4 AND [2018-2025]/py NOT ('conference abstract'/it OR 'editorial'/it OR 'letter'/it OR 'note'/it) NOT (('animal'/exp OR 'animal experiment'/exp OR 'animal model'/exp OR 'nonhuman'/exp) NOT 'human'/exp) |
7013 |
|
#6 |
'meta analysis'/exp OR 'meta analysis (topic)'/exp OR metaanaly*:ti,ab OR 'meta analy*':ti,ab OR metanaly*:ti,ab OR 'systematic review'/de OR 'cochrane database of systematic reviews'/jt OR prisma:ti,ab OR prospero:ti,ab OR (((systemati* OR scoping OR umbrella OR 'structured literature') NEAR/3 (review* OR overview*)):ti,ab) OR ((systemic* NEAR/1 review*):ti,ab) OR (((systemati* OR literature OR database* OR 'data base*') NEAR/10 search*):ti,ab) OR (((structured OR comprehensive* OR systemic*) NEAR/3 search*):ti,ab) OR (((literature NEAR/3 review*):ti,ab) AND (search*:ti,ab OR database*:ti,ab OR 'data base*':ti,ab)) OR (('data extraction':ti,ab OR 'data source*':ti,ab) AND 'study selection':ti,ab) OR ('search strategy':ti,ab AND 'selection criteria':ti,ab) OR ('data source*':ti,ab AND 'data synthesis':ti,ab) OR medline:ab OR pubmed:ab OR embase:ab OR cochrane:ab OR (((critical OR rapid) NEAR/2 (review* OR overview* OR synthes*)):ti) OR ((((critical* OR rapid*) NEAR/3 (review* OR overview* OR synthes*)):ab) AND (search*:ab OR database*:ab OR 'data base*':ab)) OR metasynthes*:ti,ab OR 'meta synthes*':ti,ab |
1070871 |
|
#7 |
'randomized controlled trial'/exp OR random*:ti,ab OR (((pragmatic OR practical) NEAR/1 'clinical trial*'):ti,ab) OR ((('non inferiority' OR noninferiority OR superiority OR equivalence) NEAR/3 trial*):ti,ab) OR rct:ti,ab,kw |
2257783 |
|
#8 |
#5 AND #6 - SR |
646 |
|
#9 |
#5 AND #7 NOT #8 - RCT |
505 |
|
#10 |
#8 OR #9 - Totaal |
1151 |
Ovid/Medline
|
# |
Searches |
Results |
|
1 |
Venous Thromboembolism/ or exp Venous Thrombosis/ or exp Pulmonary Embolism/ or ((venous or vein* or lung* or pulmon*) adj3 (embol* or microembol* or thromboembol*)).ti,ab,kf. or vte.ti,ab,kf. or dvt.ti,ab,kf. or (deep adj3 (thromb* or 'blood clot*' or embol*)).ti,ab,kf. |
159254 |
|
2 |
exp Neoplasms/ or adenoma*.ti,ab,kf. or anticarcinogen*.ti,ab,kf. or blastoma*.ti,ab,kf. or cancer*.ti,ab,kf. or carcinogen*.ti,ab,kf. or carcinom*.ti,ab,kf. or carcinosarcoma*.ti,ab,kf. or chordoma*.ti,ab,kf. or germinoma*.ti,ab,kf. or gonadoblastom*.ti,ab,kf. or hepatoblastom*.ti,ab,kf. or (hodgkin* adj1 disease*).ti,ab,kf. or leukemi*.ti,ab,kf. or lymphangioma*.ti,ab,kf. or lymphangiomyoma*.ti,ab,kf. or lymphangiosarcom*.ti,ab,kf. or lymphom*.ti,ab,kf. or malign*.ti,ab,kf. or melanom*.ti,ab,kf. or meningiom*.ti,ab,kf. or mesenchymom*.ti,ab,kf. or mesonephrom*.ti,ab,kf. or metasta*.ti,ab,kf. or neoplas*.ti,ab,kf. or neuroma*.ti,ab,kf. or nsclc.ti,ab,kf. or oncogen*.ti,ab,kf. or oncolog*.ti,ab,kf. or paraneoplastic*.ti,ab,kf. or plasmacytom*.ti,ab,kf. or precancer*.ti,ab,kf. or sarcoma*.ti,ab,kf. or teratocarcinom*.ti,ab,kf. or teratom*.ti,ab,kf. or tumor*.ti,ab,kf. or tumour*.ti,ab,kf. |
5600681 |
|
3 |
exp Anticoagulants/ or exp Platelet Aggregation Inhibitors/ or exp Dabigatran/ or exp Rivaroxaban/ or exp Factor Xa Inhibitors/ or thromboprophyla*.ti,ab,kf. or ((thrombo* or antithrombo* or vte) adj3 (prophylaxis or prophylactic or prevention)).ti,ab,kf. or (anti coagulant* or anticoagulant* or anticoagulat* or anti coagulat* or antithrombotic* or anti thrombotic* or 'antithrombos*' or 'anti thrombos*' or antithrombocytic* or anti thrombocytic* or 'antiplatelet agent*' or 'antiplatelet drug*' or 'platelet aggregation inhibitor*' or 'platelet inhibitor*' or platelet antagonist* or 'thrombocyte aggregation inhibiting agent*' or 'thrombocyte aggregation inhibitor*' or doac*).ti,ab,kf. or oac.ti,ab,kf. or oacs.ti,ab,kf. or aboxoma.ti,ab,kf. or apixaban.ti,ab,kf. or apixaben.ti,ab,kf. or bms 562247.ti,ab,kf. or "bms 562247 01".ti,ab,kf. or bms562247.ti,ab,kf. or eliques.ti,ab,kf. or eliquis.ti,ab,kf. or lunast.ti,ab,kf. or "pf 0465257".ti,ab,kf. or pf0465257.ti,ab,kf. or tah 3311.ti,ab,kf. or tah 3341.ti,ab,kf. or tah3311.ti,ab,kf. or tah3341.ti,ab,kf. or bibr 953.ti,ab,kf. or bibr953.ti,ab,kf. or dabigatran.ti,ab,kf. or aban.ti,ab,kf. or assubex.ti,ab,kf. or ast 8294.ti,ab,kf. or ast8294.ti,ab,kf. or bay 59 7939.ti,ab,kf. or bay 597939.ti,ab,kf. or bay59 7939.ti,ab,kf. or bay597939.ti,ab,kf. or bs 112.ti,ab,kf. or bs112.ti,ab,kf. or dst 8294.ti,ab,kf. or dst8294.ti,ab,kf. or jnj 39039039.ti,ab,kf. or jnj39039039.ti,ab,kf. or kriva.ti,ab,kf. or naxat.ti,ab,kf. or rivaro.ti,ab,kf. or rivarolto.ti,ab,kf. or rivaroxaban.ti,ab,kf. or rivaxa.ti,ab,kf. or throsaben.ti,ab,kf. or xanirva.ti,ab,kf. or xarelto.ti,ab,kf. or xerdoxo.ti,ab,kf. or xindus.ti,ab,kf. or du 176.ti,ab,kf. or du 176b.ti,ab,kf. or du176.ti,ab,kf. or du176b.ti,ab,kf. or edoxaban.ti,ab,kf. or edoxaban tosilate.ti,ab,kf. or edoxaban tosylate.ti,ab,kf. or endoxaban.ti,ab,kf. or lixiana.ti,ab,kf. or roteas.ti,ab,kf. or savaysa.ti,ab,kf. or ((factor 10a or factor xa) adj3 inhibit*).ti,ab,kf. |
455799 |
|
4 |
1 and 2 and 3 |
8718 |
|
5 |
limit 4 to yr="2018 -Current" |
3568 |
|
6 |
5 not (comment/ or editorial/ or letter/) not ((exp animals/ or exp models, animal/) not humans/) |
3399 |
|
7 |
meta-analysis/ or meta-analysis as topic/ or (metaanaly* or meta-analy* or metanaly*).ti,ab,kf. or systematic review/ or cochrane.jw. or (prisma or prospero).ti,ab,kf. or ((systemati* or scoping or umbrella or "structured literature") adj3 (review* or overview*)).ti,ab,kf. or (systemic* adj1 review*).ti,ab,kf. or ((systemati* or literature or database* or data-base*) adj10 search*).ti,ab,kf. or ((structured or comprehensive* or systemic*) adj3 search*).ti,ab,kf. or ((literature adj3 review*) and (search* or database* or data-base*)).ti,ab,kf. or (("data extraction" or "data source*") and "study selection").ti,ab,kf. or ("search strategy" and "selection criteria").ti,ab,kf. or ("data source*" and "data synthesis").ti,ab,kf. or (medline or pubmed or embase or cochrane).ab. or ((critical or rapid) adj2 (review* or overview* or synthes*)).ti. or (((critical* or rapid*) adj3 (review* or overview* or synthes*)) and (search* or database* or data-base*)).ab. or (metasynthes* or meta-synthes*).ti,ab,kf. |
782315 |
|
8 |
exp randomized controlled trial/ or randomized controlled trials as topic/ or random*.ti,ab. or rct?.ti,ab. or ((pragmatic or practical) adj "clinical trial*").ti,ab,kf. or ((non-inferiority or noninferiority or superiority or equivalence) adj3 trial*).ti,ab,kf. |
1762443 |
|
9 |
6 and 7 - SR |
403 |
|
10 |
(6 and 8) not 9 - RCT |
333 |
|
11 |
9 or 10 - Totaal |
736 |
23 april 2018